Biosensor Techniques Used for Determination of Telomerase Activity in Cancer Cells
Abstract
:1. Introduction
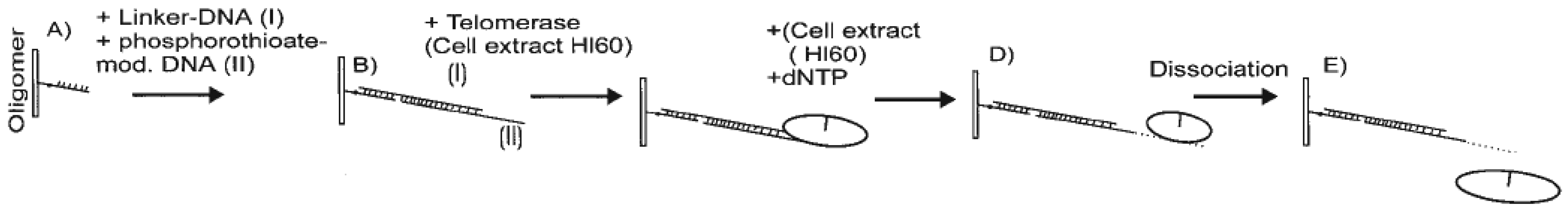
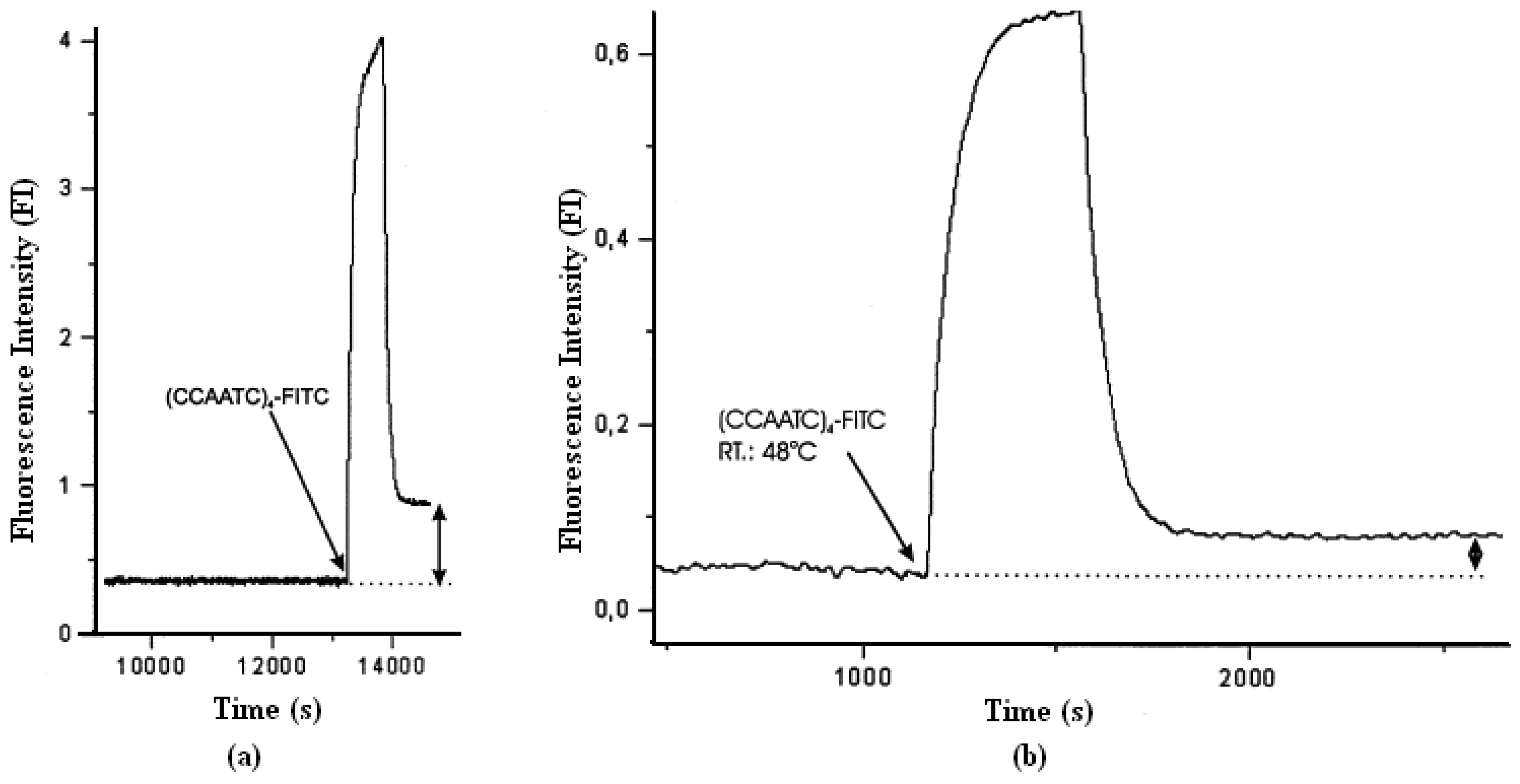
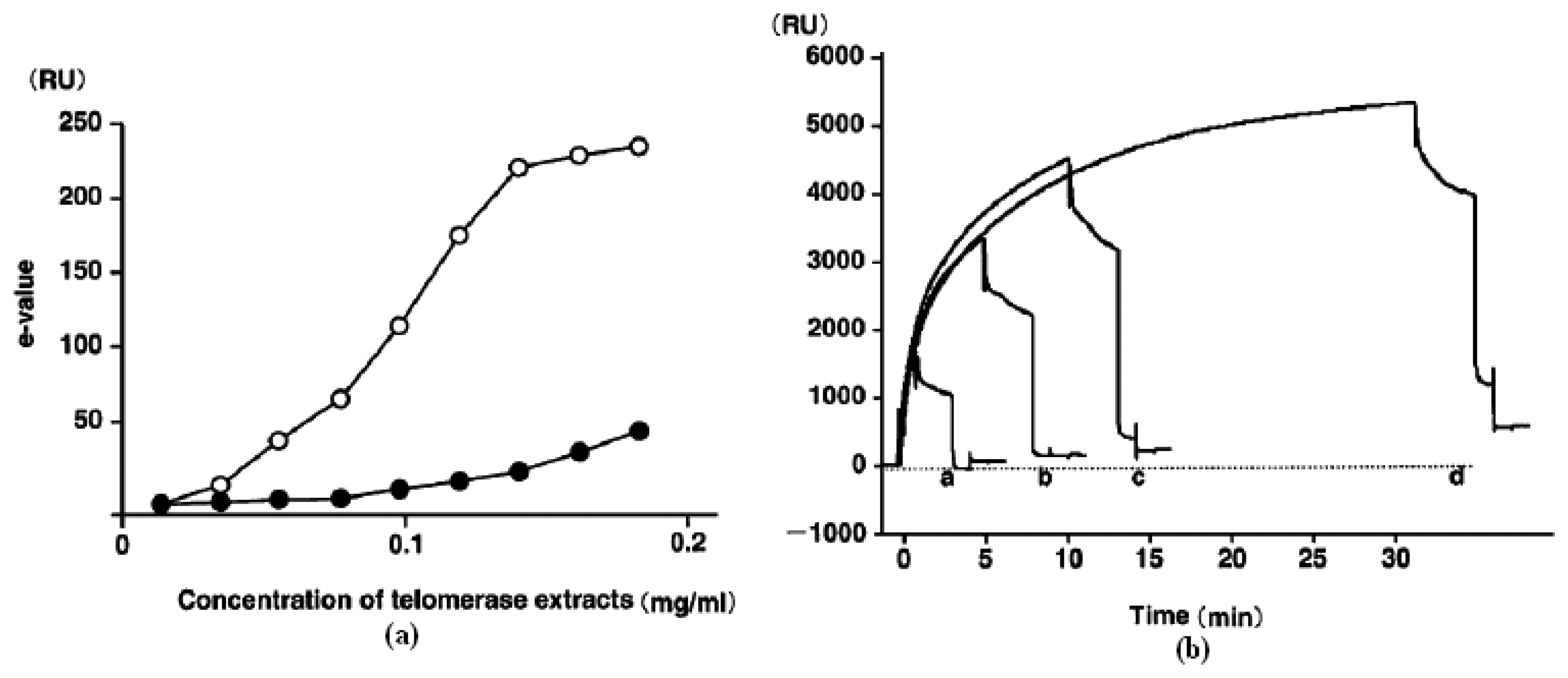
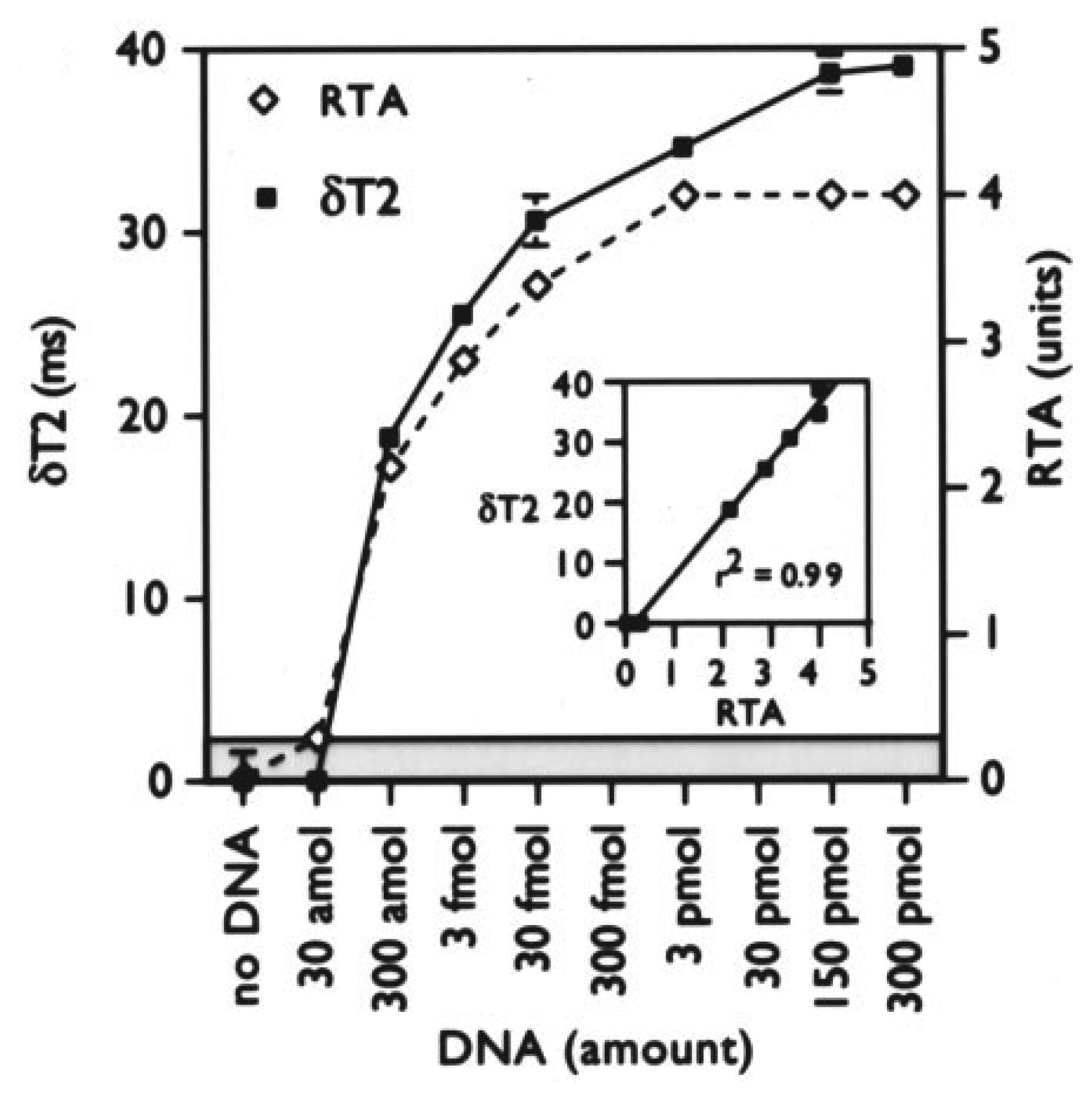
2. Label free detection of telomerase activity during hybridization step
3. Biocatalytic enzymes
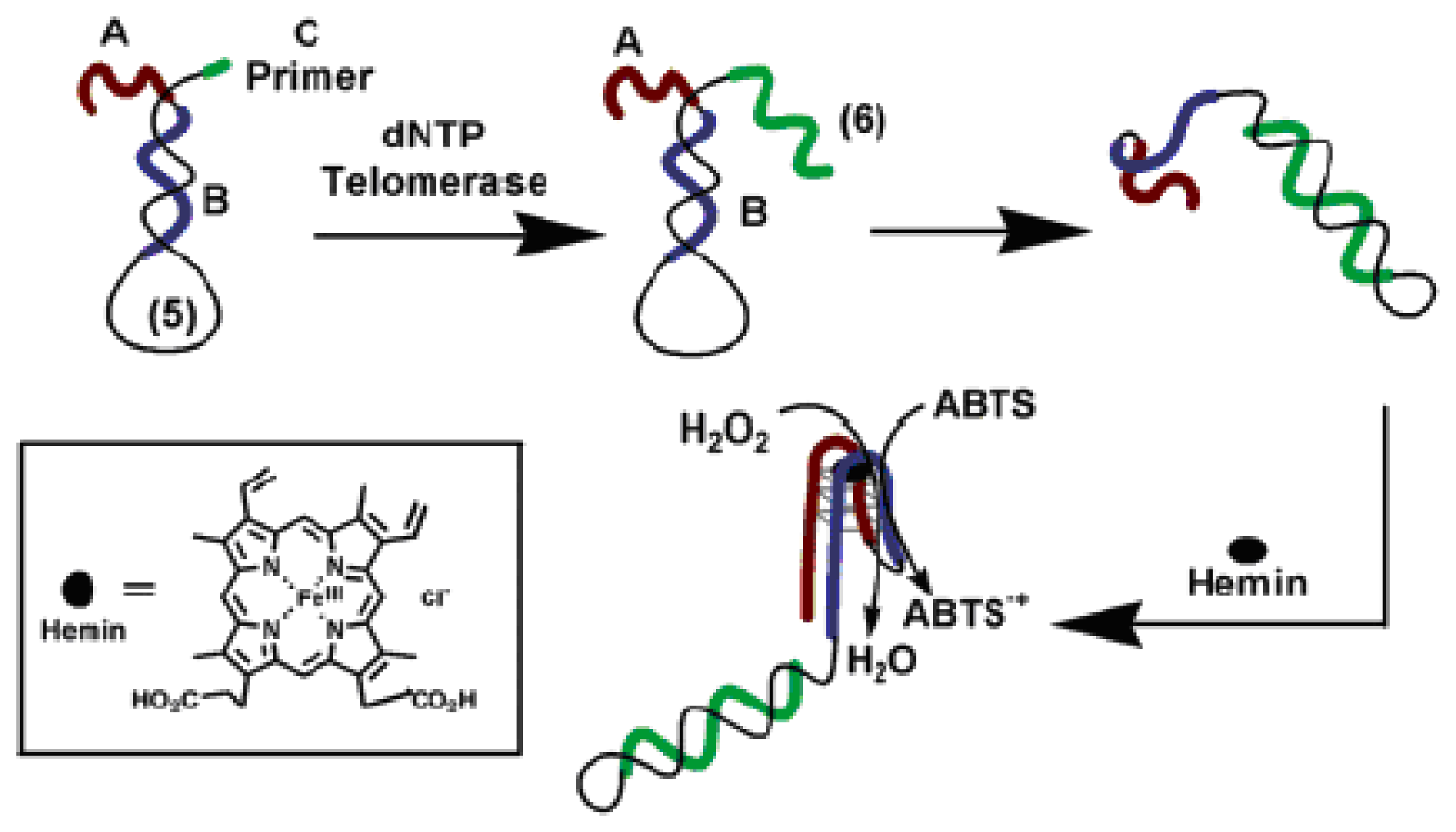
3.1. Catalytic beacons
3.2. DNAzyme
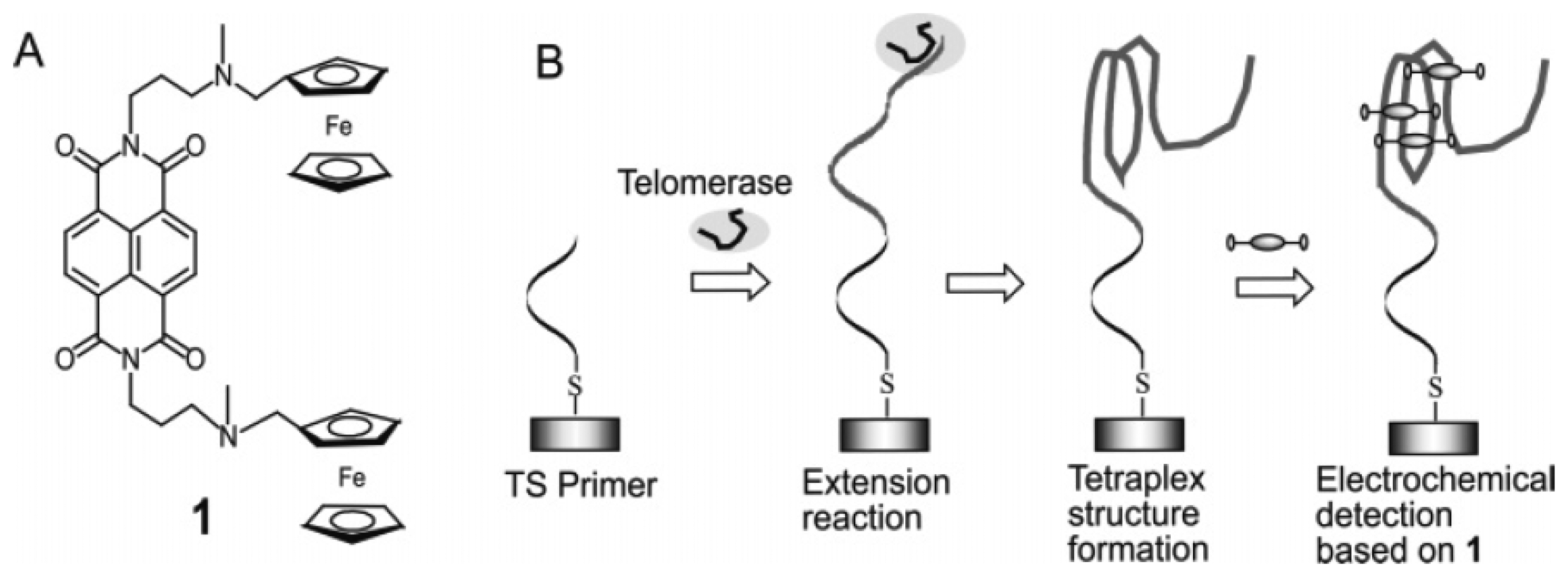
3.3. Ferrocenylnaphthalene diimide
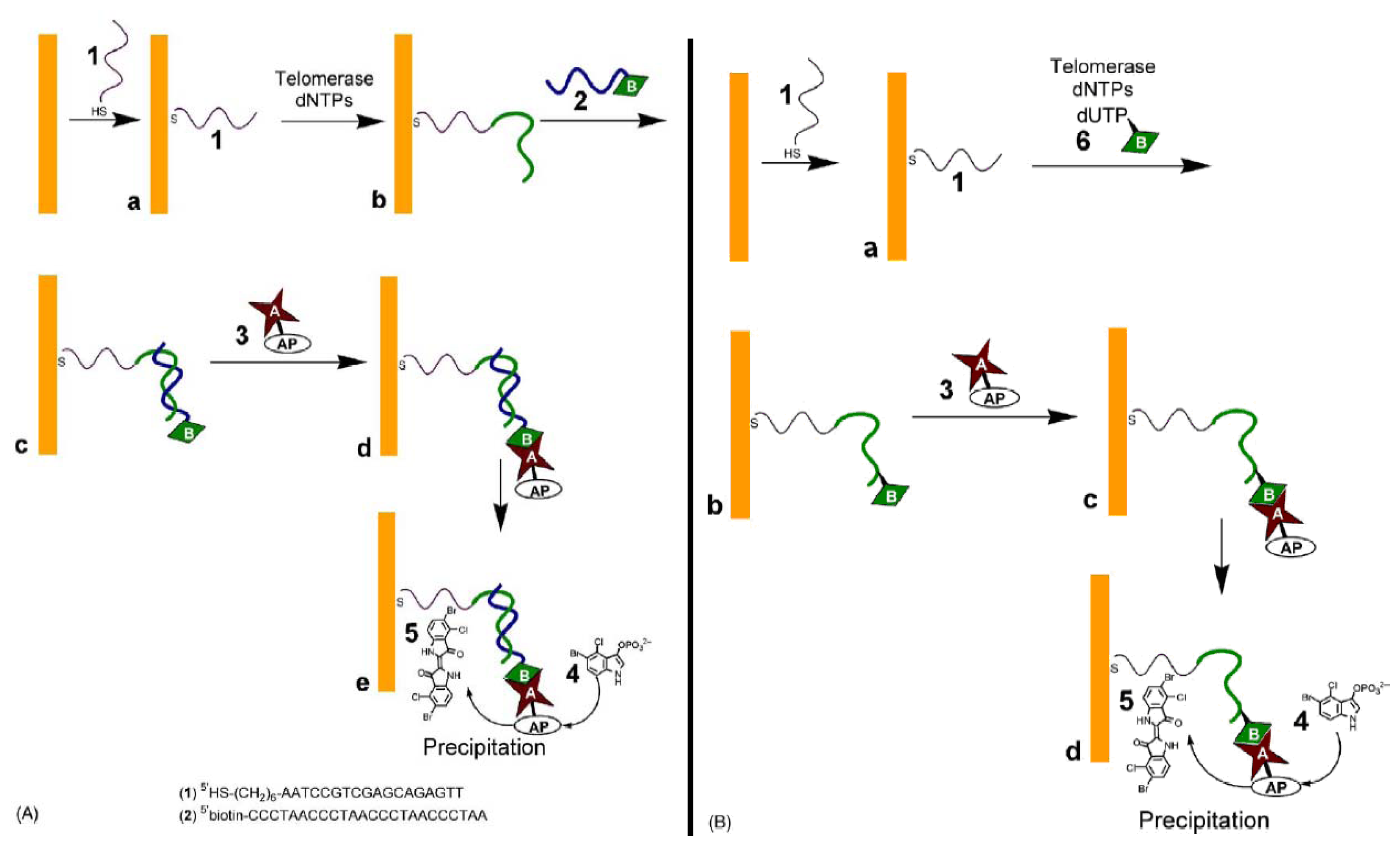
3.4. Avidin-alkaline Phosphatase conjugate
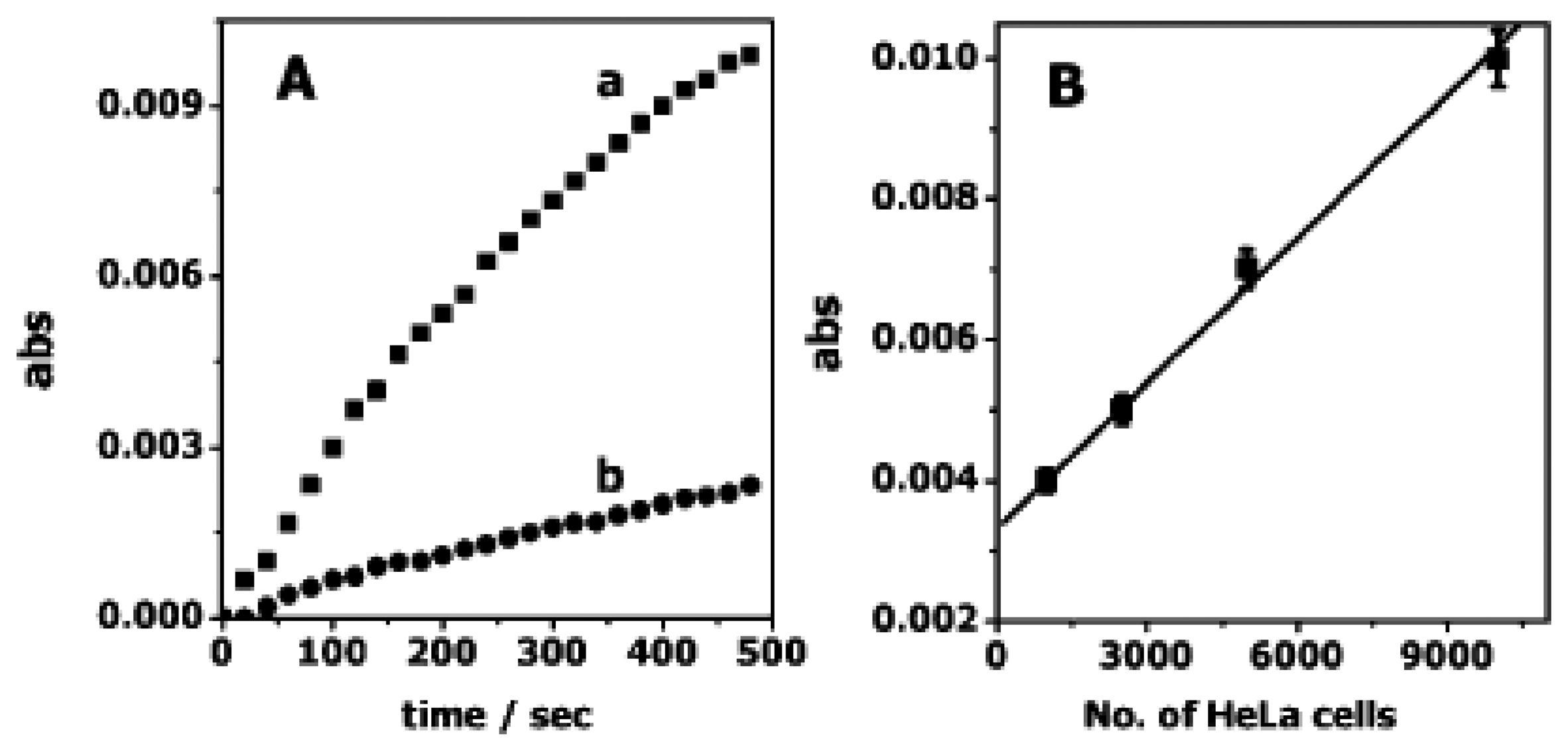
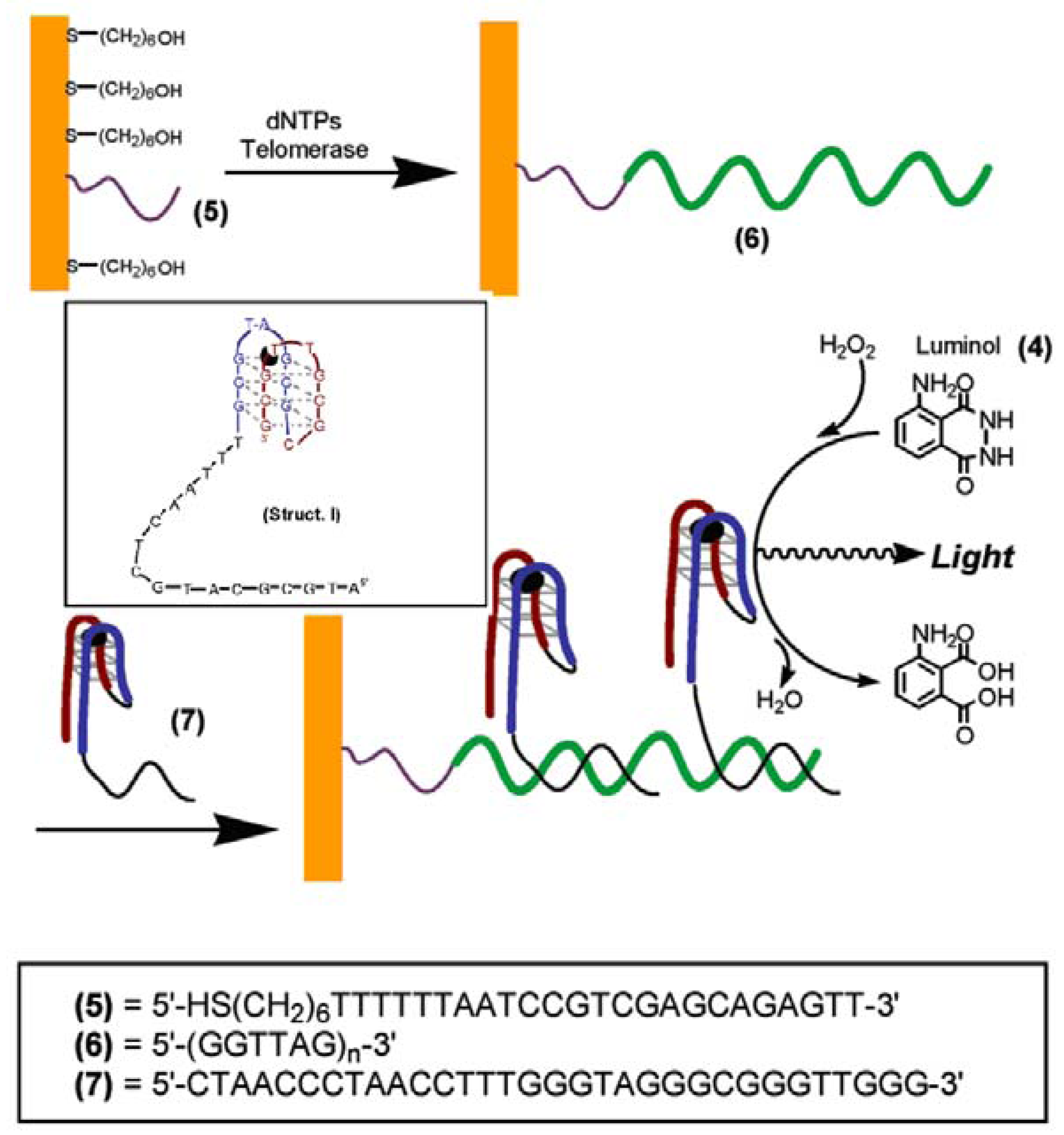
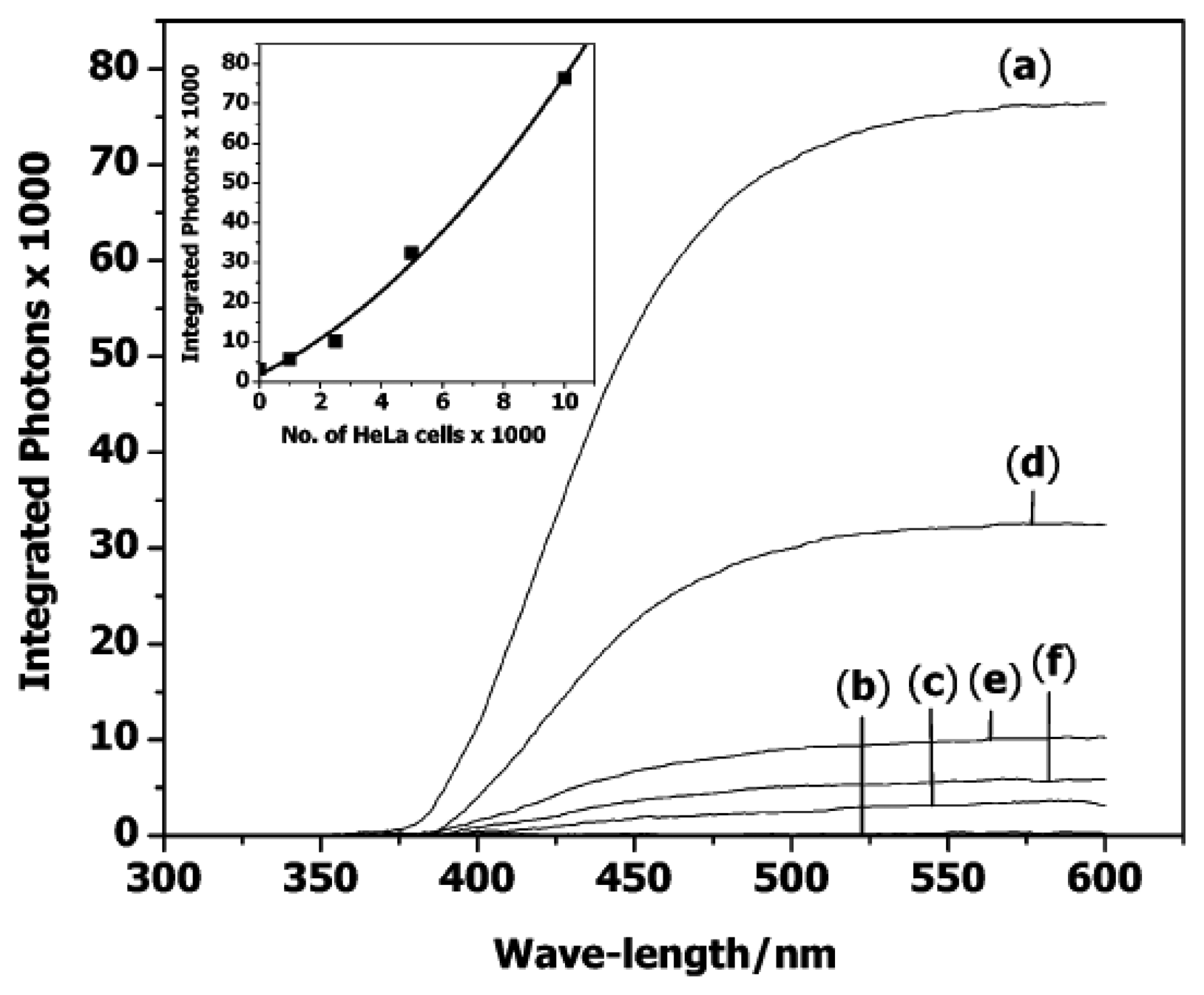
4. Nanoparticles labels for detection of telomerase activity
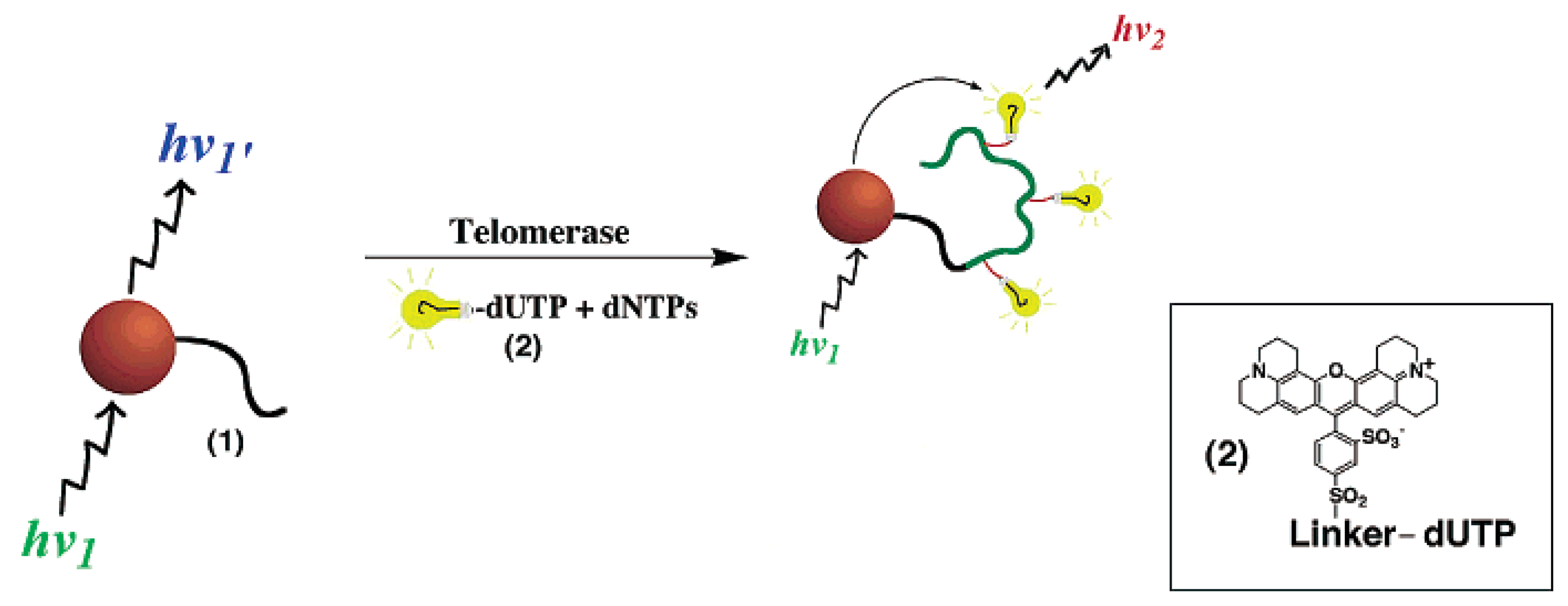
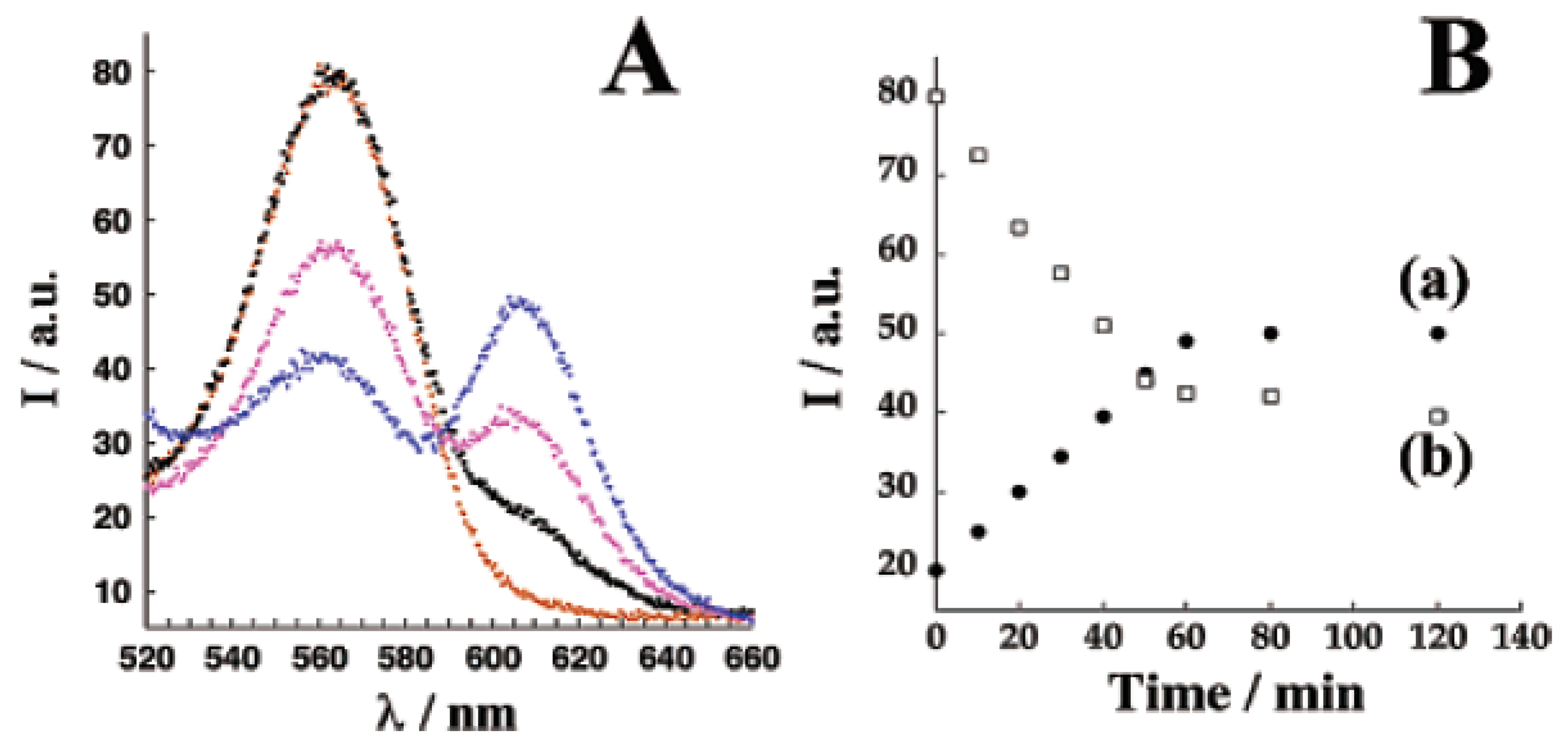
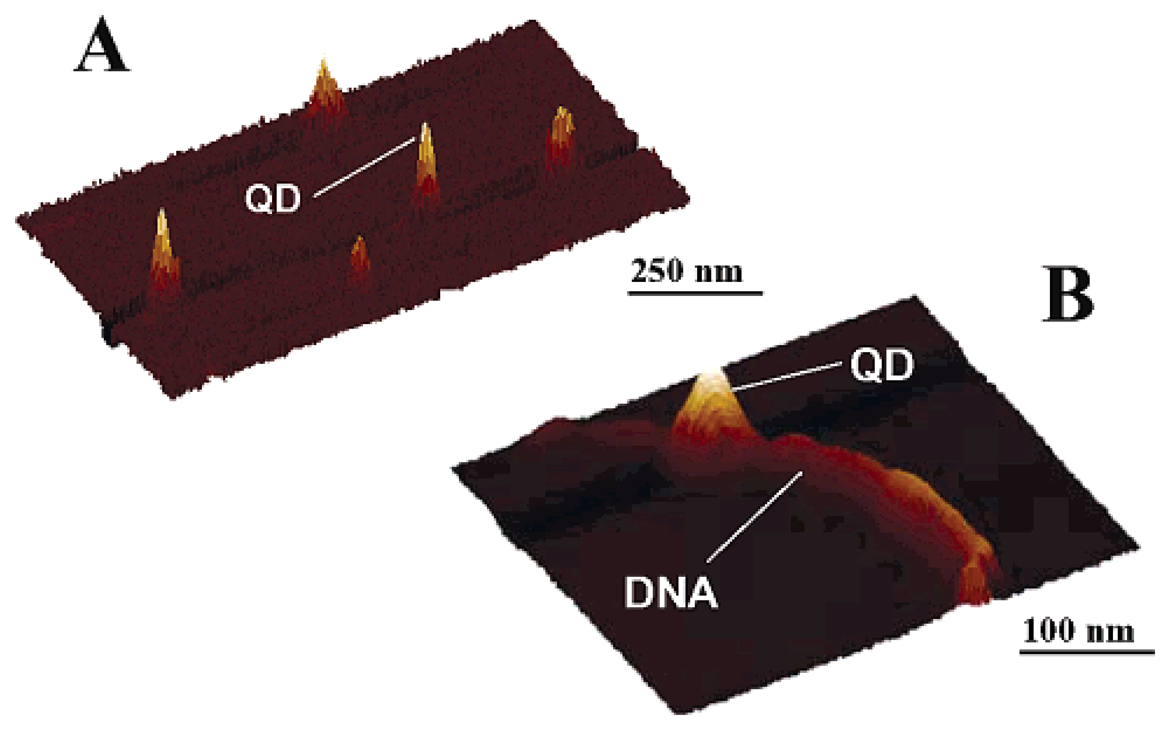
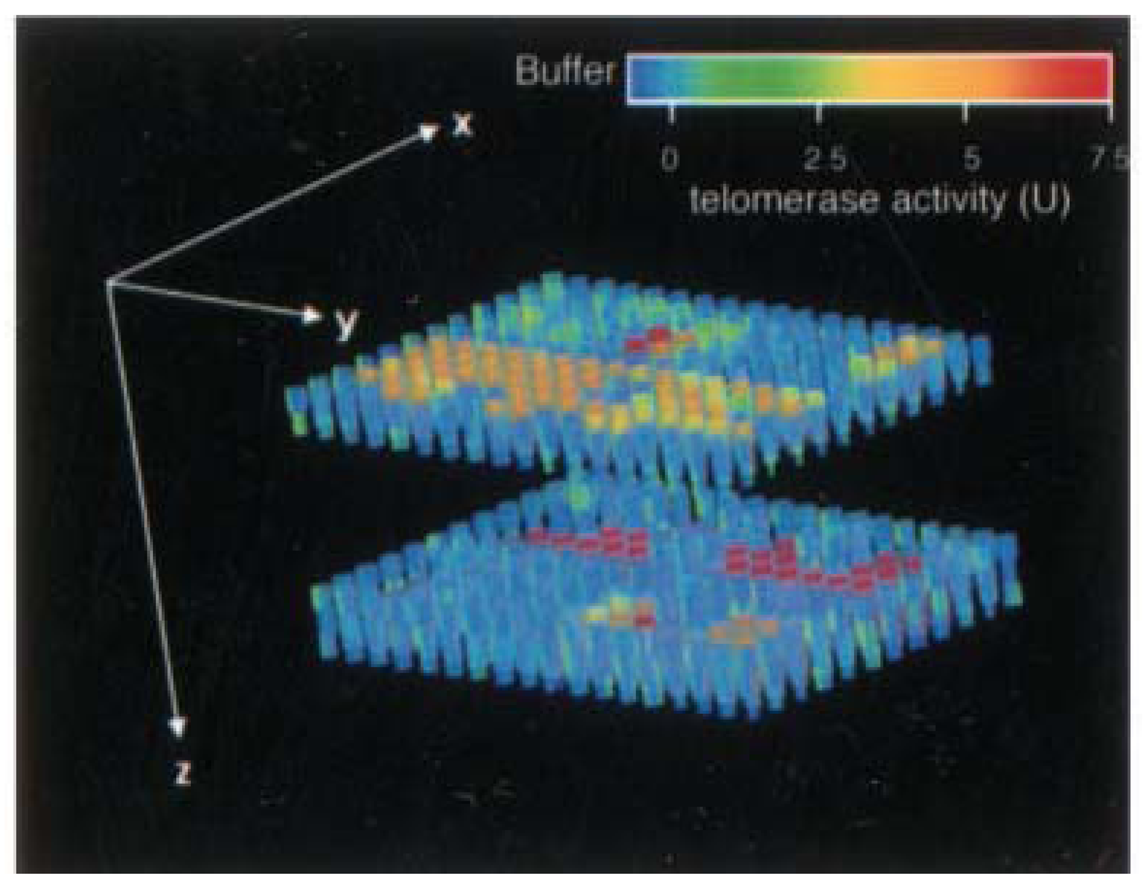
4.1. Semiconductor quantum dots (ODs)
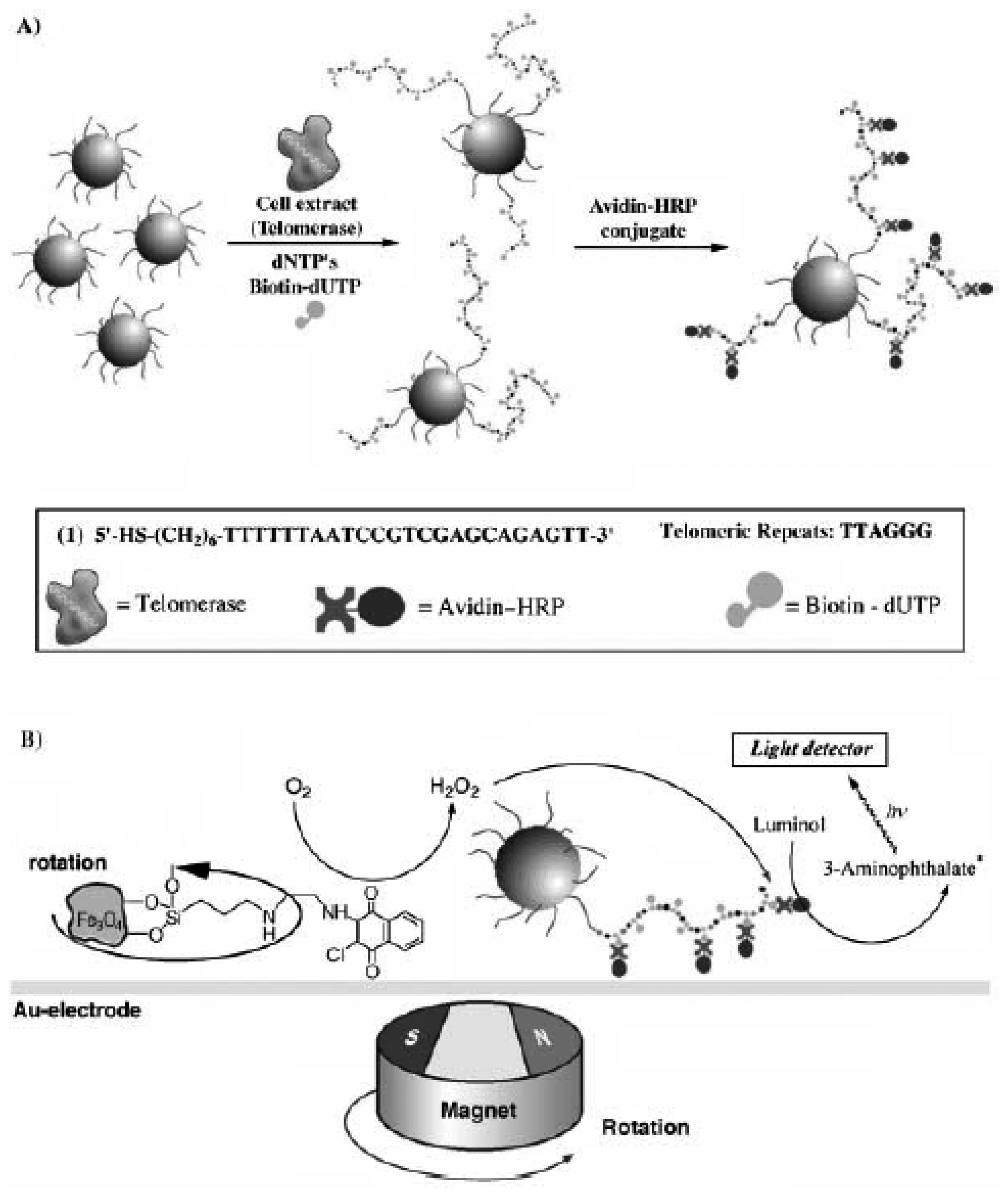
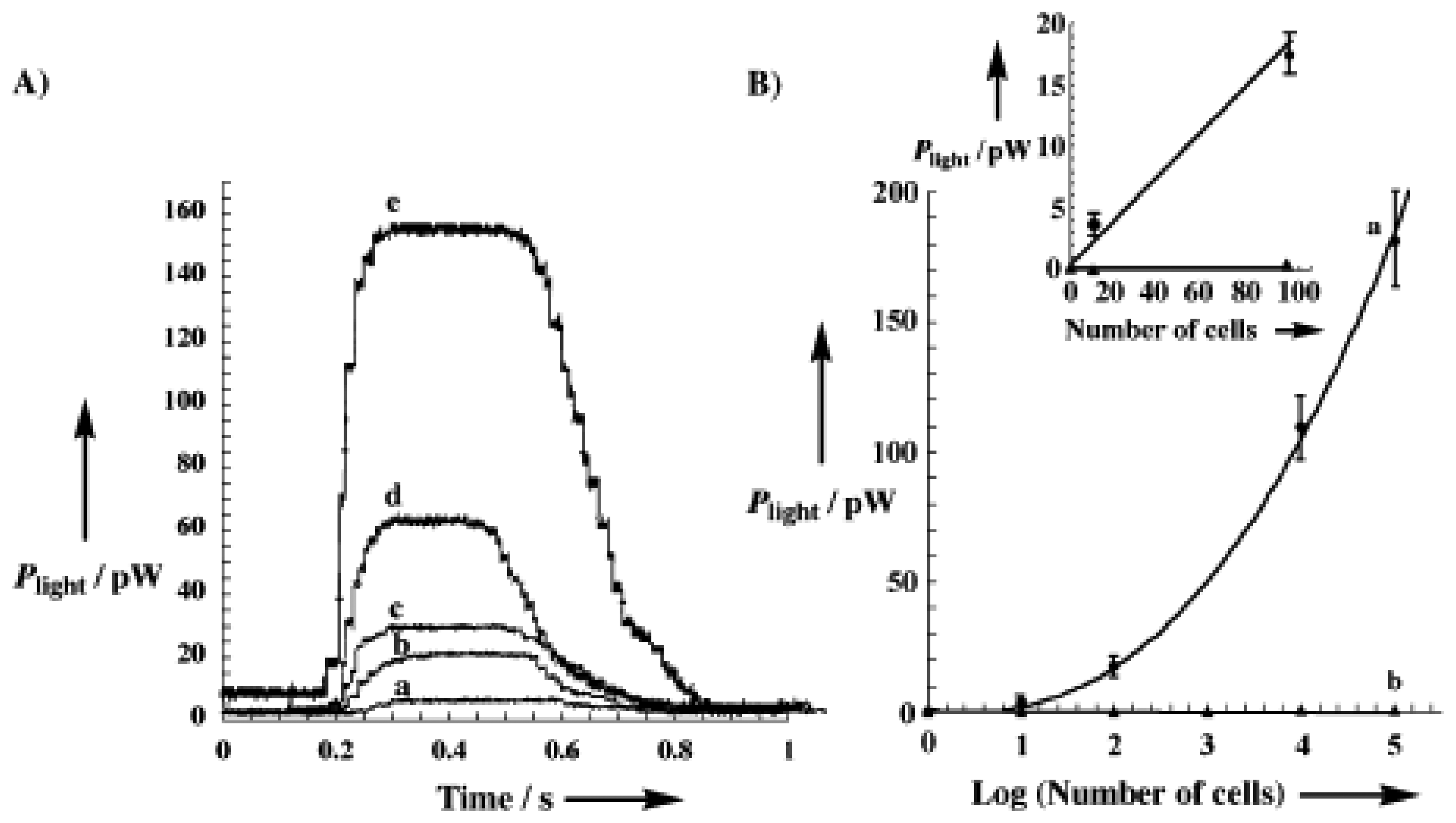
4.2. Magnetic Particles
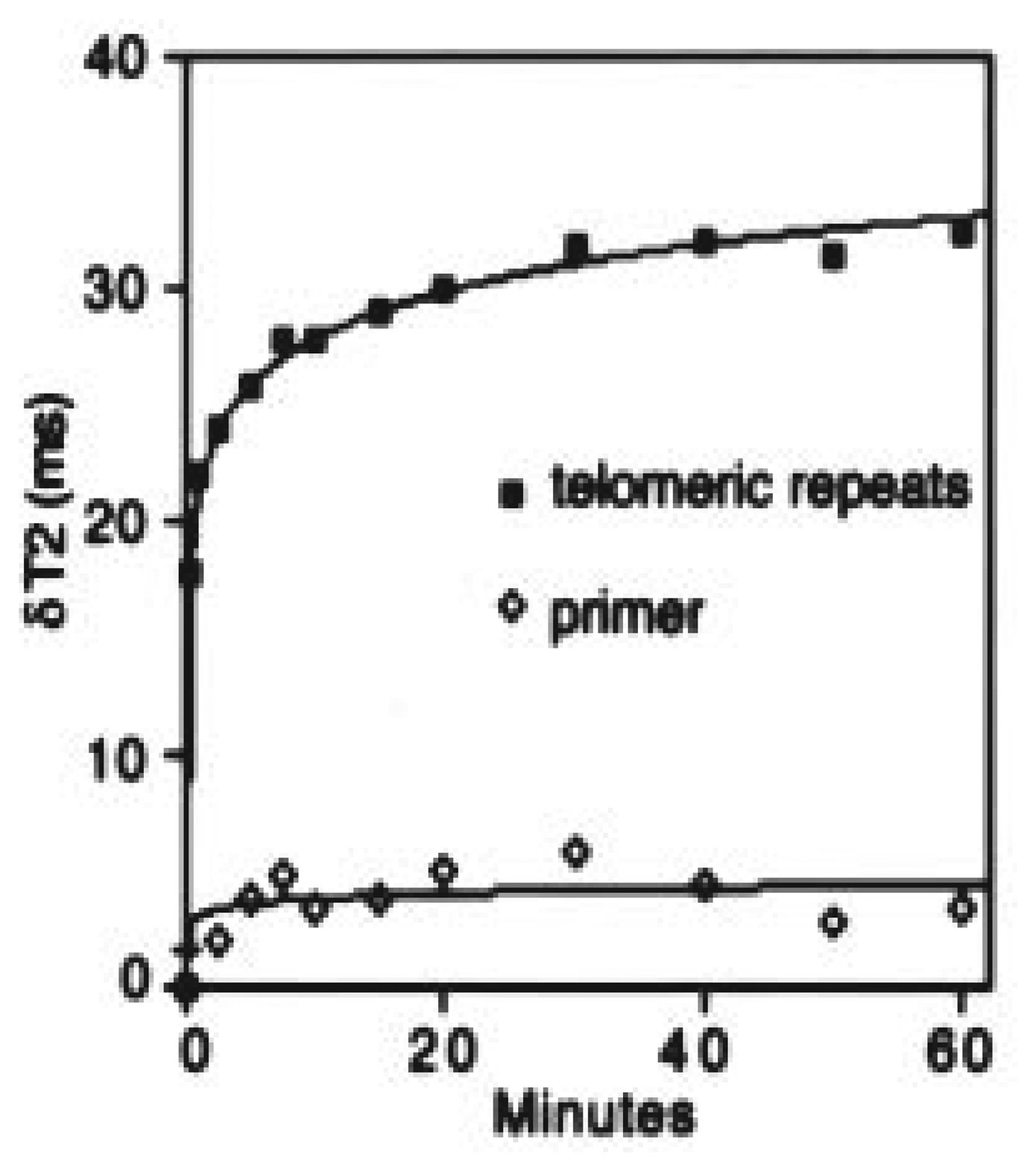
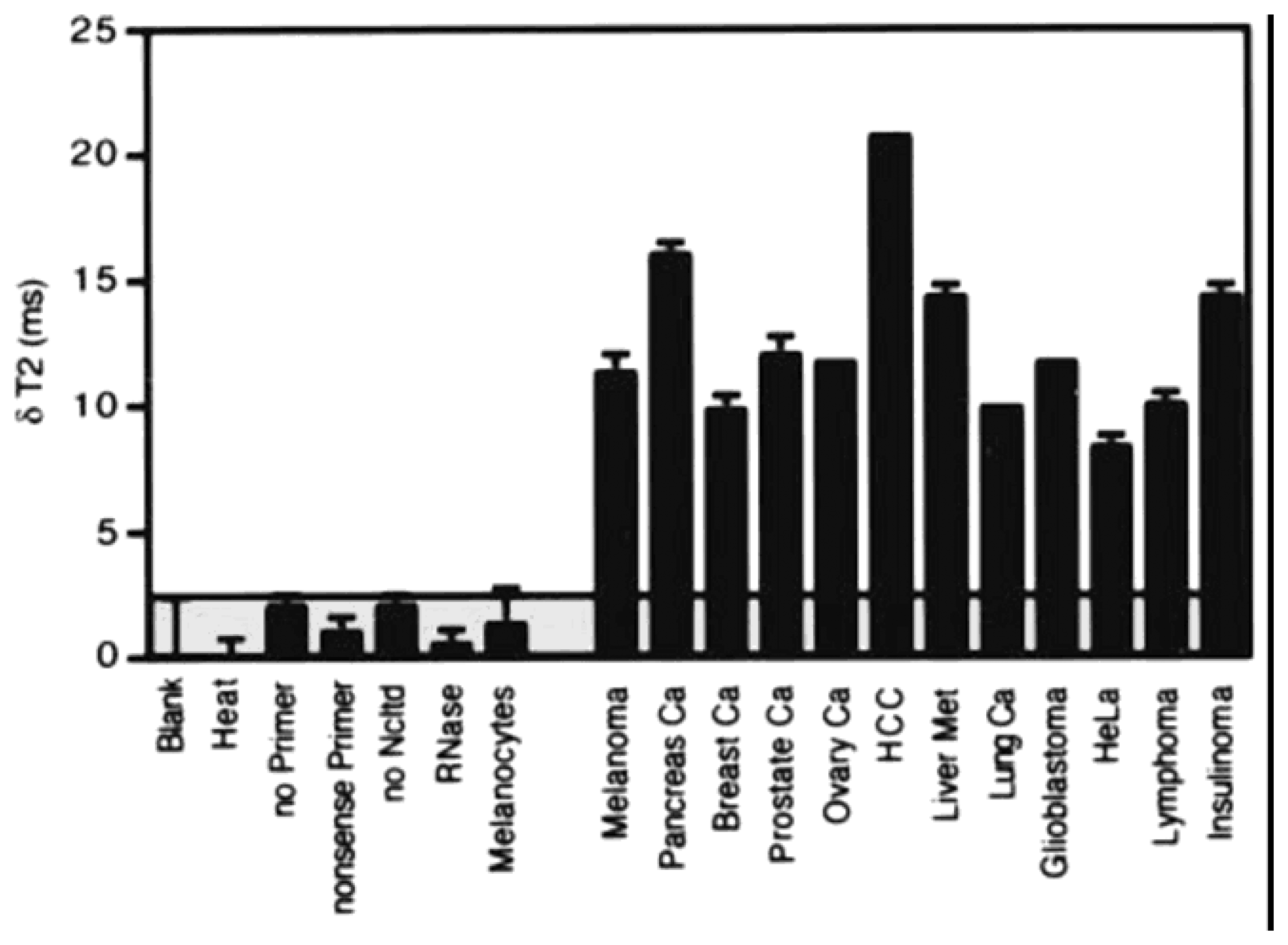
4.3. Magnetic nanosensor
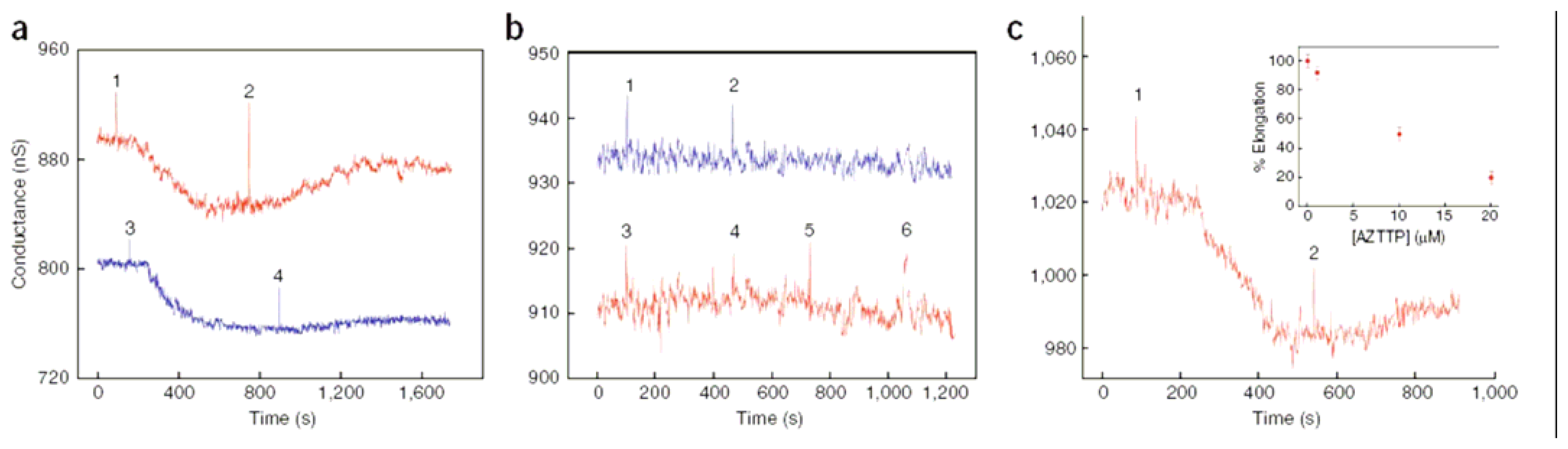
4.4. Nanowire sensor chip
5. Conclusions
Acknowledgments
References and Notes
- Hiyama, E.; Hiyama, K. Telomerase as Tumor Marker. Cancer Letters. 2003, 194, 221–233. [Google Scholar] [CrossRef]
- Greenlee, R.T.; Hill-Harmon, M. B.; Murray, T.; Thun, M. Cancer Statistics. Cancer J. Clin. 2001, 51, 15–36. [Google Scholar]
- Kosary, C. L. FIGO Stage, History, Histology, Histologic Grade, Age and Race as Prognostic Factors in Determining Survival for Cancer of the Female Gynecological System: an Analysis of Cancer of the Female Gynocological System: an Analysis of 1973-87 SEER Cases of Cancer of the Endometrium, Cervix, Ovary, Vulva, and Vagina. Semin. Surg. Oncol. 1994, 10, 31–46. [Google Scholar] [CrossRef]
- Nalwa, H. S.; Webster, T. Cancer Nanotechnology: Nanomaterials for Cancer Diagnosis and Therapy; 2007; American Scientific Publishers: California. [Google Scholar]
- Chaerkady, R.; Pandey, A. Quantitative Proteomics for Identification of Cancer Biomarkers. Proteomics Clinical Applications 2007, 1, 1080–1089. [Google Scholar] [CrossRef]
- Zimmermann, S.; Martens, U. M. Telomers and Telomerase as Targets for Cancer Therapy. Cellular and Molecular Life Sciences. 2007, 64, 906–921. [Google Scholar] [CrossRef]
- Hiyama, E.; Gollahon, L.; Katacka, T.; Kuroi, K.; Yokoyama, T.; Gazdar, A. F.; Hiyama, K.; Piatyszek, M. A.; Shay, J. W. Telomerase Activity in Human Breast Tumors. J. Natl. Cancer. Inst. 1996, 88, 116–122. [Google Scholar] [CrossRef]
- Specific Association of Human Telomerase Activity with Immortal Cells and Cancer. Science. 1994, 226, 2011–2015.
- Hiyama, E.; Hiyama, K.; Yokoyama, T.; Matsuura, Y.; Piatyszek, M. A.; Shay, J. W. Correlating Telomerase activity Levels with Human Neuroblastoma Outcomes. Nature Med. 1995, 1, 249–255. [Google Scholar] [CrossRef]
- Hiyama, K.; Hiyama, E.; Ishioka, S.; Yamekido, M.; Inai, K.; Gazdar, A. F.; Piatyszek, M. A.; Shay, J. W. Telomerase Activity in Small-Cell and Non-Small-Cell Lung Cancers. J. Natl. Cancer Inst. 1995, 87, 895–902. [Google Scholar] [CrossRef]
- Hiyama, E.; Yokoyama, T.; Tatsumoto, N.; Hiyama, K.; Imamura, Y.; Murakami, Y.; Kodama, T.; Piatyszek, M. A.; Shay, J. W.; Matsuura, Y. Telomerase Activity in Gastric Cancer. Cancer Res. 1995, 55, 3258–3262, PubMed 7614459. [Google Scholar]
- Hayflick, L.; Moorhead, P. S. The Serial Cultivation of Human Diploid Cell Strains. Exp. Cell. Res. 1961, 25, 585–621. [Google Scholar] [CrossRef]
- Olevnikov, A. M. A. Theory of Marginotomy: The Incomplete Copying of Template Margin in Enzymic Synthesis of Polynucleotides and Biological Significance of the Phenomenon. J. Theor. Biol. 1973, 41, 181–190. [Google Scholar] [CrossRef]
- Greider, C. W. Telomere Length Regulation. Ann. Rev. Biochem. 1996, 65, 337–365. [Google Scholar]
- Saldanha, S. N.; Andrews, L. G.; Tellefsbol, T. O. Analysis of Telomerase Activity and Detection of its Catalytic Subunit, hTERT. Analytical Biochemistry. 2003, 315, 1–21. [Google Scholar] [CrossRef]
- Potter, J. D. Morphogens, Morphostats, Microarchitecture and Malignancy. Nat. Rev. Cancer. 2007, 7, 464–474. [Google Scholar] [CrossRef]
- Sack, G. H. Human Cell Transformation by Simian Virus 40. In Vitro. 1981, 17, 1–19. [Google Scholar] [CrossRef]
- Di Paulo, J. A. Asbestos-Associated Chromosomal Changes in Human. J. Natl. Cancer Inst. 1983, 70, 3–8, PubMed 6571918. [Google Scholar]
- Chang, S. E. In Vitro Transformation of Human Epithelial Cells. Biochim. Biophys. Acta. 1986, 823, 161–194, PubMed 2423124. [Google Scholar]
- Greider, C. W.; Blackburn, E. H. Identification of a Specific Telomere Terminal Transferase Activity in Tetrahymena Extracts. Cell. 1985, 43, 405–413. [Google Scholar] [CrossRef]
- Greider, C. W.; Blackburn, E. H. A. Telomeric Sequence in the RNA of Tetrahymena Telomerase Required for Telomere Repeat Synthesis. Nature. 1989, 337, 331–337. [Google Scholar] [CrossRef]
- Zahler, A.M.; Prescott, D. M. Telomere Terminal Transferase Activity in the Hypotrichous Ciliate Oxytricha Nova and a Model for Replication of the Ends of Linear DNA molecules. Nucleic Acids Res. 1988, 16, 6953–6872. [Google Scholar] [CrossRef]
- Shippen-Lentz, D.; Blackburn, E. H. Telomere Terminal Transferase Activity for Euplotes Crassus Adds Large Numbers of TTTTGGGG Repeats only Telomeric Primers. Mol. Cell. Biol. 1989, 9, 2761–2764, PubMed 2474761. [Google Scholar]
- Autexier, C.; Greider, C. W. Telomerase and Cancer: Revisiting the Telomere Hypothesis. Trends Biochem. 1998, 21, 387–391. [Google Scholar] [CrossRef]
- Counter, C. M.; Hirte, H. W.; Bacchetti, S.; Harley, C. B. Telomerase Activity in Human Ovarian Carcinoma. Proc. Natl. Acad. Sci. U. S. A. 1994, 91, 2900–2904. [Google Scholar] [CrossRef]
- Counter, C. M.; Botelho, F. M.; Wang, P.; Harley, C. B.; Bacchetti, S. Stabilization of Short Telomers and Telomerase Activity Accompany Immortalization of Epstein-Barr Virus-Transformed Human B Lymphocytes. J. Virol. 1994, 68, 3410–3414, PubMed 8151802. [Google Scholar]
- Fajkus, J. Detection of Telomerase Activity by the TRAP Assay and its Variants and Alternatives. Clinica Chimica Acta. 2006, 371, 25–31. [Google Scholar] [CrossRef]
- Fajkus, J.; Koppova, K.; Kunicka, Z. Dual-Color Real-Time Telomeric Repeat Amplification Protocol. Biotechniques. 2003, 23, 912–914. [Google Scholar]
- Aldous, W. K.; Grabill, N. R. A Fluorescent Method for Detection of Telomerase Activity. Diagn. Mol. Pathol. 1997, 6, 102–110. [Google Scholar] [CrossRef]
- Savoysky, E.; Akamatsu, K.; Tsuchiya, M.; Yamazaki, T. Detection of Telomerase Activity by Combination of TRAP Method and Scintillation Proximity Assay (SPA). Nucleic Acids Res. 1996, 24, 1175–1176. [Google Scholar] [CrossRef]
- Ohyashiki, K.; Ohyashiki, J. H.; Nishimaki, J.; et al. Cytological Detection of Telomerase Activity Using an in situ Telomeric Repeat Amplification Protocol Assay. Cancer Res. 1997, 57, 2100–2103, PubMed 9187102. [Google Scholar]
- Elmore, L. W.; Forsythe, H. L.; Ferreira-Gonzalez, A.; Garrett, C. T.; Clark, G. M.; Holt, S. E. Real-Time Quantitative Analysis of Telomerase Activity in Breast Tumor Specimens Using a Highly Specific and Sensitive Fluorescent-Based Assay. Diagn. Mo.l Pathol. 2002, 11, 177–185. [Google Scholar] [CrossRef]
- Mayfield, M.P.; Shah, T.; Flannigan, G.M.; Hamilton Stewart, P. A.; Bibby, M. C. Telomerase Activity in Malignant and Benign Bladder Conditions. Int. J. Mol. Med. 1998, 1, 835–840, PubMed 9852304. [Google Scholar]
- Hoos, A.; Hepp, H. H.; Kaul, S.; Ahlert, T. Telomerase Activity Correlates With Tumor Aggressiveness and Teflects Therapy Effect in Breast Cancer. Int. J. Cancer. 1998, 79, 8–12. [Google Scholar] [CrossRef]
- Thurnher, D.; Knerer, B. Non-Radioactive Semiquantitative Testing for the Expression Levels of Telomerase Activity in Head and Neck Squamous cell Carcinomas may be Indicative for Biological Tumour Behaviour. Acta Otolaryngol. 1998, 118, 423–427. [Google Scholar] [CrossRef]
- Hirose, M.; Abe-Hashimoto, J.; Ogura, K.; Tahara, H. A Rapid, Useful and Quantitative Method to Measure Telomerase Activity by Hybridization Protection Assay Connected with a Telomeric Repeat Amplification Protocol. J. Cancer Res. Clin. Oncol. 1997, 123, 337–344, PubMed 9222300. [Google Scholar]
- Takata, M.; Kerman, K.; Nagatani, N.; Konaka, H.; Namiki, M.; Tamiya, E. Label-Free Bioelectronic Immunoassay for the Detection of Human Telomerase Reverse Transcriptase in Urine. Journal of Electroanalytical Chemistry. 2006, 596, 109–116. [Google Scholar] [CrossRef]
- Nilsson, P.; Persson, B.; Uhlen, M.; Nygren, P.A. Real-Time Monitoring of DNA Manipulations Using Biosensor Technology. Analytical Biochemistry. 1995, 224, 400–408, PubMed 7710099. [Google Scholar]
- Schmidt, P. M.; Matthes, E.; Scheller, F. W. Real-Time Determination of Telomerase Activity in Cell Extracts Using an Optical Biosensor. Biol. Chem. 2002, 383, 1659–1666. [Google Scholar] [CrossRef]
- Matthes, E.; Lehmann, C. Telomerase Protein Rather than its RNA in the Target of Phosphorothioate-Modified Oligonucleotides. Nucleic Acids Res. 1999, 27, 1152–1158. [Google Scholar] [CrossRef]
- Schmidt, P. M.; Lehmann, C.; Matthes, E.; Bier, F. F. Detection of Activity of Telomerase in Tumor Cells Using Fiber Optical Biosensors. Biosensors & Bioelectronics. 2002, 17, 1081–1087. [Google Scholar] [CrossRef]
- Maesawa, C.; Inaba, T.; Sato, H.; Injima, S.; Ishida, K.; Terashima, M.; Sato, R.; Suzuki, M.; Yashima, A.; Ogasawara, S.; Oikawa, H.; Soto, N.; Saito, K.; Masuda, T. A Rapid Biosensor Chip Assay for Measuring the Telomerase Activity Using Surface Plasmon Resonance. Nucleic Acids Research. 2003, 31, 1–6. [Google Scholar] [CrossRef]
- Xiao, Y.; Pavlov, V.; Niazov, T.; Dishon, A.; Kotler, M.; Willher, I. Catalytic Beacons for the Detection of DNA and Telomerase Activity. J. Am. Chem. Soc. 2004, 126, 7430–7431, PubMed 15198576. [Google Scholar]
- Pavlov, V.; Xiao, Y.; Ron, G.; Dishon, A.; Kotler, M.; Willner, I. Amplified Chemiluminescence Surface Detection of DNA and Telomerase Activity Using Catalytic Nucleic Acid Labels. Anal. Chem. 2004, 76, 2152–2156. [Google Scholar] [CrossRef]
- Soto, S.; Kondo, H.; Nojima, T.; Takenaka, S. Electrochemical Telomerase Assay with Ferrocenylnaphthalene Diimide as a Tetraplex DNA-Specific Binder. Anal Chem. 2005, 77, 7304–7309. [Google Scholar] [CrossRef]
- Pavlov, V.; Willner, I.; Dishon, A.; Kotler, M. Amplified Detection of Telomerase Activity Using Electrochemical and Quartz Crystal Microbalance Measurements. Biosensors & Bioelectronics. 2004, 20, 1011–1021. [Google Scholar] [CrossRef]
- Paolsky, F.; Gill, R.; Weizmann, Y.; Mokari, T.; Banin, U.; Willner, I. Lighting-Up the Dynamics of Telomerization and DNA Replication by CdSe-ZnS Quantum Dots. J. Am. Chem. Soc. 2003, 125, 13918–13919. [Google Scholar] [CrossRef]
- Li, Y. Cds Nanocrystal Induced Chemiluminescence: Reaction Mechanism and Applications. Nanotechnology. 2007, 18, 1–8. [Google Scholar]
- Weizmann, Y.; Patolsky, F.; Katz, E.; Willner, I. Amplified Telomerase Analysis by Using Rotating Magnetic Particles: The Rapid and Sensitive Detection of Cancer Cells. ChemBioChem. 2004, 5, 943–948. [Google Scholar] [CrossRef]
- Grimm, J.; Perez, J. M.; Josephson, L.; Weissleder, R. Novel Nanosensors for Rapid Analysis of Telomerase Activity. Cancer Research. 2004, 64, 639–643. [Google Scholar] [CrossRef]
- Zheng, G.; Patolsky, F.; Cui, Y. Multiplexed Electrical Detection of Cancer Markers with Nanowire Sensor Arrays. Nature Biotechnology. 2005, 23, 1294–1301. [Google Scholar] [CrossRef]

© 2008 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
Kulla, E.; Katz, E. Biosensor Techniques Used for Determination of Telomerase Activity in Cancer Cells. Sensors 2008, 8, 347-369. https://doi.org/10.3390/s8010347
Kulla E, Katz E. Biosensor Techniques Used for Determination of Telomerase Activity in Cancer Cells. Sensors. 2008; 8(1):347-369. https://doi.org/10.3390/s8010347
Chicago/Turabian StyleKulla, Eliona, and Evgeny Katz. 2008. "Biosensor Techniques Used for Determination of Telomerase Activity in Cancer Cells" Sensors 8, no. 1: 347-369. https://doi.org/10.3390/s8010347
APA StyleKulla, E., & Katz, E. (2008). Biosensor Techniques Used for Determination of Telomerase Activity in Cancer Cells. Sensors, 8(1), 347-369. https://doi.org/10.3390/s8010347



