Cyclodextrin Derivatives as Chiral Supramolecular Receptors for Enantioselective Sensing
Abstract
:1. Introduction
1.1. General Considerations
1.2. Chiral recognition principle
- ▪
- “An essential requirement is that the chiral macrocycles form reasonably stable complexes with the guest enantiomers so that the repulsive interactions can effectively lessen the stability of the complex of one enantiomer.
- ▪
- Large chiral barrier(s) result in a high degree of enantiomeric recognition.
- ▪
- Low conformational flexibility of diastereomeric complexes plays an important role in good enantiomeric recognition. Two factors, rigid macrocycles and multipoint interactions, ensure a fixed conformation of the complexes.
- ▪
- The structural complementarity between chiral macrocycles and enantiomers ensures that the chiral barriers of macrocycles make full use of steric repulsion for enantiomeric recognition.
- ▪
- Macrocyclic receptors possessing C2, C3, and D2 symmetry usually show higher enantioselectivity than those of C1 and D3 symmetry”
1.3. Scope of the review
2. Cyclodextrins for Chiral Sensing
2.1. Generalities
2.2. Chiral recognition properties of cyclodextrins
2.3. Cyclodextrins in chiral sensing layer
2.3.1. Chiral sensing at the solid-liquid interface
2.3.2. In the gas phase
3. Other chiral macrocycles, potential candidates for chiral sensing
3.1. Calixarenes and Calix-resorcinarenes
3.2. Crown-ethers
4. Concluding remarks
References and Notes
- Buckingham, A. D.; Fowler, P. W.; Hutson, J. M. Theoretical studies of van der Waals molecules and intermolecular forces. Chem. Rev. 1988, 88, 963–88. [Google Scholar]
- Brammer, L. Developments in inorganic crystal engineering. Chem. Soc. Rev. 2004, 33, 476–489. [Google Scholar]
- Belkova, N. V.; Shubina, E. S.; Epstein, L. M. Diverse world of unconventional hydrogen bonds. Acc. Chem. Res. 2005, 38, 624–631. [Google Scholar]
- Bushey, M. L.; Nguyen, T.-Q.; Zhang, W.; Horoszewski, D.; Nuckolls, C. Using hydrogen bonds to direct the Assembly of Crowded Aromatics. Angew. Chem., Int. Ed. 2004, 43, 5446–5453. [Google Scholar]
- Desiraju, G. R. Hydrogen bridges in crystal engineering: interactions without borders. Acc. Chem. Res. 2002, 35, 565–573. [Google Scholar]
- Sherrington, D. C.; Taskinen, K. A. Self-assembly in synthetic macromolecular systems via multiple hydrogen bonding interactions. Chem. Soc. Rev. 2001, 30, 83–93. [Google Scholar]
- Desiraju, G. R. The C-H⋯O Hydrogen bond: structural implications and supramolecular design. Acc. Chem. Res. 1996, 29, 441–449. [Google Scholar]
- Eisenberg, D.; Kauzmann, W. The Structure and Properties of Water; Oxford University Press: Oxford, 2005. [Google Scholar]
- Alvarez, S.; Palacios, A. A.; Aullon, G. Ligand orientation effects on metal-metal, ligand-ligand and metal-ligand interactions. Coord. Chem. Rev 1999, 185-186, 431–450. [Google Scholar]
- Cory, M. G.; Zerner, M. C. Metal-ligand exchange coupling in transition-metal complexes. Chem. Rev. 1991, 91, 813–822. [Google Scholar]
- Ruchon, T.; Vallet, M.; Thepot, J.-Y.; Le Floch, A.; Boyd, R. W. Experimental evidence of magnetochiral interaction in Pasteur's tartrates. Comptes Rendus Physique 2004, 5, 273–277. [Google Scholar]
- Reddy, I. K.; Mehvar, R. Chirality in Drug Design and Development; Marcel Dekker, Inc.: New-York, 1994. [Google Scholar]
- Cope, M. J. Understanding chirality: how molecules that are mirror images of each other can act differently in the Body. Anal. Proceed. 1993, 30, 498–500. [Google Scholar]
- Wolf, C. Stereolabile Chiral compounds: analysis by dynamic chromatography and stopped-flow methods. Chem. Soc. Rev. 2005, 34, 595–608. [Google Scholar]
- Roussel, C.; Del Rio, A.; Pierrot-Sanders, J.; Piras, P.; Vanthuyne, N. Chiral liquid chromatography contribution to the determination of the absolute configuration of enantiomers. J. Chromatography A 2004, 1037, 311–328. [Google Scholar]
- Ward, T. J. Chiral separations. Anal. Chem. 2002, 74, 2863–2872. [Google Scholar]
- Ward, T. J. Chiral separations. Anal. Chem. 2000, 72, 4521–4528. [Google Scholar]
- vander Heyden, Y.; Mangelings, D.; Matthijs, N.; Perrin, C. Chiral Separations 2005, Vol. 6, p 447–498.
- Natishan, T. K. Recent progress in the analysis of pharmaceuticals by capillary electrophoresis. J. Liq. Chromatogr. Relat. Technol. 2005, 28, 1115–1160. [Google Scholar]
- Laemmerhofer, M. Chiral separations by capillary electromigration techniques in nonaqueous media. II. enantioselective nonaqueous capillary electrochromatography. J. Chromatography, A 2005, 1068, 31–57. [Google Scholar]
- Veuthey, J.-L. Capillary electrophoresis in pharmaceutical and biomedical analysis. Anal. Bioanal. Chem. 2005, 381, 93–95. [Google Scholar]
- Guebitz, G.; Schmid, M. G. Recent advances in chiral separation principles in capillary electrophoresis and capillary electrochromatography. Electrophoresis 2004, 25, 3981–3996. [Google Scholar]
- Ward, T. J.; Hamburg, D.-M. Chiral separations. Anal. Chem. 2004, 76, 4635–4644. [Google Scholar]
- Riekkola, M.-L.; Siren, H. Chiral separation by capillary electrophoresis in nonaqueous medium. Methods Mol Biol. 2004, 243, 365–373. [Google Scholar]
- Schurig, V. Practice and theory of enantioselective complexation gas chromatography. J. Chromatography A 2002, 965, 315–356. [Google Scholar]
- Eiceman, G. A.; Hill, H. H., Jr.; Gardea-Torresdey, J. Gas chromatography. Anal. Chem. 1998, 70, 321R–339R. [Google Scholar]
- Schurig, V. Enantiomer separation by gas chromatography on chiral stationary phases. J. Chromatography A 1994, 666, 111–29. [Google Scholar]
- Kasper, M.; Busche, S.; Gauglitz, G. Frontiers in Chemical Sensors, Novel Principles and Techniques; Orellana, G., Moreno-Bondi, M. C., Eds.; Springer-Verlag: Berlin, 2005; Vol. 3, pp. p 323–341. [Google Scholar]
- Hillberg, A. L.; Brain, K. R.; Allender, C. J. Molecular imprinted polymer sensors: implications for therapeutics. Adv. Drug Deliv. Rev. 2005, 57, 1875–1889. [Google Scholar]
- Mahony, J. O.; Nolan, K.; Smyth, M. R.; Mizaikoff, B. Molecularly imprinted polymers-potential and challenges in analytical chemistry. Anal. Chim. Acta 2005, 534, 31–39. [Google Scholar]
- Zhang, X. X.; Bradshaw, J. S.; Izatt, R. M. Enantiomeric recognition of amine compounds by chiral macrocyclic receptors. Chem. Rev. 1997, 97, 3313–3361. [Google Scholar]
- Szejtli, J. Introduction and general overview of cyclodextrin chemistry. Chem. Rev. 1998, 98, 1743–1754. [Google Scholar]
- Szejtli, J. Past, present, and future of cyclodextrin Research. Pure Appl. Chem. 2004, 76, 1825–1845. [Google Scholar]
- Szejtli, J. Encyclopedia of Nanoscience and Nanotechnology; Marcel Dekker: New York, 2004; Vol. 2, pp. p 283–304. [Google Scholar]
- Villiers, A. Sur la transformation de la fécule en dextrine par le ferment butyrique. Compt. Rend. Fr. Acad. Sci. 1891, 112, 435–438. [Google Scholar]
- Freudenberg, K.; G, B.; Ewald, L.; Soff, K. Hydrolysis and acetolysis of starch and of the Schardinger dextrins. Ber. Deutsch. Chem. Ges. 1936, 69, 1258. [Google Scholar]
- Khan, A. R.; Forgo, P.; Stine, K. J.; D'Souza, V. T. Methods for selective modifications of cyclodextrins. Chem. Rev. 1998, 98, 1977–1996. [Google Scholar]
- Connors, K. A. The stability of cyclodextrin complexes in solution. Chem. Rev. 1997, 97, 1325–1357. [Google Scholar]
- Schneider, H.-J.; Hacket, F.; Ruediger, V.; Ikeda, H. NMR studies of cyclodextrins and cyclodextrin complexes. Chem. Rev. 1998, 98, 1755–1785. [Google Scholar]
- Szejtli, J. Utilization of cyclodextrins in industrial products and processes. J. Mater. Chem. 1997, 7, 575–587. [Google Scholar]
- Cramer, F.; Dietsche, W. Occlusion compounds. XV. Resolution of racemates with cyclodextrins. Chem. Ber. 1959, 92, 378–84. [Google Scholar]
- Kano, K.; Nishiyabu, R. General mechanism for chiral recognition by native and modified cyclodextrins. J. Incl. Phenom. Macrocyclic Chem. 2002, 44, 355–359. [Google Scholar]
- Kano, K. Mechanisms for chiral recognition by cyclodextrins. J. Phys. Org. Chem. 1997, 10, 286–291. [Google Scholar]
- Easton, C. J.; Lincoln, S. F. Chiral discrimination by modified cyclodextrins. Chem. Soc. Rev. 1996, 25, 163–170. [Google Scholar]
- Ogston, A. G. Interpretation of experiments on metabolic processes, using isotopic tracer elements. Nature 1948, 162, 963. [Google Scholar]
- Behr, J.-P. The Lock and Key Principle. The State of the Art-100 Years on; Wiley: Chichester, 1994; Vol. 1. [Google Scholar]
- Kataky, R.; Bates, P. S.; Parker, D. Functionalized alpha-cyclodextrins as potentiometric chiral sensors. Analyst 1992, 117, 1313–1317. [Google Scholar]
- Bates, P. S.; Kataky, R.; Parker, D. A chiral sensor based on a peroctylated alpha-Cyclodextrin. J. Chem. Soc., Chem. Commun. 1992, 153–155. [Google Scholar]
- Bates, P. S.; Kataky, R.; Parker, D. Chiral sensors based on lipophilic cyclodextrins: interrogation of enantioselectivity by combined NMR, structural correlation and electrode response studies. J. Chem. Soc., Perkin Trans. 1994, 2, 669–75. [Google Scholar]
- Kataky, R.; Parker, D.; Kelly, P. M. Potentiometric, enantioselective sensors for alkyl and aryl ammonium-ions of pharmaceutical significance, based on lipophilic cyclodextrins. Scand. J. Clinical Lab. Invest. 1995, 55, 409–419. [Google Scholar]
- Stefan, R. I.; van Staden, J. F.; Baiulescu, G. E.; Aboul-Enein, H. Y. A potentiometric, enantioselective membrane electrode for S-ramipril assay. Chem. Anal. 1999, 44, 417–422. [Google Scholar]
- Stefan, R.-I.; Van Staden, J. F.; Aboul-Enein, H. Y. Detection of S-enantiomers of cilazapril, pentopril, and trandolapril using a potentiometric, enantioselective membrane electrode. Electroanalysis 1999, 11, 192–194. [Google Scholar]
- Ozoemena, K. I.; Stefan, R.-I.; Van Staden, J. F.; Aboul-Enein, H. Y. Enantioanalysis of S-perindopril using different cyclodextrin-based potentiometric sensors. Sens. Actuators, B 2005, B105, 425–429. [Google Scholar]
- Stefan, R.-I.; Van Staden, J. F.; Aboul-Enein, H. Y. S-Perindopril assay using a potentiometric, enantioselective membrane electrode. Chirality 1999, 11, 631–634. [Google Scholar]
- Stefan, R. I.; Van Staden, J. F.; Aboul-Enein, H. Y. A new construction for potentiometric, enantioselective membrane electrodes, and use for L-proline assay. Anal. Lett. 1998, 31, 1787–1794. [Google Scholar]
- Shahgaldian, P.; Hegner, M.; Pieles, U. A cyclodextrin self-assembled monolayer (SAM) based surface plasmon resonance (SPR) Sensor for Enantioselective Analysis of Thyroxine. J. Inclusion Phenom. Macrocyclic Chem. 2005, 53, 35–39. [Google Scholar]
- Maeda, Y.; Fukuda, T.; Yamamoto, H.; Kitano, H. Regio- and stereoselective complexation by a self-assembled monolayer of thiolated cyclodextrin on a gold electrode. Langmuir 1997, 13, 4187–4189. [Google Scholar]
- Fukuda, T.; Maeda, Y.; Kitano, H. Stereoselective inclusion of DOPA derivatives by a self-assembled monolayer of thiolated cyclodextrin on a gold electrode. Langmuir 1999, 15, 1887–1890. [Google Scholar]
- Stefan, R.-I.; Van Staden, J. F.; Aboul-Enein, H. Y. A new construction for a potentiometric, enantioselective membrane electrode-its utilization to the S-Captopril Assay. Talanta 1999, 48, 1139–1143. [Google Scholar]
- Ozoemena, K. I.; Stefan, R.-I. Enantioselective potentiometric membrane electrodes based on α-, β- and γ-cyclodextrins as chiral selectors for the assay of L-proline. Talanta 2005, 66, 501–504. [Google Scholar]
- Beulen, M. W. J.; Bugler, J.; De Jong, M. R.; Lammerink, B.; Huskens, J.; Schonherr, H.; Vancso, G. J.; Boukamp, B. A.; Wieder, H.; Offenhauser, A.; Knoll, W.; Van Veggel, F. C. J. M.; Reinhoudt, D. N. Host-guest interactions at self-assembled monolayers of cyclodextrins on gold. Chem. Eur. J. 2000, 6, 1176–1183. [Google Scholar]
- Auletta, T.; Dordi, B.; Mulder, A.; Sartori, A.; Onclin, S.; Bruinink, C. M.; Peter, M.; Nijhuis, C. A.; Beijleveld, H.; Schoenherr, H.; Vansco, G. J.; Casnati, A.; Ungaro, R.; Ravoo, B. J.; Huskens, J.; Reinhoudt, D. N. Writing patterns of molecules on molecular printboards. Angew. Chem., Int. Ed. 2004, 43, 369–373. [Google Scholar]
- Yamamoto, H.; Maeda, Y.; Kitano, H. Molecular recognition by self-assembled monolayers of cyclodextrin on Ag. J. Phys. Chem. B 1997, 101, 6855–6860. [Google Scholar]
- Schalley, C. A. Molecular recognition and supramolecular chemistry in the gas phase. Mass Spec. Rev. 2002, 20, 253–309. [Google Scholar]
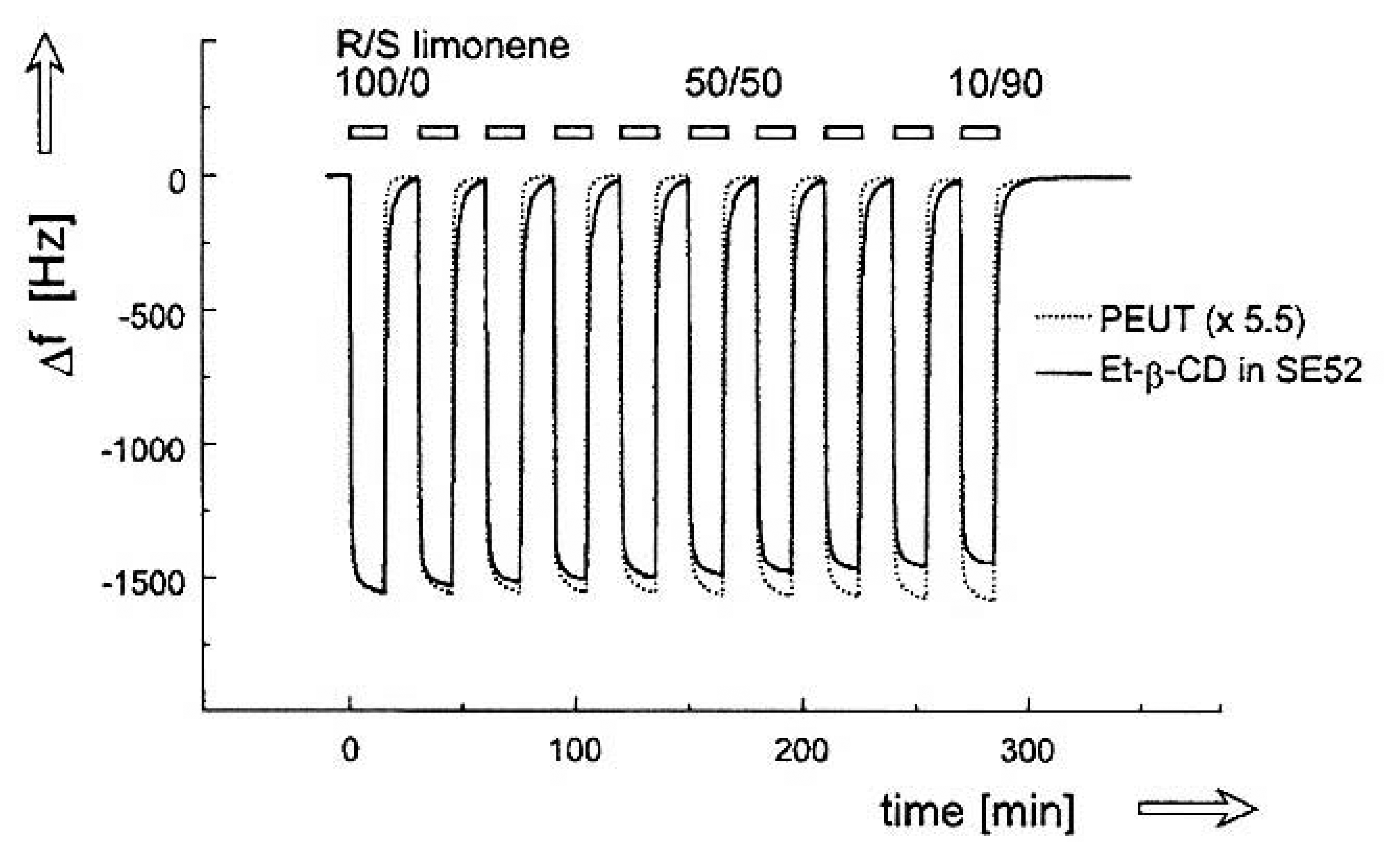
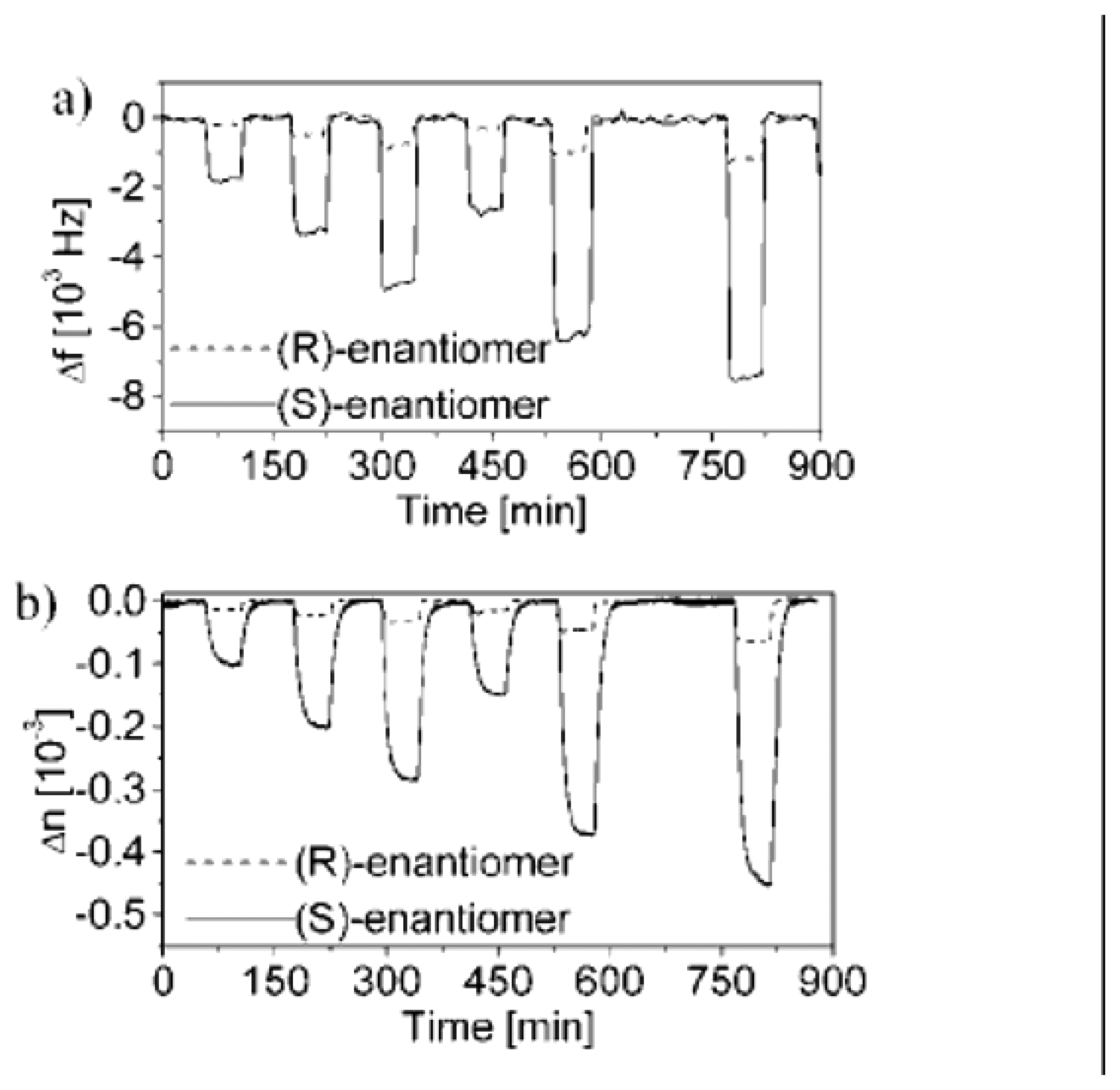
- Ide, J.; Nakamoto, T.; Moriizumi, T. Discrimination of aromatic optical isomers using quartz-resonator sensors. Sens. Actuators, A 1995, 49, 73–78. [Google Scholar]
- Fietzek, C.; Hermle, T.; Rosenstiel, W.; Schurig, V. Chiral discrimination of limonene by use of beta-cyclodextrin-coated quartz-crystal-microbalances (QCMs) and data evaluation by artificial neuronal networks. Fresenius J. Anal. Chem. 2001, 371, 58–63. [Google Scholar]
- May, L. P.; Byfield, M. P.; Lindstrom, M.; Wunsche, L. F. Chiral discrimination using a quartz crystal microbalance and comparison with gas chromatographic retention data. Chirality 1997, 9, 225–232. [Google Scholar]
- Ng, S. C.; Sun, T.; Chan, H. S. O. Chiral discrimination of enantiomers with a self-assembled monolayer of functionalized beta-cyclodextrins on Au surfaces. Tetrahedron Lett. 2002, 43, 2863–2866. [Google Scholar]
- Ng, S. C.; Sun, T.; Chan, H. S. O. Durable chiral sensor based on quartz crystal microbalance using self-assembled monolayer of permethylated beta-cyclodextrin. Macromol. Symp. 2003, 192, 171–181. [Google Scholar]
- Hierlemann, A.; Ricco, A. J.; Bodenhofer, K.; Gopel, W. Effective use of molecular recognition in gas sensing: results from acoustic wave and in situ FT-IR measurements. Anal. Chem. 1999, 71, 3022–3035. [Google Scholar]
- Kieser, B.; Fietzek, C.; Schmidt, R.; Belge, G.; Weimar, U.; Schurig, V.; Gauglitz, G. Use of a modified cyclodextrin host for the enantioselective detection of a halogenated diether as chiral guest via optical and electrical transducers. Anal. Chem. 2002, 74, 3005–3012. [Google Scholar]
- Bodenhofer, K.; Hierlemann, A.; Juza, N.; Schurig, V.; Gopel, W. Chiral discrimination of inhalation anesthetics and methyl propionates by thickness shear mode resonators: new insights into the mechanisms of enantioselectivity by cyclodextrins. Anal. Chem. 1997, 69, 4017–4031. [Google Scholar]
- Sallas, F.; Kovacs, J.; Pinter, I.; Jicsinszky, L.; Marsura, A. One-step synthesis of new urea-linked b-cyclodextrin dimers. Tetrahedron Letters 1996, 37, 4011–4014. [Google Scholar]
- Arduini, A.; Casnati, A. Macrocycle Synthesis; Parker, D., Ed.; Oxford University Press: Oxford, 1996; pp. 145–173. [Google Scholar]
- Atwood, J. L.; Steed, J. W. Encyclopedia of Supramolecular Chemistry; Marcel Dekker: New York, 2004. [Google Scholar]
- Gutsche, C. D. Calixarenes Revisited; The Royal Society of Chemistry: Letchworth, 1998; p. p 233 pp. [Google Scholar]
- Diamond, D.; Nolan, K. Calixarenes: designer ligands for chemical sensors. Anal. Chem. 2001, 73, 22A–29A. [Google Scholar]
- Vysotsky, M.; Schmidt, C.; Bohmer, V. Chirality in Calixarenes and Calixarene Assemblies. Advances in Supramolecular Chemistry 2000, 7, 139–233. [Google Scholar]
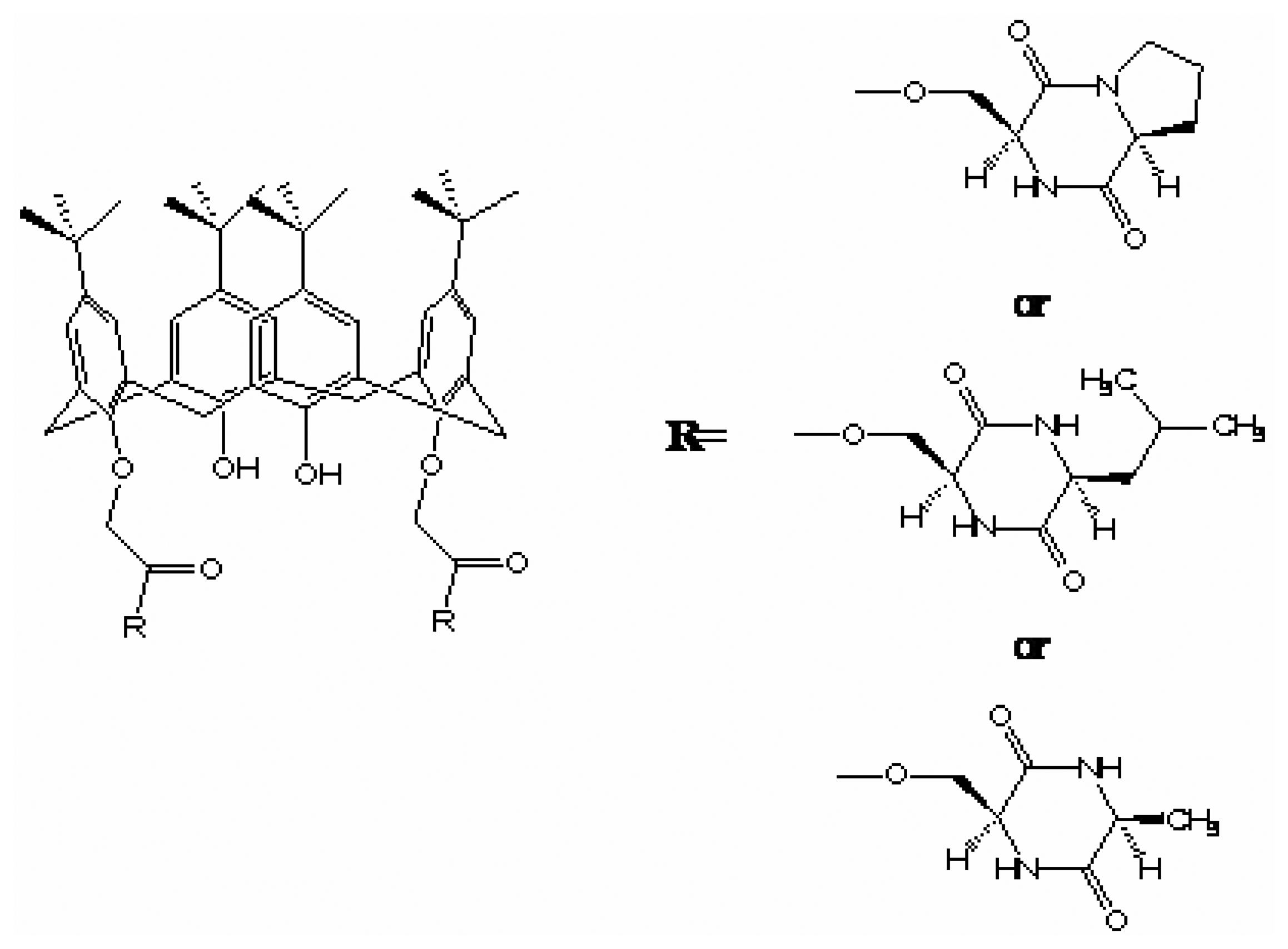
- Guo, W.; Wang, J.; Wang, C.; He, J. Q.; He, X. W.; Cheng, J. P. Design, synthesis, and enantiomeric recognition of dicyclodipeptide-bearing Calix[4]arenes: a promising family for chiral gas sensor coatings. Tetrahedron Lett. 2002, 43, 5665–5667. [Google Scholar]
- Liu, Y.; You, C. C.; Kang, S. Z.; Wang, C.; Chen, F.; He, X. W. Synthesis of novel beta-cyclodextrin and calixarene derivatives and their use in gas sensing on the basis of molecular recognition. Eur. J. Org. Chem 2002, 607–613. [Google Scholar]
- Pietraszkiewicz, M.; Prus, P.; Bilewicz, R. pH dependent enantiomeric recognition of amino acids by Mannich-type calix[4]resorcinarenes in Langmuir monolayers. Polish J. Chem. 1999, 73, 1845–1853. [Google Scholar]
- Prus, P.; Pietraszkiewicz, M.; Bilewicz, R. Monolayers of chiral calix[4]resorcinarenes. surface pressure and surface Potential Studies. Supramol. Chem. 1998, 10, 17–25. [Google Scholar]
- Pietraszkiewicz, M.; Prus, P.; Bilewicz, R. pH dependent enantioselection of amino acids by phosphorous-containing calix[4]resorcinarene in Langmuir monolayers. Polish J. Chem. 1999, 73, 2035–2042. [Google Scholar]
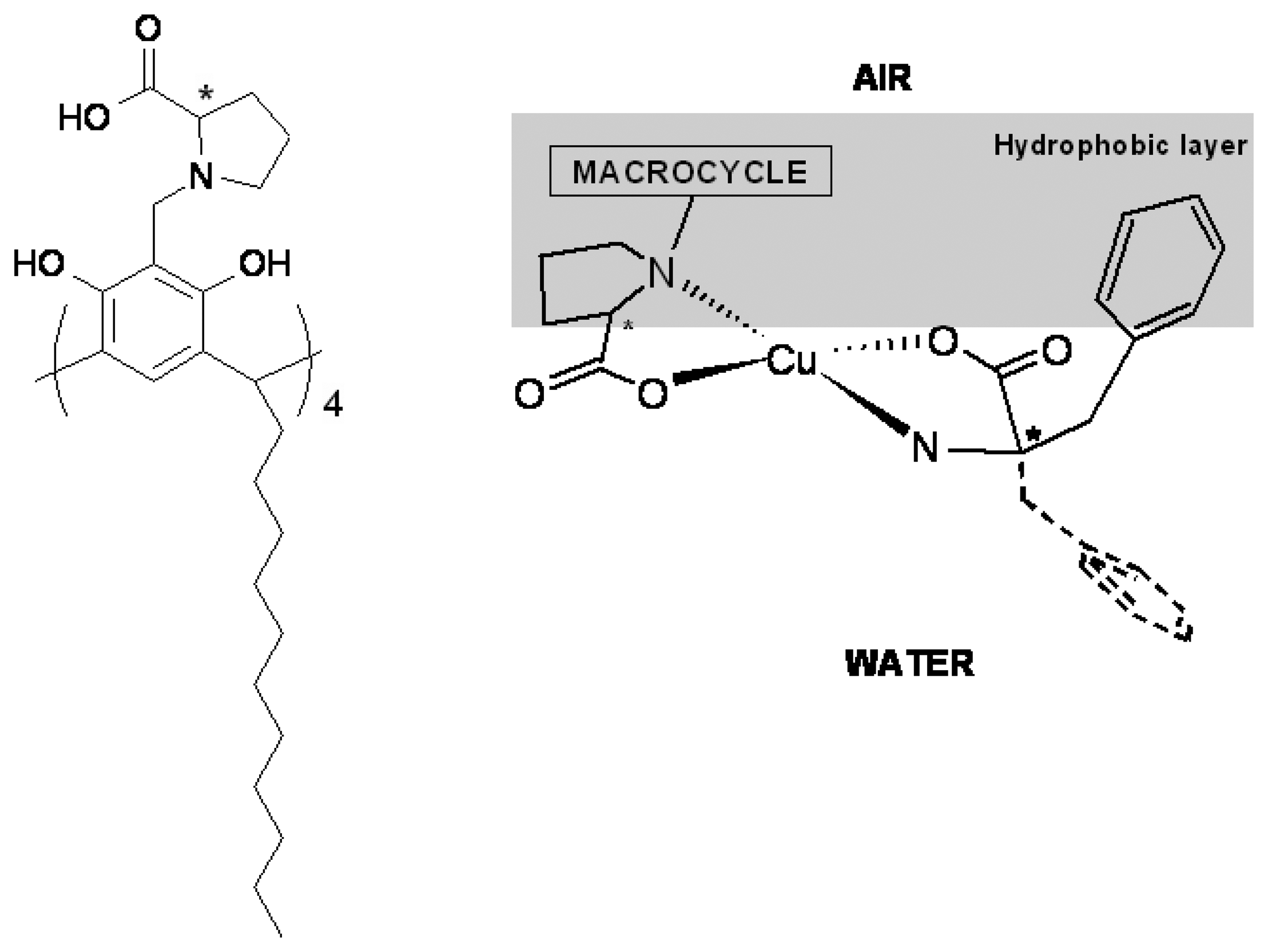
- Shahgaldian, P.; Pieles, U.; Hegner, M. Enantioselective reognition of phenylalanine by a chiral amphiphilic macrocycle at the air-water interface: a copper-mediated mechanism. Langmuir 2005, 21, 6503–6507. [Google Scholar]
- Gokel, G. W.; Leevy, W. M.; Weber, M. E. Crown ethers: sensors for ions and molecular scaffolds for materials and biological models. Chem. Rev. 2004, 104, 2723–2750. [Google Scholar]
- Bradshaw, J. M.; Izatt, R. M.; Bordunov, A. V.; Zhu, C. Y.; Hathaway, J. K. Comprehensive Supramolecular Chemistry; Gokel, G. W., Ed.; Pergamon: Oxford, 1996; Vol. 1, p. p 35. [Google Scholar]
- Pedersen, C. J. The discovery of crown ethers. Science 1988, 241, 536–540. [Google Scholar]
- Yasaka, Y.; Yamamoto, T.; Kimura, K.; Shono, T. Simple evaluation of enantiomer selectivity of crown ether using a membrane electrode. Chem. Lett 1980, 769–72. [Google Scholar]
- Shinbo, T.; Yamaguchi, T.; Nishimura, K.; Kikkawa, M.; Sugiura, M. Enantiomer-selective membrane electrode for amino acid methyl esters. Anal. Chim. Acta 1987, 193, 367–71. [Google Scholar]
- Badis, M.; Tomaszkiewicz, I.; Joly, J.-P.; Rogalska, E. Enantiomeric recognition of amino acids by amphiphilic crown ethers in Langmuir monolayers. Langmuir 2004, 20, 6259–6267. [Google Scholar]
- Horvath, V.; Takacs, T.; Horvai, G.; Huszthy, P.; Bradshaw, J. S.; Izatt, R. M. Enantiomer selectivity of ion-selective electrodes based on a chiral crown-ether ionophore. Anal. Lett. 1997, 30, 1591–1609. [Google Scholar]
- Kawabata, H.; Shinkai, S. Chiral recognition of α-amino acid derivatives by a steroidal crown Ether at the air-water interface. Chem. Lett 1994, 375–378. [Google Scholar]
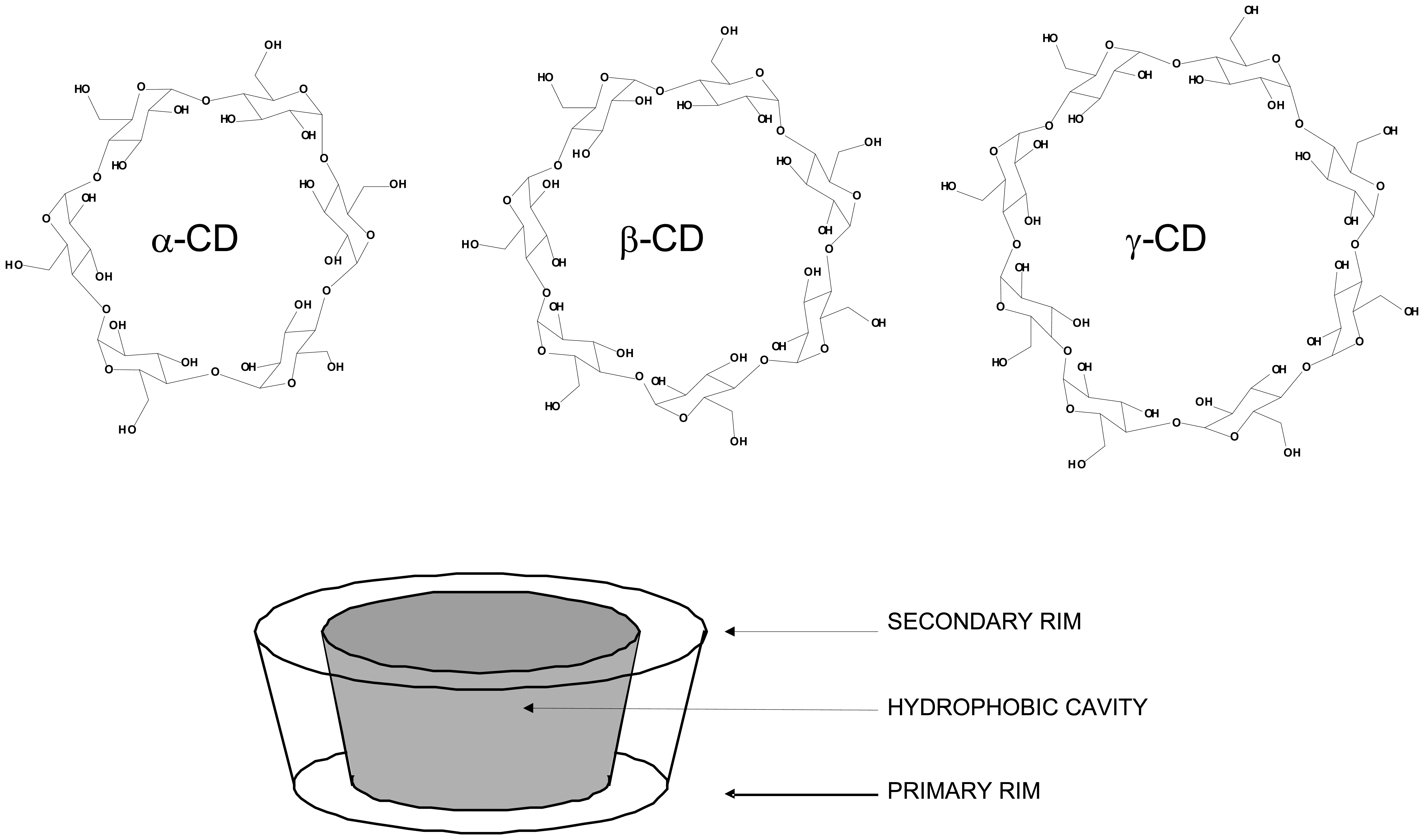

| Alpha | Beta | Gamma | |
|---|---|---|---|
| Glucose monomers | 6 | 7 | 8 |
| Molecular weight | 972 | 1135 | 1297 |
| Height of torus, Å | 7.9 | 7.9 | 7.9 |
| Internal cavity volume, Å3 | 174 | 262 | 427 |
| Internal cavity diameter, Å | 4.7-5.3 | 6.0-6.5 | 8 |
| [α]D 25°C | 150 | 162 | 177 |
| Water solubility (g/100mL: 25° C) | 14.2 | 1.85 | 23.2 |
| Water molecules in cavity | 6 | 11 | 17 |
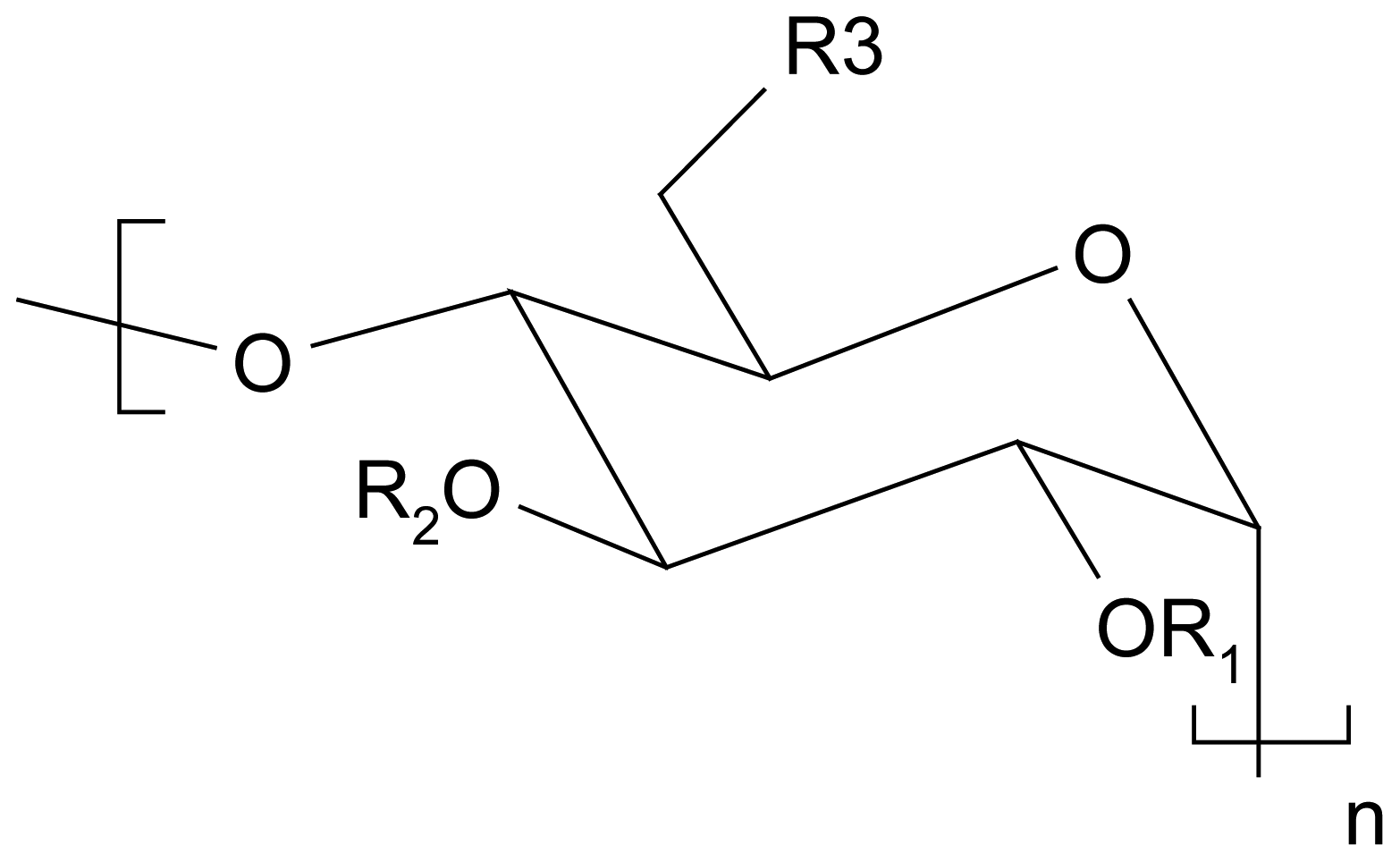 | ||||||
|---|---|---|---|---|---|---|
| Ref | n | R1 | R2 | R3 | Guest | Sensing method |
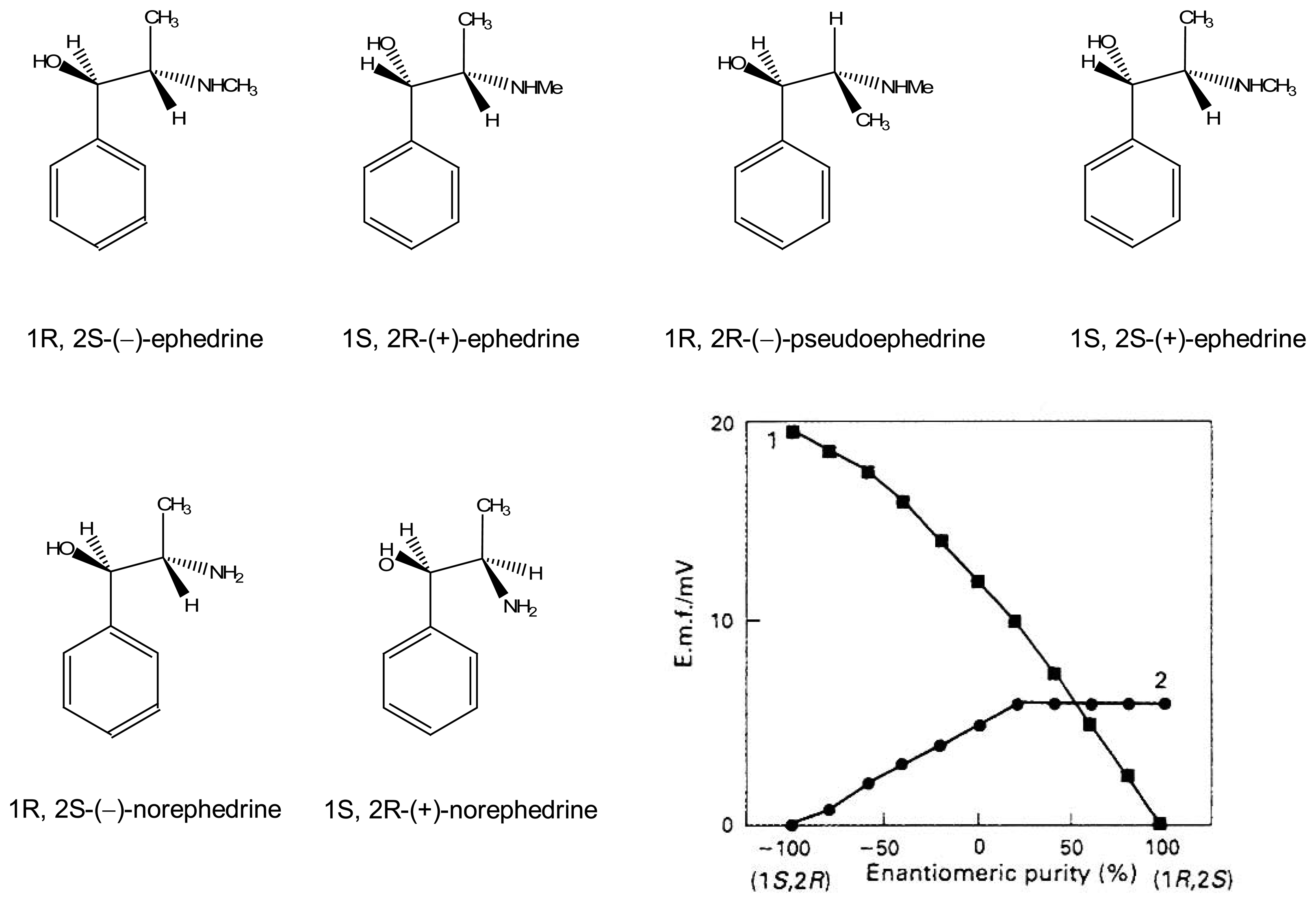
| [47] | 6 | octyl | Octyl, H | O-octyl | ephedrine | Potentiometric |
| [48] | 6 | octyl | octyl | O-octyl | ehedrine | Potentiometric |
| [49] | 6 | H, Me, Ac or Octyl | Octyl | O-octyl | Ephedrinium ion | Potentiometric |
| [50] | Octyl | octyl or dodecyl | O-octyl or O-dodecyl | amphetamine propanalol | Potentiometric | |
| [51] | 7 | trimethylammoniopropyl | H | OH | ramipril | Potentiometric |
| [52] | 7 | trimethylammoniopropyl | H | OH | cilazapril pentopril trandolapril | Potentiometric |
| [53] | 6, 7 or 8 | H | H | OH | perindopril | Potentiometric |
| [54] | 7 | trimethylammoniopropyl | H | OH | perindopril | Potentiometric |
| [55] | 7 | trimethylammoniopropyl | H | OH | proline | Potentiometric |
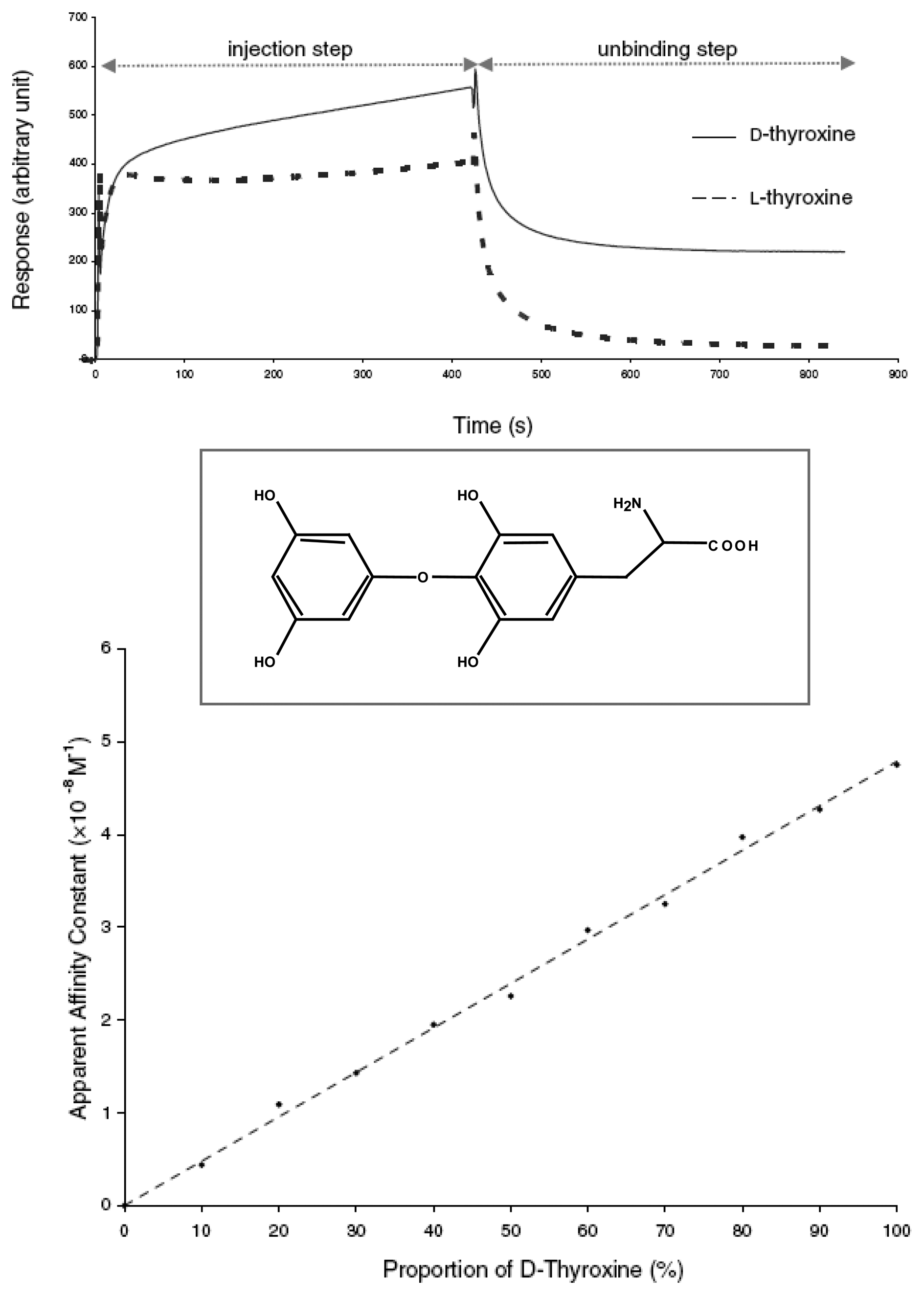
| [56] | 7 | H | H | NH-CO-(CH2)nS(CH2)-nCH3 | thyroxine | SPR |
| [57] | 6 | H | H | NH(CH2)2SH | Phenyl-ethylamine | CV |
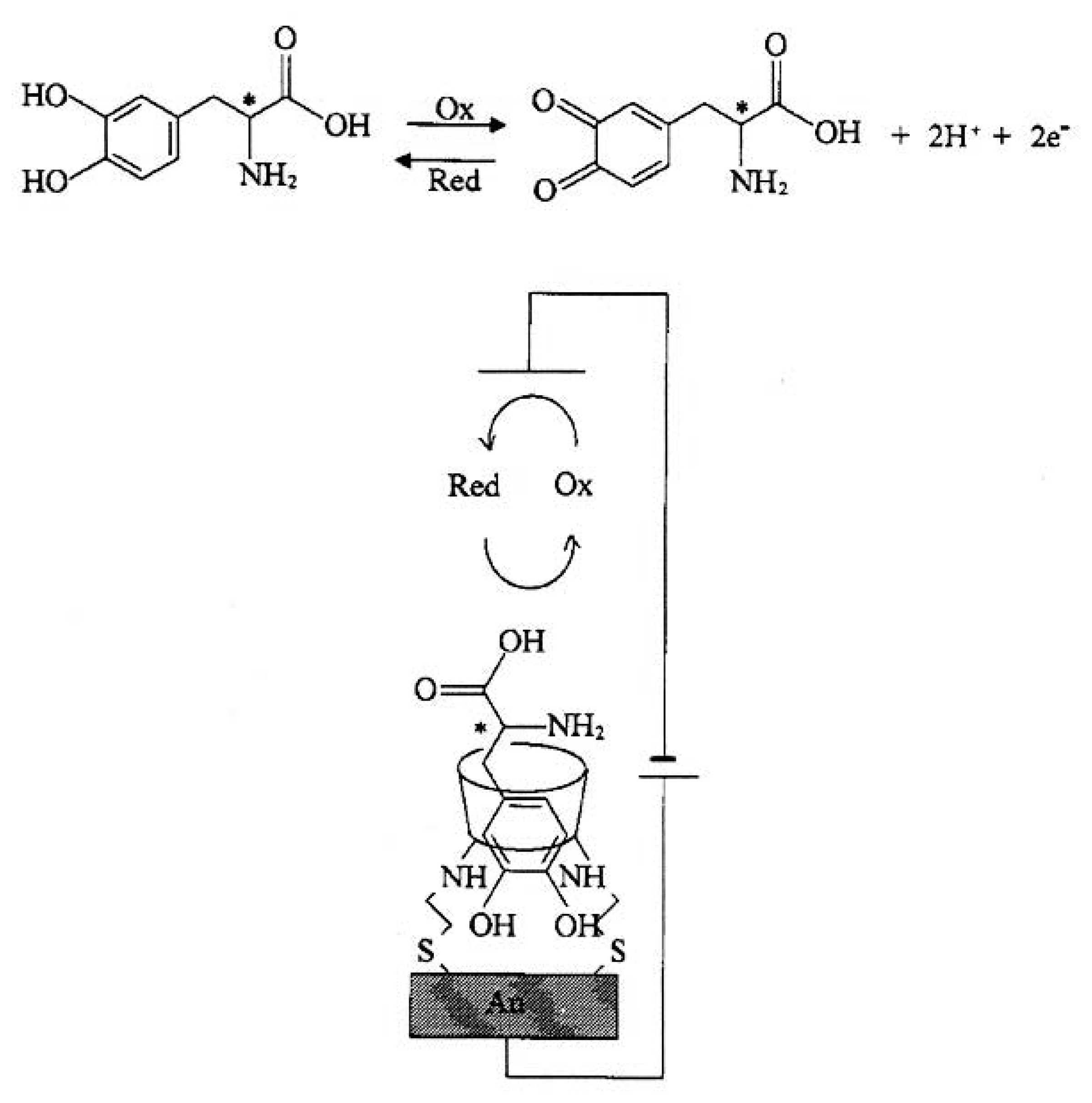
| [58] | 6 | H | H | NH(CH2)2SH | DOPA-derivatives | CV |
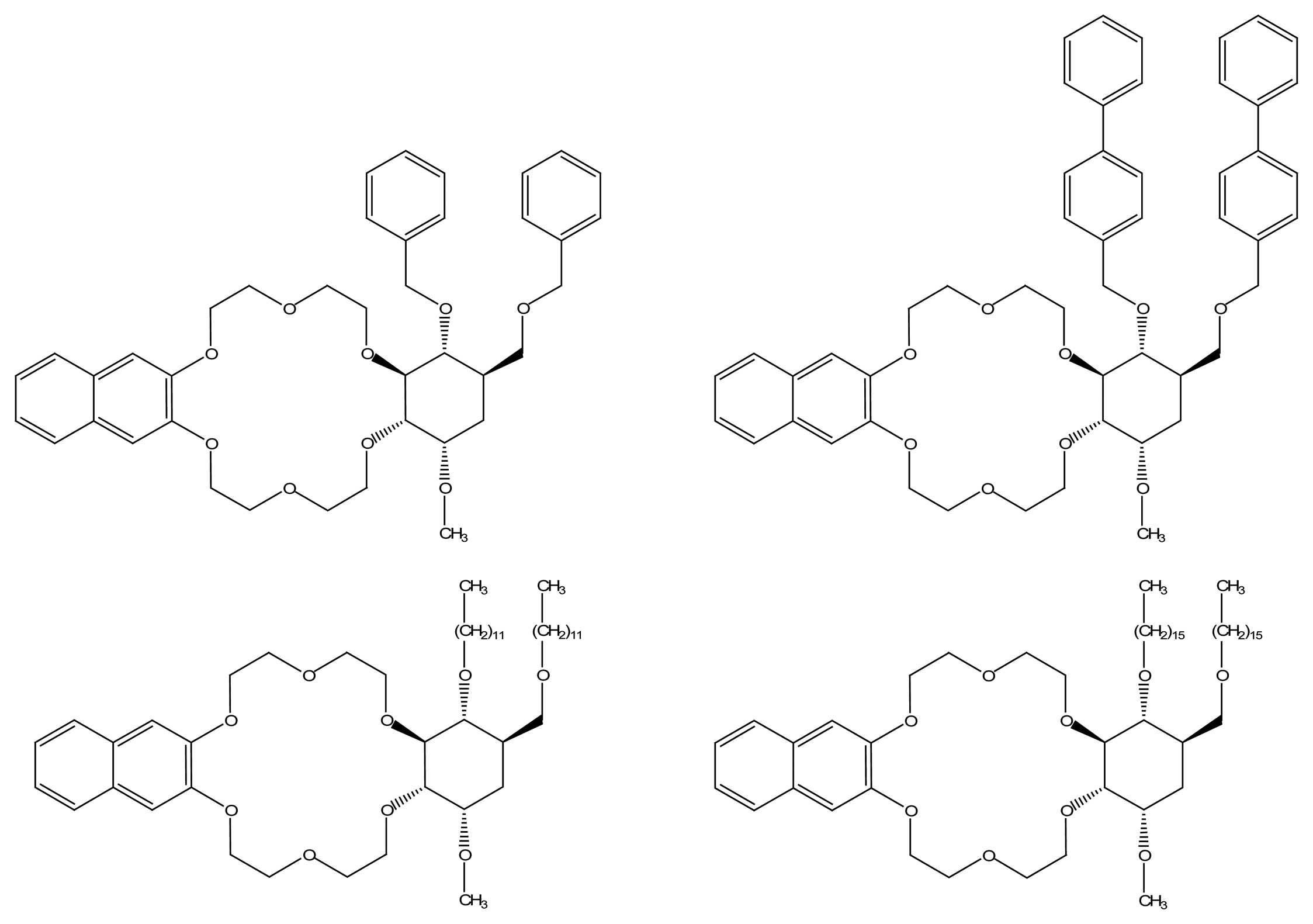
 | |||||
|---|---|---|---|---|---|
| n | R1 | R2 | R3 | Guest | Sensing method |
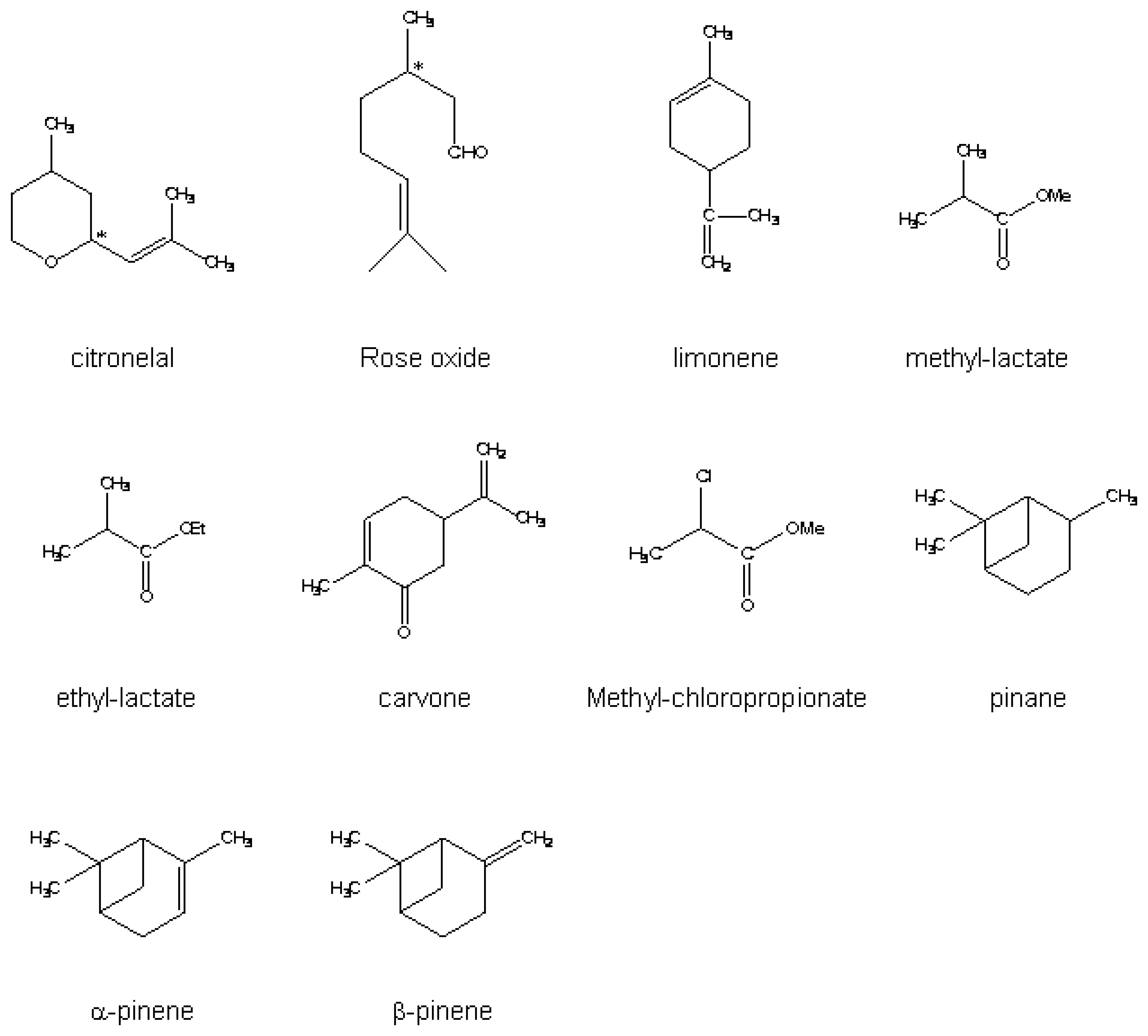
| 6,7,8 | Me | Me, pentyl | O-Me | citronellal rose oxide | QCM |
| 7 | Ethyl, methyl, acetyl | Ethyl, methyl, acetyl | TBDMS | limonene | QCM |
| 7, 8 | pentyl | pentyl | O-Me | Pinene pinane | QCM |
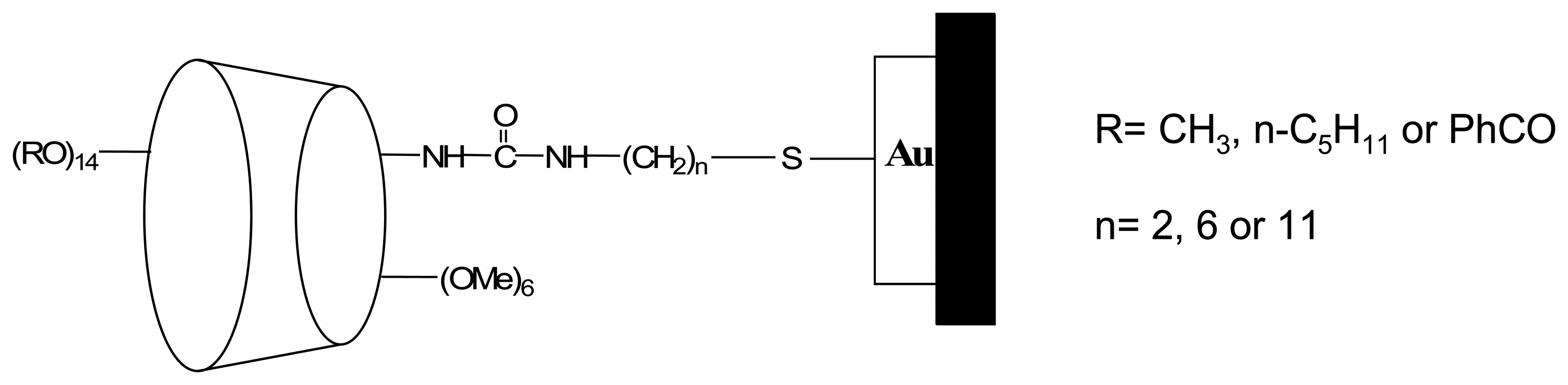
| 7 | Me, Pentyl, φCHO | Me, Pentyl, φCHO | mono-ONHCONH(CH2)nSH | methyl-lactate | QCM |
| 7 | (OMe)6 ONHCONH(CH2)nSH n= 2, 6 or 11 | 2-butanol 2-octanol methyl-lactate ethyl-lactate carvone | QCM | ||
| 7 | pentyl | Butanoyl | O-pentyl | Methyl lactate Methyl chloropropionate | SAW |
| 8 | pentyl | butanoyl | O-pentyl | Sevoflurane degradation product | TSMR, SAW, SPR |
| 8 | butanoyl | pentyl | O-pentyl | methyl-lactate methyl-chloropropionate flurane derivatives | SMR |
© 2006 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Shahgaldian, P.; Pieles, U. Cyclodextrin Derivatives as Chiral Supramolecular Receptors for Enantioselective Sensing. Sensors 2006, 6, 593-615. https://doi.org/10.3390/s6060593
Shahgaldian P, Pieles U. Cyclodextrin Derivatives as Chiral Supramolecular Receptors for Enantioselective Sensing. Sensors. 2006; 6(6):593-615. https://doi.org/10.3390/s6060593
Chicago/Turabian StyleShahgaldian, Patrick, and Uwe Pieles. 2006. "Cyclodextrin Derivatives as Chiral Supramolecular Receptors for Enantioselective Sensing" Sensors 6, no. 6: 593-615. https://doi.org/10.3390/s6060593




