A Microsystem Based on Porous Silicon-Glass Anodic Bonding for Gas and Liquid Optical Sensing
Abstract
:Introduction
Theory
Experimental Section
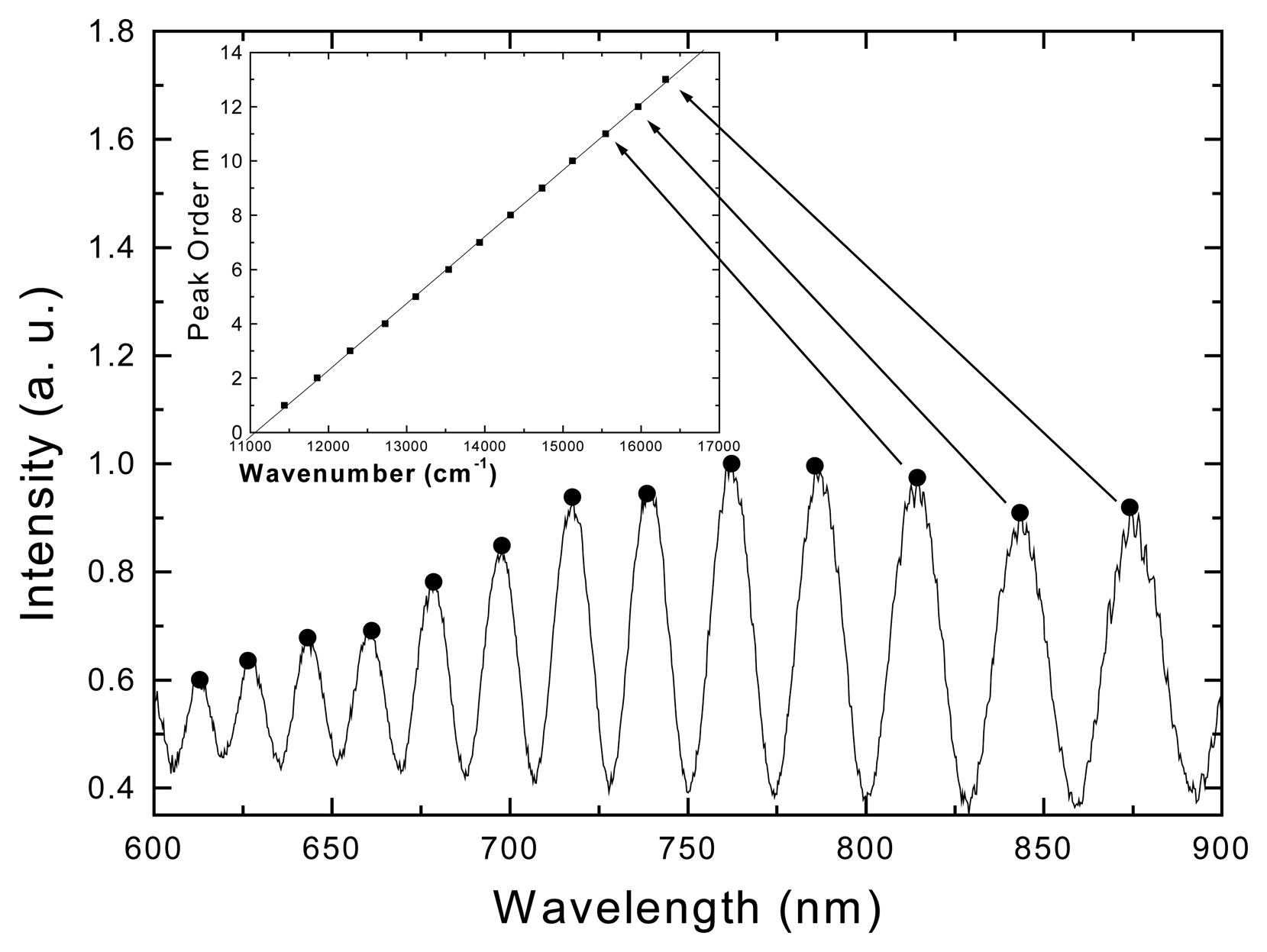
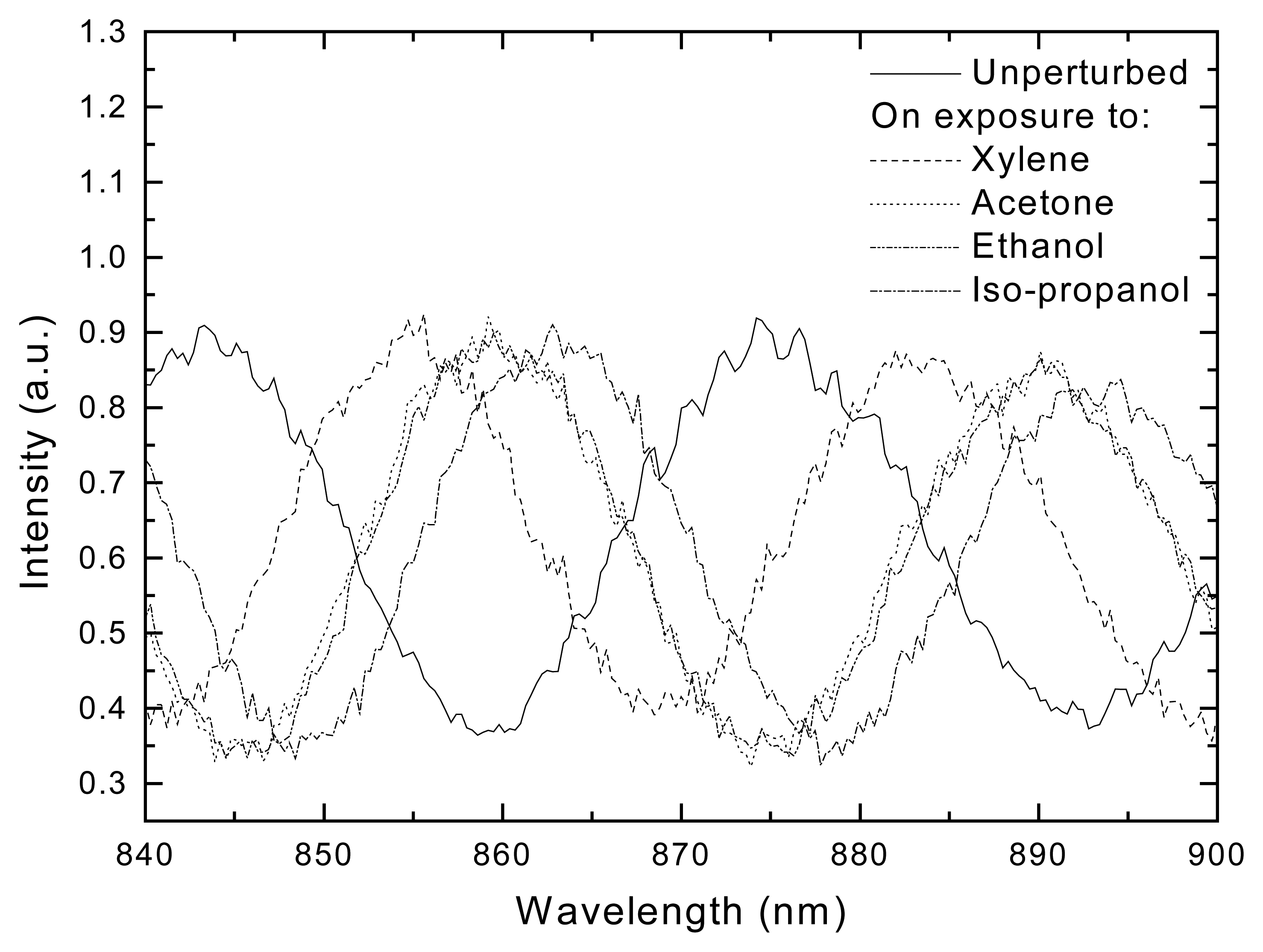
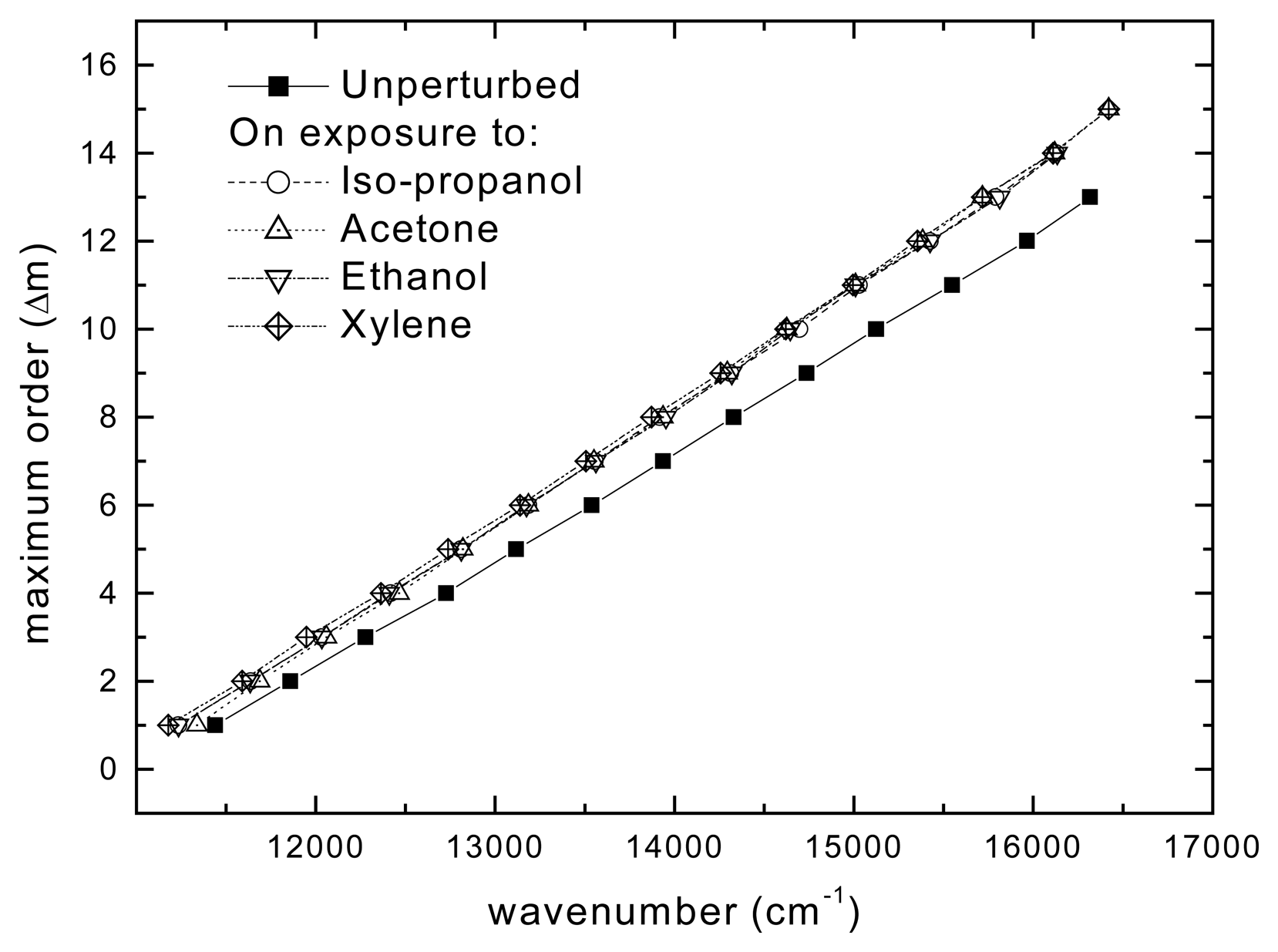
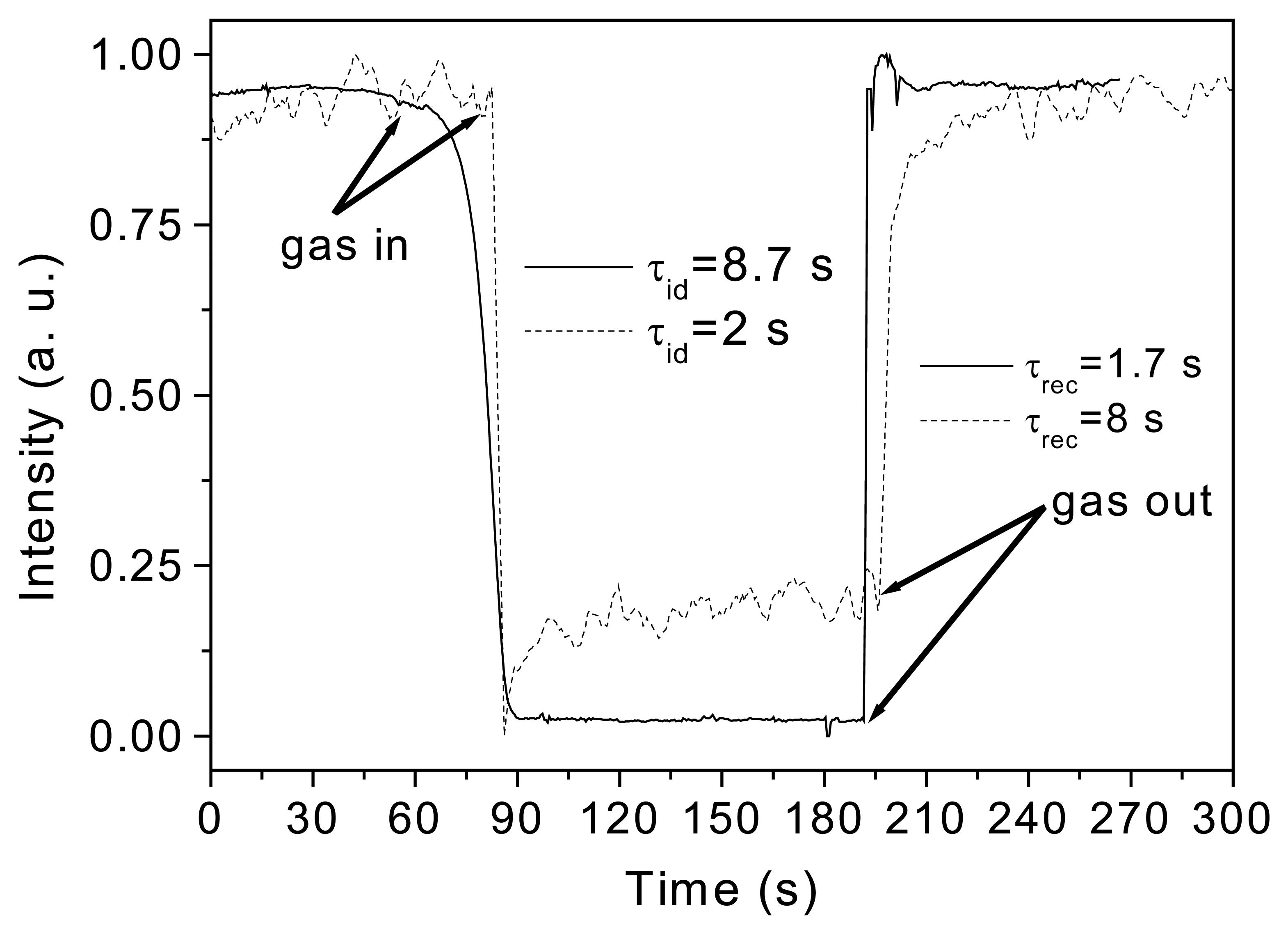
Results and Discussion
Conclusions
Acknowledgments
References
- Mulloni, V.; Pavesi, L. Porous silicon microcavities as optical chemical sensors. Appl. Phys. Lett. 2000, 76, 2523–2525. [Google Scholar]
- Gao, J.; Gao, T.; Sailor, M.J. Porous-silicon vapor sensor based on laser interferometry. Appl. Phys. Lett. 2000, 77, 901–903. [Google Scholar]
- Benson, T. M.; Arrand, H. F.; Sewell, P.; Niemeyer, D.; Loni, A.; Bozeat, R. J.; Kruger, M.; Arens-Fischer, R.; Thoenissen, M.; Luth, H. Progress towards achieving integrated circuit functionality using porous silicon optoelectronic components. Mat. Sci. Eng. B. 1999, 69-70, 92–95. [Google Scholar]
- Wongmanerod, C.; Zangooie, S.; Arwin, H. Determination of pore size distribution and surface area of thin porous silicon layers by spectroscopic ellipsometry. Applied Surface Science 2001, 172, 117–125. [Google Scholar]
- Puers, B.; Peeters, E.; Van Den Bossche, A.; Sansen, W. A capacitive pressure sensor with low impedance output and active suppression of parasitic effects. Sens. Actuat. A. 1990, 21-23, 108–114. [Google Scholar]
- Obermeier, E. Proc. Semiconductor Wafer Bonding. Science, Technology and Applications. Electrochem. Soc. 1995, 212–220. [Google Scholar]
- Malecki, K.; Della Corte, F.G. Silicon-glass anodic bonding at low temperature. Proc. of Micromachining and Microfabrication Process Technology X, SPIE 2005, Vol. 5715, 180–189. [Google Scholar]
- Lin, V.S.-Y.; Motesharei, K.; Dancil, K.-P. S.; Sailor, M. J.; Ghadiri, M. R. A porous silicon-based optical interferometric biosensor. Science 1997, 278, 840–843. [Google Scholar]
- Anderson, M.A.; Tinsley-Brown, A.; Allcock, P.; Perkins, E. A.; Snow, P.; Hollings, M.; Smith, R. G.; Reeves, C.; Squirrell, D. J.; Nicklin, S.; Cox, T. I. Sensitivity of the optical properties of porous silicon layers to the refractive index of liquid in the pores. Phys. Stat. Sol. A. 2003, 197, 528–533. [Google Scholar]
- Neimark, V.; Ravikovitch, P. I. Capillary condensation in MMS and pore structure characterization. Microporous Mesoporous Mater. 2001, 44-45, 697–707. [Google Scholar]
- Spanier, J.E.; Herman, I. P. Use of hybrid phenomenological and statistical effective-medium theories of dielectric functions to model the infrared reflectance of porous SiC films. Phys. Rev. B. 2000, 61, 10437–10450. [Google Scholar]
- De Stefano, L.; Malecki, K.; Moretti, L.; Rendina, I. Anodically bonded silicon-glass optical chip for biochemical sensing applications. Proc. SPIE. Int. Soc. Opt. Eng. 2005. [Google Scholar]
- De Stefano, L.; Rendina, I.; Moretti, L.; Rossi, A.M.; Tundo, S. Smart optical sensors for chemical substances based on porous silicon technology. Applied Optics 2004, 43/1, 167–172. [Google Scholar]
- Allcock, P.; Snow, P.A. Time-resolved sensing of organic vapors in low modulating porous silicon dielectric mirrors. J. Appl. Phys. 2001, 90, 50525057. [Google Scholar]
- De Stefano, L.; Moretti, L.; Rendina, I.; Rossi, A.M. Time-resolved sensing of chemical species in porous silicon optical microcavity. Sensor and Actuators B. 2004, 100, 168–172. [Google Scholar]

| Chemical compounds | n | ρ(g/cm3) | STC (mN/m) | VP (kPa) | BP (°C) | Δ<nd> (nm) | LLF |
|---|---|---|---|---|---|---|---|
| Iso-propanol | 1.377 | 0.785 | 20.93 | 6.8 | 82.4 | 1000 | 0.22 |
| Ethanol | 1.360 | 0.785 | 22.8 | 5.8 | 78 | 950 | 0.23 |
| Xylene | 1.501 | 1.454 | 38.8 | 0.046 | 214 | 1400 | 0.22 |
| Acetone | 1.359 | 0.791 | 23.46 | 30.8 | 56 | 998 | 0.23 |
© 2006 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
De Stefano, L.; Malecki, K.; Della Corte, F.G.; Moretti, L.; Rea, I.; Rotiroti, L.; Rendina, I. A Microsystem Based on Porous Silicon-Glass Anodic Bonding for Gas and Liquid Optical Sensing. Sensors 2006, 6, 680-687. https://doi.org/10.3390/s6060680
De Stefano L, Malecki K, Della Corte FG, Moretti L, Rea I, Rotiroti L, Rendina I. A Microsystem Based on Porous Silicon-Glass Anodic Bonding for Gas and Liquid Optical Sensing. Sensors. 2006; 6(6):680-687. https://doi.org/10.3390/s6060680
Chicago/Turabian StyleDe Stefano, Luca, Krzysztof Malecki, Francesco G. Della Corte, Luigi Moretti, Ilaria Rea, Lucia Rotiroti, and Ivo Rendina. 2006. "A Microsystem Based on Porous Silicon-Glass Anodic Bonding for Gas and Liquid Optical Sensing" Sensors 6, no. 6: 680-687. https://doi.org/10.3390/s6060680
APA StyleDe Stefano, L., Malecki, K., Della Corte, F. G., Moretti, L., Rea, I., Rotiroti, L., & Rendina, I. (2006). A Microsystem Based on Porous Silicon-Glass Anodic Bonding for Gas and Liquid Optical Sensing. Sensors, 6(6), 680-687. https://doi.org/10.3390/s6060680





