UV Absorption Spectroscopy in Water-Filled Antiresonant Hollow Core Fibers for Pharmaceutical Detection
Abstract
:1. Introduction
2. Materials and Methods
2.1. Antiresonant Hollow Core Fiber
2.2. Optofluidic Chip
2.3. Spectroscopic Setup
2.4. Sample Substances
2.5. Measurement Procedure and Data Processing
3. Results
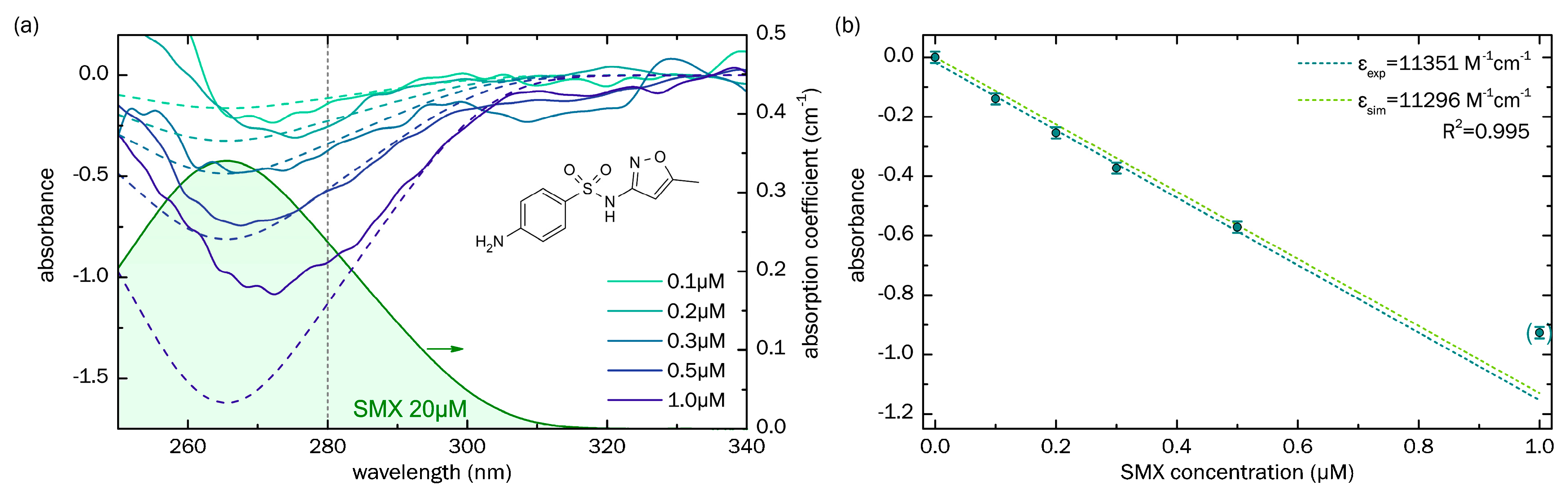
3.1. Sensing of Sulfamethoxazole
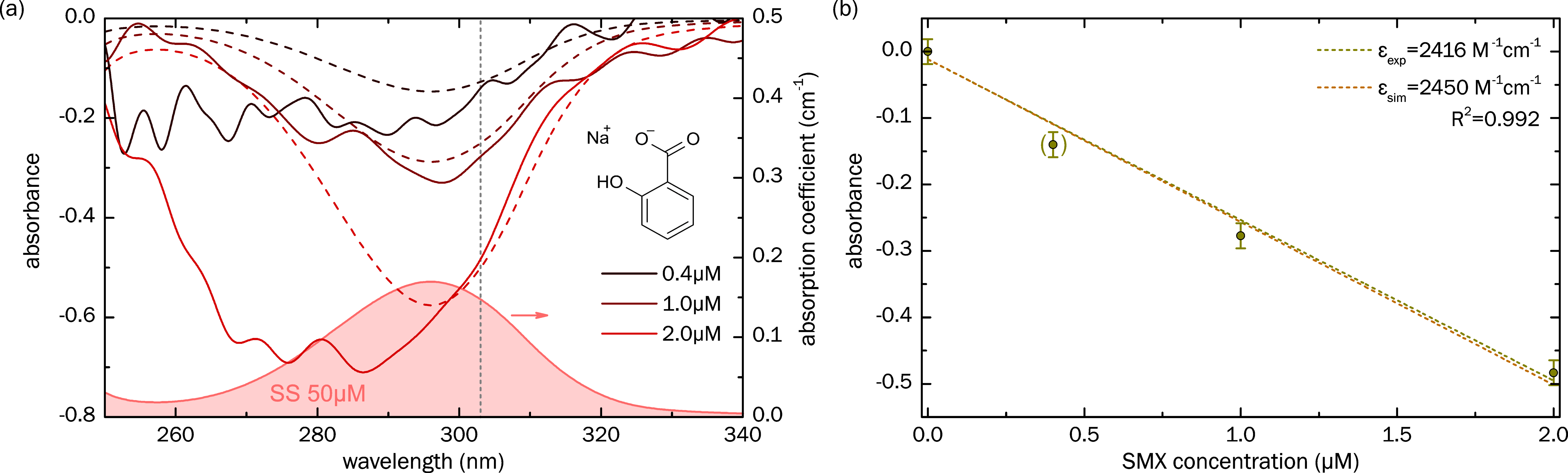
3.2. Sensing of Sodium Salicylate
4. Discussion and Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sarmah, A.K.; Meyer, M.T.; Boxall, A.B.A. A global perspective on the use, sales, exposure pathways, occurrence, fate and effects of veterinary antibiotics (VAs) in the environment. Chemosphere 2006, 65, 725–759. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, I.T.; Santos, L. Antibiotics in the aquatic environments: A review of the European scenario. Environ. Int. 2016, 94, 736–757. [Google Scholar] [CrossRef] [PubMed]
- Hannappel, S.; Köpp, C.; Zühlke, S. Aufklärung der Ursachen von Tierarzneimittelfunden im Grundwasser—Untersuchung eintragsgefährdeter Standorte in Norddeutschland; Umweltbundesamt: Dessau-Roßlau, Germany, 2016. [Google Scholar]
- Dasgupta, D.; Sengupta, T.K. Techniques and methods: Detection of antibiotics in environmental samples. In The Battle against Microbial Pathogens: Basic Science, Technological Advances and Educational Programs; Formatex: Badajoz, Spain, 2015; pp. 1084–1090. [Google Scholar]
- Palmisano, T.; Prudenzano, F.; Warren-Smith, S.C.; Monro, T.M. Design of exposed-core fiber for methadone monitoring in biological fluids. J. Non-Cryst. Solids 2011, 357, 2000–2004. [Google Scholar] [CrossRef]
- Ahrer, W.; Scherwenk, E.; Buchberger, W. Determination of drug residues in water by the combination of liquid chromatography or capillary electrophoresis with electrospray mass spectrometry. J. Chromatogr. A 2001, 910, 69–78. [Google Scholar] [CrossRef]
- Öllers, S.; Singer, H.P.; Fässler, P.; Müller, S.R. Simultaneous quantification of neutral and acidic pharmaceuticals and pesticides at the low-ng/l level in surface and waste water. J. Chromatogr. A 2001, 911, 225–234. [Google Scholar] [CrossRef]
- Luthria, D.L.; Mukhopadhyay, S.; Robbins, R.J.; Finley, J.W.; Banuelos, G.S.; Harnly, J.M. UV spectral fingerprinting and analysis of variance-principal component analysis: A useful tool for characterizing sources of variance in plant materials. J. Agric. Food Chem. 2008, 56, 5457–5462. [Google Scholar] [CrossRef] [PubMed]
- Ojeda, C.B.; Rojas, F.S. Recent applications in derivative ultraviolet/visible absorption spectrophotometry: 2009–2011: A review. Microchem. J. 2013, 106, 1–16. [Google Scholar] [CrossRef]
- Behera, S.; Ghanty, S.; Ahmad, F.; Santra, S.; Banerjee, S. UV-Visible Spectrophotometric Method Development and Validation of Assay of Paracetamol Tablet Formulation. J. Anal. Bioanal. Tech. 2012, 3, 2–6. [Google Scholar] [CrossRef]
- Kröckel, L.; Frosch, T.; Schmidt, M.A. Multiscale spectroscopy using a monolithic liquid core waveguide with laterally attached fiber ports. Anal. Chim. Acta 2015, 875, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Cregan, R.F.; Mangan, B.J.; Knight, J.C.; Birks, T.A.; Russell, P.S.J.; Roberts, P.J.; Allan, D.C. Single-mode photonic band gap guidance of light in air. Science 1999, 285, 1537–1539. [Google Scholar] [CrossRef] [PubMed]
- Hart, S.D.; Maskaly, G.R.; Temelkuran, B.; Prideaux, P.H.; Joannopoulos, J.D.; Fink, Y. External reflection from omnidirectional dielectric mirror fibers. Science 2002, 296, 510–513. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.G.; Ibanescu, M.; Skorobogatiy, M.; Weisberg, O.; Engeness, T.D.; Soljačić, M.; Jacobs, S.A.; Joannopoulos, J.D.; Fink, Y. Low-loss asymptotically single-mode propagation in large-core OmniGuide fibers. Opt. Express 2001, 9, 748–779. [Google Scholar] [CrossRef] [PubMed]
- Zeisberger, M.; Tuniz, A.; Schmidt, M.A. Analytic model for the complex effective index dispersion of metamaterial-cladding large-area hollow core fibers. Opt. Express 2016, 24, 20515–20528. [Google Scholar] [CrossRef] [PubMed]
- Tuniz, A.; Zeisberger, M.; Schmidt, M.A. Tailored loss discrimination in indefinite metamaterial-clad hollow-core fibers. Opt. Express 2016, 24, 15702–15709. [Google Scholar] [CrossRef] [PubMed]
- Couny, F.; Benabid, F.; Light, P.S. Large-pitch kagome-structured hollow-core photonic crystal fiber. Opt. Lett. 2006, 31, 3574–3576. [Google Scholar] [CrossRef] [PubMed]
- Sollapur, R.; Kartashov, D.; Zürch, M.; Hoffmann, A.; Grigorova, T.; Sauer, G.; Hartung, A.; Schwuchow, A.; Bierlich, J.; Kobelke, J.; et al. Resonance-enhanced multi-octave supercontinuum generation in antiresonant hollow-core fibers. Light Sci. Appl. 2017, 6, e17124. [Google Scholar] [CrossRef]
- Debord, B.; Amsanpally, A.; Chafer, M.; Baz, A.; Maurel, M.; Blondy, J.M.; Hugonnot, E.; Scol, F.; Vincetti, L.; Gérôme, F.; et al. Ultralow transmission loss in inhibited-coupling guiding hollow fibers. Optica 2017, 4, 209–217. [Google Scholar] [CrossRef]
- Hartung, A.; Kobelke, J.; Schwuchow, A.; Bierlich, J.; Popp, J.; Schmidt, M.A.; Frosch, T. Low-loss single-mode guidance in large-core antiresonant hollow-core fibers. Opt. Lett. 2015, 40, 3432–3435. [Google Scholar] [CrossRef] [PubMed]
- Hartung, A.; Kobelke, J.; Schwuchow, A.; Wondraczek, K.; Bierlich, J.; Popp, J.; Frosch, T.; Schmidt, M.A. Double antiresonant hollow core fiber–guidance in the deep ultraviolet by modified tunneling leaky modes. Opt. Express 2014, 22, 19131–19140. [Google Scholar] [CrossRef] [PubMed]
- Rindorf, L.; Høiby, P.E.; Jensen, J.B.; Pedersen, L.H.; Bang, O.; Geschke, O. Towards biochips using microstructured optical fiber sensors. Anal. Bioanal. Chem. 2006, 385, 1370–1375. [Google Scholar] [CrossRef] [PubMed]
- Washburn, E.W. The dynamics of capillary flow. Phys. Rev. 1921, 17, 273–283. [Google Scholar] [CrossRef]
- Zeisberger, M.; Schmidt, M.A. Analytic model for the complex effective index of the leaky modes of tube-type anti-resonant hollow core fibers. Sci. Rep. 2017, 7, 11761–11767. [Google Scholar] [CrossRef] [PubMed]
- Litchinitser, N.M.; Abeeluck, A.K.; Headley, C.; Eggleton, B.J. Antiresonant reflecting photonic crystal optical waveguides. Opt. Lett. 2002, 27, 1592–1594. [Google Scholar] [CrossRef] [PubMed]
- Daimon, M.; Masumura, A. Measurement of the refractive index of distilled water from the near-infrared region to the ultraviolet region. Appl. Opt. 2007, 46, 3811–3820. [Google Scholar] [CrossRef] [PubMed]
- Kröckel, L.; Schmidt, M.A. Extinction properties of ultrapure water down to deep ultraviolet wavelengths. Opt. Mater. Express 2014, 4, 1932–1942. [Google Scholar] [CrossRef]
- Li, W.C. Occurrence, sources, and fate of pharmaceuticals in aquatic environment and soil. Environ. Pollut. 2014, 187, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Loos, R.; Wollgast, J.; Huber, T.; Hanke, G. Polar herbicides, pharmaceutical products, perfluorooctanesulfonate (PFOS), perfluorooctanoate (PFOA), and nonylphenol and its carboxylates and ethoxylates in surface and tap waters around Lake Maggiore in Northern Italy. Anal. Bioanal. Chem. 2007, 387, 1469–1478. [Google Scholar] [CrossRef] [PubMed]
- Dibbern, H.W.; Müller, R.M.; Wirbitzki, E. UV and IR Spectra—Pharmaceutical Substances (UV and IR) and Pharmaceutical and Cosmetic Excipients (IR); Editio Cantor Verlag: Aulendorf, Germany, 2002; ISBN 3-87193-238-8. [Google Scholar]
- Verhoeven, J.W. Glossary of terms used in photochemistry (IUPAC Recommendations 1996). Pure Appl. Chem. 1996, 68, 2223–2286. [Google Scholar] [CrossRef]
- Tamtam, F.; Mercier, F.; Eurin, J.; Chevreuil, M.; Le Bot, B. Ultra performance liquid chromatography tandem mass spectrometry performance evaluation for analysis of antibiotics in natural waters. Anal. Bioanal. Chem. 2009, 393, 1709–1718. [Google Scholar] [CrossRef] [PubMed]
- Botitsi, E.; Frosyni, C.; Tsipi, D. Determination of pharmaceuticals from different therapeutic classes in wastewaters by liquid chromatography–electrospray ionization–tandem mass spectrometry. Anal. Bioanal. Chem. 2006, 387, 1317–1327. [Google Scholar] [CrossRef] [PubMed]
- Wittkamp, B.L.; Hawthorne, S.B.; Tilotta, D.C. Determination of Aromatic Compounds in Water by Solid Phase Microextraction and Ultraviolet Absorption Spectroscopy. 1. Methodology. Anal. Chem. 1997, 69, 1197–1203. [Google Scholar] [CrossRef]
- Merschman, S.A.; Tilotta, D.C. Fiber-Optic Sensor for Aromatic Compounds in Water Based on Solid-Phase Microextraction and Ultraviolet Evanescent Wave Absorption Spectroscopy. Appl. Spectrosc. 1998, 52, 106–111. [Google Scholar] [CrossRef]




| Method | Substance | LoD 1 | Probe Volume | Source |
|---|---|---|---|---|
| UP 2 LC/MS/MS 3 | SMX | 0.5 ng/L | 100 mL | Tamtam 2008 [32] |
| SPE 4 LC/MS/MS | 0.1 ng/L | 1 L | Loos 2007 [29] | |
| SPE LC/MS/MS | 7 ng/L | 50 mL | Botitsi 2006 [33] | |
| Abs. 5 spectroscopy in 1 cm cuvette | 26 µg/L 7 | 2 mL | Jasco “V-700” spectrophotometer | |
| Abs. spectroscopy in ARHCF | 12 µg/L | 10 µL | This paper | |
| UV abs. spectroscopy | Aromatic compounds | 64.900 µg/L | 3.5 mL | Wittkamp 1997 [34] |
| SPME 4 + UV abs. spectroscopy | 0.4.12 µg/L | 50 mL | Wittkamp 1997 [34] | |
| SPME + UV-EWA 6 spectroscopy | 1..18 µg/L | 10 mL | Merschman 1998 [35] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nissen, M.; Doherty, B.; Hamperl, J.; Kobelke, J.; Weber, K.; Henkel, T.; Schmidt, M.A. UV Absorption Spectroscopy in Water-Filled Antiresonant Hollow Core Fibers for Pharmaceutical Detection. Sensors 2018, 18, 478. https://doi.org/10.3390/s18020478
Nissen M, Doherty B, Hamperl J, Kobelke J, Weber K, Henkel T, Schmidt MA. UV Absorption Spectroscopy in Water-Filled Antiresonant Hollow Core Fibers for Pharmaceutical Detection. Sensors. 2018; 18(2):478. https://doi.org/10.3390/s18020478
Chicago/Turabian StyleNissen, Mona, Brenda Doherty, Jonas Hamperl, Jens Kobelke, Karina Weber, Thomas Henkel, and Markus A. Schmidt. 2018. "UV Absorption Spectroscopy in Water-Filled Antiresonant Hollow Core Fibers for Pharmaceutical Detection" Sensors 18, no. 2: 478. https://doi.org/10.3390/s18020478




