Electronic Nose Based on Metal Oxide Semiconductor Sensors as an Alternative Technique for the Spoilage Classification of Red Meat
Abstract
:1. Introduction
2. Experimental
2.1. Sample preparation and sampling
2.2. Microbiological population enumeration
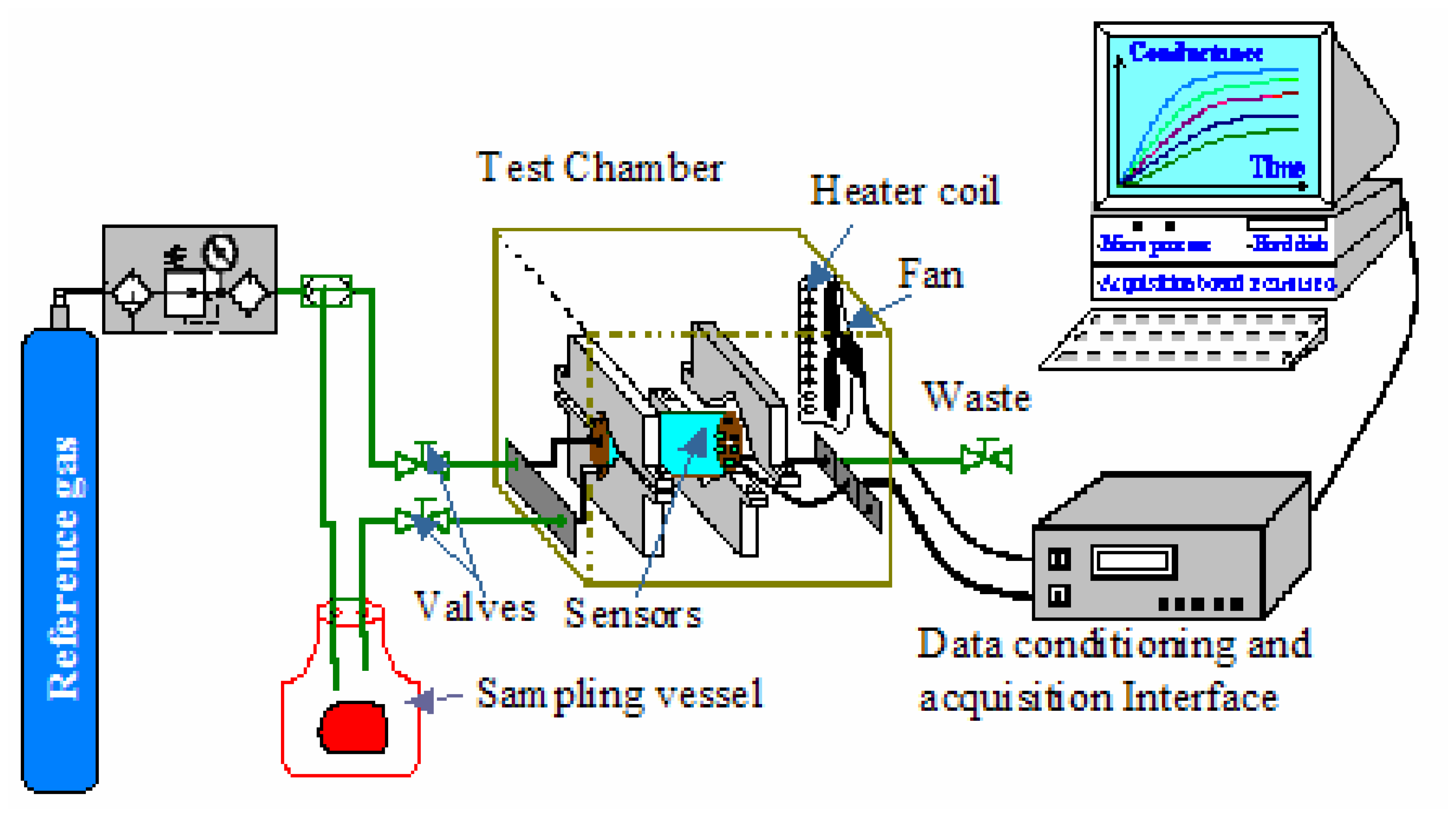
2.3. Electronic nose system
2.3.1. Feature extraction and pre-processing
- G0: the initial conductance of a sensor calculated as the average value of its conductance during the first 15 minutes of a measurement (using the definition in Eq. 1).
- Gs: the steady-state conductance calculated as the average value of its conductance during the last 5 minutes of a measurement.
- dG/dt: the dynamic slope of the conductance calculated between minute 15 and 35 of a measurement. This corresponds to a phase where a fast increase of sensor conductance is observed.
- A: the area below the conductance curve in a time interval defined between 15 and 40 min of a measurement. This area is estimated by the trapeze method.
2.3.2. Data analysis
2.3.2.1. Principal component analysis (PCA)
2.3.2.2 Partial least squares regression (PLS)
2.3.2.3. Support vector machines (SVM)
3. Results and Discussion
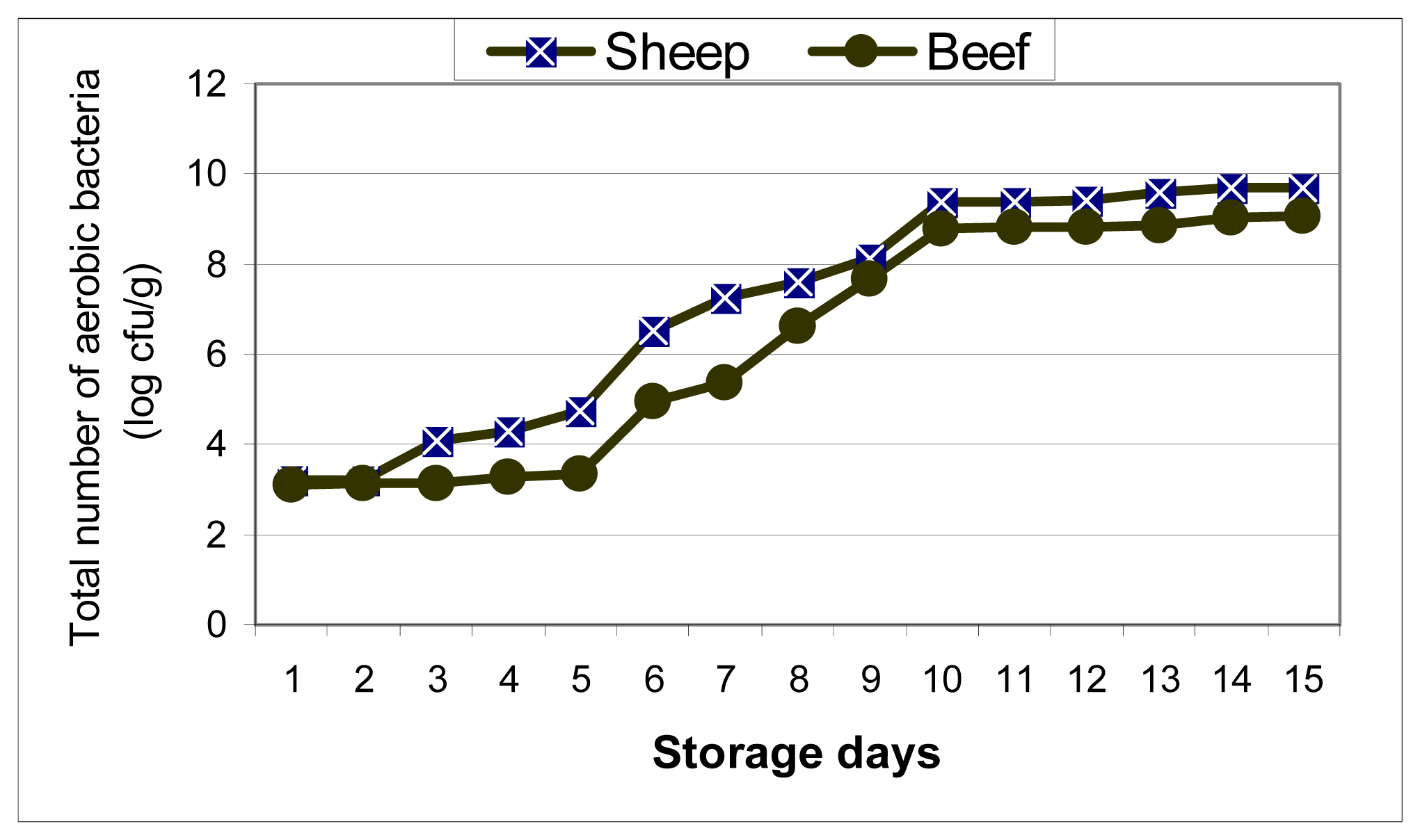
3.1. Bacterial analysis
3.2. Electronic nose analysis
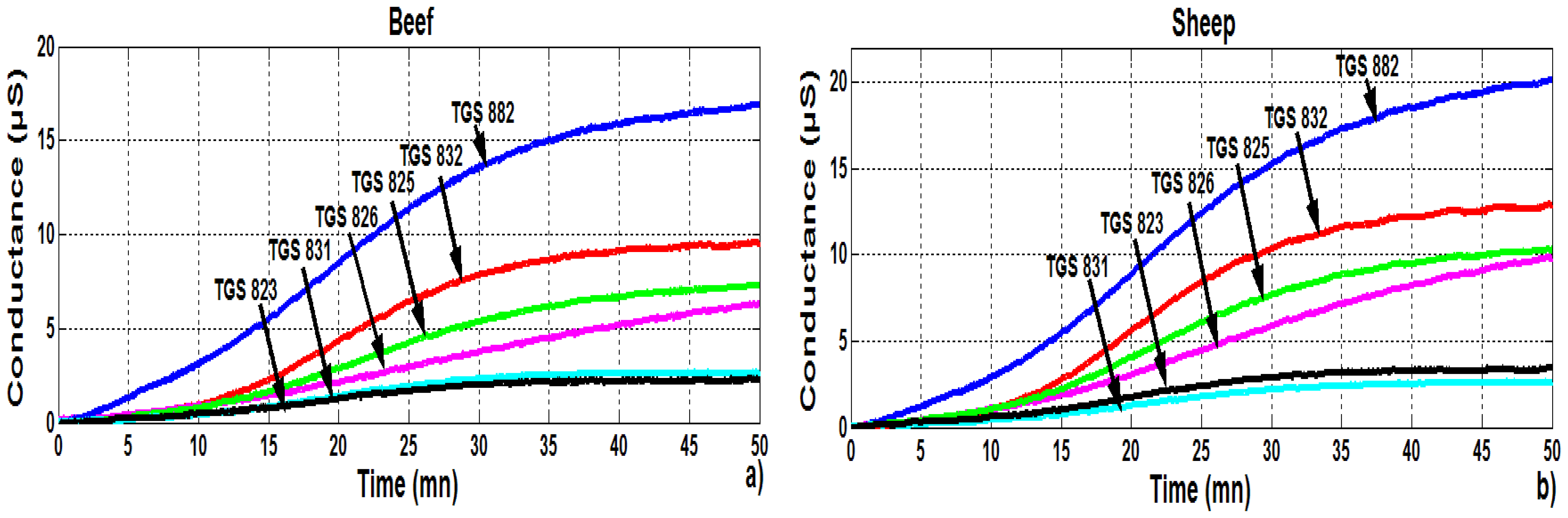
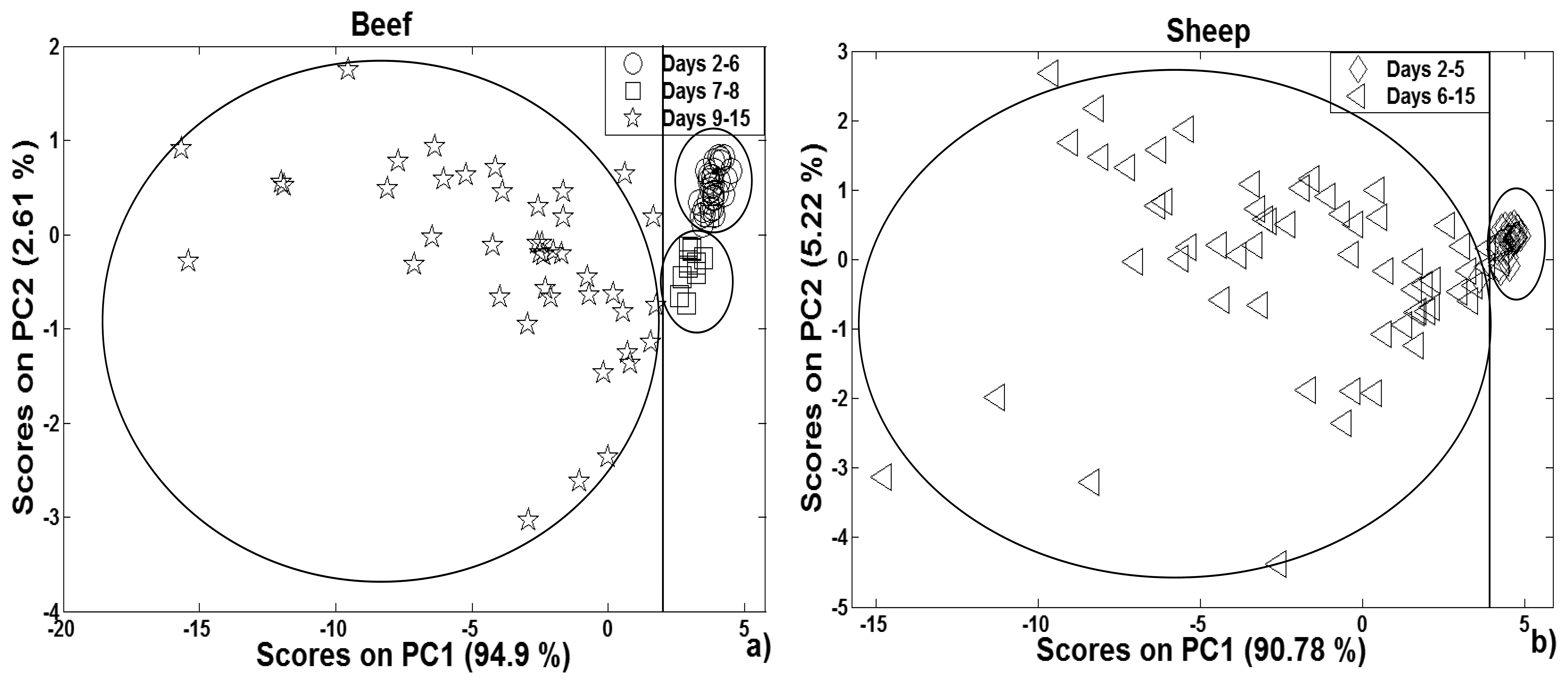
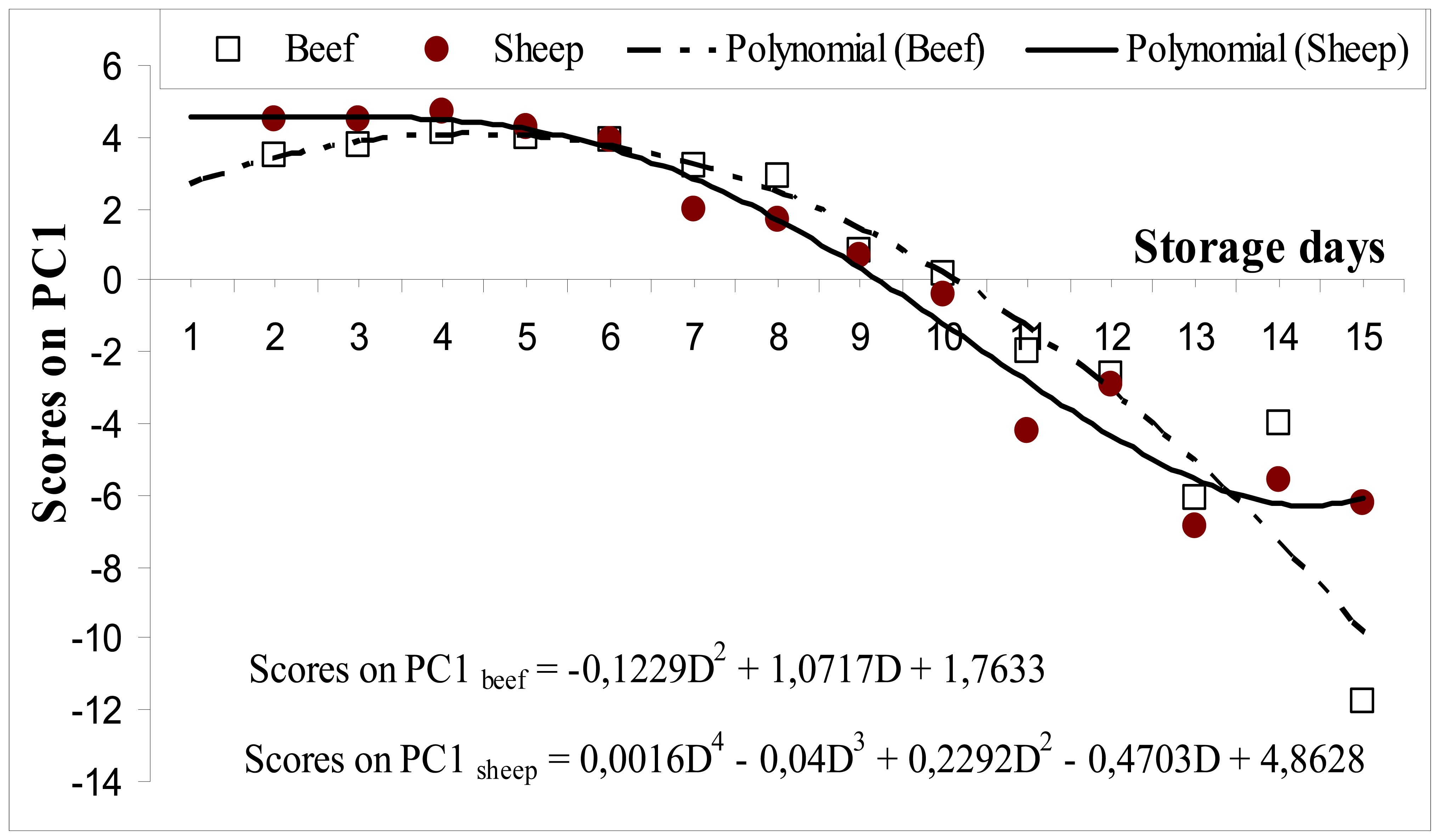
3.2.1. PCA analysis
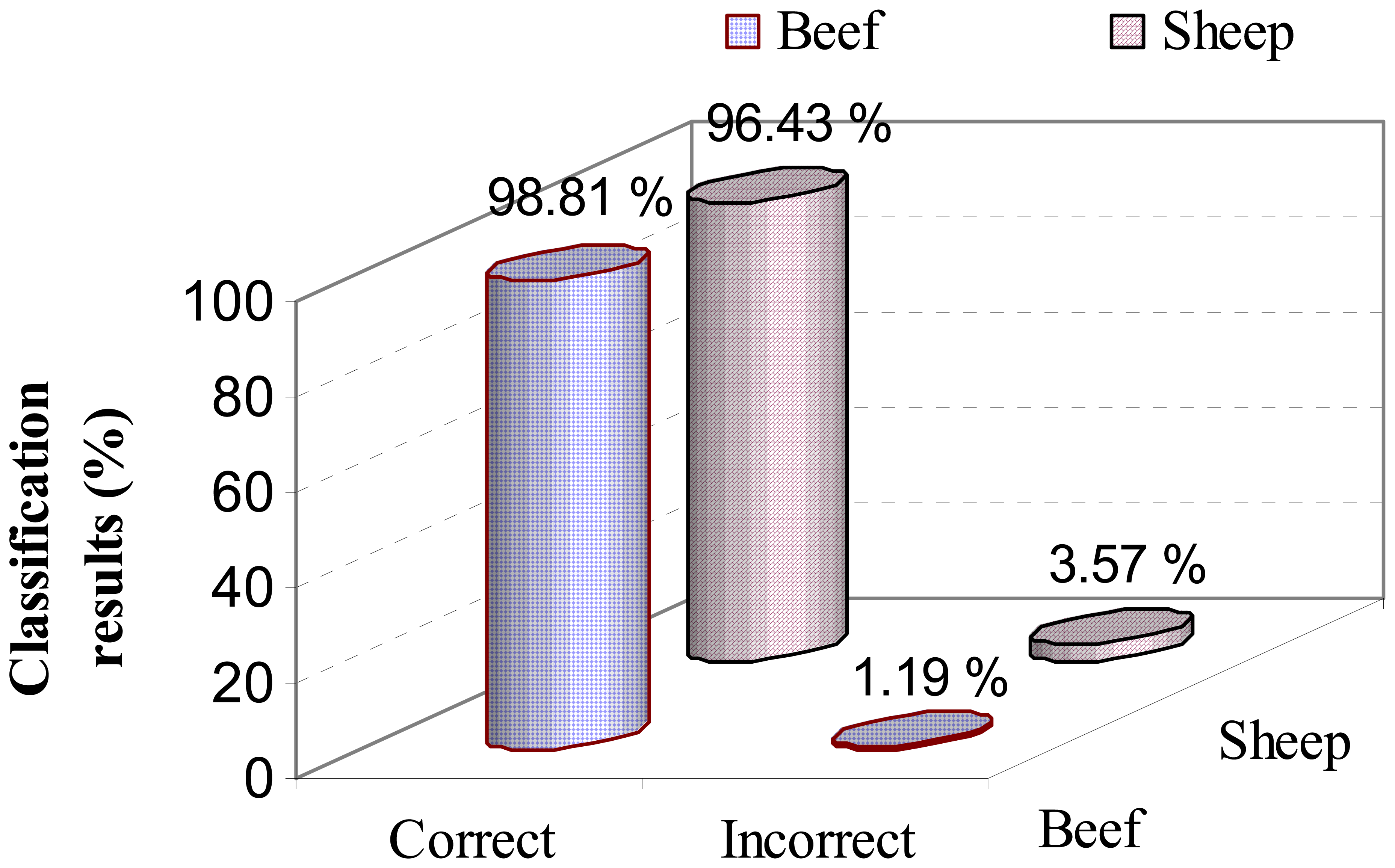
3.2.2. SVM analysis
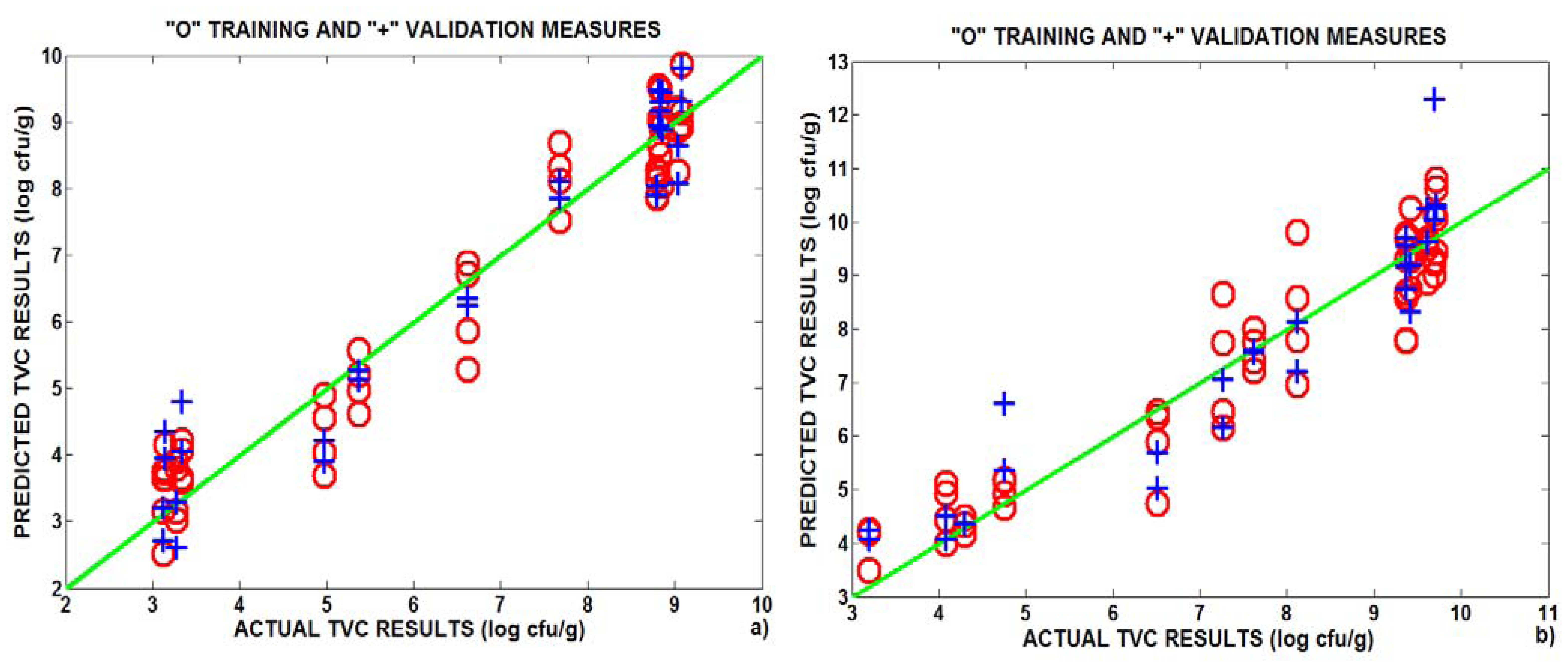
3.3. Correlation between e-nose and bacterial analysis
4. Conclusion
Acknowledgments
References and Notes
- Egan, A.F. Lactic acid bacteria of meat and meat products. Antonie Van Leeuwenhoek 1983, 49, 327–336. [Google Scholar]
- Dainty, R.H.; Mackey, B.M. The relationship between the phenotypic properties of bacteria from chill-stored meat and spoilage processes. J. Appl. Bacteriol. 1992, 73, 103S–114S. [Google Scholar]
- Borch, E.; Kant-Muermans, M.-L.; Blixt, Y. Bacterial spoilage of meat and cured meat products. Int. J. Food of Microbiol. 1996, 33, 103–120. [Google Scholar]
- Stutz, H.K.; Silverman, G. J.; Angelini, P.; Levin, R. E. Bacteria and other volatile compounds associated with ground beef spoilage. Journal of Food Science 1991, 56, 1147–1153. [Google Scholar]
- Schreurs, F.J. G. Post-mortem changes in chicken muscle. Worlds Poultry Science Journal 2000, 56, 319–346. [Google Scholar]
- Yano, Y.; Numata, M.; Hachiya, H.; Ito, S.; Masadome, T.; Ohkubo, S.; Asano, Y.; Imato, T. Application of a microbial sensor to the quality control of meat freshness. Talanta 2001, 54, 255–262. [Google Scholar]
- Panigrahi, S.; Balasubramanian, S.; Gu, H.; Logue, C.; Marchello, M. Neural-network-integrated electronic nose system for identification of spoiled beef. LWT 2006, 39, 135–145. [Google Scholar]
- Borch, E.; Agerhem, H. Chemical, microbial and sensory changes during the anaerobic cold storage of beef inoculated with homofermentative Lactobacillus sp. or a Leuconostoc sp. Int. J. Food Microbiol. 1992, 15, 99–108. [Google Scholar]
- Gardner, J.W.; Bartlett, P. N. A Brief History of Electronic Noses. Sensors Actuators B 1994, 18, 211–220. [Google Scholar]
- El Barbri, N.; Bouchikhi, B.; Llobet, E.; El Bari, N.; Correig, X. Differentiation of red meat using an electronic nose based on metal oxide semiconductor sensors and support vector machines. International Symposium on Olfaction and Electronic Nose ISOEN'07, Saint-Petersburg, Russia, 3- 5 May 2007; pp. 156–157.
- Rossi, V. V.; Garcia, C.; Talon, R.; Denoyer, C.; Berdagué, J.-L. Rapid discrimination of meat products and bacterial strains using semiconductor gas sensors. Sensors Actuators B 1996, 37, 43–48. [Google Scholar]
- Blixt, Y.; Borch, E. Using an electronic nose for determining the spoilage of vacuum packaged beef. International Journal of Food Microbiology 1999, 46, 123–134. [Google Scholar]
- Panigrahi, S.; Balasubramanian, S.; Gu, H.; Logue, C.M.; Marchello, M. Design and development of a metal oxide based electronic nose for spoilage classification of beef. Sensors Actuators B 2006, 119, 2–14. [Google Scholar]
- O'Connell, M.; Valdora, G.; Peltzer, G.; Negri, R.M. A practical approach for fish freshness determinations using a portable electronic nose. Sensors Actuators B 2001, 80, 149–154. [Google Scholar]
- Amari, A.; El Barbri, N.; Llobet, E.; El Bari, N.; Correig, X.; Bouchikhi, B. Monitoring the Freshness of Moroccan Sardines with a Neural-Network Based Electronic Nose. Sensors 2006, 6, 1209–1223. [Google Scholar]
- Sarry, F.; Lumbreras, M. Gas discrimination in an air conditioned system. IEEE Trans. Instrument. Measurement 2000, 49(4), 809–812. [Google Scholar]
- Gardner, J.W. Detection of vapours and odours from a multisensor array using pattern recognition Part 1. Principal component and cluster analysis. Sensors Actuators B 1991, 4, 109–115. [Google Scholar]
- Geladi, P.; Kowalski, B.R. Partial least squares regression: a tutorial. Anal. Chim. Acta 1986, 185, 1–17. [Google Scholar]
- Vapnik, V. Statistical Learning Theory; Wiley, 1998. [Google Scholar]
- Knerr, S.; Personnaz, L.; Dreyfus, G. Single-layer learning revisited: a stepwise procedure for building and training a neural network. Fogelman, J., Ed.; In Neurocomputing: Algorithms, Architectures and Applications; Springer-Verlag, 1990. [Google Scholar]
- Friedman, J. Another approach to polychotomous classification, technical report. Dept. of Statistics, Stanford Univ., 1996. [Google Scholar]
- Krebel, U. Pairwise classification and support vector machines. Schölkopf, B., Burges, C. J. C., Smola, A. J., Eds.; In Advances in Kernel Methods – Support Vector Learning; Cambridge, MA, 1999; pp. 255–268. [Google Scholar]
- Senter, S.D.; Arnold, J. W.; Chew, V. APC values and volatile compounds formed in commercially processed raw chicken parts during storage at 4 and 13 °C and under simulated temperature abuse conditions. Journal of the Science of Food and Agriculture 2000, 80(10), 1559–1564. [Google Scholar]
- Gram, L.; Ravn, L.; Rasch, M.; Bruhn, J.B.; Christensen, A. B.; Givskov, M. Food spoilage-interactions between food spoilage bacteria. International Journal of Food Microbiology 2002, 78(1–2), 79–97. [Google Scholar]
- Moh'd Abdullah, B. Beef and sheep mortadella: formulation, processing and quality aspects. International Journal of Food Science & Technology 2004, 39, 177–182. [Google Scholar]
- Vinaixa, M.; Llobet, E.; Brezmes, J.; Vilanova, X.; Correig, X. A fuzzy ARTMAP- and PLS-based MS e-nose for the qualitative and quantitative assessment of rancidity in crisps. Sensors Actuators B 2004, 106, 677–686. [Google Scholar]
- Dalgaard, P. Qualitative and quantitative characterization of spoilage bacteria from packed fish. Int. J. Food Microbiol. 1995, 26, 319–333. [Google Scholar]
- Gram, L.; Huss, H. H. Microbiological spoilage of fish and fish products. Int. J. Food Microbiol. 1996, 33, 121–137. [Google Scholar]
| Beef | Sheep | |||||
|---|---|---|---|---|---|---|
| Training | Validation | LVs | Training | Validation | LVs | |
| Fold 1 | 0.95 | 0.88 | 7 | 0.93 | 0.80 | 11 |
| Fold 2 | 0.78 | 0.70 | 7 | 0.93 | 0.84 | 11 |
| Fold 3 | 0.94 | 0.93 | 7 | 0.9 | 0.86 | 9 |
| Average | 0.89 | 0.84 | 7 | 0.92 | 0.83 | 10 |
© 2008 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
El Barbri, N.; Llobet, E.; El Bari, N.; Correig, X.; Bouchikhi, B. Electronic Nose Based on Metal Oxide Semiconductor Sensors as an Alternative Technique for the Spoilage Classification of Red Meat. Sensors 2008, 8, 142-156. https://doi.org/10.3390/s8010142
El Barbri N, Llobet E, El Bari N, Correig X, Bouchikhi B. Electronic Nose Based on Metal Oxide Semiconductor Sensors as an Alternative Technique for the Spoilage Classification of Red Meat. Sensors. 2008; 8(1):142-156. https://doi.org/10.3390/s8010142
Chicago/Turabian StyleEl Barbri, Noureddine, Eduard Llobet, Nezha El Bari, Xavier Correig, and Benachir Bouchikhi. 2008. "Electronic Nose Based on Metal Oxide Semiconductor Sensors as an Alternative Technique for the Spoilage Classification of Red Meat" Sensors 8, no. 1: 142-156. https://doi.org/10.3390/s8010142
APA StyleEl Barbri, N., Llobet, E., El Bari, N., Correig, X., & Bouchikhi, B. (2008). Electronic Nose Based on Metal Oxide Semiconductor Sensors as an Alternative Technique for the Spoilage Classification of Red Meat. Sensors, 8(1), 142-156. https://doi.org/10.3390/s8010142





