Laser Spectroscopy for Atmospheric and Environmental Sensing
Abstract
:1. Introduction
2. Laser-induced Fluorescence Spectroscopy
2.1. Introduction
2.2. Review of the Technique
2.2.1. Laser-induced Fluorescence (LIF) for Gas Phase Samples
2.2.2. Laser-Excited Atomic Fluorescence Spectroscopy (LEAFS) for Solids
2.2.3. Laser-induced Breakdown Spectroscopy (LIBS) for Solids, Liquids, and Aerosols
2.3. Applications
2.3.1. Solids
2.3.2. Liquids
2.3.3. Aerosols and Gases
3. Atmospheric Sensing based on Cavity Ring-Down Spectroscopy
3.1. Introduction
3.2. General Principles and Techniques
3.3. Sensing Applications of CRDS
3.3.1. Atmospheric Sensing
3.3.1.1. Trace Gas Detection
3.3.1.1.1. Water
3.2.1.1.2. Carbon Dioxide, Methane, and Carbon Monoxide
3.2.1.1.3. Oxides of Nitrogen
3.2.1.1.4. HOx Radicals
3.3.1.2. Aerosol Measurement
3.3.1.3. Isotope Sensing
3.3.2. Sensing of Atmospherically-relevant Compounds from Biological Sources
3.3.2.1. CO2 Stable Isotope
3.3.2.2. Nitric Oxide
3.3.2.3. Ethane
3.4. Future Outlook
4. Photoluminescence Sensing
4.1. Introduction
4.2. Transition Metal Complexes for VOC Sensing
4.2.1. Gold(I) Complexes
4.2.2. Mixed Gold-Silver Complexes
4.2.3. Mixed Gold-Thallium Complexes
4.2.4. Copper(I) Complexes
4.2.5. Platinum(II) Complexes
4.2.6. Other Metal Complexes
4.3. On/Off Photoluminescence Switching
4.4. Host-guest Molecules
4.5. Photoluminescent Porphyrins
4.6. Additional Remarks
Acknowledgments
References and Notes
- Skoog, D.A.; Hollar, F.J.; Nieman, T.A. Principles of Instrumental Analysis, 5th ed.; Sauders College Publishing: Philadelphia, PA, USA, 1998. [Google Scholar]
- Demchenki, A.P. Introduction to Fluorescence Sensing; Springer Science and Business Media: New York, NY, USA, 2009. [Google Scholar]
- Guilbault, G.G. (Ed.) Practical Fluorescence, 2nd ed.; Marcel Dekker, Inc.: New York, NY, USA, 1990.
- Naik, P.D.; Kumar, A.; Upadhyaya, H.P.; Bajaj, P.N.; Sarkar, S.K. Laser-induced fluorescence (LIF) spectroscopy. Lasers Chem. 2008, 1, 463–502. [Google Scholar]
- Butler, O.T.; Cook, J.M.; Davidson, C.M.; Harrington, C.F.; Miles, D.L. Atomic spectrometry update. Environmental analysis. J. Anal. At. Spectrom. 2009, 24, 131–177. [Google Scholar]
- Butler, O.T.; Cook, J.M.; Harrington, C.F.; Hill, S.J.; Rieuwerts, J.; Miles, D.L. Atomic spectrometry update. Environmental analysis. J. Anal. At. Spectrom. 2005, 20, 130–157. [Google Scholar]
- Butler, O.T.; Cook, J.M.; Harrington, C.F.; Hill, S.J.; Rieuwerts, J.; Miles, D.L. Atomic spectrometry update. Environmental analysis. J. Anal. At. Spectrom. 2006, 21, 217–243. [Google Scholar]
- Butler, O.T.; Cook, J.M.; Davidson, C.M.; Harrington, C.F.; Miles, D.L. Atomic spectrometry update. Environmental analysis. J. Anal. At. Spectrom. 2008, 23, 249–286. [Google Scholar]
- Butler, O.T.; Cook, J.M.; Davidson, C.M.; Harrington, C.F.; Miles, D.L. Atomic spectrometry update. Environmental analysis. J. Anal. At. Spectrom. 2007, 22, 187–221. [Google Scholar]
- Charlton, B.; Fisher, A.S.; Goodall, P.S.; Hinds, M.W.; Lancaster, S.; Salisbury, M. Atomic spectrometry update. Industrial analysis: metals, chemicals and advanced materials. J. Anal. At. Spectrom. 2007, 22, 1517–1560. [Google Scholar]
- Charlton, B.; Fisher, A.S.; Goodall, P.S.; Hinds, M.W.; Lancaster, S.; Shore, S. Atomic spectrometry update. Industrial analysis: metals, chemicals and advanced materials. J. Anal. At. Spectrom. 2008, 23, 1636–1692. [Google Scholar]
- Charlton, B.; Fisher, A.S.; Goodall, P.S.; Hinds, M.W.; Lancastera, S.; Salisbury, M. Atomic spectrometry update. Industrial analysis: Metals, chemicals and advanced materials. J. Anal. At. Spectrom. 2006, 21, 1431–1471. [Google Scholar]
- Evans, E.H.; Day, J.A.; Palmer, C.D.; Price, W.J.; Smith, C.M.M.; Tyson, J.F. Atomic spectrometry update. Advances in atomic emission, absorption and fluorescence spectrometry, and related techniques. J. Anal. At. Spectrom. 2005, 20, 562–590. [Google Scholar]
- Evans, E.H.; Day, J.A.; Palmer, C.D.; Price, W.J.; Smith, C.M.M.; Tyson, J.F. Atomic spectrometry update. Advances in atomic emission, absorption, and fluorescence spectrometry, and related techniques. J. Anal. At. Spectrom. 2008, 23, 889–918. [Google Scholar]
- Evans, E.H.; Day, J.A.; Palmer, C.D.; Smith, C.M.M. Atomic spectrometry update. Advances in atomic spectrometry and related techniques. J. Anal. At. Spectrom. 2009, 24, 711–733. [Google Scholar]
- Hill, S.J.; Arowolo, T.A.; Butler, O.T.; Cook, J.M.; Cresser, M.S.; Harrington, C.; Miles, D.L. Atomic spectrometry update. Environmental analysis. J. Anal. At. Spectrom. 2003, 18, 170–202. [Google Scholar]
- Hill, S.J.; Arowolo, T.A.; Butler, O.T.; Cook, J.M.; Cresser, M.S.; Harrington, C.; Miles, D.L. Atomic spectrometry update. Environmental analysis. J. Anal. At. Spectrom. 2004, 19, 301–330. [Google Scholar]
- Günther, D.; Hattendorf, B. Solid sample analysis using laser ablation inductively coupled plasma mass spectrometry. Trac-Trends Anal. Chem. 2005, 24, 255–265. [Google Scholar]
- Sneddon, J.; Hardaway, C.; Bobbadi, K.; Reddy, A. Sample preparation of solid samples for metal determination by atomic spectroscopy - An overview and selected recent applications. Appl. Spectrosc. Rev. 2006, 41, 1–14. [Google Scholar]
- Hou, X.; Tsai, S.J.; Zhou, J.X.; Yang, K.X.; Lonardo, R.F.; Michel, R.G. Laser-Excited Atomic Fluorescence Spectrometry: Principles, Instrumentation, and Applications; Wiley-VCH: New York, NY, USA, 1987. [Google Scholar]
- Sjöström, S. Laser-excited fluorescence spectrometry in a graphite furnace electrothermal atomizer. Spectrochim. Acta Rev. 1990, 13, 407–465. [Google Scholar]
- Sjöström, S.; Mauchien, P. Laser atomic spectroscopic techniques - The analytical performance for trace-element analysis of solid and liquid samples. Spectroc. Acta Rev. 1993, 15, 153–180. [Google Scholar]
- Stchur, P.; Yang, K.X.; Hou, X.D.; Sun, T.; Michel, R.G. Laser excited atomic fluorescence spectrometry - A review. Spectroc. Acta Pt. B-Atom. Spectr. 2001, 56, 1565–1592. [Google Scholar]
- Vadillo, J.M.; Laserna, J.J. Laser-induced plasma spectrometry: truly a surface analytical tool. Spectroc. Acta Pt. B-Atom. Spectr. 2004, 59, 147–161. [Google Scholar]
- Sneddon, J.; Lee, Y.-I.; Hou, X.; Zhou, J.X.; Michel, R.G. Lasers in Analytical Atomic Spectroscopy; John Wiley & Sons-VCH: New York, NY, USA, 1997. [Google Scholar]
- Butcher, D.J.; Dougherty, J.P.; Preli, F.R.; Walton, A.P.; Wei, G.T.; Irwin, R.L.; Michel, R.G. Laser-excited atomic fluorescence spectrometry in flames, plasmas and electrothermal atomizers. J. Anal. At. Spectrom. 1988, 3, 1059–1078. [Google Scholar]
- Demtroder, W. Laser Spectroscopy: Basic Concepts and Instrumentation, 2nd ed.; Springer: Berlin, Germany, 1998. [Google Scholar]
- Butcher, D.J. Advances in laser-excited atomic fluorescence and ionization. Appl. Spectrosc. Rev. 2005, 40, 147–164. [Google Scholar]
- Butcher, D.J. Lasers as light sources for analytical atomic spectrometry. Appl. Spectrosc. Rev. 2007, 42, 543–562. [Google Scholar]
- Fang, Z. Flow Injection Atomic Absorption Spectrometry; John Wiley & Sons: New York, NY, USA, 1995. [Google Scholar]
- Haswell, S.J. (Ed.) Atomic Absorption Spectrometry: Theory, Design, and Applications; Elsevier: Amsterdam, Netherlands, 1991; Volume 5.
- Welz, B. Atomic Absorption Spectrometry, 3rd ed.; John Wiley & Sons: Chichester, UK, 1991. [Google Scholar]
- Butcher, D.J.; Sneddon, J. A Practical Guide to Graphite Furnace Atomic Absorption Spectrometry; John Wiley & Sons: New York, NY, USA, 1998. [Google Scholar]
- Jackson, K.W. (Ed.) Electrothermal Atomization for Analytical Atomic Spectrometry; John Wiley & Sons: New York, NY, USA, 1999.
- Montaser, A. (Ed.) Inductively Coupled Plasms Mass Spectrometry; John Wiley & Sons-VCH: New York, NY, USA, 1998.
- Montaser, A.; Golightly, D.W. (Eds.) Inductively Coupled Plasmas in Analytical Atomic Spectrometry, 2nd ed.; VCH: New York, NY, USA, 1992.
- Michel, A.P.M.; Chave, A.D. Analysis of laser-induced breakdown spectroscopy spectra: the case for extreme value statistics. Spectroc. Acta Pt. B-Atom. Spectr. 2007, 62, 1370–1378. [Google Scholar]
- Voigtman, E. Limits of detection and decision. Part 1. Spectroc. Acta Pt. B-Atom. Spectr. 2008, 63, 115–128. [Google Scholar]
- Voigtman, E. Limits of detection and decision. Part 2. Spectroc. Acta Pt. B-Atom. Spectr. 2008, 63, 129–141. [Google Scholar]
- Voigtman, E. Limits of detection and decision. Part 3. Spectroc. Acta Pt. B-Atom. Spectr. 2008, 63, 142–153. [Google Scholar]
- Voigtman, E. Limits of detection and decision. Part 4. Spectroc. Acta Pt. B-Atom. Spectr. 2008, 63, 154–165. [Google Scholar]
- Mermet, J.M. Limit of quantitation in atomic spectrometry: An unambiguous concept? Spectroc. Acta Pt. B-Atom. Spectr. 2008, 63, 166–182. [Google Scholar]
- Laville, S.; Sabsabi, M.; Doucet, F.R. Multi-elemental analysis of solidified mineral melt samples by laser-induced breakdown spectroscopy coupled with a linear multivariate calibration. Spectroc. Acta Pt. B-Atom. Spectr. 2007, 62, 1557–1566. [Google Scholar]
- Cremers, D.A.; Radziemski, L.J. Handbook of Laser-Induced Breakdown Spectroscopy; John Wiley & Sons: New York, NY, USA, 2006. [Google Scholar]
- Russo, R.E.; Mao, X.L.; Liu, C.; Gonzalez, J. Laser assisted plasma spectrochemistry: laser ablation. J. Anal. At. Spectrom. 2004, 19, 1084–1089. [Google Scholar]
- Lee, W.B.; Wu, J.Y.; Lee, Y.I.; Sneddon, J. Recent applications of laser-induced breakdown spectrometry: a review of material approaches. Appl. Spectrosc. Rev. 2004, 39, 27–97. [Google Scholar]
- Pasquini, C.; Cortez, J.; Silva, L.M.C.; Gonzaga, F.B. Laser induced breakdown spectroscopy. J. Braz. Chem. Soc. 2007, 18, 463–512. [Google Scholar]
- Buckley, S.G. Laser-induced breakdown spectroscopy for toxic metal emission measurements: Experimental considerations and oxygen quenching. Environ. Eng. Sci. 2005, 22, 195–204. [Google Scholar]
- Taschuk, M.T.; Godwal, Y.; Tsui, Y.Y.; Fedosejevs, R.; Tripathi, M.; Kearton, B. Absolute characterization of laser-induced breakdown spectroscopy detection systems. Spectroc. Acta Pt. B-Atom. Spectr. 2008, 63, 525–535. [Google Scholar]
- Ferrero, A.; Laserna, J.J. A theoretical study of atmospheric propagation of laser and return light for stand-off laser induced breakdown spectroscopy purposes. Spectroc. Acta Pt. B-Atom. Spectr. 2008, 63, 305–311. [Google Scholar]
- Alvarez-Trujillo, L.A.; Ferrero, A.; Laserna, J.J.; Hahn, D.W. Alternative statistical methods for spectral data processing: applications to laser-induced breakdown spectroscopy of gaseous and aerosol systems. Appl. Spectrosc. 2008, 62, 1144–1152. [Google Scholar]
- Carranza, J.E.; Hahn, D.W. Sampling statistics and considerations for single-shot analysis using laser-induced breakdown spectroscopy. Spectroc. Acta Pt. B-Atom. Spectr. 2002, 57, 779–790. [Google Scholar]
- Hettinger, B.; Hohreiter, V.; Swingle, M.; Hahn, D.W. Laser-induced breakdown spectroscopy for ambient air particulate monitoring: correlation of total and speciated aerosol particle counts. Appl. Spectrosc. 2006, 60, 237–245. [Google Scholar]
- Alvarez-Trujillo, L.A.; Ferrero, A.; Laserna, J.J. Preliminary studies on stand-off laser induced breakdown spectroscopy detection of aerosols. J. Anal. At. Spectrom. 2008, 23, 885–888. [Google Scholar]
- Mermet, J.M.; Mauchien, P.; Lacour, J.L. Processing of shot-to-shot raw data to improve precision in laser-induced breakdown spectrometry microprobe. Spectroc. Acta Pt. B-Atom. Spectr. 2008, 63, 999–1005. [Google Scholar]
- Robert, P.; Fabre, C.; Dubessy, J.; Flin, M.; Boiron, M.-C. Optimization of micro-laser induced breakdown spectroscopy analysis and signal processing. Spectroc. Acta Pt. B-Atom. Spectr. 2008, 63, 1109–1116. [Google Scholar]
- Mukherjee, D.; Cheng, M.D. Quantitative analysis of carbonaceous aerosols using laser-induced breakdown spectroscopy: a study on mass loading induced plasma matrix effects. J. Anal. At. Spectrom. 2008, 23, 119–128. [Google Scholar]
- Diwakar, P.K.; Jackson, P.B.; Hahn, D.W. The effect of multi-component aerosol particles on quantitative laser-induced breakdown spectroscopy: consideration of localized matrix effects. Spectroc. Acta Pt. B-Atom. Spectr. 2007, 62, 1466–1474. [Google Scholar]
- Fantoni, R.; Caneve, L.; Colao, F.; Fornarini, L.; Lazic, V.; Spizzichino, V. Methodologies for laboratory laser induced breakdown spectroscopy semi-quantitative and quantitative analysis-A review. Spectroc. Acta Pt. B-Atom. Spectr. 2008, 63, 1097–1108. [Google Scholar]
- Lithgow, G.A.; Buckley, S.G. Influence of particle location within plasma and focal volume on precision of single-particle laser-induced breakdown spectroscopy measurements. Spectroc. Acta Pt. B-Atom. Spectr. 2005, 60, 1060–1069. [Google Scholar]
- Lithgow, G.A.; Buckley, S.G. Effects of focal volume and spatial inhomogeneity on uncertainty in single-aerosol laser-induced breakdown spectroscopy measurements. Appl. Phys. Lett. 2005, 87, 011501:1–011501:3. [Google Scholar]
- Winefordner, J.D.; Gornushkin, I.B.; Correll, T.; Gibb, E.; Smith, B.W.; Omenetto, N. Comparing several atomic spectrometric methods to the super stars: special emphasis on laser induced breakdown spectrometry, LIBS, a future super star. J. Anal. At. Spectrom. 2004, 19, 1061–1083. [Google Scholar]
- Meissner, K.; Lippert, T.; Wokaun, A.; Guenther, D. Analysis of trace metals in comparison of laser-induced breakdown spectroscopy with LA-ICP-MS. Thin Solid Films 2004, 453, 316–322. [Google Scholar]
- Song, K.; Kim, D.; Cha, H.K.; Kim, Y.; Jung, E.C.; Choi, I.; Yoo, H.S.; Oh, S. Characterization of laser-induced plasma in a vacuum using laser ablation mass spectrometry and laser-induced breakdown spectrometry. Microchem. J. 2004, 76, 95–103. [Google Scholar]
- Latkoczy, C.; Ghislain, T. Simultaneous LIBS and LA-ICP-MS analysis of industrial samples. J. Anal. At. Spectrom. 2006, 21, 1152–1160. [Google Scholar]
- Salle, B.; Mauchien, P.; Maurice, S. Laser-induced breakdown spectroscopy in open-path configuration for the analysis of distant objects. Spectroc. Acta Pt. B-Atom. Spectr. 2007, 62, 739–768. [Google Scholar]
- Harmon, R.S.; DeLucia, F.C.; McManus, C.E.; McMillan, N.J.; Jenkins, T.F.; Walsh, M.E.; Miziolek, A. Laser-induced breakdown spectroscopy—an emerging chemical sensor technology for real-time field-portable, geochemical, mineralogical, and environmental applications. Appl. Geochem. 2006, 21, 730–747. [Google Scholar]
- Pershin, S.M.; Colao, F.; Spizzichino, V. Quantitative analysis of bronze samples by laser-induced breakdown spectroscopy (LIBS): a new approach, model, and experiment. Laser Phys. 2006, 16, 455–467. [Google Scholar]
- Dreyer, C.B.; Mungas, G.S.; Thanh, P.; Radziszewski, J.G. Study of sub-mJ-excited laser-induced plasma combined with Raman spectroscopy under Mars atmosphere-simulated conditions. Spectroc. Acta Pt. B-Atom. Spectr. 2007, 62, 1448–1459. [Google Scholar]
- Giakoumaki, A.; Osticioli, I.; Anglos, D. Spectroscopic analysis using a hybrid LIBS-Raman system. Appl. Phys. A-Mater. Sci. Process. 2006, 83, 537–541. [Google Scholar]
- Wiens, R.C.; Sharma, S.K.; Thompson, J.; Misra, A.; Lucey, P.G. Joint analyses by laser-induced breakdown spectroscopy (LIBS) and Raman spectroscopy at stand-off distances. Spectroc. Acta Pt. A-Molec. Biomolec. Spectr. 2005, 61, 2324–2334. [Google Scholar]
- Lazic, V.; Colao, F.; Fantoni, R.; Spizzichino, V.; Jovicevic, S. Underwater sediment analyses by laser induced breakdown spectroscopy and calibration procedure for fluctuating plasma parameters. Spectroc. Acta Pt. B-Atom. Spectr. 2007, 62, 30–39. [Google Scholar]
- Lazic, V.; Colao, F.; Fantoni, R.; Spizzicchino, V. Recognition of archeological materials underwater by laser induced breakdown spectroscopy. Spectroc. Acta Pt. B-Atom. Spectr. 2005, 60, 1014–1024. [Google Scholar]
- Lazic, V.; Colao, F.; Fantoni, R.; Spizzicchino, V. Laser-induced breakdown spectroscopy in water: Improvement of the detection threshold by signal processing. Spectroc. Acta Pt. B-Atom. Spectr. 2005, 60, 1002–1013. [Google Scholar]
- Cheam, V.; Lechner, J.; Desrosiers, R. Laser-induced fluorescence determination of thallium in sediments. Fresenius J. Anal. Chem. 1998, 360, 81–85. [Google Scholar]
- Barbini, R.; Colao, F.; Lazic, V.; Fantoni, R.; Palucci, A.; Angelone, M. On board LIBS analysis of marine sediments collected during the XVI Italian campaign in Antarctica. Spectroc. Acta Pt. B-Atom. Spectr. 2002, 57, 1203–1218. [Google Scholar]
- Kaski, S.; Hakkanen, H.; Korppi-Tommola, J. Sulfide mineral identification using laser-induced plasma spectroscopy. Miner. Eng. 2003, 16, 1239–1243. [Google Scholar]
- Kaski, S.; Hakkanen, H.; Korppi-Tommola, J. Determination of Cl/C and Br/C ratios in pure organic solids using laser-induced plasma spectroscopy in near vacuum ultraviolet. J. Anal. At. Spectrom. 2004, 19, 474–478. [Google Scholar]
- Solo-Gabriele, H.M.; Townsend, T.G.; Hahn, D.W.; Moskal, T.M.; Hosein, N.; Jambeck, J.; Jacobi, G. Evaluation of XRF and LIBS technologies for on-line sorting of CCA-treated wood waste. Waste Manage. 2004, 24, 413–424. [Google Scholar]
- Martin, M.Z.; Labbe, N.; Rials, T.G.; Wullschleger, S.D. Analysis of preservative-treated wood by multivariate analysis of laser-induced breakdown spectroscopy spectra. Spectroc. Acta Pt. B-Atom. Spectr. 2005, 60, 1179–1185. [Google Scholar]
- Gondal, M.A.; Hussain, T.; Ahmed, Z.; Bakry, A.H. Detection of contaminants in ore samples using laser-induced breakdown spectroscopy. J. Environ. Sci. Health Part A-Toxic/Hazard. Subst. Environ. Eng. 2007, 42, 879–887. [Google Scholar]
- Gondal, M.A.; Hussain, T.; Yamani, Z.H.; Ahmed, Z. Determination of toxic metals in petroleum, cultivated land and ore samples using laser-induced breakdown spectroscopy. Bull. Environ. Contam. Toxicol. 2007, 78, 270–274. [Google Scholar]
- Asimellis, G.; Giannoudakos, A.; Kompitsas, M. Phosphate ore beneficiation via determination of phosphorus-to-silica ratios by laser induced breakdown spectroscopy. Spectroc. Acta Pt. B-Atom. Spectr. 2006, 61, 1253–1259. [Google Scholar]
- Death, D.L.; Cunningham, A.P.; Pollard, L.J. Multi-element analysis of iron ore pellets by laser-induced breakdown spectroscopy and principal components regression. Spectroc. Acta Pt. B-Atom. Spectr. 2008, 63, 763–769. [Google Scholar]
- McMillan, N.J.; McManus, C.E.; Harmon, R.S.; De Lucia, F.C.; Miziolek, A.W. Laser-induced breakdown spectroscopy analysis of complex silicate minerals-beryl. Anal. Bioanal. Chem. 2006, 385, 263–271. [Google Scholar]
- Gondal, M.A.; Hussain, T.; Yamani, Z.H.; Bakry, A.H. Study of hazardous metals in iron slag waste using laser induced breakdown spectroscopy. J. Environ. Sci. Health Part A-Toxic/Hazard. Subst. Environ. Eng. 2007, 42, 767–775. [Google Scholar]
- Gehl, R.J.; Rice, C.W. Emerging technologies for in situ measurement of soil carbon. Clim. Change 2007, 80, 43–54. [Google Scholar]
- Martin, M.Z.; Wullschleger, S.D.; Garten, C.T.; Palumbo, A.V. Laser-induced breakdown spectroscopy for the environmental determination of total carbon and nitrogen in soils. Appl. Optics 2003, 42, 2072–2077. [Google Scholar]
- Hussain, T.; Gondal, M.A.; Yamani, Z.H.; Baig, M.A. Measurement of nutrients in green house soil with laser induced breakdown spectroscopy. Environ. Monit. Assess. 2007, 124, 131–139. [Google Scholar]
- Bousquet, B.; Sirven, J.B.; Canioni, L. Towards quantitative laser-induced breakdown spectroscopy analysis of soil samples. Spectroc. Acta Pt. B-Atom. Spectr. 2007, 62, 1582–1589. [Google Scholar]
- Hussain, T.; Gondal, M.A. Monitoring and assessment of toxic metals in Gulf War oil spill contaminated soil using laser-induced breakdown spectroscopy. Environ. Monit. Assess. 2008, 136, 391–399. [Google Scholar]
- Lopez-Moreno, C.; Palanco, S.; Laserna, J.J. Remote laser-induced plasma spectrometry for elemental analysis of samples of environmental interest. J. Anal. At. Spectrom. 2004, 19, 1479–1484. [Google Scholar]
- Caneve, L.; Colao, F.; Fabbri, F.; Fantoni, R.; Spizzichino, V.; Striber, J. Laser-induced breakdown spectroscopy analysis of asbestos. Spectroc. Acta Pt. B-Atom. Spectr. 2005, 60, 1115–1120. [Google Scholar]
- Cheam, V.; Reynoldson, T.; Garbai, G.; Rajkumar, J.; Milani, D. Local impacts of coal mines and power plants across Canada. II. Metals, organics and toxicity in sediments. Water Qual. Res. J. Canada 2000, 35, 609–631. [Google Scholar]
- Cheam, V.; Garbai, G.; Lechner, J.; Rajkumar, J. Local impacts of coal mines and power plants across Canada. I. Thallium in waters and sediments. Water Qual. Res. J. Canada 2000, 35, 581–607. [Google Scholar]
- Cheam, V. Thallium contamination of water in Canada. Water Qual. Res. J. Canada 2001, 36, 851–878. [Google Scholar]
- Borgmann, U.; Cheam, V.; Norwood, W.P.; Lechner, J. Toxicity and bioaccumulation of thallium in Hyalella azteca, with comparison to other metals and prediction of environmental impact. Environ. Pollut. 1998, 99, 105–114. [Google Scholar]
- Simeonsson, J.B.; Elwood, S.A.; Ezer, M.; Pacquette, H.L.; Swart, D.J.; Beach, H.D.; Thomas, D.J. Development of ultratrace laser spectrometry techniques for measurements of arsenic. Talanta 2002, 58, 189–199. [Google Scholar]
- Hou, X.D.; Levine, K.E.; Salido, A.; Jones, B.T.; Ezer, M.; Elwood, S.; Simeonsson, J.B. Tungsten coil devices in atomic spectrometry: Absorption, fluorescence, and emission. Anal. Sci. 2001, 17, 175–180. [Google Scholar]
- Simeonsson, J.B.; Ezer, M.; Pacquette, H.L.; Preston, S.L.; Swart, D.J. Laser-induced fluorescence of As, Se and Sb in the inductively coupled plasma. Spectroc. Acta Pt. B-Atom. Spectr. 1997, 52, 1955–1963. [Google Scholar]
- Swart, D.J.; Ezer, M.; Pacquette, H.L.; Simeonsson, J.B. Laser-induced fluorescence of Se, As, and Sb in an electrothermal atomizer. Anal. Chem. 1998, 70, 1324–1330. [Google Scholar]
- Pacquette, H.L.; Elwood, S.A.; Ezer, M.; Simeonsson, J.B. A comparison of continuous flow hydride generation laser-induced fluorescence and laser-enhanced ionization spectrometry approaches for parts per trillion level measurements of arsenic, selenium and antimony. J. Anal. At. Spectrom. 2001, 16, 152–158. [Google Scholar]
- Ezer, M.; Elwood, S.A.; Jones, B.T.; Simeonsson, J.B. Evaluation of a tungsten coil atomization-laser-induced fluorescence detection approach for trace elemental analysis. Anal. Chim. Acta 2006, 571, 136–141. [Google Scholar]
- Heitmann, U.; Sy, T.; Hese, A.; Schoknecht, G. High-sensitivity detection of selenium and arsenic by laser-excited atomic fluorescence spectrometry using electrothermal atomization. J. Anal. At. Spectrom. 1994, 9, 437–442. [Google Scholar]
- Cornelis, R.; Sabbioni, E.; Vandervenne, M.T. Trace-element reference values in tissues from inhabitants of the European community. 7. Review of trace-elements in blood, serum, and urine of the Belgian population and critical evaluation of their possible use as reference values. Sci. Total Environ. 1994, 158, 191–226. [Google Scholar]
- Swart, D.J.; Simeonsson, J.B. Direct determination of selenium in serum by electrothermal atomization laser-induced fluorescence spectrometry. J. Anal. At. Spectrom. 1999, 14, 929–932. [Google Scholar]
- Versieck, J. Trace-elements in human-body fluids and tissues. CRC Crit. Rev. Clin. Lab. Sci. 1985, 22, 97–184. [Google Scholar]
- Francesconi, K.A.; Kuehnelt, D. Determination of arsenic species: A critical review of methods and applications, 2000-2003. Analyst 2004, 129, 373–395. [Google Scholar]
- Hung, D.Q.; Nekrassova, O.; Compton, R.G. Analytical methods for inorganic arsenic in water: a review. Talanta 2004, 64, 269–277. [Google Scholar]
- Leermakers, M.; Baeyens, W.; De Gieter, M.; Smedts, B.; Meert, C.; de Bisschop, H.C.; Morabito, R.; Quevauviller, P. Toxic arsenic compounds in environmental samples: Speciation and validation. Trac-Trends Anal. Chem. 2006, 25, 1–10. [Google Scholar]
- Terlecka, E. Arsenic speciation analysis in water samples: A review of the hyphenated techniques. Environ. Monit. Assess. 2005, 107, 259–284. [Google Scholar]
- Fortes, F.J.; Cunat, J.; Cabalin, L.M.; Laserna, J.J. In situ analytical assessment and chemical imaging of historical buildings using a man-portable laser system. Appl. Spectrosc. 2007, 61, 558–564. [Google Scholar]
- Nicolas, G.; Mateo, M.P.; Pinon, V. 3D chemical maps of non-flat surfaces by laser-induced breakdown spectroscopy. J. Anal. At. Spectrom. 2007, 22, 1244–1249. [Google Scholar]
- Lopez-Moreno, C.; Palanco, S.; Laserna, J.J. Stand-off analysis of moving targets using laser-induced breakdown spectroscopy. J. Anal. At. Spectrom. 2007, 22, 84–87. [Google Scholar]
- Anzano, J.M.; Villoria, M.A.; Ruiz-Medina, A.; Lasheras, R.J. Laser-induced breakdown spectroscopy for quantitative spectrochemical analysis of geological materials: Effects of the matrix and simultaneous determination. Anal. Chim. Acta 2006, 575, 230–235. [Google Scholar]
- Baudelet, M.; Boueri, M.; Yu, J.; Mao, S.S.; Piseltelli, V.; Mao, X.L.; Russo, R.E. Time-resolved ultraviolet laser-induced breakdown spectroscopy for organic material analysis. Spectroc. Acta Pt. B-Atom. Spectr. 2007, 62, 1329–1334. [Google Scholar]
- Galiová, M.; Kaiser, J.; Novotný, K.; Samek, O.; Reale, L.; Malina, R.; Páleníková, K.; Liska, M.; Cudek, V.; Kanický, V.; Otruba, V.; Poma, A.; Tucci, A. Utilization of laser induced breakdown spectroscopy for investigation of the metal accumulation in vegetal tissues. Spectroc. Acta Pt. B-Atom. Spectr. 2007, 62, 1597–1605. [Google Scholar]
- Bossu, M.; Hao, Z.Q.; Baudelet, M.; Yu, J.; Zhang, Z.; Zhang, J. Femtosecond laser-induced breakdown spectroscopy for detection of trace elements in Sophora leaves. Chin. Phys. Lett. 2007, 24, 3466–3468. [Google Scholar]
- Michel, A.P.M.; Lawrence-Snyder, M.; Angel, S.M.; Chave, A.D. Laser-induced breakdown spectroscopy of bulk aqueous solutions at oceanic pressures: Evaluation of key measurement parameters. Appl. Optics 2007, 46, 2507–2515. [Google Scholar]
- Lawrence-Snyder, M.; Scaffidi, J.; Angel, S.M.; Michel, A.P.M.; Chave, A.D. Sequential-pulse laser-induced breakdown spectroscopy of high-pressure bulk aqueous solutions. Appl. Spectrosc. 2007, 61, 171–176. [Google Scholar]
- Huang, J.S.; Ke, C.B.; Lin, K.C. Matrix effect on emission/current correlated analysis in laser-induced breakdown spectroscopy of liquid droplets. Spectroc. Acta Pt. B-Atom. Spectr. 2004, 59, 321–326. [Google Scholar]
- Lui, S.L.; Godwal, Y.; Taschuk, M.T.; Tsui, Y.Y.; Fedosejevs, R. Detection of lead in water using laser-induced breakdown spectroscopy and laser-induced fluorescence. Anal. Chem. 2008, 80, 1995–2000. [Google Scholar]
- Godwal, Y.; Lui, S.L.; Taschuk, M.T.; Tsui, Y.; Fedosejevs, R. Determination of lead in water using laser ablation-laser induced fluorescence. Spectroc. Acta Pt. B-Atom. Spectr. 2007, 62, 1443–1447. [Google Scholar]
- Dockery, C.R.; Pender, J.E.; Goode, S.R. Speciation of chromium via laser-induced breakdown spectroscopy of ion exchange polymer membranes. Appl. Spectrosc. 2005, 59, 252–257. [Google Scholar]
- Le Bihan, A.; Lijour, Y.; Giamarchi, P.; Burel-Deschamps, L.; Stephan, L. Direct determination of aluminum content in seawater by electrothermal atomization-laser excited atomic fluorescence. Spectroc. Acta Pt. B-Atom. Spectr. 2003, 58, 15–26. [Google Scholar]
- Le Bihan, A.; Lijour, Y.; Giamarchi, P.; Burel-Deschamps, L.; Stephan, L. Laser-induced fluorescence with an OPO system. Part II: Direct determination of lead content in seawater by electrothermal atomization-laser-excited atomic fluorescence (ETA-LEAF). Anal. Bioanal. Chem. 2003, 375, 815–819. [Google Scholar]
- Koch, S.; Garen, W.; Muller, M.; Neu, W. Detection of chromium in liquids by laser induced breakdown spectroscopy (LIBS). Appl. Phys. A-Mater. Sci. Process. 2004, 79, 1071–1073. [Google Scholar]
- Mohamed, W.T.Y. Calibration free laser-induced breakdown spectroscopy (LIBS) identification of seawater salinity. Opt. Appl. 2007, 37, 5–19. [Google Scholar]
- Gondal, M.A.; Hussain, T. Determination of poisonous metals in wastewater collected from paint manufacturing plant using laser-induced breakdown spectroscopy. Talanta 2007, 71, 73–80. [Google Scholar]
- Gondal, M.A.; Hussain, T.; Yamani, Z.H.; Baig, M.A. Detection of heavy metals in Arabian crude oil residue using laser induced breakdown spectroscopy. Talanta 2006, 69, 1072–1078. [Google Scholar]
- Chen, Z.J.; Li, H.K.; Liu, M.; Li, R.H. Fast and sensitive trace metal analysis in aqueous solutions by laser-induced breakdown spectroscopy using wood slice substrates. Spectroc. Acta Pt. B-Atom. Spectr. 2008, 63, 64–68. [Google Scholar]
- Idris, N.; Kagawa, K.; Sakan, F.; Tsuyuki, K.; Miura, S. Analysis of heavy metal pollution in soil using transversely excited atmospheric CO2 laser-induced plasma by trapping the soil in microstructured holes on metal subtargets. Appl. Spectrosc. 2007, 61, 1344–1351. [Google Scholar]
- Beddows, D.C.S.; Telle, H.H. Prospects of real-time single-particle biological aerosol analysis: a comparison between laser-induced breakdown spectroscopy and aerosol time-of-flight mass spectrometry. Spectroc. Acta Pt. B-Atom. Spectr. 2005, 60, 1040–1059. [Google Scholar]
- Bauer, D.; Campuzano-Jost, P.; Hynes, A.J. Rapid, ultra-sensitive detection of gas phase elemental mercury under atmospheric conditions using sequential two-photon laser induced fluorescence. J. Environ. Monit. 2002, 4, 339–343. [Google Scholar]
- Lithgow, G.A.; Robinson, A.L.; Buckley, S.G. Ambient measurements of metal-containing PM2.5 in an urban environment using laser-induced breakdown spectroscopy. Atmos. Environ. 2004, 38, 3319–3328. [Google Scholar]
- Kuhlen, T.; Frickee-Begemann, C.; Strauss, N.; Noll, R. Analysis of size-classified fine and ultrafine particulate matter on substrates with laser-induced breakdown spectroscopy. Spectroc. Acta Pt. B-Atom. Spectr. 2008, 63, 1171–1176. [Google Scholar]
- Potgieter-Vermaak, S.S.; van Grieken, R. Preliminary evaluation of micro-Raman spectrometry for the characterization of individual aerosol particles. Appl. Spectrosc. 2006, 60, 39–47. [Google Scholar]
- Godoi, R.H.M.; Potgieter-Vermaak, S.; De Hoog, J.; Kaegi, R.; Grieken, R. Substrate selection for optimum qualitative and quantitative single atmospheric particles analysis using nano-manipulation, sequential thin-window electron probe X-ray microanalysis and micro-Raman spectrometry. Spectroc. Acta Pt. B-Atom. Spectr. 2006, 61, 375–388. [Google Scholar]
- Heard, D.E. Atmospheric field measurements of the hydroxyl radical using laser-induced fluorescence spectroscopy. Annu. Rev. Phys. Chem. 2006, 57, 191–216. [Google Scholar]
- Heard, D.E.; Pilling, M.J. Measurement of OH and HO2 in the troposphere. Chem. Rev. 2003, 103, 5163–5198. [Google Scholar]
- Baardsen, E.L.; Terhune, R.W. Detection of OH in atmosphere using a dye laser. Appl. Phys. Lett. 1972, 21, 209–211. [Google Scholar]
- Schlosser, E.; Bohn, B.; Brauers, T.; Dorn, H.P.; Fuchs, H.; Haseler, R.; Hofzumahaus, A.; Holland, F.; Rohrer, F.; Rupp, L.O.; Siese, M.; Tillmann, R.; Wahner, A. Intercomparison of two hydroxyl radical measurement techniques at the atmosphere simulation chamber SAPHIR. J. Atmos. Chem. 2007, 56, 187–205. [Google Scholar]
- Dusanter, S.; Vimal, D.; Stevens, P.S. Technical note: Measuring tropospheric OH and HO2 by laser-induced fluorescence at low pressure. A comparison of calibration techniques. Atmos. Chem. Phys. 2008, 8, 321–340. [Google Scholar]
- Bloss, W.J.; Lee, J.D.; Bloss, C.; Heard, D.E.; Pilling, M.J.; Wirtz, K.; Martin-Reviejo, M.; Siese, M. Validation of the calibration of a laser-induced fluorescence instrument for the measurement of OH radicals in the atmosphere. Atmos. Chem. Phys. 2004, 4, 571–583. [Google Scholar]
- Kwon, B.G. In-situ calibration of the hydroperoxyl radical using an immobilized TiO2 photocatalyst in the atmosphere. Bull. Korean Chem. Soc. 2008, 29, 785–789. [Google Scholar]
- Cantrell, C.A.; Shetter, R.E.; McDaniel, A.H.; Calvert, J.G. Measurement Challenges in Atmospheric Chemistry; American Chemical Society: Washington, DC, USA, 1993. [Google Scholar]
- Lightfoot, P.D.; Cox, R.A.; Crowley, J.N.; Destriau, M.; Hayman, G.D.; Jenkin, M.E.; Moortgat, G.K.; Zabel, F. Organic peroxy radicals: Kinetics, spectroscopy and tropospheric chemistry. Atmos. Environ. A-Gen. Topics 1992, 26, 1805–1961. [Google Scholar]
- Fuchs, H.; Holland, F.; Hofzumahaus, A. Measurement of tropospheric RO2 and HO2 radicals by a laser-induced fluorescence instrument. Rev. Sci. Instrum. 2008, 79, 084104:1–084104:12. [Google Scholar]
- Bloss, W.J.; Lee, J.D.; Heard, D.E.; Salmon, R.A.; Bauguitte, S.J.B.; Roscoe, H.K.; Jones, A.E. Observations of OH and HO2 radicals in coastal Antarctica. Atmos. Chem. Phys. 2007, 7, 4171–4185. [Google Scholar]
- Dusanter, S.; Vimal, D.; Stevens, P.S.; Volkamer, R.; Molina, L.T. Measurements of OH and HO2 concentrations during the MCMA-2006 field campaign - Part 1: Deployment of the Indiana University laser-induced fluorescence instrument. Atmos. Chem. Phys. 2009, 9, 1665–1685. [Google Scholar]
- Wood, E.C.; Herndon, S.C.; Onasch, T.B.; Kroll, J.H.; Canagaratna, M.R.; Kolb, C.E.; Worsnop, D.R.; Neuman, J.A.; Seila, R.; Zavala, M.; Knighton, W.B. A case study of ozone production, nitrogen oxides, and the radical budget in Mexico City. Atmos. Chem. Phys. 2009, 9, 2499–2516. [Google Scholar]
- Yoshino, A.; Sadanaga, Y.; Watanabe, K.; Kato, S.; Miyakawa, Y.; Matsumoto, J.; Kajii, Y. Measurement of total OH reactivity by laser-induced pump and probe technique—comprehensive observations in the urban atmosphere of Tokyo. Atmos. Environ. 2006, 40, 7869–7881. [Google Scholar]
- O'Keefe, A.; Deacon, D.A.G. Cavity ring-down optical spectrometer for absorption measurements using pulsed laser sources. Rev. Sci. Inst. 1988, 59, 2544–2551. [Google Scholar]
- Berden, G.; Peeters, R.; Meijer, G. Cavity ring-down spectroscopy: experimental schemes and applications. Int. Rev. Phys. Chem. 2000, 19, 565–607. [Google Scholar]
- Scherer, J.J.; Paul, J.B.; O'Keefe, A.; Saykally, R.J. Cavity ringdown laser absorption spectroscopy: history, development, and application to pulsed molecular beams. Chem. Rev. 1997, 97, 25–52. [Google Scholar]
- Looney, J.P.; Hodges, J.T.; van Zee, R.D. Quantitative absorption measurements using cavity-ringdown spectroscopy with pulsed lasers. ACS Symposium Series 1999, 720, 93–105. [Google Scholar]
- Paldus, B.A.; Kachanov, A.A. An historical overview of cavity-enhanced methods. Can. J. Phys. 2005, 83, 975–999. [Google Scholar]
- Brown, S.S. Absorption spectroscopy in high-finesse cavities for atmospheric studies. Chem. Rev. 2003, 103, 5219–5238. [Google Scholar]
- Atkinson, D.B. Solving chemical problems of environmental importance using cavity ring-down spectroscopy. Analyst 2003, 128, 117–125. [Google Scholar]
- Ball, S.M.; Jones, R.L. Broad-band cavity ring-down spectroscopy. Chem. Rev. 2003, 103, 5239–5262. [Google Scholar]
- Mazurenka, M.; Orr-Ewing, A.J.; Peverall, R.; Ritchie, G.A.D. 4 Cavity ring-down and cavity enhanced spectroscopy using diode lasers. Annu. Rep. Prog. Chem. Sect. C: Phys. Chem. 2005, 101, 100–142. [Google Scholar]
- Scherer, J.J.; Paul, J.B.; Jiao, H.; O'Keefe, A. Broadband ringdown spectral photography. Appl. Opt. 2001, 40, 6725–6732. [Google Scholar]
- Engeln, R.; von Helden, G.; Berden, G.; Meijer, G. Phase shift cavity ring down absorption spectroscopy. Chem. Phys. Lett. 1996, 262, 105–109. [Google Scholar]
- Czyzewski, A.; Chudzynski, S.; Ernst, K.; Karasinski, G.; Kilianek, L.S.; Pietruczuk, A.; Skubiszak, W.; Stacewicz, T.; Stelmaszczyk, K.; Koch, B.; Rairoux, P. Cavity ring-down spectrography. Opt. Commun. 2001, 191, 271–275. [Google Scholar]
- He, Y.; Orr, B.J. Rapid measurement of cavity ringdown absorption spectra with a swept-frequency laser. Appl. Phys. B 2004, 79, 941–945. [Google Scholar]
- Romanini, D.; Kachanov, A.A.; Sadeghi, N.; Stoeckel, F. CW cavity ring down spectroscopy. Chem. Phys. Lett. 1997, 264, 316–322. [Google Scholar]
- He, Y.; Orr, B.J. Rapidly swept, continuous-wave cavity ringdown spectroscopy with optical heterodyne detection: Single- and multi-wavelength sensing of gases. Appl. Phys. B 2002, 75, 267–280. [Google Scholar]
- He, Y.; Orr, Brian J. Continuous-wave cavity ringdown absorption spectroscopy with a swept-frequency laser: Rapid spectral sensing of gas-phase molecules. Appl. Opt. 2005, 44, 6752–6761. [Google Scholar]
- Engeln, R.; Berden, G.; Peeters, R.; Meijer, G. Cavity enhanced absorption and cavity enhanced magnetic rotation spectroscopy. Rev. Sci. Inst. 1998, 69, 3763–3769. [Google Scholar]
- O'Keefe, A. Integrated cavity output analysis of ultra-weak absorption. Chem. Phys. Lett. 1998, 293, 331–336. [Google Scholar]
- Paul, J.B.; Lapson, L.; Anderson, J.G. Ultrasensitive absorption spectroscopy with a high-finesse optical cavity and off-axis alignment. Appl. Opt. 2001, 40, 4904–4910. [Google Scholar]
- Hamers, E.; Schram, D.; Engeln, R. Fourier transform phase shift cavity ring down spectroscopy. Chem. Phys. Lett. 2002, 365, 237–243. [Google Scholar]
- Lewis, E.K.; Moehnke, C.J.; Manzanares, C.E. Phase shift cavity ring down and FT-VIS measurements of C-H (Δv = 5) vibrational overtone absorptions. Chem. Phys. Lett. 2004, 394, 25–31. [Google Scholar]
- van der Sneppen, L.; Ariese, F.; Gooijer, C.; Ubachs, W. Liquid-phase and evanescent-wave cavity ring-down spectroscopy in analytical chemistry. Ann. Rev. Anal. Chem. 2009, 2, 13–35. [Google Scholar]
- Ball, S.M.; Langridge, J.M.; Jones, R.L. Broadband cavity enhanced absorption spectroscopy using light emitting diodes. Chem. Phys. Lett. 2004, 398, 68–74. [Google Scholar]
- Langridge, J.M.; Ball, S.M.; Shillings, A.J.L.; Jones, R.L. A broadband absorption spectrometer using light emitting diodes for ultrasensitive, in situ trace gas detection. Rev. Sci. Inst. 2008, 79, 123110–123114. [Google Scholar]
- Triki, M.; Cermak, P.; Méjean, G.; Romanini, D. Cavity-enhanced absorption spectroscopy with a red LED source for NOx trace analysis. Appl. Phys. B 2008, 91, 195–201. [Google Scholar]
- Wu, T.; Zhao, W.; Chen, W.; Zhang, W.; Gao, X. Incoherent broadband cavity enhanced absorption spectroscopy for in situ measurements of NO2 with a blue light emitting diode. Appl. Phys. B 2009, 94, 85–94. [Google Scholar]
- Gherman, T.; Venables, D.S.; Vaughan, S.; Orphal, J.; Ruth, A.A. Incoherent broadband cavity-enhanced absorption spectroscopy in the near-ultraviolet: application to HONO and NO2. Environ. Sci. Technol. 2007, 42, 890–895. [Google Scholar]
- Johnston, P.S.; Lehmann, K.K. Cavity enhanced absorption spectroscopy using a broadband prism cavity and a supercontinuum source. Opt. Express 2008, 16, 15013–15023. [Google Scholar]
- Langridge, J.M.; Laurila, T.; Watt, R.S.; Jones, R.L.; Kaminski, C.F.; Hult, J. Cavity enhanced absorption spectroscopy of multiple trace gas species using a supercontinuum radiation source. Opt. Express 2008, 16, 10178–10188. [Google Scholar]
- Stelmaszczyk, K.; Rohwetter, P.; Fechner, M.; Queißer, M.; Czyzewski, A.; Stacewicz, T.; Wöste, L. Cavity ring-down absorption spectrography based on filament-generated supercontinuum light. Opt. Express 2009, 17, 3673–3678. [Google Scholar]
- Stelmaszczyk, K.; Fechner, M.; Rohwetter, P.; Queißer, M.; Czyżewski, A.; Stacewicz, T.; Wöste, L. Towards supercontinuum cavity ring-down spectroscopy. Appl. Phys. B 2009, 94, 369–373. [Google Scholar]
- Crunaire, S.; Tarmoul, J.; Fittschen, C.; Tomas, A.; Lemoine, B.; Coddeville, P. Use of cw-CRDS for studying the atmospheric oxidation of acetic acid in a simulation chamber. Appl. Phys. B 2006, 85, 467–476. [Google Scholar]
- Wheeler, M.D.; Newman, S.M.; Orr-Ewing, A.J.; Ashfold, M.N.R. Cavity ring-down spectroscopy. J. Chem. Soc. Faraday Trans. 1998, 94, 337–351. [Google Scholar]
- Thiébaud, J.; Fittschen, C. Near infrared cw-CRDS coupled to laser photolysis: spectroscopy and kinetics of the HO2 radical. Appl. Phys. B 2006, 85, 383–389. [Google Scholar]
- Brown, S.S.; Wilson, R.W.; Ravishankara, A.R. Absolute intensities for third and fourth overtone absorptions in HNO3 and H2O2 measured by cavity ring down spectroscopy. J. Phys. Chem. A 2000, 104, 4976–4983. [Google Scholar]
- Feierabend, K.J.; Havey, D.K.; Brown, S.S.; Vaida, V. Experimental absolute intensities of the 4v9 and 5v9 O-H stretching overtones of H2SO4. Chem. Phys. Lett. 2006, 420, 438–442. [Google Scholar]
- Havey, D.K.; Feierabend, K.J.; Takahashi, K.; Skodje, R.T.; Vaida, V. Experimental and theoretical investigation of vibrational overtones of glycolic acid and its hydrogen bonding interactions with water. J. Phys. Chem. A 2006, 110, 6439–6446. [Google Scholar]
- Lane, J.R.; Kjaergaard, H.G.; Plath, K.L.; Vaida, V. Overtone spectroscopy of sulfonic acid derivatives. J. Phys. Chem. A 2007, 111, 5434–5440. [Google Scholar]
- Tiger Optics. Available online: http://www.tigeroptics.com (accessed on August 22, 2009).
- Los Gatos Research. Available online: http://www.lgrinc.com/ (accessed on August 22, 2009).
- Picarro. Available online: http://www.picarro.com/ (accessed on August 22, 2009).
- Dupre, P.; Gherman, T.; Zobov, N.F.; Tolchenov, R.N.; Tennyson, J. Continuous-wave cavity ringdown spectroscopy of the 8v polyad of water in the 25,195– 25,340 cm–1 range. J. Chem. Phys. 2005, 123, 154307–154311. [Google Scholar]
- Lisak, D.; Hodges, J.T. High-resolution cavity ring-down spectroscopy measurements of blended H2O transitions. Appl. Phys. B 2007, 88, 317–325. [Google Scholar]
- Crosson, E.R. A cavity ring-down analyzer for measuring atmospheric levels of methane, carbon dioxide, and water vapor. Appl. Phys. B 2008, 92, 403–408. [Google Scholar]
- Kassi, S.; Chenevier, M.; Gianfrani, L.; Salhi, A.; Rouillard, Y.; Ouvrard, A.; Romanini, D. Looking into the volcano with a Mid-IR DFB diode laser and cavity enhanced absorption spectroscopy. Opt. Express 2006, 14, 11442–11452. [Google Scholar]
- Malara, P.; Maddaloni, P.; Gagliardi, G.; De Natale, P. Combining a difference-frequency source with an off-axis high-finesse cavity for trace-gas monitoring around 3 μm. Opt. Express 2006, 14, 1304–1313. [Google Scholar]
- Orphal, J.; Ruth, A.A. High-resolution Fourier-transform cavity-enhanced absorption spectroscopy in the near-infrared using an incoherent broad-band light source. Opt. Express 2008, 16, 19232–19243. [Google Scholar]
- Welzel, S.; Lombardi, G.; Davies, P.B.; Engeln, R.; Schram, D.C.; Ropcke, J. Trace gas measurements using optically resonant cavities and quantum cascade lasers operating at room temperature. J. Appl. Phys. 2008, 104, 093115–093115. [Google Scholar]
- Fallows, E.A.; Cleary, T.G.; Miller, J.H. Development of a multiple gas analyzer using cavity ringdown spectroscopy for use in advanced fire detection. Appl. Opt. 2009, 48, 695–703. [Google Scholar]
- Pradhan, M.; Aziz, M.S.I.; Grilli, R.; Orr-Ewing, A.J. Automated system for monitoring trace C2H2 in ambient air by cavity ring-down spectroscopy combined with sample preconcentration. Environ. Sci. Technol. 2008, 42, 7354–7359. [Google Scholar]
- Pradhan, M.; Lindley, R.E.; Grilli, R.; White, I.R.; Martin, D.; Orr-Ewing, A.J. Trace detection of C2H2 in ambient air using continuous wave cavity ring-down spectroscopy combined with sample pre-concentration. Appl. Phys. B 2008, 90, 1–9. [Google Scholar]
- Crow, M.B.; Gilchrist, A.; Hancock, G.; Peverall, R.; Richmond, G.; Ritchie, G.A.D.; Taylor, S.R. High-resolution absorption studies of the Á1A2-X1A1 202401 band of formaldehyde. J. Phys. Chem. A 2009, 113, 6689–6696. [Google Scholar]
- Schuster, G.; Labazan, I.; Crowley, J.N. A cavity ring down/cavity enhanced absorption device for measurement of ambient NO3 and N2O5. Atmos. Meas. Tech. 2009, 2, 1–13. [Google Scholar]
- Sato, J.; Kikukawa, M.; Yamaguchi, S.; Endo, M.; Tei, K.; Nanri, K.; Fujioka, T. Flexible laser diode trace gas sensor based on cavity ringdown spectroscopy with an optical fiber-coupled high-finesse external-cavity diode laser. Appl. Phys. B 2009, 96, 741–744. [Google Scholar]
- Venables, D.S.; Gherman, T.; Orphal, J.; Wenger, J.C.; Ruth, A.A. High sensitivity in situ monitoring of NO3 in an atmospheric simulation chamber using incoherent broadband cavity-enhanced absorption spectroscopy. Environ. Sci. Technol. 2006, 40, 6758–6763. [Google Scholar]
- Varma, R.M.; Venables, D.S.; Ruth, A.A.; Heitmann, U.; Schlosser, E.; Dixneuf, S. Long optical cavities for open-path monitoring of atmospheric trace gases and aerosol extinction. Appl. Opt. 2009, 48, B159–B171. [Google Scholar]
- Liu, Z.-W.; Xu, Y.; Yang, X.-F.; Zhu, A.-M.; Zhao, G.-L.; Wang, W.-G. Determination of the HO2 radical in dielectric barrier discharge plasmas using near-infrared cavity ring-down spectroscopy. J. Phys. D: Appl. Phys. 2008, 41, 045203:1–045203:7. [Google Scholar]
- Srivastava, N.; Wang, C.; Dibble, T.S. A study of OH radicals in an atmospheric AC discharge plasma using near infrared diode laser cavity ringdown spectroscopy combined with optical emission spectroscopy. Eur. Phys. J. D 2009, 54, 77–86. [Google Scholar]
- Moosmüller, H.; Chakrabarty, R.K.; Arnott, W.P. Aerosol light absorption and its measurement: A review. J. Quant. Spectrosc. Radiat. Transfer 2009, 110, 844–878. [Google Scholar]
- Sappey, A.D.; Hill, E.S.; Settersten, T.; Linne, M.A. Fixed-frequency cavity ringdown diagnostic for atmospheric particulate matter. Opt. Lett. 1998, 23, 954–956. [Google Scholar]
- van der Wal, R.L.; Ticich, T.M. Cavity ringdown and laser-induced incandescence measurements of soot. Appl. Opt. 1999, 38, 1444–1451. [Google Scholar]
- Bulatov, V.; Fisher, M.; Schechter, I. Aerosol analysis by cavity-ring-down laser spectroscopy. Anal. Chim. Acta 2002, 466, 1–9. [Google Scholar]
- Thompson, J.E.; Smith, B.W.; Winefordner, J.D. Monitoring atmospheric particulate matter through cavity ring-down spectroscopy. Anal. Chem. 2002, 74, 1962–1967. [Google Scholar]
- Bulatov, V.; Khalmanov, A.; Schechter, I. Study of the morphology of a laser-produced aerosol plume by cavity ringdown laser absorption spectroscopy. Anal. Bioanal. Chem. 2003, 375, 1282–1286. [Google Scholar]
- Strawa, A.W.; Castaneda, R.; Owano, T.; Baer, D.S.; Paldus, B.A. The measurement of aerosol optical properties using continuous wave cavity ring-down techniques. J. Atmos. Oceanic Technol. 2003, 20, 454–465. [Google Scholar]
- Hallar, A.G.; Strawa, A.W.; Schmid, B.; Andrews, E.; Ogren, J.; Sheridan, P.; Ferrare, R.; Covert, D.; Elleman, R.; Jonsson, H.; Bokarius, K.; Luu, A. Atmospheric radiation measurements aerosol intensive operating period: Comparison of aerosol scattering during coordinated flights. J. Geophys. Res.-Atmos. 2006, 111, D05S09:01–D05S09:17. [Google Scholar]
- Schmid, B.; Ferrare, R.; Flynn, C.; Elleman, R.; Covert, D.; Strawa, A.; Welton, E.; Turner, D.; Jonsson, H.; Redemann, J.; Eilers, J.; Ricci, K.; Hallar, A.G.; Clayton, M.; Michalsky, J.; Smirnov, A.; Holben, B.; Barnard, J. How well do state-of-the-art techniques measuring the vertical profile of tropospheric aerosol extinction compare? J. Geophys. Res.-Atmos. 2006, 111, D05S07:01–D05S07:25. [Google Scholar]
- Pettersson, A.; Lovejoy, E.R.; Brock, C.A.; Brown, S.S.; Ravishankara, A.R. Measurement of aerosol optical extinction at 532 nm with pulsed cavity ring down spectroscopy. J. Aerosol Sci. 2004, 35, 995–1011. [Google Scholar]
- Baynard, T.; Lovejoy, E.R.; Pettersson, A.; Brown, S.S.; Lack, D.; Osthoff, H.; Massoli, P.; Ciciora, S.; Dube, W.P.; Ravishankara, A.R. Design and application of a pulsed cavity ring-down aerosol extinction spectrometer for field measurements. Aerosol Sci. Technol. 2007, 41, 447–462. [Google Scholar]
- Sheridan, P.J.; Arnott, W.P.; Ogren, J.A.; Andrews, E.; Atkinson, D.B.; Covert, D.S.; Moosmuller, H.; Petzold, A.; Schmid, B.; Strawa, A.W.; Varma, R.; Virkkula, A. The Reno Aerosol Optics Study: an evaluation of aerosol absorption measurement methods. Aerosol Sci. Technol. 2005, 39, 1–16. [Google Scholar]
- Smith, J.D.; Atkinson, D.B. A portable pulsed cavity ring-down transmission meter for measurement of the optical extinction of the atmospheric aerosol. Analyst 2001, 126, 1216–1220. [Google Scholar]
- Moosmüller, H.; Varma, R.; Arnott, W.P. Cavity ring-down and cavity-enhanced detection techniques for the measurement of aerosol extinction. Aerosol Sci. Technol. 2005, 39, 30–39. [Google Scholar]
- Richman, B.; Kachanov, A.; Paldus, B.; Strawa, A. Novel detection of aerosols: combined cavity ring-down and fluorescence spectroscopy. Opt. Express 2005, 13, 3376–3387. [Google Scholar]
- Riziq, A.A.; Erlick, C.; Dinar, E.; Rudich, Y. Optical properties of absorbing and non-absorbing aerosols retrieved by cavity ring down (CRD) spectroscopy. Atmos. Chem. Phys. Discuss. 2006, 6, 12347–12387. [Google Scholar]
- Spindler, C.; Riziq, A.A.; Rudich, Y. Retrieval of aerosol complex refractive index by combining cavity ring down aerosol spectrometer measurements with full size distribution information. Aerosol Sci. Technol. 2007, 41, 1011–1017. [Google Scholar]
- Lang-Yona, N.; Rudich, Y.; Segre, E.; Dinar, E.; Abo-Riziq, A. Complex refractive indices of aerosols retrieved by continuous wave-cavity ring down aerosol spectrometer. Anal. Chem. 2009, 81, 1762–1769. [Google Scholar]
- Butler, T.J.A.; Miller, J.L.; Orr-Ewing, A.J. Cavity ring-down spectroscopy measurements of single aerosol particle extinction. I. The effect of position of a particle within the laser beam on extinction. J. Chem. Phys. 2007, 126, 174302–174307. [Google Scholar]
- Butler, T.J.A.; Mellon, D.; Kim, J.; Litman, J.; Orr-Ewing, A.J. Optical-feedback cavity ring-down spectroscopy measurements of extinction by aerosol particles. J. Phys. Chem. A 2009, 113, 3963–3972. [Google Scholar]
- Kerstel, E.; Gianfrani, L. Advances in laser-based isotope ratio measurements: Selected applications. Appl. Phys. B 2008, 92, 439–449. [Google Scholar]
- Crosson, E.R.; Ricci, K.N.; Richman, B.A.; Chilese, F.C.; Owano, T.G.; Provencal, R.A.; Todd, M.W.; Glasser, J.; Kachanov, A.A.; Paldus, B.A.; Spence, T.G.; Zare, R.N. Stable isotope ratios using cavity ring-down spectroscopy: determination of 13C/12C for carbon dioxide in human breath. Anal. Chem. 2002, 74, 2003–2007. [Google Scholar]
- Kasyutich, V.L.; Martin, P.A.; Holdsworth, R.J. An off-axis cavity-enhanced absorption spectrometer at 1605 nm for the 12CO2/13CO2 measurement. Appl. Phys. B 2006, 85, 413–420. [Google Scholar]
- Wahl, H.; Fidric, B.; Rella, C.W.; Koulikov, S.; Kharlamov, B.; Tan, S.; Kachanov, A.A.; Richman, B.A.; Crosson, E.R.; Paldus, B.A.; Kalaskar, S.; Bowling, D.R. Applications of cavity ring-down spectroscopy to high precision isotope ratio measurement of 13C/12C in carbon dioxide. Isot. Environ. Health Stud. 2006, 42, 21–35. [Google Scholar]
- Wehr, R.; Kassi, S.; Romanini, D.; Gianfrani, L. Optical feedback cavity-enhanced absorption spectroscopy for in situ measurements of the ratio 13C:12C in CO2. Appl. Phys. B 2008, 92, 459–465. [Google Scholar]
- Wang, C.; Srivastava, N.; Jones, B.A.; Reese, R.B. A novel multiple species ringdown spectrometer for in situ measurements of methane, carbon dioxide, and carbon isotope. Appl. Phys. B 2008, 92, 259–270. [Google Scholar]
- Lis, G.; Wassenaar, L.I.; Hendry, M.J. High-precision laser spectroscopy D/H and 18O/16O measurements of microliter natural water samples. Anal. Chem. 2007, 80, 287–293. [Google Scholar]
- Gupta, P.; Noone, D.; Galewsky, J.; Sweeney, C.; Vaughn, B.H. Demonstration of high-precision continuous measurements of water vapor isotopologues in laboratory and remote field deployments using wavelength-scanned cavity ring-down spectroscopy (WS-CRDS) technology. Rapid Commun. Mass Spectrom. 2009, 23, 2534–2542. [Google Scholar]
- Risby, T.H.; Solga, S.F. Current status of clinical breath analysis. Appl. Phys. B 2006, 85, 421–426. [Google Scholar]
- Kleine, D.; Dahnke, H.; Urban, W.; Hering, P.; Mürtz, M. Real-time detection of 13CH4 in ambient air by use of mid-infrared cavity leak-out spectroscopy. Opt. Lett. 2000, 25, 1606–1608. [Google Scholar]
- Paldus, B.A.; Harb, C.C.; Spence, T.G.; Zare, R.N.; Gmachl, C.; Capasso, F.; Sivco, D.L.; Baillargeon, J.N.; Hutchinson, A.L.; Cho, A.Y. Cavity ringdown spectroscopy using mid-infrared quantum-cascade lasers. Opt. Lett. 2000, 25, 666–668. [Google Scholar]
- Menzel, L.; Kosterev, A.A.; Curl, R.F.; Tittel, F.K.; Gmachl, C.; Capasso, F.; Sivco, D.L.; Baillargeon, J.N.; Hutchinson, A.L.; Cho, A.Y.; Urban, W. Spectroscopic detection of biological NO with a quantum cascade laser. Appl. Phys. B 2001, 72, 859–863. [Google Scholar]
- Marshall, B.J.; Warren, J.R. Unidentified curved bacilli in the stomach of patients with gastritis and peptic ulceration. Lancet 1984, 323, 1311–1315. [Google Scholar]
- Thorpe, M.J.; Balslev-Clausen, D.; Kirchner, M.S.; Ye, J. Cavity-enhanced optical frequency comb spectroscopy: application to human breath analysis. Opt. Express 2008, 16, 2387–2397. [Google Scholar]
- Kosterev, A.A.; Malinovsky, A.L.; Tittel, F.K.; Gmachl, C.; Capasso, F.; Sivco, D.L.; Baillargeon, J.N.; Hutchinson, A.L.; Cho, A.Y. Cavity ringdown spectroscopic detection of nitric oxide with a continuous-wave quantum-cascade laser. Appl. Opt. 2001, 40, 5522–5529. [Google Scholar]
- Bakhirkin, Y.A.; Kosterev, A.A.; Roller, C.; Curl, R.F.; Tittel, F.K. Mid-infrared quantum cascade laser based off-axis integrated cavity output spectroscopy for biogenic nitric oxide detection. Appl. Opt. 2004, 43, 2257–2266. [Google Scholar]
- Halmer, D.; von Basum, G.; Horstjann, M.; Hering, P.; Mürtz, M. Time resolved simultaneous detection of 14NO and 15NO via mid-infrared cavity leak-out spectroscopy. Isot. Environ. Health Stud. 2005, 41, 303–311. [Google Scholar]
- McCurdy, M.R.; Bakhirkin, Y.A.; Tittel, F.K. Quantum cascade laser-based integrated cavity output spectroscopy of exhaled nitric oxide. Appl. Phys. B 2006, 85, 445–452. [Google Scholar]
- McCurdy, M.R.; Bakhirkin, Y.; Wysocki, G.; Tittel, F.K. Performance of an exhaled nitric oxide and carbon dioxide sensor using quantum cascade laser-based integrated cavity output spectroscopy. J. Biomed. Opt. 2007, 12, 034034:1–034034:9. [Google Scholar]
- Heinrich, K.; Fritsch, T.; Hering, P.; Mürtz, M. Infrared laser-spectroscopic analysis of 14NO and 15NO in human breath. Appl. Phys. B 2009, 95, 281–286. [Google Scholar]
- von Basum, G.; Dahnke, H.; Halmer, D.; Hering, P.; Murtz, M. Online recording of ethane traces in human breath via infrared laser spectroscopy. J. Appl. Physiol. 2003, 95, 2583–2590. [Google Scholar]
- Popp, A.; Müller, F.; Kühnemann, F.; Schiller, S.; von Basum, G.; Dahnke, H.; Hering, P.; Mürtz, M. Ultra-sensitive mid-infrared cavity leak-out spectroscopy using a CW optical parametric oscillator. Appl. Phys. B 2002, 75, 751–754. [Google Scholar]
- von Basum, G.; Halmer, D.; Hering, P.; Mürtz, M.; Schiller, S.; Müller, F.; Popp, A.; Kühnemann, F. Parts per trillion sensitivity for ethane in air with an optical parametricoscillator cavity leak-out spectrometer. Opt. Lett. 2004, 29, 797–799. [Google Scholar]
- Halmer, D.; Thelen, S.; Hering, P.; Mürtz, M. Online monitoring of ethane traces in exhaled breath with a difference frequency generation spectrometer. Appl. Phys. B 2006, 85, 437–443. [Google Scholar]
- Parameswaran, K.R.; Rosen, D.I.; Allen, M.G.; Ganz, A.M.; Risby, T.H. Off-axis integrated cavity output spectroscopy with a mid-infrared interband cascade laser for real-time breath ethane measurements. Appl. Opt. 2009, 48, B73–B79. [Google Scholar]
- Thelen, S.; Miekisch, W.; Halmer, D.; Schubert, J.; Hering, P.; Murtz, M. Intercomparison of infrared cavity leak-out spectroscopy and gas chromatography-flame ionization for trace analysis of ethane. Anal. Chem. 2008, 80, 2768–2773. [Google Scholar]
- Wang, C.; Mbi, A. A new acetone detection device using cavity ringdown spectroscopy at 266 nm: evaluation of the instrument performance using acetone sample solutions. Meas. Sci. Technol. 2007, 18, 2731–2741. [Google Scholar]
- Wang, C.; Surampudi, A.B. An acetone breath analyzer using cavity ringdown spectroscopy: an initial test with human subjects under various situations. Meas. Sci. Technol. 2008, 19, 105604:1–105604:10. [Google Scholar]
- Halmer, D.; von Basum, G.; Hering, P.; Mürtz, M. Mid-infrared cavity leak-out spectroscopy for ultrasensitive detection of carbonyl sulfide. Opt. Lett. 2005, 30, 2314–2316. [Google Scholar]
- Stamyr, K.; Vaittinen, O.; Jaakola, J.; Guss, J.; Metsälä, M.; Johanson, G.; Halonen, L. Background levels of hydrogen cyanide in human breath measured by infrared cavity ring down spectroscopy. Biomarkers 2009, 14, 285–291. [Google Scholar]
- McDonagh, C.; Burke, C.S.; MacCraith, B.D. Optical chemical sensors. Chem. Rev. 2008, 108, 400–422. [Google Scholar]
- Lakowicz, J.R. Principles of fluorescence spectroscopy, 3rd ed.; Springer: Berlin, Germany, 2006; p. 954. [Google Scholar]
- Ropp, R.C. Luminescence and the solid state, 2nd ed.; Elsevier: Amsterdam, Netherlands, 2004. [Google Scholar]
- Lefebvre, J.; Katz, M.I.; Leznoff, D.B. Vapochromic coordination polymers for use in analyte detection. US Patent No. 0130768, 2009. [Google Scholar]
- McGee, K.A.; Marquardt, B.J.; Mann, K.R. Concurrent sensing of benzene and oxygen by a crystalline salt of tris(5,6-dimethyl-1,10-phenanthroline)ruthenium(II). Inorg. Chem. 2008, 47, 9143–9145. [Google Scholar]
- Cariati, E.; Bu, X.; Ford, P.C. Solvent- and vapor-induced isomerization between the luminescent solids [CuI(4-pic)]4 and [CuI(4-pic)]∞ (pic = methylpyridine). The structural basis for the observed luminescence vapochromism. Chem. Mater. 2000, 12, 3385–3391. [Google Scholar]
- Grate, J.W.; Moore, L.K.; Janzen, D.E.; Veltkamp, D.J.; Kaganove, S.; Drew, S.M.; Mann, K.R. Steplike response behavior of a new vapochromic platinum complex observed with simultaneous acoustic wave sensor and optical reflectance measurements. Chem. Mater. 2002, 14, 1058–1066. [Google Scholar]
- Lefebvre, J.; Batchelor, R.J.; Leznoff, D.B. Cu[Au(CN)2]2(DMSO)2: Golden polymorphs that exhibit vapochromic behavior. J. Am. Chem. Soc. 2004, 126, 16117–16125. [Google Scholar]
- Elosúa, C.; Bariain, C.; Matias, I.R.; Arregui, FJ.; Luquin, A.; Laguna, M. Volatile alcoholic compounds fibre optic nanosensor. Sens. Actuators, B 2006, 115, 444–449. [Google Scholar]
- Forniés, J.; Fuertes, S.; Lopez, J.A.; Martin, A.; Sicilia, V. New water soluble and luminescent platinum(II) compounds, vapochromic behavior of [K(H2O)][Pt(bzq)(CN)2], new examples of the influence of the counterion on the photophysical properties of d8 square-planar complexes. Inorg. Chem. 2008, 47, 7166–7176. [Google Scholar]
- Buss, C.E.; Anderson, C.E.; Pomije, M.K.; Lutz, C.M.; Britton, D.; Mann, K.R. Structural investigations of vapochromic behavior. X-ray single-crystal and powder diffraction studies of [Pt(CN-iso-C3H7)4][M(CN)4] for M = Pt or Pd. J. Am. Chem. Soc. 1998, 120, 7783–7790. [Google Scholar]
- White-Morris, R.L.; Marilyn, M.O.; Saeed, A.; Balch, A.L. Intermolecular interactions in polymorphs of trinuclear gold(I) complexes: insight into the solvoluminescence of AuI3(MeN=COMe)3. Inorg. Chem. 2005, 44, 5021–5029. [Google Scholar]
- Lu, W.; Chan, M.C.W.; Zhu, N.; Che, C.-M.; He, Z.; Wong, K.-Y. Structural basis for vapoluminescent organoplatinum materials derived from noncovalent interactions as recognition components. Chem. Eur. J. 2003, 9, 6155–6166. [Google Scholar]
- MacCraith, B.D.; Polerecky, L. Luminescence based sensor. US Patent No. 0124336, 2004. [Google Scholar]
- Bariain, C.; Matias, I.R.; Fdez-Valdivielso, C.; Elosúa, C.; Luquin, A.; Garrido, J.; Laguna, M. Optical fibre sensors based on vapochromic gold complexes for environmental applications. Sens. Actuators, B 2005, 108, 535–541. [Google Scholar]
- Luquin, A.; Elosúa, C.; Vergara, E.; Estella, J.; Cerrada, E.; Bariáin, C.; Matías, I.R.; Garrido, J.; Laguna, M. Application of gold complexes in the development of sensors for volatile organic compounds. Gold. Bull. 2006, 40, 225–233. [Google Scholar]
- Keefe, M.H.; Benkstein, K.D.; Hupp, J.T. Luminescent sensor molecules based on coordinated metals: a review of recent developments. Coord. Chem. Rev. 2000, 205, 201–228. [Google Scholar]
- Balch, A.L. Polymorphism and luminescent behavior of linear, two-coordinate gold(I) complexes. Gold. Bull. 2004, 37, 45–50. [Google Scholar]
- White-Morris, R.L.; Olmstead, M.M.; Balch, A.L.; Elbjeirami, O.; Omary, M.A. Orange luminescence and structural properties of three isostructural halocyclohexylisonitrilegold(I) complexes. Inorg. Chem. 2003, 42, 6741–6748. [Google Scholar]
- Leznoff, D.B.; Lefebvre, J. Coordination polymers with cyanoaurate building blocks: potential new industrial applications for gold. Gold. Bull. 2005, 38, 47–54. [Google Scholar]
- Tzeng, B.-C.; Liu, W.-H.; Liao, J.-H.; Lee, G.-H.; Peng, S.-M. Self-assembly of gold(I) compounds with (aza-15-crown-5)dithiocarbamate and 2-mercapto-4-methyl-5-thiazoleacetic acid. Crys. Growth Des. 2004, 4, 573–577. [Google Scholar]
- Pyykkö, P. Theoretical chemistry of gold. II. Inorg. Chim. Acta 2005, 358, 4113–4130. [Google Scholar]
- Pyykkö, P. Theoretical chemistry of gold. Angew. Chem. Int. Ed. 2004, 43, 4412–4456. [Google Scholar]
- White-Morris, R.L.; Olmstead, M.M.; Jiang, F.; Tinti, D.S.; Balch, A.L. Remarkable variations in the luminescence of frozen solutions of [Au{C(NHMe)2}2](PF6).0.5(acetone). Structural and spectroscopic studies of the effects of anions and solvents on gold(I) carbene complexes. J. Am. Chem. Soc. 2002, 124, 2327–2336. [Google Scholar]
- White-Morris, R.L.; Olmstead, M.M.; Balch, A.L. Aurophilic interactions in cationic gold complexes with two isocyanide ligands. Polymorphic yellow and colorless forms of [(cyclohexyl isocyanide)2AuI](PF6) with distinct luminescence. J. Am. Chem. Soc. 2003, 125, 1033–1040. [Google Scholar]
- Balch, A.L. Remarkable luminescence behaviors and structural variations of two-coordinate gold(I) complexes. Struct. Bond. 2007, 123, 1–40. [Google Scholar]
- Tzeng, B.-C.; Yeh, H.-T.; Wu, Y.-L.; Kuo, J.-H.; Lee, G.-H.; Peng, S.-M. Supramolecular assembly of gold(I) complexes of diphosphines and N,N′-Bis-4-methylpyridyl oxalamide. Inorg. Chem. 2006, 45, 591–598. [Google Scholar]
- Hunks, W.J.; Jennings, M.C.; Puddephatt, R.J. Supramolecular gold(I) thiobarbiturate chemistry: Combining aurophilicity and hydrogen bonding to make polymers, sheets, and networks. Inorg. Chem. 2002, 41, 4590–4598. [Google Scholar]
- Assefa, Z.; Omary, M.A.; McBurnett, B.G.; Mohamed, A.A.; Patterson, H.H.; Staples, R.J.; Fackler, J.P. Syntheses, structure, and photoluminescence properties of the 1-dimensional chain compounds [(TPA)2Au][Au(CN)2] and (TPA)AuCl (TPA = 1,3,5-Triaza-7-phosphaadamantane). Inorg. Chem. 2002, 41, 6274–6280. [Google Scholar]
- Yip, S.-K.; Cheng, E.C.-C.; Li-Hua Yuan, N.Z.; Yam, V.W.-W. Supramolecular assembly of luminescent gold(I) alkynylcalix[4]crown-6 complexes with planar η2,η2-coordinated gold(I) centers. Angew. Chem. Int. Ed. 2004, 43, 4954–4957. [Google Scholar]
- Lee, Y.-A.; Eisenberg, R. Luminescence tribochromism and bright emission in gold(I) thiouracilate complexes. J. Am. Chem. Soc. 2003, 125, 7778–7779. [Google Scholar]
- Mansour, M.A.; Connick, W.B.; Lachicotte, R.J.; Gysling, H.J.; Eisenberg, R. Linear chain Au(I) dimer compounds as environmental sensors: A luminescent switch for the detection of volatile organic compounds. J. Am. Chem. Soc. 1998, 120, 1329–1330. [Google Scholar]
- Tzeng, B.-C.; Yeh, H.-T.; Huang, Y.-C.; Chao, H.-Y.; Lee, G.-H.; Peng, S.-M. A luminescent supermolecule with gold(I) quinoline-8-thiolate: crystal structure, spectroscopic and photophysical properties. Inorg. Chem. 2003, 42, 6008–6014. [Google Scholar]
- Fernández, E.J.; Lopez-de-Luzuriaga, J.M.; Monge, M.; Olmos, M.E.; Puelles, R.C.; Laguna, A.; Mohamed, A.A.; Fackler, J.J.P. Vapochromic behavior of {Ag2(Et2O)2[Au(C6F5)2]2}n with volatile organic compounds. Inorg. Chem. 2008, 47, 8069–8076. [Google Scholar]
- Catalano, V.J.; Horner, S.J. Luminescent gold(I) and silver(I) complexes of 2-(diphenylphosphino)-1-methylimidazole (dpim): Characterization of a three-coordinate Au(I) Ag(I) dimer with a short metal-metal separation. Inorg. Chem. 2003, 42, 8430–8438. [Google Scholar]
- Fernández, E.J.; Laguna, A.; López-de-Luzuriaga, José M.; Monge, M.; Pyykkö, P.; Runeberg, N. A study of the interactions in an extended unsupported gold-silver chain. Eur. J. Inorg. Chem. 2002, 2002, 750–753. [Google Scholar]
- Luquin, A.; Bariáin, C.; Vergara, E.; Cerrada, E.; Garrido, J.; Matias, I.R.; Laguna, M. New preparation of gold-silver complexes and optical fibre environmental sensors based on vapochromic [Au2Ag2(C6F5)4(phen)2]. Appl. Organomet. Chem. 2005, 19, 1232–1238. [Google Scholar]
- Bezunartea, M.; Estella, J.; Echeverría, J.C.; Elosúa, C.; Bariáin, C.; Laguna, M.; Luquin, A.; Garrido, J.J. Optical fibre sensing element based on xerogel-supported [Au2Ag2(C6F5)4(C14H10)]n for the detection of methanol and ethanol in the vapour phase. Sens. Actuators, B 2008, 134, 966–973. [Google Scholar]
- Elosúa, C.; Bariain, C.; Matias, I.R.; Arregui, F.J.; Luquin, A.; Vergara, E.; Laguna, M. Indicator immobilization on Fabry-Perot nanocavities towards development of fiber optic sensors. Sens. Actuators, B 2008, 130, 158–163. [Google Scholar]
- Mohamed, A.A.; Burini, A.; Fackler, J.P. Mixed-metal triangular trinuclear complexes: Dimers of gold-silver mixed-metal complexes from gold(I) carbeniates and silver(I) 3,5-diphenylpyrazolates. J. Am. Chem. Soc. 2005, 127, 5012–5013. [Google Scholar]
- Mohamed, A.A.; Galassi, R.; Papa, F.; Burini, A.; Fackler, J.P. Gold(I) and silver(I) mixed-metal trinuclear complexes: Dimeric products from the reaction of gold(I) carbeniates or benzylimidazolates with silver(I) 3,5-diphenylpyrazolate. Inorg. Chem. 2006, 45, 7770–7776. [Google Scholar]
- Fernández, E.J.; Laguna, A.; Lopez-de-Luzuriaga, J.M.; Monge, M.; Montiel, M.; Olmos, M.E.; Rodriguez-Castillo, M. Photophysical and theoretical studies on luminescent tetranuclear coinage metal building blocks. Organomet. 2006, 25, 3639–3646. [Google Scholar]
- Colis, J.C.F.; Larochelle, C.; Fernandez, E.J.; Lopez-de-Luzuriaga, J.M.; Monge, M.; Laguna, A.; Tripp, C.; Patterson, H. Tunable photoluminescence of closed-shell heterobimetallic Au-Ag dicyanide layered systems. Phys. Chem. B 2005, 109, 4317–4323. [Google Scholar]
- Wang, Q.-M.; Lee, Y.-A.; Crespo, O.; Deaton, J.; Tang, C.; Gysling, H.J.; Concepcion Gimeno, M.; Larraz, C.; Villacampa, M.D.; Laguna, A.; Eisenberg, R. Intensely luminescent gold(I)-silver(I) cluster complexes with tunable structural features. J. Am. Chem. Soc. 2004, 126, 9488–9489. [Google Scholar]
- Elosúa, C.; Bariain, C.; Matias, I.R.; Arregui, F.J.; Vergara, E.; Laguna, M. Optical fiber sensing devices based on organic vapor indicators towards sensor array implementation. Sens. Actuators, B 2009, 137, 139–146. [Google Scholar]
- Stork, J.R.; Rios, D.; Pham, D.; Bicocca, V.; Olmstead, M.M.; Balch, A.L. Metal-metal interactions in platinum(II)/gold(I) or platinum(II)/silver(I) salts containing planar cations and linear anions. Inorg. Chem. 2005, 44, 3466–3472. [Google Scholar]
- Fernández, E.J.; Laguna, A.; López-de-Luzuriaga, J.M.; Mendizabal, F.; Monge, M.; Olmos, M.E.; Pérez, J. Theoretical and photoluminescence studies on the d10-s2 Au1-Tl1 interaction in extended unsupported chains. Chem. Eur. J. 2003, 9, 456–465. [Google Scholar]
- Catalano, V.J.; Etogo, A.O. Luminescent coordination polymers with extended Au(I)–Ag(I) interactions supported by a pyridyl-substituted NHC ligand. Organomet. Chem. 2005, 690, 6041–6050. [Google Scholar]
- Chiu, P.L.; Chen, C.Y.; Zeng, J.Y.; Lu, C.Y.; Lee, H.M. Coordination polymers of silver(I) with bis(N-heterocyclic carbene): Structural characterization and carbene transfer. J. Organomet. Chem. 2005, 690, 1682–1687. [Google Scholar]
- Zhou, Y.; Zhang, X.; Chen, W.; Qiu, H. Synthesis, structural characterization, and luminescence properties of multinuclear silver complexes of pyrazole-functionalized NHC ligands containing Ag-Ag and Ag-π interactions. J. Organomet. Chem. 2008, 693, 205–215. [Google Scholar]
- Lee, C.K.; Vasam, C.S.; Huang, T.W.; Wang, H.M.J.; Yang, R.Y.; Lee, C.S.; Lin, I.J.B. Silver(I) N-heterocyclic carbenes with long N-alkyl chains. Organomet. 2006, 25, 3768–3775. [Google Scholar]
- Che, C.-M.; Tse, M.-C.; Chan, M.C.W.; Cheung, K.-K.; Phillips, D.L.; Leung, K.-H. Spectroscopic evidence for argentophilicity in structurally characterized luminescent binuclear silver(I) complexes. J. Am. Chem. Soc. 2000, 122, 2464–2468. [Google Scholar]
- Kanan, S.M.; Kanan, M.C.; Patterson, H.H. Photoluminescence spectroscopy as a probe of silver doped zeolites as photocatalysts. Curr. Opin. Solid State Mater. Sci. 2003, 7, 443–449. [Google Scholar]
- Rawashdeh-Omary, M.A.; Omary, M.A.; Patterson, H.H. Oligomerization of Au(CN)2− and Ag(CN)2− ions in solution via ground-state aurophilic and argentophilic bonding. J. Am. Chem. Soc. 2000, 122, 10371–10380. [Google Scholar]
- Rawashdeh-Omary, M.A.; Omary, M.A.; Shankle, G.E.; Patterson, H.H. Luminescence thermochromism in dicyanoargentate(I) ions doped in alkali halide crystals. J. Phys. Chem. B 2000, 104, 6143–6151. [Google Scholar]
- Omary, M.A.; Patterson, H.H. Luminescent homoatomic exciplexes in dicyanoargentate(I) ions doped in alkali halide crystals. 1. “Exciplex tuning” by site-selective excitation. J. Am. Chem. Soc. 1998, 120, 7696–7705. [Google Scholar]
- Omary, M.A.; Webb, T.R.; Assefa, Z.; Shankle, G.E.; Patterson, H.H. Crystal structure, electronic structure, and temperature-dependent Raman spectra of Tl[Ag(CN)2]: evidence for ligand-unsupported argentophilic interactions. Inorg. Chem. 1998, 37, 1380–1386. [Google Scholar]
- Fernández, E.J.; Jones, P.G.; Laguna, A.; Lopez-de-Luzuriaga, J.M.; Monge, M.; Perez, J.; Olmos, M.E. Synthesis, structure, and photophysical studies of luminescent two and three-dimensional gold-thallium supramolecular arrays. Inorg. Chem. 2002, 41, 1056–1063. [Google Scholar]
- Fernández, E.J.; Laguna, A.; López-de-Luzuriaga, J.M.; Olmos, M.E.; Pérez, J. Gold-thallium supramolecular arrays with 4,4′-bipyridine. Solvent induction of luminescent networks. Dalton Trans. 2004, 1801–1806. [Google Scholar]
- Fernández, E.J.; Laguna, A.; Lopez-de-Luzuriaga, J.M.; Monge, M.; Montiel, M.; Olmos, M.E.; Perez, J. Thallium(I) acetylacetonate as building blocks of luminescent supramolecular architectures. Organomet. 2004, 23, 774–782. [Google Scholar]
- Fernández, E.J.; Lopez-de-Luzuriaga, J.M.; Monge, M.; Montiel, M.; Olmos, M.E.; Perez, J. A detailed study of the vapochromic behavior of {Tl[Au(C6Cl5)2]}n. Inorg. Chem. 2004, 43, 3573–3581. [Google Scholar]
- Fernández, E.J.; Laguna, A.; Lopez-de-Luzuriaga, J.M.; Montiel, M.; Olmos, M.E.; Perez, J. Dimethylsulfoxide gold–thallium complexes. Effects of the metal–metal interactions in the luminescence. Inorg. Chim. Acta 2005, 358, 4293–4300. [Google Scholar]
- Fernández, E.J.; Lopez-de-Luzuriaga, J.M.; Olmos, M.E.; Perez, J. A family of Au-Tl loosely bound butterfly clusters. Inorg. Chem. 2005, 44, 6012–6018. [Google Scholar]
- Fernández, E.J.; Laguna, A.; Lopez-de-Luzuriaga, J.M.; Montiel, M.; Olmos, M.E.; Perez, J. Easy ketimine formation assisted by heteropolynuclear gold-thallium complexes. Organomet. 2006, 25, 1689–1695. [Google Scholar]
- Fernández, E.J.; Laguna, A.; Lopez-de-Luzuriaga, J.M.; Monge, M.; Montiel, M.; Olmos, M.E. Photophysical studies and excited-state structure of a blue phosphorescent gold-thallium complex. Inorg. Chem. 2007, 46, 2953–2955. [Google Scholar]
- van Zyl, W.E.; López-de-Luzuriaga, J.M.; Fackler, J.P. Luminescence studies of dinuclear gold(I) phosphor-1,1-dithiolate complexes. J. Mol. Struct. 2000, 516, 99–106. [Google Scholar]
- Fernández, E.J.; Lopez-de-Luzuriaga, J.M.; Monge, M.; Olmos, M.E.; Perez, J.; Laguna, A.; Mohamed, A.A.; Fackler, J.P. {Tl[Au(C6Cl5)2]}n: A vapochromic complex. J. Am. Chem. Soc. 2003, 125, 2022–2023. [Google Scholar]
- Fernández, E.J.; Laguna, A.; Lopez-de-Luzuriaga, J.M.; Monge, M.; Montiel, M.; Olmos, M.E. Unsupported gold(I)-copper(I) interactions through η1 Au-[Au(C6F5)2]- coordination to Cu+ lewis acid sites. Inorg. Chem. 2005, 44, 1163–1165. [Google Scholar]
- Dias, H.V.R.; Diyabalanage, H.V.K.; Eldabaja, M.G.; Elbjeirami, O.; Rawashdeh-Omary, M.A.; Omary, M.A. Brightly phosphorescent trinuclear copper(I) complexes of pyrazolates: Substituent effects on the supramolecular structure and photophysics. J. Am. Chem. Soc. 2005, 127, 7489–7501. [Google Scholar]
- Yam, V.W.-W.; Lo, K.K.-W. Luminescent polynuclear d10 metal complexes. Chem. Soc. Rev. 1999, 28, 323–334. [Google Scholar]
- Wang, F.; Yu, R.; Zhang, Q.-S.; Zhao, Z.-G.; Wu, X.-Y.; Xie, Y.-M.; Qin, L.; Chen, S.-C.; Lu, C.-Z. Solvent-dependent luminescent Cu(I) framework based on 5-(4-pyridyl)tetrazole. Solid State Chem. 2009, 182, 2555–2559. [Google Scholar]
- Cariati, E.; Bourassa, J. Luminescence response of the solid state polynuclear copper(I) iodide materials [CuI(4-picoline)]x to volatile organic compounds. Chem. Commun. 1998, 1623–1624. [Google Scholar]
- Ford, P.C.; Cariati, E.; Bourassa, J. Photoluminescence properties of multinuclear copper(I) compounds. Chem. Rev. 1999, 99, 3625–3648. [Google Scholar]
- Dias, H.V.R.; Diyabalanage, H.V.K.; Rawashdeh-Omary, M.A.; Franzman, M.A.; Omary, M.A. Bright phosphorescence of a trinuclear copper(I) complex: luminescence thermochromism, solvatochromism, and “concentration luminochromism”. J. Am. Chem. Soc. 2003, 125, 12072–12073. [Google Scholar]
- Kato, M. Luminescent platinum complexes having sensing functionalities. Bull. Chem. Soc. Jpn. 2007, 80, 287–294. [Google Scholar]
- Field, J.S.; Haines, R.J.; McMillin, D.R.; Summerton, G.C. Tuning solid emission by salts of the [Pt{4-(o-CH3–Ph)trpy}Cl]+ and [Pt{4-(o-CF3–Ph)trpy}Cl]+ luminophores: Crystal structures of [Pt{4′-(o-CH3–Ph)trpy}Cl]A (A = BF4 or SbF6) and [Pt{4-(o-CF3–Ph)trpy}Cl]SbF6 (trpy = 2,2′:6′,2″-terpyridine). Dalton Trans. 2002, 1369–1376. [Google Scholar]
- Yang, Q.-Z.; Tong, Q.-X.; Wu, L.-Z.; Wu, Z.-X.; Zhang, L.-P.; Tung, C.-H. Switch of the lowest excited-states of terpyridylplatinum(II) acetylide complexes bearing amino or azacrown moieties by proton and cations. Eur. J. Inorg. Chem. 2004, 1948–1954. [Google Scholar]
- Du, P.; Schneider, J.; Brennessel, W.W.; Eisenberg, R. Synthesis and structural characterization of a new vapochromic Pt(II) complex based on the 1-terpyridyl-2,3,4,5,6-pentaphenylbenzene (TPPPB) ligand. Inorg. Chem. 2008, 47, 69–77. [Google Scholar]
- Kui, S.C.F.; Chui, S.S.-Y.; Che, C.-M.; Zhu, N. Structures, photoluminescence, and reversible vapoluminescence properties of neutral platinum(II) complexes containing extended π-conjugated cyclometalated ligands. J. Am. Chem. Soc. 2006, 128, 8297–8309. [Google Scholar]
- Lu, W.; Chan, M.C.W.; Cheung, K.-K.; Che, C.-M. π-π interactions in organometallic systems. Crystal structures and spectroscopic properties of luminescent mono-, bi-, and trinuclear trans-cyclometalated platinum(II) complexes derived from 2,6-diphenylpyridine. Organomet. 2001, 20, 2477–2486. [Google Scholar]
- Wong, K.M.-C.; Yam, V.W.-W. Luminescence platinum(II) terpyridyl complexes—from fundamental studies to sensory functions. Coord. Chem. Rev. 2007, 251, 2477–2488. [Google Scholar]
- Wadas, T.J.; Wang, Q.-M.; Kim, Y.-J.; Flaschenreim, C.; Blanton, T.N.; Eisenberg, R. Vapochromism and its structural basis in a luminescent Pt(II) terpyridine-nicotinamide complex. J. Am. Chem. Soc. 2004, 126, 16841–16849. [Google Scholar]
- Drew, S.M.; Janzen, D.E.; Buss, C.E.; MacEwan, D.I.; Dublin, K.M.; Mann, K.R. An electronic nose transducer array of vapoluminescent platinum(II) double salts. J. Am. Chem. Soc. 2001, 123, 8414–8415. [Google Scholar]
- Sun, Y.; Ye, K.; Zhang, H.; Zhang, J.; Zhao, L.; Li, B.; Yang, G.; Yang, B.; Wang, Y.; Lai, S.-W.; Che, C.-M. Luminescent one-dimensional nanoscale materials with PtII⋯PtII interactions. Angew. Chem. Int. Ed. 2006, 45, 5610–5613. [Google Scholar]
- Dylla, A.G.; Janzen, D.E.; Pomije, M.K.; Mann, K.R. A comparison of isomers: trans- and cis-dicyanobis(para-ethylisocyanobenzene)platinum. Organomet. 2007, 26, 6243–6247. [Google Scholar]
- Stork, J.R.; Olmstead, M.M.; Balch, A.L. Effects of Pt⋯Pt bonding, anions, solvate molecules, and hydrogen bonding on the self-association of Chugaev's cation, a platinum complex with a chelating carbene ligand. Inorg. Chem. 2004, 43, 7508–7515. [Google Scholar]
- Lu, W.; Zhu, N.; Che, C.-M. Tethered trinuclear cyclometalated platinum(II) complexes: from crystal engineering to tunable emission energy. Chem. Commun. 2002, 900–901. [Google Scholar]
- Kato, M.; Omura, A.; Toshikawa, A.; Kishi, S.; Sugimoto, Y. Vapor-induced luminescence switching in crystals of the syn isomer of a dinuclear (bipyridine)platinum(II) complex bridged with pyridine-2-thiolate ions. Angew. Chem. Int. Ed. 2002, 41, 3183–3185. [Google Scholar]
- Grove, L.J.; Rennekamp, J.M.; Jude, H.; Connick, W.B. A new class of platinum(II) vapochromic salts. J. Am. Chem. Soc. 2004, 126, 1594–1595. [Google Scholar]
- Grove, L.J.; Oliver, A.G.; Krause, J.A.; Connick, W.B. Structure of a crystalline vapochromic platinum(II) salt. Inorg. Chem. 2008, 47, 1408–1410. [Google Scholar]
- Drew, S.M.; Smith, L.I.; McGee, K.A.; Mann, K.R. A platinum(II) extended linear chain material that selectively uptakes benzene. Chem. Mater. 2009, 21, 3117–3124. [Google Scholar]
- Sluch, I.M.; Miranda, A.J.; Slaughter, L.M. Channeled polymorphs of cis-M(CNPh)2Cl2 (M = Pt, Pd) with extended metallophilic interactions. Crys. Growth Des. 2009, 9, 1267–1270. [Google Scholar]
- Gussenhoven, E.M.; Jevric, M.; Olmstead, M.M.; Fettinger, J.C.; Mascal, M.; Balch, A.L. Crystal packing in planar platinum(II) and palladium(II) complexes. Hydrogen-bond-mediated supramolecular assembly of ten wedge-shaped molecules into a cyclic array. Crys. Growth Des. 2009, 9, 1786–1792. [Google Scholar]
- Baldauff, E.A.; Buriak, J.M. Optical sensing of amine vapors with a series of tin compounds. Chem. Commun. 2004, 2028–2029. [Google Scholar]
- Navale, S.C.; Mulla, I.S. Photoluminescence and gas sensing study of nanostructured pure and Sn doped ZnO. Mat. Sci. Eng. C 2009, 29, 1317–1320. [Google Scholar]
- Gu, F.; Wang, S.F.; Lü, M.K.; Cheng, X.F.; Liu, S.W.; Zhou, G.J.; Xu, D.; Yuan, D.R. Luminescence of SnO2 thin films prepared by spin-coating method. Crys. Growth 2004, 262, 182–185. [Google Scholar]
- Gu, F.; Wang, S.F.; Song, C.F.; Lü, M.K.; Qi, Y.X.; Liu, S.W.; Zhou, G.J.; Xu, D.; Yuan, D.R. Synthesis and luminescence properties of SnO2 nanoparticles. Chem. Phys. Lett. 2003, 372, 451–454. [Google Scholar]
- Gu, F.; Wang, S.F.; Lü, M.K.; Zhou, G.J.; Xu, D.; Yuan, D.R. Photoluminescence properties of SnO2 nanoparticles synthesized by sol-gel method. Phys. Chem. B 2004, 108, 8119–8123. [Google Scholar]
- Rao, G.; Chang, Q.; Lokowicz, R.; Murtaza, Z. Method using luminescent transition metal-ligand complex for detecting polar solvents. US Patent No. 6699717, 2004. [Google Scholar]
- Abe, T.; Shinozaki, K. Tuning of luminescence spectra of neutral ruthenium(II) complexes by crystal waters. Inorg. Chem. 2005, 44, 849–851. [Google Scholar]
- Liu, Z.; Bian, Z.; Bian, J.; Li, Z.; Nie, D.; Huang, C. Acetonitrile-vapor-induced color and luminescence changes in a cyclometalated heteroleptic iridium complex. Inorg. Chem. 2008, 47, 8025–8030. [Google Scholar]
- Beauvais, L.G.; Shores, M.P.; Long, J.R. Cyano-bridged Re6Q8 (Q = S, Se) cluster-cobalt(II) framework materials: Versatile solid chemical sensors. J. Am. Chem. Soc. 2000, 122, 2763–2772. [Google Scholar]
- Baho, N.; Zargarian, D. Syntheses, structures, spectroscopy, and chromotropism of new complexes arising from the reaction of nickel(II) nitrate with diphenyl(dipyrazolyl)methane. Inorg. Chem. 2007, 46, 299–308. [Google Scholar]
- An, B.-K.; Kwon, S.-K.; Park, S.Y. Highly sensitive fluorescence probes for organic vapors: on/off and dual color fluorescence switching. Bull. Korean Chem. Soc. 2005, 26, 1555–1559. [Google Scholar]
- An, B.-K.; Kwon, S.-K.; Jung, S.-D.; Park, S.Y. Enhanced emission and its switching in fluorescent organic nanoparticles. J. Am. Chem. Soc. 2002, 124, 14410–14415. [Google Scholar]
- Ooyama, Y.; Kumaoka, H.; Uwada, K.; Yoshida, K. Photophysical properties of phenanthro[9,10-d]imidazole-type fluorescent hosts upon inclusion of organic solvent molecules. Tetrahedron 2009, 65, 8336–8343. [Google Scholar]
- Ooyama, Y.; Nagano, S.; Yoshida, K. Drastic solid-state fluorescence enhancement behaviour of imidazo[4,5-a]naphthalene-type fluorescent hosts upon inclusion of polyethers and tert-butyl alcohol. Tetrahedron 2009, 65, 1467–1474. [Google Scholar]
- Horiuchi, T.; Iki, N.; Hoshino, H. Detection of cationic guest molecules by quenching of luminescence of a self-assembled host molecule consisting of terbium(III) and calix[4]arene-p-tetrasulfonates. Anal. Chim. Acta. 2009, 650, 258–263. [Google Scholar]
- Buss, C.E.; Mann, K.R. Synthesis and characterization of Pt(CN-p-(C2H5)C6H4)2(CN)2, a crystalline vapoluminescent compound that detects vapor-phase aromatic hydrocarbons. J. Am. Chem. Soc. 2002, 124, 1031–1039. [Google Scholar]
- Das, S.; Bharadwaj, P.K. Supramolecular host-guest systems of luminescent Zn(II) complexes with benzene, nitrobenzene, and ethanol: Selectivity of guest inclusion and solid-state fluorescence modulation studies. Crys. Growth Des. 2007, 7, 1192–1197. [Google Scholar]
- Das, S.; Bharadwaj, P.K. Self-assembly of a luminescent zinc(II) complex: A supramolecular host-guest fluorescence signaling system for selective nitrobenzene inclusion. Inorg. Chem. 2006, 45, 5257–5259. [Google Scholar]
- Pang, J.; Marcotte, E.J.-P.; Seward, C.; Brown, R.S.; Wang, S. A blue luminescent star-shaped ZnII complex that can detect benzene. Angew. Chem. Int. Ed. 2001, 40, 4042–4045. [Google Scholar]
- Hou, L.; Lin, Y.-Y.; Chen, X.-M. Porous metal-organic framework based on μ4-oxo tetrazinc clusters: sorption and guest-dependent luminescent properties. Inorg. Chem. 2008, 47, 1346–1351. [Google Scholar]
- Di Natale, C.; Salimbeni, D.; Paolesse, R.; Macagnano, A.; D'Amico, A. Porphyrins-based opto-electronic nose for volatile compounds detection. Sens. Actuators, B 2000, 65, 220–226. [Google Scholar]
- Spadavecchia, J.; Ciccarella, G.; Siciliano, P.; Capone, S.; Rella, R. Spin-coated thin films of metal porphyrin-phthalocyanine blend for an optochemical sensor of alcohol vapours. Sens. Actuators, B 2004, 100, 88–93. [Google Scholar]
- Yusoff, N.H.; Salleh, M.M.; Yahaya, M. Fluorescence gas sensor using TiO2 nanoparticles coated with porphyrin dye thin Films. Solid State Sci. Technol. 2008, 16, 63–74. [Google Scholar]
- Ni, R.; Tong, R.-B.; Guo, C.-C.; Shen, G.-L.; Yu, R.-Q. An anthracene/porphyrin dimer fluorescence energy transfer sensing system for picric acid. Talanta 2004, 63, 251–257. [Google Scholar]
- Regulska, E.; Tarasiewicz, M.; Puzanowska-Tarasiewicz, H. Extractive-spectrophotometric determination of some phenothiazines with dipicrylamine and picric acid. J. Pharm. Biomed. Anal. 2002, 27, 335–340. [Google Scholar]
- Levitsky, I.A.; Krivoshlykov, S.G. Method for detection of organic vapors based on fluorescence enhancement in porphyrin aggregates. US Patent No. 6623973, 2003. [Google Scholar]
- Ogoshi, T.; Harada, A. Chemical sensors based on cyclodextrin derivatives. Sensors 2008, 8, 4961–4982. [Google Scholar]
- Tanabe, T.; Touma, K.; Hamasaki, K.; Ueno, A. Immobilized fluorescent cyclodextrin on a cellulose membrane as a chemosensor for molecule detection. Anal. Chem. 2001, 73, 3126–3130. [Google Scholar]
- Surpateanu, G.G.; Becuwe, M.; Lungu, N.C.; Dron, P.I.; Fourmentin, S.; Landy, D.; Surpateanu, G. Photochemical behaviour upon the inclusion for some volatile organic compounds in new fluorescent indolizine β-cyclodextrin sensors. J. Photochem. Photobiol. A 2007, 185, 312–320. [Google Scholar]
- Surpateanu, G.G.; Landy, D.; Lungu, C.N.; Fourmentin, S.; Surpateanu, G.; Réthoré, C.; Avarvari, N. Synthesis and inclusion capability of a β-cyclodextrin-tetrathiafulvalene derivative. Tetrahedron 2006, 62, 9701–9704. [Google Scholar]
- Surpateanu, G.G.; Landy, D.; Lungu, N.C.; Fourmentin, S.; Surpateanu, G. New fluorescent bis-β-cyclodextrin-indolizine sensor. Synthesis and sensing ability. J. Heterocycl. Chem. 2007, 44, 783–786. [Google Scholar]
- Lungu, N.C.; Dépret, A.; Delattre, F.; Surpateanu, G.G.; Cazier, F.; Woisel, P.; Shirali, P.; Surpateanu, G. Synthesis of a new fluorinated fluorescent β-cyclodextrin sensor. J. Fluorine Chem. 2005, 126, 385–388. [Google Scholar]
- Delattre, F.; Woisel, P.; Surpateanu, G.; Cazier, F.; Blach, P. 1-(4-nitrophenoxycarbonyl)-7-pyridin-4-yl indolizine: A new versatile fluorescent building block. Application to the synthesis of a series of fluorescent β-cyclodextrins. Tetrahedron 2005, 61, 3939–3945. [Google Scholar]
- Delattre, F.; Woisel, P.; Surpateanu, G.; Bria, M.; Caziera, F.; Decocka, P. 1,3-dipolar cycloaddition reaction of bipyridinium ylides with the propynamido-β-cyclodextrin. A regiospecific synthesis of a new class of fluorescent β-cyclodextrins. Tetrahedron 2004, 60, 1557–1562. [Google Scholar]
- Delattre, F.; Woisel, P.; Cazier, F.; Decock, P.; Surpateanu, G. Adamantanol inclusion in fluorescent β–cyclodextrin derivatives. Theoretical study by molecular mechanics and quantum semi–empirical methods. Internet Electron. J. Mol. Des 2005, 4, 1–8. [Google Scholar]
- Delattre, F.; Woisel, P.; Briab, M.; Surpateanu, G. Structural investigations of pyridin-4-yl indolizine modified β-cyclodextrin derivatives as fluorescent chemosensors for organic guest molecules. Carbohydr. Res. 2005, 340, 1706–1713. [Google Scholar]
- Becuwe, M.; Landy, D.; Delattre, F.; Cazier, F.; Fourmentin, S. Fluorescent indolizine-β-cyclodextrin derivatives for the detection of volatile organic compounds. Sensors 2008, 8, 3689–3705. [Google Scholar]
- Vrkoslava, V.; Jelinek, I.; Trojan, T.; Jindrich, J.; Dian, J. Porous silicon with β-cyclodextrin modified surface for photoluminescence sensing of organic molecules in gas and liquid phase. Phys. E 2007, 38, 200–204. [Google Scholar]
- Fourmentin, S.; Surpateanu, G.G.; Blach, P.; Landy, D.; Decock, P.; Surpateanu, G. Experimental and theoretical study on the inclusion capability of a fluorescent indolizine β-cyclodextrin sensor towards volatile and semi-volatile organic guest. J. Incl. Phenom. Macro. 2006, 55, 263–269. [Google Scholar]
- Narita, M.; Naranchimeg, D.; Teranishi, K.; Hamada, F. Selective fluorescent molecular sensing based on 2A,2D-disulfonyl dibenzosulfolane-diphenyl-capped β-cyclodextrin for phenolic guests. Anal. Sci. 2002, 18, 711–713. [Google Scholar]
- Aernecke, M.J.; Guo, J.; Sonkusale, S.; Walt, D.R. Design, implementation, and field testing of a portable fluorescence-based vapor sensor. Anal. Chem. 2009, 81, 5281–5290. [Google Scholar]
- Aernecke, M.J.; Walt, D.R. Temporally resolved fluorescence spectroscopy of a microarray-based vapor sensing system. Anal. Chem. 2009, 81, 5762–5769. [Google Scholar]
- Albert, K.J.; Lewis, N.S.; Schauer, C.L.; Sotzing, G.A.; Stitzel, S.E.; Vaid, T.P.; Walt, D.R. Cross-reactive chemical sensor arrays. Chem. Rev. 2000, 100, 2595–2626. [Google Scholar]
- Albert, K.J.; Walt, D.R.; Gill, D.S.; Pearce, T.C. Optical multibead arrays for simple and complex odor discrimination. Anal. Chem. 2001, 73, 2501–2508. [Google Scholar]
- Epstein, J.R.; Walt, D.R. Fluorescence-based fibre optic arrays: A universal platform for sensing. Chem. Soc. Rev. 2003, 32, 203–214. [Google Scholar]
- Stitzel, S.E.; Cowen, L.J.; Albert, K.J.; Walt, D.R. Array-to-array transfer of an artificial nose classifier. Anal. Chem. 2001, 73, 5266–5271. [Google Scholar]
- Dickinson, T.A.; Michael, K.L.; Kauer, J.S.; Walt, D.R. Convergent, self-encoded bead sensor arrays in the design of an artificial nose. Anal. Chem. 1999, 71, 2192–2198. [Google Scholar]
- Stevens, N.; Akins, D.L. Dye-doped inorganic/organic composite films as fluorescence sensors for methanol vapor. Sens. Actuators, B 2007, 123, 59–64. [Google Scholar]
- Xia, H.; Peng, J.; Liu, K.; Li, C.; Fang, Y. Preparation and gas sensing properties of novel CdS-supramolecular organogel hybrid films. J. Phys. D: Appl. Phys. 2008, 41, 1–8. [Google Scholar]
- Chen, J.; Cao, Y. Fluorescence responses of 1-methyl-1,2,3,4,5-pentaphenylsilole thin layer to vapors of common solvents. Sens. Actuators, B 2006, 114, 65–70. [Google Scholar]
- Katsumata, T.; Aizawa, H.; Takechi, W.; Ito, K.; Komuro, S.; Morikawa, T. Gas Sensing Using Photoluminescence from Phosphor Contained Silica Gel; Toyo University: Saitama, Japan, 2006; pp. 1–4. [Google Scholar]
- Katsumata, T.; Aizawa, H.; Kobayashi, C.; Ito, K. Fluorescence sensor application of phosphor contained silica gel. SICE Annual Conference in Sapporo; Hokkaido Institute of Technology: Hokkaido, Japan, 2004. [Google Scholar]
- Yuan, M.; Li, Y.; Liu, H.; Li, Y. Chemical sensors based on π-conjugated organic molecules and gold nanoparticles. Sci. China Ser. B Chem. 2009, 52, 715–730. [Google Scholar]
- Quaranta, A.; Carturan, S.; Bonafini, M.; Maggioni, G.; Tonezzer, M.; Mattei, G.; de Julián Fernandez, C.; Della Mea, G.; Mazzoldi, P. Optical sensing to organic vapors of fluorinated polyimide nanocomposites containing silver nanoclusters. Sens. Actuators, B 2006, 118, 418–424. [Google Scholar]
- Che, Y.; Yang, X.; Loser, S.; Zang, L. Expedient vapor probing of organic amines using fluorescent nanofibers fabricated from an n-type organic semiconductor. Nano Lett. 2008, 8, 2219–2223. [Google Scholar]
- Butcher, K.S.A.; Ferris, J.M.; Phillips, M.R.; Wintrebert-Fouquet, M.; Wah, J.W.J.; Jovanovic, N.; Vyverman, W.; Chepurnov, V.A. A luminescence study of porous diatoms. Mat. Sci. Eng. C 2005, 25, 658–663. [Google Scholar]
- Stefano, L.D.; Rendina, I.; Castellino, V.P.; Stefano, M.D.; Bismuto, A.; Maddalena, P. Marine diatoms as optical chemical sensors. Appl. Phys. Lett. 2005, 87, 1–3. [Google Scholar]
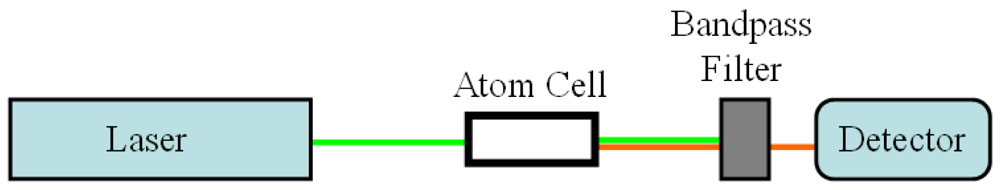
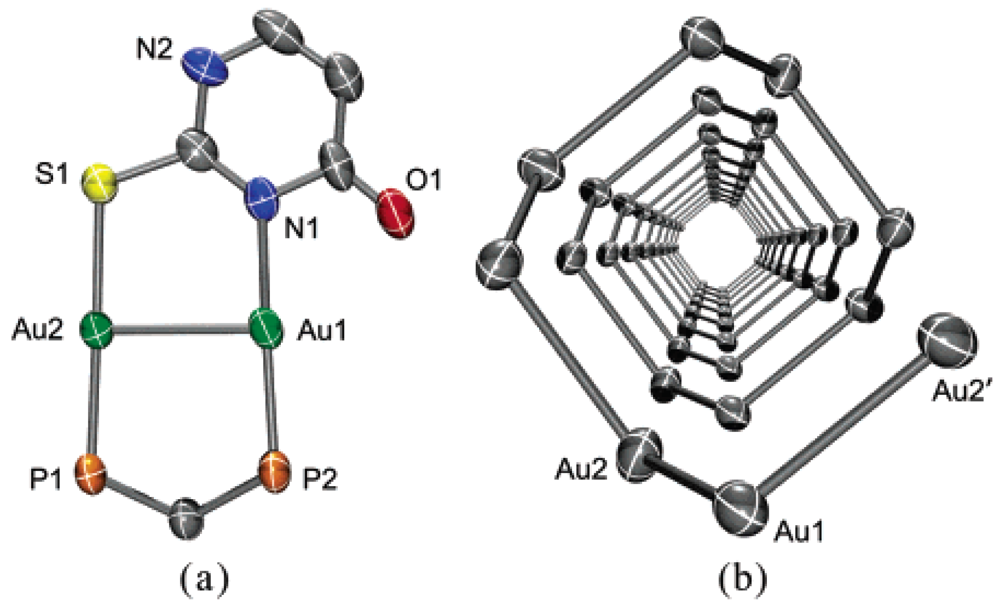
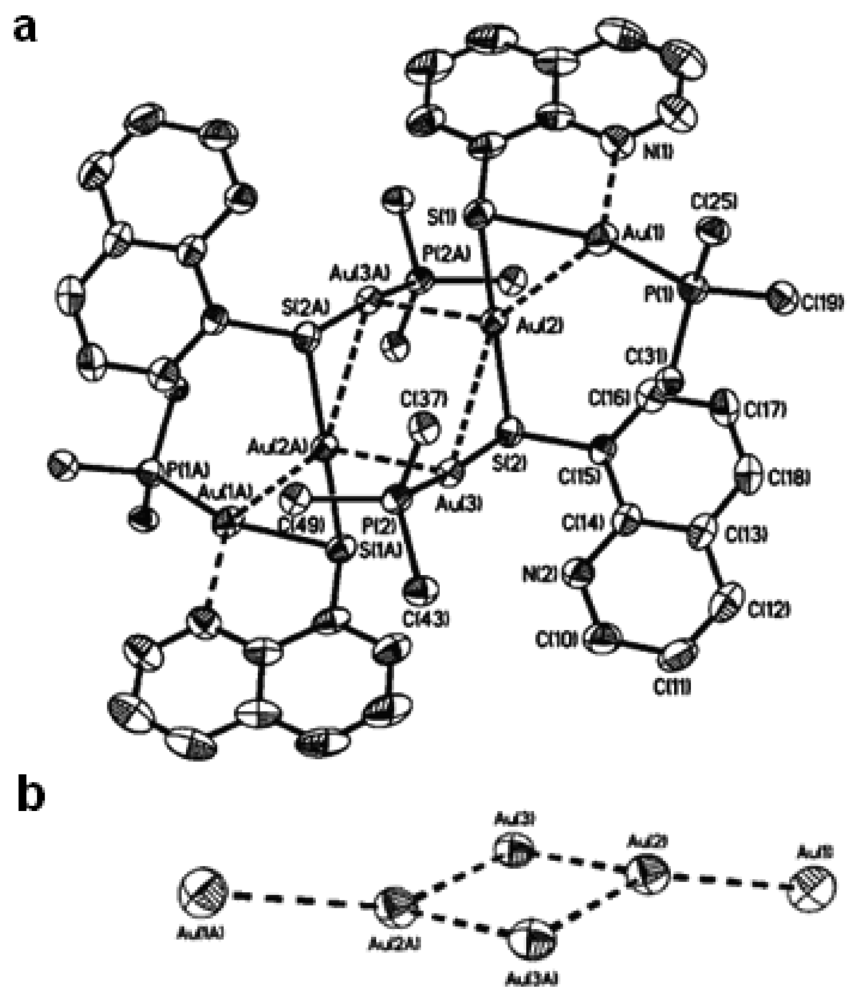
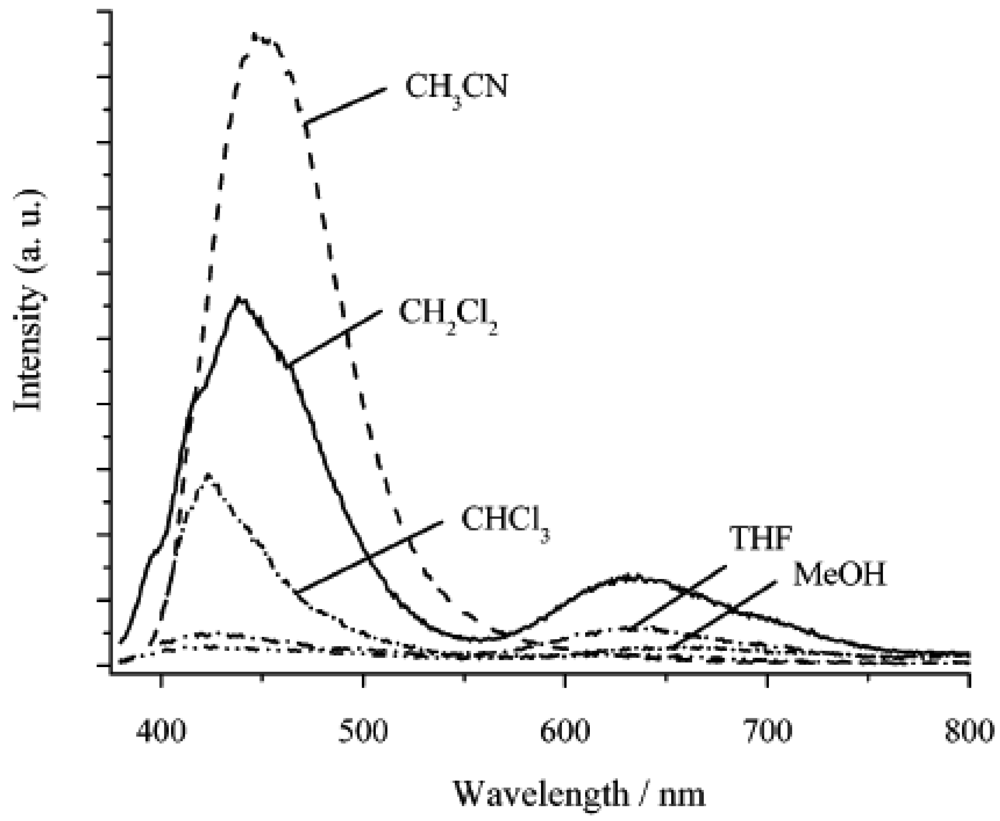




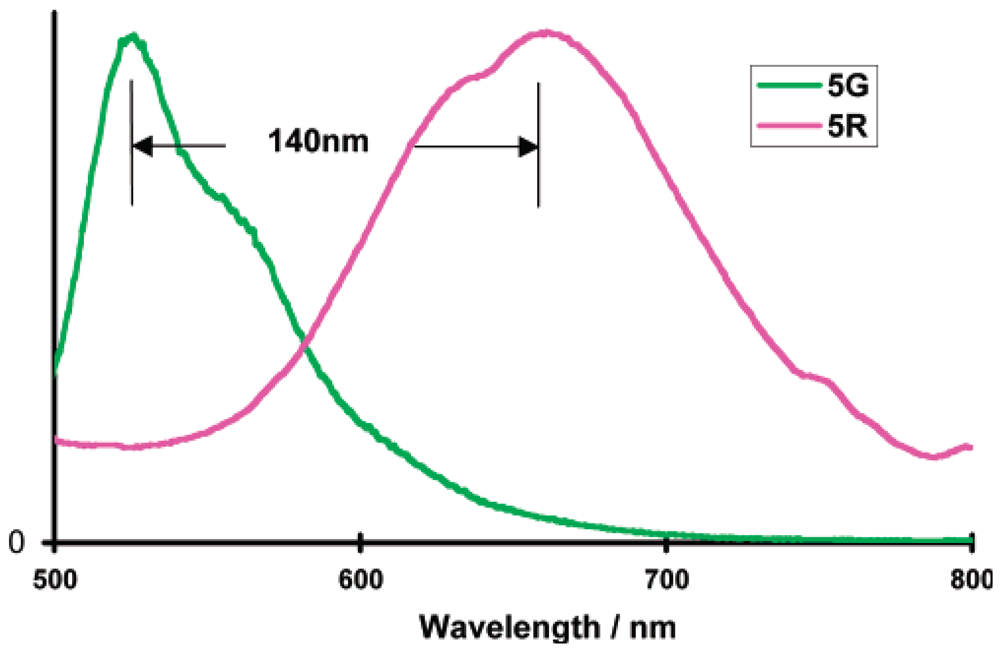

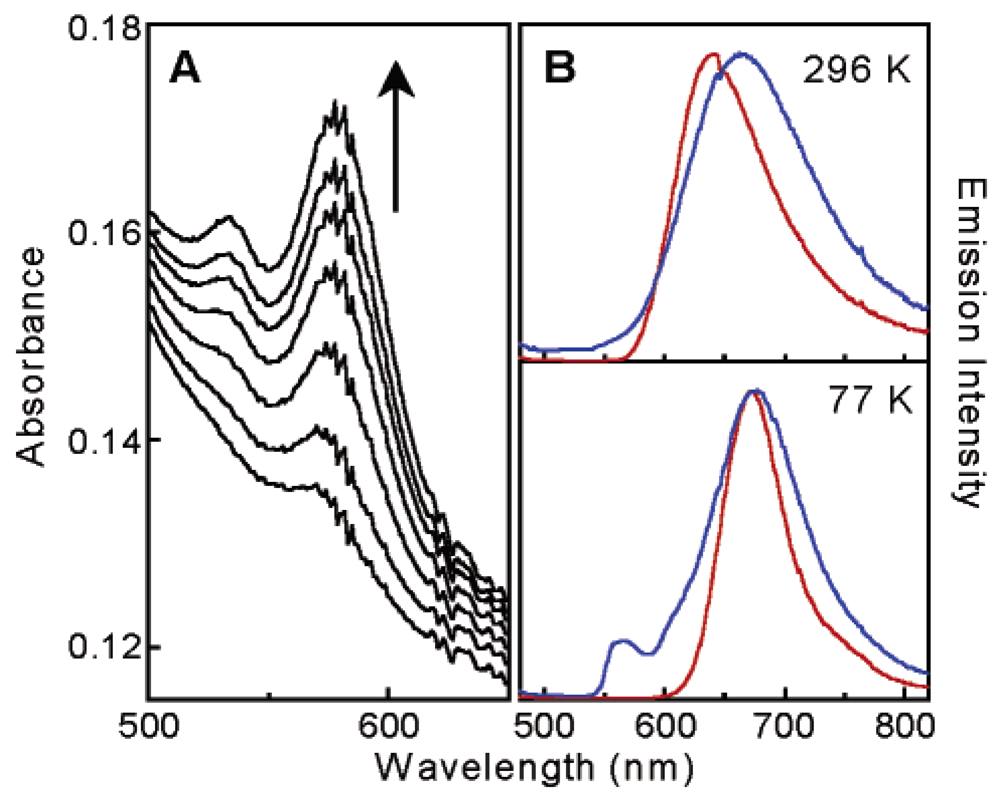

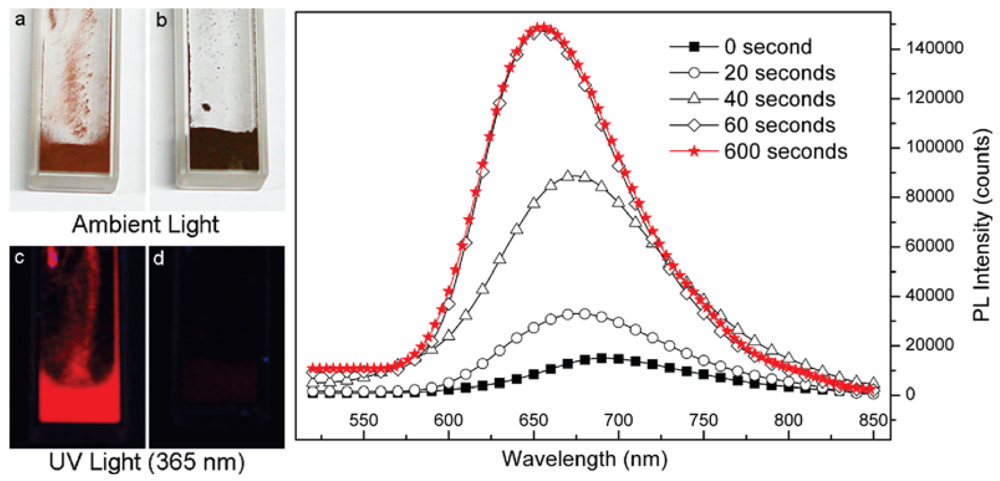

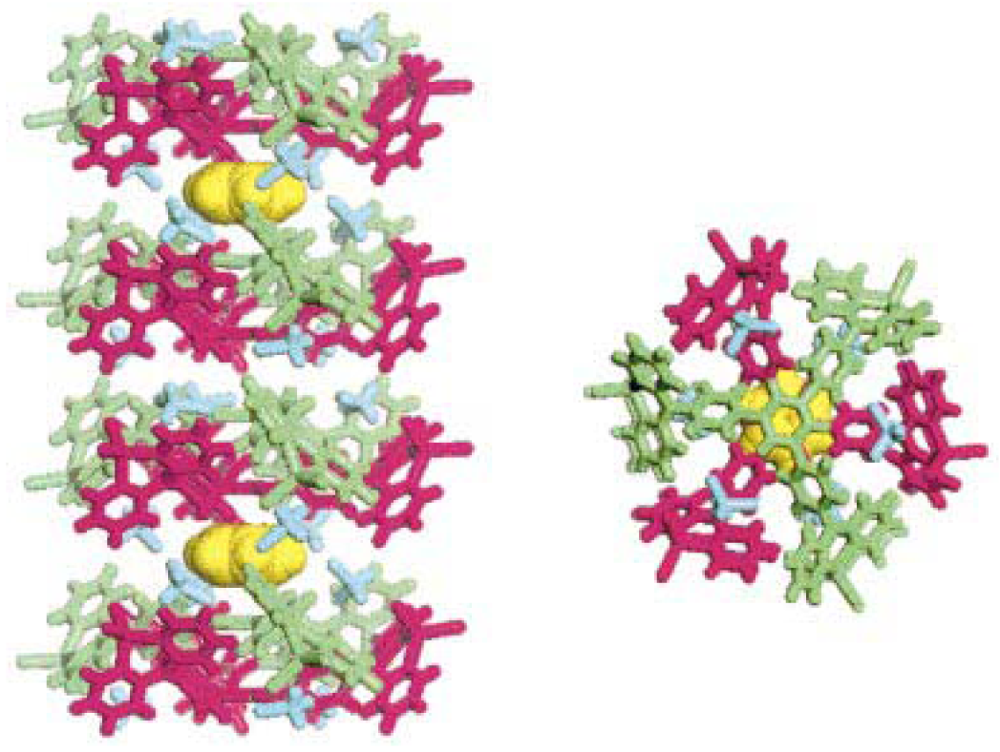
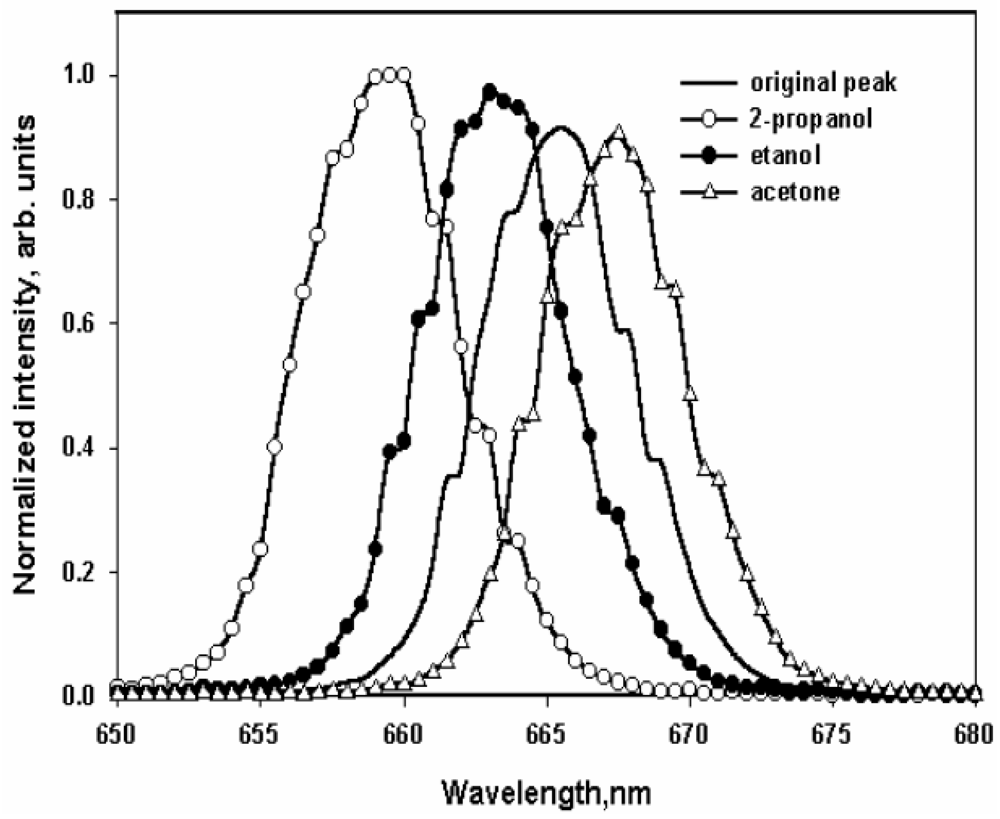
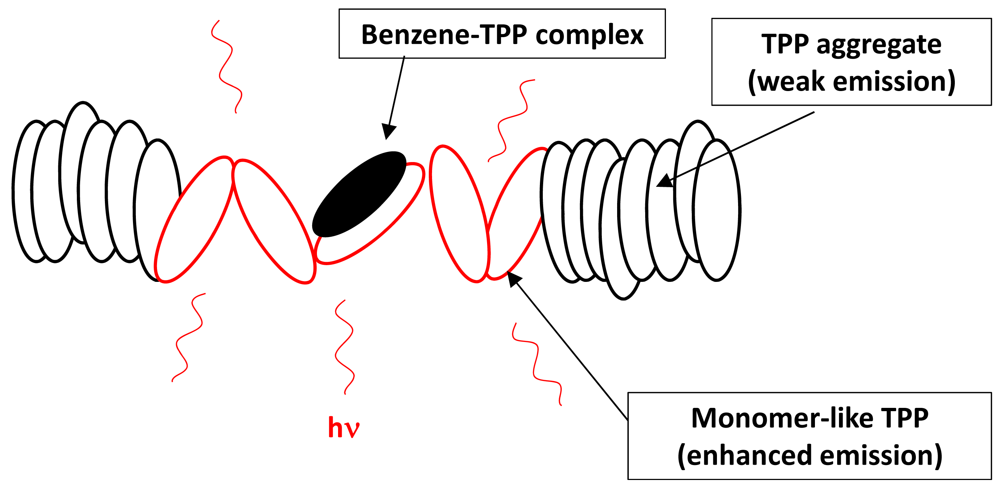
| Element | Sample | Method | LOD | Notes | Reference |
|---|---|---|---|---|---|
| Tl | Sediment | LEAFS | 0.5 ppb | HNO3-HF Dissolution | [75] |
| Al, Ba, Ca, Fe, Mg, Mn, Na, Si, Sr, Ti | Sediment | LIBS | Not given | Various sampling systems evaluated, samples were dried, instrument was ship-board | [76] |
| Al, Ba, C, Ca, Fe, K, Li, Mn, Na, Si, Sr, Ti | Sediment | LIBS | 114-566 ppm | Underwater measurement using many techniques | [72] |
| Ba, C, Ca, Cu, Fe, O, S, Zn | Minerals | LIBS | Not given | [77] | |
| C, Br, Cl | Organic solids | LIBS | 0.011, 0.054 | LODs refer to Br/C and Cl/C ratio, respectively | [78] |
| As, Cr, Cu | Treated wood waste | LIBS | Not given | X-ray fluorescence also evaluated | [79] |
| As, Ca, Cr, Cu, Na, Zn | Treated wood | LIBS | Not given | Wood discriminated based on treatment, compared to digestion and AAS | [80] |
| Many (21) | Ore | LIBS | 2-16 ppm | Contaminant screening in open pit mines | [81,82] |
| P, Si | Phosphate ore | LIBS | Not given | Removal of high silica pebbles, single shot analysis | [83] |
| Al, Fe, K, Si, Mn, P | Iron ore | LIBS | Not given | Principle component regression, samples powdered, pressed | [84] |
| Many (34) | Beryl | LIBS | Not given | Differentiation of country of origin | [85] |
| Ba, Ca, Cd, Cr, Mg, Mn, S, Ti | Slag | LIBS | 6-16 ppm | Risk assessment of steel plant waste | [86] |
| C | Soil | LIBS | 300 ppm | Also examines near-IR spec. and inelastic neutron scattering | [87] |
| C, N | Soil | LIBS | Not given | Acid washing used to distinguish organic and inorganic C | [88] |
| Ba, Ca, Fe, K, Mg, Ni, P, S | Soil | LIBS | 7-12 ppm | 2% precision | [89] |
| Cr | Soil | LIBS | Not given | Principle component regression, samples pressed into pellets | [90] |
| Al, Ba, Ca, Cr, Fe, K, Mg, Na, Sr, Ti, V, Zr | Soil, contaminated | LIBS | 2-12 ppm | Soil contaminated by coastal oil spill, compared to ICP- emission spectroscopy | [91] |
| Fe, Cr | Soil, rocks, vegetation | LIBS | 2 ppth | Laboratory-based assessment on contamination of remote coastal areas from industrial activity | [92] |
| Al, B, Ba, C, Ca, Fe, H, Mg, Mn, Na, Si, Sr | Marble and wood | LIBS | Not given | Immersed in seawater, only marble yielded quantitative results | [73] |
| Fe, Mg, Si | Asbestos | LIBS | Not given | Discrimination between serpentine asbestos, amphibole asbestos, and cement | [93] |
| Element | Sample | Sample Preparation | Method | LOD | Notes | Reference |
|---|---|---|---|---|---|---|
| Tl | Water | Direct analysis | LEAFS | 0.03 ng/L | [96] | |
| Pb | Water | Direct analysis | LA-LEAFS | 35 ppb | [122] | |
| Pb | Water | Pneumatically sprayed | LIBS, LA-LEAFS | 75 and 4.3 ppm | [123] | |
| Cr | Water | Preconcentration using cation exchange resin | LIBS | 500 ng/L | Sample preparation allowed speciation of CrIII and CrIV | [124] |
| Al | Seawater | Autosampler | ETA-LEAFS | 5 ng/L | CCD camera detector | [125] |
| Pb | Seawater | Autosampler | ETA-LEAFS | 0.3 ng/L | CCD camera detector | [126] |
| Cr | Seawater | Direct analysis | LIBS | 60 ppm | [127] | |
| Ca, Mg, Na | Seawater | Direct analysis | LIBS | Not given | Sampled at the surface, in air at atmospheric pressure | [128] |
| Al, Ba, Cr, Cu, K, Mg, Na, Ni, Pb, Si, Ti, Zr | Wastewater | Evaporation of water | LIBS | 1-301 ppm | Paint manufacturing plant effluent | [129] |
| Ca, Cu, Fe, K, Mg, Mo, Na, Ni, Zn | Crude oil | Serial distillation, heating in furnace to form pellet | LIBS | 2-4 ppm | Compared to ICP-OES | [130] |
| Element | Sample | Sample Preparation | Method | LOD | Notes | Reference |
|---|---|---|---|---|---|---|
| Hg | Air | Direct analysis | LEAFS | 0.1 ng/m3 | Double resonance excitation, quartz cell | [134] |
| Al, Ca, Cr, Cu, Mg, Mn, Na | Urban aerosol | PM2.5 cyclonic inlet and virtual impactor | LIBS | 15-185 fg | Field experiment at Pittsburgh Aerosol Supersite | [135] |
| Al, Ba, Ca, Cl, Fe, Mg, Na, P | Urban bioaerosol | Direct analysis | LIBS | Not given | Comparison to aerosol mass spectrometry, 0.1–2 μm aerosols can be quantified | [133] |
| Many (14) | Aerosol | Cascade impactor, greased aluminum substrate | LIBS | Not given | Particles from steel making, composition shows size dependence | [136] |
| Al, C, Ca, Fe, N, Na, O, P, S, Si | Inorganic aerosol | Impactor or passive deposition | micro-Raman | Not given | >1 μm diameter, organic and mixed particles more problematic | [137] |
| Species | Technique | Laser | Reference |
|---|---|---|---|
| 13/12C in CO2 | CRDS | Near IR-External Cavity Diode Laser | [232] |
| 13/12C in CO2 | OA-CEAS | DF Diode Laser | [233] |
| 13/12C in CO2 | CRDS | Near IR Diode Laser | [234] |
| 13/12C in CO2 | OF-CEAS | DF Diode Laser | [235] |
| 13C in CO2 | NIR-CW-CRDS | DF Laser Diodes | [236] |
| D/H, 18O/16O | OA-ICOS | Not known | [237] |
| Water | WS-CRDS | Tunable Diode Laser | [238] |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fiddler, M.N.; Begashaw, I.; Mickens, M.A.; Collingwood, M.S.; Assefa, Z.; Bililign, S. Laser Spectroscopy for Atmospheric and Environmental Sensing. Sensors 2009, 9, 10447-10512. https://doi.org/10.3390/s91210447
Fiddler MN, Begashaw I, Mickens MA, Collingwood MS, Assefa Z, Bililign S. Laser Spectroscopy for Atmospheric and Environmental Sensing. Sensors. 2009; 9(12):10447-10512. https://doi.org/10.3390/s91210447
Chicago/Turabian StyleFiddler, Marc N., Israel Begashaw, Matthew A. Mickens, Michael S. Collingwood, Zerihun Assefa, and Solomon Bililign. 2009. "Laser Spectroscopy for Atmospheric and Environmental Sensing" Sensors 9, no. 12: 10447-10512. https://doi.org/10.3390/s91210447
APA StyleFiddler, M. N., Begashaw, I., Mickens, M. A., Collingwood, M. S., Assefa, Z., & Bililign, S. (2009). Laser Spectroscopy for Atmospheric and Environmental Sensing. Sensors, 9(12), 10447-10512. https://doi.org/10.3390/s91210447





