Immunoanalysis Methods for the Detection of Dioxins and Related Chemicals
Abstract
: With the development of biotechnology, approaches based on antibodies, such as enzyme-linked immunosorbent assay (ELISA), active aryl hydrocarbon immunoassay (Ah-I) and other multi-analyte immunoassays, have been utilized as alternatives to the conventional techniques based on gas chromatography and mass spectroscopy for the analysis of dioxin and dioxin-like compounds in environmental and biological samples. These screening methods have been verified as rapid, simple and cost-effective. This paper provides an overview on the development and application of antibody-based approaches, such as ELISA, Ah-I, and multi-analyte immunoassays, covering the sample extraction and cleanup, antigen design, antibody preparation and immunoanalysis. However, in order to meet the requirements for on-site fast detection and relative quantification of dioxins in the environment, further optimization is needed to make these immuno-analytical methods more sensitive and easy to use.1. Introduction
Dioxin and dioxin-like compounds, including polychlorinated dibenzo-p-dioxins (PCDDs), polychlorinated dibenzofurans (PCDFs) and polychlorinated biphenyls (PCBs) are a group of chemicals classified as persistent organic pollutants (POPs). Most of them are man-made and released from waste incinerators [1,2] and many other industry processes, such as iron ore sintering, power plants and secondary aluminum smelters [3–5]. They are found ubiquitously in the environment, even in very remote locations, such as the Alaskan Arctic and Antarctica [6], and located in soils, sediments, air and water [7,8]. Because dioxins bioaccumulate in the food chain, they can have serious ecological effects and can impact human health, impairing fertility and development, attacking the immune system, posing neurological risks and causing cancer [9–11]. The multiple long-term health impacts of dioxins have been found in victims of exposure accidents. For example, in 1976, an industrial accident that occurred in Seveso (Italy) exposed a large population to substantial amounts of relatively pure 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD). In early health investigations, chloracne was the only effect established with certainty. In long-term studies, however, an excess of diabetes and cancer cases were associated with dioxin exposure. Additionally, an excess mortality from cardiovascular and respiratory diseases was uncovered [12]. Furthermore, Koopman-Esseboom et al.[13] reported that elevated levels of dioxins and PCBs can alter the thyroid hormone status of pregnant women and their infants. Moreover, Weisglas-Kuperus et al.[14] found that Dutch preschool children who were exposed to PCBs and dioxins persisting into childhood might have a greater susceptibility to infectious diseases. In order to provide guidance on acceptable levels of exposure, the World Health Organization (WHO) has determined a tolerable daily intake of 1–4 pg·kg−1 body weight expressed as dioxin toxic equivalency (TEQ) [15]. TEQ represents total toxicity of a mixture of dioxins, each with its own toxic equivalency factor (TEF), expressing the toxicity relative to the most toxic dioxin congener TCDD. At the same time, scientists have been developing methods to analyze dioxins in environmental and biological samples, resulting in a variety of chemical analytical and bio-analytical detection methods for dioxins [16,17].
In the 1990s, the US Environmental Protection Agency (US EPA) published standard determination methods for dioxins: EPA Methods 1613 and 1618. These methods, viewed as the “gold standard”, employ high-resolution gas chromatography/mass spectrometry (HRGC/HRMS). They are considered to be the most reliable and sensitive methods for determining the congener-specific concentrations of dioxins. These methods were also adopted and standardized by other counties and organizations, such as Japan, WHO and European Union, but HRGC/HRMS has obvious disadvantages. First, this analytical method can only be performed by highly trained specialists. Second, it is very time-consuming. Usually, it takes three or four days to analyze one sample. Third, it requires extremely complicated and expensive pretreatments, costly instrumentation and high daily running cost to maintain a standardized laboratory. Between 1970 and 1993, it has been estimated that more than one billion US$ was spent on determining the toxicity of PCDD/PCDFs in samples in America [18]. Currently, it is estimated that one dioxin analysis costs in the range from $900 to $1,800. Moreover, there is an easily ignored problem on how to treat the large amount of solid matter and liquids polluted by dioxins during the analysis process. For example, analysis of one soil sample usually produces about four liters of liquid waste, and due to the high expense and complex detection procedures, it is hard to perform high throughput dioxin screening in large scale environmental surveillance using instrumental analysis. Therefore, it is urgent to develop more rapid, simple, cost-effective and sensitive methods. With the development of biotechnology, bioassays which could be rapid, simple and offer moderate sensitivity are being developed to detect dioxins [19]. Similar to instrumental analyses, TEQ of a particular class of dioxins can be analyzed and estimated according to the concentrations obtained by these methods (Table 1). The present bioassays of dioxins are divided into two categories: one is based on the aryl hydrocarbon receptor (AhR) signaling pathway activated by dioxins; the other, also called immunoassay, is based on antibodies. Because the former is correlated to the toxicological effects of dioxins in cells, making it physiologically relevant, total TEQ of dioxins in samples can be obtained. The latter, except for Ah-immunoassay (Ah-I) using antibodies to recognize AhR directly, use specific antibodies to identify dioxins, so it is not able to directly reflect the total toxicological and biological effects of mixtures of dioxins.
The detection methods for dioxin bioassays include quantitative polymerase chain reaction (Q-PCR) for CYP1A1 mRNA expression, the 7-ethoxyresorufin-O-deethylase (EROD) bioassay, the aryl hydrocarbon hydroxylase (AHH) bioassay, chemical-activated luciferase gene expression (CALUX), chemical-activated green fluorescent protein luciferase gene expression (CAFLUX), the gel retardation of AhR (GRAB) assay, cell proliferation-based assay, DNA binding assay, AhR ligand binding, enzyme-linked immunesorbent assay (ELISA), radioimmunoassay (RIA), dissociation enhanced lanthanide fluorescence immunoassay (DELFIA) and Ah-I (Table 1). Some of them have been standardized by different countries and organizations, such as the United States of America, Japan and European Union (Table 2). Each of these methods has its advantages and drawbacks. EROD is not useful for detecting trace dioxins due to its low sensitivity. Though CALUX is considered to be the best bio-analytical method for detecting TEQ concentrations of dioxins due to its high sensitivity and rapid detection [30], cell culture and other cellular biology facilities are necessary. CAFLUX does not require addition of an external substrate, unlike CALUX, but its background noise is high, which often prohibits its use. GRAB assay uses a 32P labeled DNA probe, requiring a radioactivity use license and facility and is mainly used in research. ELISA and RIA are based on the interaction of antigen and antibody therefore they have relative high specificity, and have the added benefit that cell culture is not required, unlike EROD or CALUX, but RIA requires antibodies labeled with 125I or 32P, again requiring a radioactivity use license, and is seldom employed. DELFIA is similar to the ELISA assay, but is more sensitive. A major drawback is its expense due to the requirement for special antigens labeled with lanthanide. An added concern is the risk of lanthanide ion contamination. Differing from the other antibody-based assays, the Ah-I assay, which is designed based on AhR signaling pathway, allows an estimation of the TEQ of mixtures of dioxins. All of these antibody-based bioassays require expensive antibodies. Despite this, antibody-based methods are the simplest, have moderate sensitivity, and are less expensive than HRGC/HRMS, leading to the development of commercial ELISA and Ah-I kits which have been utilized for dioxins screening [46–51]. At the same time, in order to improve detective sensitivity, several detection technologies can be combined as multi-analysis immunoassays, which will be described in detail later.
Developing detection methods suitable for screening dioxins on-site is of great importance for dealing with exposure accidents and daily surveillance. To fulfill the special requirement for on-site fast screening, the detection system must be simple, rapid, sensitivity and practical. Thus, bioassays based on antibodies may be the best suited for this purpose. Therefore, the present review focuses on several practical bio-analytical methods based on antibodies for dioxins such as ELISA, Ah-I and other multi-analysis immunoassays. First, the methodology and optimization of ELISA are summarized, in terms of the samples preparation, antigen design and antibody productions. In the following part, the Ah-I bioassay and its applications are introduced. Finally multi-analyte immunoassays are described. Other screening methods based on antibody-antigen reactions have been highlighted in numerous excellent reviews [19,52,53].
2. Detection Methods
2.1. ELISA
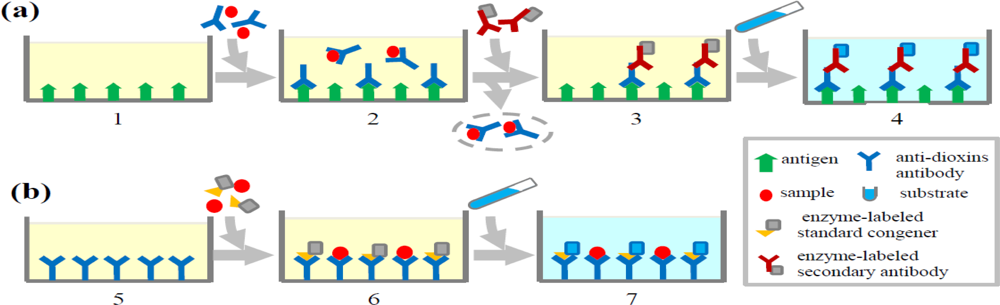
ELISA is based on the specific reaction between dioxins and their antibodies. Currently the predominant ELISA systems used for detecting dioxins are direct competitive and indirect competitive ELISAs which are illustrated in Figure 1. The basic principle of competitive ELISA is that dioxins in environmental and biological samples compete with standard congener conjugated with enzyme for binding to antibody against a certain class of dioxin congener immobilized on a microplate. The substrate reacts with enzyme-linked standard congener and gives absorbance which reflects the amount of conjugated standard congener binding to the antibody on the plate. The absorbance is inversely in proportion to the amount of dioxins present in the tested samples. The concentrations of dioxins in the samples are calculated by comparison with a standard curve generated by addition of known amounts of standard dioxin.
Indirect competitive ELISA is based on the competition between dioxins in samples and antigens immobilized on the microplates for binding to antibodies that recognize a certain class of dioxin congener. After washing to remove soluble antigen-antibody complexes, the antibody bound to the antigen on the plate is then incubated with secondary antibody conjugated to an enzyme which can subsequently react with substrate to give an absorbance. Like the direct competitive ELISA, dioxin levels in the samples can be evaluated by comparison with a standard curve. Although ELISA is a simple assay for determination of dioxins, its respective lack of sensitivity and specificity compared to CALUX and instrumental analyses limits its applications. To make improvements, several aspects should be considered, including the optimization of sample preparation and improvement of the specificity of antibodies against dioxins. The latter is related to the haptens chosen for antigen design and the technology for generating antibodies.
2.1.1. Sample Preparation
Sample preparation is important for concentrating the dioxins in the mixture, removing potential interfering ingredients and improving the sensitivity of the assay. HRGC/HRMS requires exhaustive extraction and expensive cleanup for sample preparation which is expensive and time-consuming, while the sample preparations for ELISA assays are usually fast, simple and cost-effective. Moreover, new methods for sample extraction and cleanup are being developed for better determination of dioxins using ELISAs.
Sample Extraction
Samples from different sources are accompanied by distinct complex matrices, requiring different extraction methods. In the early days, soil and sediment samples were easily extracted by shaking with methanol or/and Soxhlet [47,54,55] or using an ultrasonic process [56], but these methods did not result in a complete extraction, leading to compound loss or the need for further cleanup. In order to solve these problems, several alternative extraction technologies were introduced, which resulted in time and reagent savings. Microwave energy has been used to extract organic compounds from contaminated soils for many years. Microwave-Assisted Extraction (MAE) is fast (i.e., the sample is statically extracted within 10–15 min) and solvent-saving (i.e., 30 mL per sample as compared to 300 mL for instrumental analysis). Lopez-Avila et al.[57] detected PCBs in soils and sediments with this method. Five gram soil samples were subjected to extractions with only 30 mL hexane-acetone (1:1), and the extraction was performed for only 10 min. The subsequent analyses showed a good match between MAE-ELISA and MAE-GC/ECD results with a correlation coefficient over 0.97.
Supercritical fluid extraction (SFE) is an alternative efficient extraction for some special samples such as oily soils. It is a technique combining the liquid-like solvating and gas-like transport properties of supercritical fluids. Because pure carbon dioxide (CO2) is used as solvent, extractions are clean enough for direct reconstitution in a solvent compatible with ELISA [58–61]. For example, Johnson et al.[51] studied the extraction of PCBs from oily soil using both a methanol shake extraction and a methanolic Soxhlet extraction, but found that the ELISA results disagreed with the GC confirmatory data. However, SFE with high pressure/temperature regime coupled to the same ELISA, resulted in a good agreement between the results obtained by GC and by ELISA. Additionally, Bøwadt et al.[62] also examined PCBs in soils using a SFE method without the need for cleanup steps after extraction. The following ELISA performed under laboratory conditions gave reasonable agreement with GC/ECD for determining total PCBs.
Accelerated solvent extraction (ASE) uses a conventional solvent to extract samples, but at elevated pressure and temperature. Like MAE, it is also fast and consumes only small amounts of solvent. Soil samples heavily contaminated by PCDD/Fs from small-scale industrial waste combustion sites in Uruguay, wood treatment sites in Sweden and a Swedish chloralkali site were put into 33 mL stainless steel extraction cells filled with solvents. The toluene fractions, containing the PCDD/Fs, were obtained in 43 min, which is considerably faster than using Soxhlet extraction with toluene (15 h). The ELISA results were strongly correlated with HRGC/HRMS results (correlation coefficient, 0.90) [63]. Schrock et al.[64] also used ASE to extract sediments and soils contaminated with PCDD/Fs. The extractions were carried out at 13.8 MPa (2,000 psi), 125 °C and three static cycles of 10 min each. There was also a high correlation between ELISA and GC results, with a coefficient at 0.99.
Selective pressurized liquid extraction (SPLE) is another relatively new technique. In this method, sulphuric acid-impregnated silica was used for the lipid free extraction of compounds by pressurized liquid extraction. Similar to SFE, it also reduced the need for post-extraction cleanup. For instance, Chuang et al.[65] adopted this method to extract sediments and soils contaminated by PCDD/Fs. The ELISA results demonstrated that this method was an effective extraction method for a wide range of PCDD/F concentrations in sediments and soils without the need for further cleanup.
For contaminated oil samples, the extraction is usually less complicated. For example, Samara et al.[66] studied industrial source oil samples contaminated by PCDD/Fs, which were cleaned up by means of Soxhlet extraction with methylene chloride (3.5 h) under restricted exposure to light. In addition, Glass et al.[67] reported a less complicated simple two-step liquid-liquid extraction which was used to detect PCBs in transformer oil, resulting in no false negatives at a 1.4 ppm nominal cutoff. Moreover, using a simple oxidation of the sample extract (including 1 mL acetonitrile, 0.5 mL isooctane, 1 mL H2SO4 and 8 mL “Quenching Buffer”) to analyze PCBs in waste oil, ELISA results were highly correlated with the results obtained by instrumental analysis [68].
With biological samples, different extraction methods are available for obtaining reliable ELISA results. Wiberg et al.[69] used SPLE to treat food and feed samples contaminated by PCDD/Fs and dioxin-like PCBs. The ELISA results showed good agreement with HRGC/HRMS. Similar to environmental samples, SPLE also reduced time and solvent consumption compared to instrumental methods. In addition, different sample preparation methods were used for fish. Fish samples were mixed with sodium sulfate, extracted with methylene chloride and fortified with the surrogate congener [70]. Tsutsumi et al.[71] reported another method for sample preparation of retail fish tissue, in which the homogenized fish sample was first subjected to alkali digestion, and then, with addition of methanol, it was extracted with hexane and finally washed with aqueous NaCl. The ELISA results correlated well with TEQ concentrations for dioxin-like PCBs obtained by HRGC/HRMS (r = 0.92, n = 26). Invertebrate animals such as mussels exposed to PCBs were also studied by Fillmann et al.[72]. The samples were Soxhlet extracted in hexane-dichloromethane (1:1), and the extracts were then concentrated to a few mL using rotary evaporation followed by pure nitrogen “blow down” The ELISA results indicated that it was a rapid and efficient extraction for detecting total PCBs in mussels.
Cleanup
In order to make the extracted sample compatible with ELISA assays, samples must be re-dissolved in dimethyl sulfoxide (DMSO) before assay. In addition, cleanup is usually needed after extraction to eliminate interfering ingredients. Several general chemical methods have been used for the cleanup. Tsutsumi et al.[73] cleaned up sample extracts from retail fish contaminated by dioxin-like PCBs by passing them through a multi-layer silica gel column and an alumina column, resulting in eluents suitable for ELISA assays. Deng et al.[55] cleaned soil and sediment samples on a column containing activated Florisil [74]. Nichkova et al.[56] utilized a multilayered silica gel column and an activated carbon column to clean up extracted sediment samples contaminated by PCDD/Fs. In addition, Van Emon et al.[75] used the same method to refine dioxins-contaminated sediment and soil sample extracts, following the EU standard methods. For the cleanup of samples extracted from oils, KOH-ethanol/sulphuric acid is suitable for oil samples containing > 35 μg·mL−1 PCB [76].
For biological samples containing extremely low concentrations of dioxins, such as human milk, Sugawara et al.[77] reported that routine purification with nonpolar solvents was incompatible with ELISA assays. They suggested extracting human milk samples by employing an alkali decomposition step, then passing the samples through a three-layer column, and finally evaporating the eluent with nitrogen before re-dissolving in MeOH-DMSO (1:1) with 100 ppm Triton X-100. A fairly good correlation between ELISA results and GC/MS results was obtained with a coefficient of 0.92.
As an alternative to traditional chemical cleanup methods, immunoaffinity purification (IAP) is a powerful biochemical technique for a single-step separation and purification of individual compounds or classes of compounds from liquid matrices [78–80]. IAP is based on the highly specific interaction of an antigen in the samples with its antibody immobilized on a solid support matrix. It was originally applied to pharmaceutical and biomedical trace analysis and in recent decades for analysis of environmental contaminants and pesticide residues [81,82]. Altstein et al.[83] first coupled ELISA with the sol-gel-based IAP method for cleanup of soil and sediment samples for determination of PCB 126, demonstrating that this method was very efficient in purifying PCB 126 and eliminating matrix interferences. Shelver et al.[84] also used an immunoaffinity column for cleanup of TCDD containing serum samples. The binding efficiencies of a spiked serum sample combined with the immunoaffinity column were over 90%.
2.1.2. Immunogen Design for PCDD/Fs and PCBs
The specificity and selectivity of ELISAs mainly depend upon the antibodies used. Many studies show that the design of the immunogens determines the features of the resulting antibodies [85–88]. Since small molecules seldom stimulate an immune response, it is necessary to design a hapten which can be covalently combined with a carrier protein to make a complex for immunization. Usually two aspects of a hapten are related to the immunogenicity: chemical structure and functional group(s). The chemical structure is sometimes the key to produce valuable antibody. The functional groups (e.g., NH2, COOH) help to combine chemical structures with carrier proteins by covalent coupling.
A lot of effort has been made to detect various PCB congeners. Chiu et al.[89] designed a hapten (6-[(3,3′,4′-trichlorobiphenyl-4-yl)oxy]hexanoic acid) coupled to bovine serum albumin (BSA) by which an appropriate monoclonal antibody for sensitive detection of PCB congeners or mixtures was obtained. The ELISA using the antibody was highly selective for PCB 77 and PCB 126, with 50% inhibition concentration (IC50) values of 0.9 and 1.2 ng·mL−1, respectively. Altstein et al.[83] used a 3,3′,4,4′-trichloro-(3-thiobiphenyl) hapten linked to keyhole limpet hemocyanin by means of 6-bromohexanoic acid to obtain a goat 2504 anti-PCB polyclonal antibody. The goat anti-PCB IgGs exhibited the highest affinity and lowest detection limit for PCB 77 and did not cross react with any other tested PCBs, Aroclors or furans in any of the ELISA formats. Fránek et al.[90] synthesized PCB 126-(CH2)3-hapten mimic-BSA and PCB 77-CH2-hapten mimic-peroxidase. The antibody was highly specific for non-ortho-substituted (coplanar) congeners, with low cross-reactivity values for PCB 189 and PCB 77 (1.3 or 1.0%, respectively) and did not recognize non-coplanar PCB congeners or organochlorine compounds. Fránek et al.[91] also synthesized a 4,4′-dichlorobiphenyl- and 3′,4,4′,5-tetrachlorobiphenyl-2-azo-BSA immunogen and obtained a sheep polyclonal antibody. Direct competitive ELISA results showed IC50 values for the commercial PCB mixture, Delor 103,104,105 and 106 (corresponding to Aroclor 1242, 1248, 1254 and 1260), were about 200, 250, 600 and 900 pg·L−1, respectively, and the cross-reactivity to Delor 103, 104, 105 and 106 was 100, 79.6, 31.7 and 13.5%, respectively. Furthermore, a 3,3′,5,5′-tetrachlorobiphenoxybutyric acid (PCBH)-keyhole limpet hemocyanin conjugate was also prepared by Inui et al.[92]. The resulting monoclonal antibodies, 0217 and 4444, were highly selective for 3,3′,5,5′-tetrachlorobiphenyl (PCB 80) in ELISA assays with IC50 values of 2.6 and 0.46 ng·mL−1, respectively. Moreover, Shan et al.[74] designed a hapten (7,8-dichlorobenzo[5,6][1,4]dioxino[2,3-b]pyridine-3-carboxylic acid) conjugate with BSA and obtained a sensitive polyclonal antibody for screening TCDD from soil and biota samples.
2.1.3. Development of Antibodies for PCDD/Fs and PCBs
Among various bioassays for dioxin determination, one of the outstanding advantages of immunoassays is their specificity, which depends on the characteristics of the antibodies recognizing target analytes (antigens). To date there are several successful antibodies being used for ELISA assays of PCDD/Fs in environmental samples. Early work in this area has been summarized by Sherry et al. [93] and Harrison et al. [94]. So far there are three main kinds of antibodies used in the ELISA: polyclonal antibodies, monoclonal antibodies and recombinant antibodies. Polyclonal antibodies are widely used and easy to be obtained and purified from serum. For example, Mapes et al. [54] utilized New Zealand white rabbit to raise polyclonal antibodies against PCBs in the development of a fast on-site immunoassay. By using this assay PCBs in soil could be detected at concentrations as low as 5 ppm. The results of cross-reactivity studies showed that the assay was highly specific. Chuang et al. [50] also used pooled rabbit anti-PCB antiserum (AC-3) to determine PCBs in soil, and obtained a correlation coefficient between GC/ECD and ELISA of 0.91 for 41 soil samples. In addition, a sheep polyclonal antibody was developed, which had no cross-reaction with 19 other structurally related compounds, and only 5% cross-reactivity with dichlorodiphenyledichloroethylene (DDE) [91]. Deng et al. [55] also applied the sheep antiserum raised against 4,4′-dichlorobiphenyl- thyroglobulin immunogen to successfully evaluate eight PCB-contaminated soil samples. Besides polyclonal antibodies used for detection of PCB congeners, Carlson et al. [95] used a novel anti-PCDD/Fs polyclonal antiserum to measure TEQ of PCDD/Fs from soil and fly ash samples.
It is generally accepted that polyclonal antibodies are not absolutely specific and often exhibit cross-reactivity with other molecules. In the study of Harrison et al.[96], a competitive ELISA assay based on a mouse monoclonal antibody, which was specific for TCDD and related congeners, was documented. The sensitivities were 100 pg/tube or 25 pg/well of TCDD. Moreover, Tsutsumi et al.[71] used a monoclonal antibody, which was highly specific for 2,3′,4,4′,5-pentachlorobiphenyl (PCB 118), to determine contamination of retail fish. The ELISA results for the fish samples correlated well with the TEQ concentrations of dioxin-like PCBs obtained by GC/MS (r = 0.92, n = 26).
In most cases, cross reactivity is viewed as undesirable, and minimizing it is a common goal. However, since a mixture of different classes of dioxin congeners is always present in environmental samples, a general environmental screening of dioxins requires detection of certain classes of compounds rather than individual congeners. Therefore cross reactivity among different classes of the dioxin congeners, rather than individual congeners, is sometimes preferably considered in the development of immunoassays for dioxin screening. A new strategy to raise suitable antibodies has been put forward based on the notion of “generic” antibodies with relatively broad cross reactivity to targeted compounds [97]. This approach involves either screening of antibodies raised against a family of target compounds or generation of specially designed analogues, often based on a fragment of the target compounds aiming at presenting the pivotal features of the entire class [98,99]. Some researchers have described the use of mixed antibodies to increase the working range of an immunoassay for detecting herbicides [100,101]. A similar idea has been used to develop immunoassays for dioxins. For instance, Glass et al.[102] first described an immunoassay for PCBs in which two different monoclonal antibodies. As a supplement, cross reactivity was used to broaden the assay reactivity. Antibodies that individually exhibit 20 or 50-fold differences in response between Kanechlor 300 and Kanechlor 600 can be mixed so that the overall difference in response to those compounds was reduced to three fold. The reported cross reactivity of a PCB immunoassay was increased, resulting in an assay system which was better able to detect total PCBs.
Polyclonal and monoclonal antibodies dominated the dioxin screening field in the past and will continue to do so in the near future. But recent years, some researchers have begun to study recombinant monoclonal antibodies. The technology for producing recombinant monoclonal antibodies is not novel, originating in the medical field for generating humanized antibodies [103–105]. Before being used in dioxin mornitoring, it was introduced for screening pesticide [106–108]. Recombinant monoclonal antibodies are created from cDNAs encoding available monoclonal antibodies or taken from human cells, then cloning them into expression vectors for production in suitable organisms such as Escherichia coli or yeast. The preparation is highlighted in an excellent review [109]. Recombinant monoclonal antibodies have some advantages over conventional polyclonal antibodies and monoclonal antibodies. They are stable and have high affinity and low production cost. However, the procedure for their production is very complicated and requires highly specialized bioengineering techniques. The main advantage of recombinant monoclonal antibodies for ELISA analysis is its high tolerance towards solvents such as methanol and DMSO, alleviating the usual problem of having to greatly dilute the samples before assay [72,110]. Although some studies reported that ELISA system for coplanar PCBs were tolerant to organic solvents such as 5% of methanol and 1.3% of DMSO [89,90], these tolerances still decreased sensitivity for PCBs with standard antibodies.
2.2. Ah-I
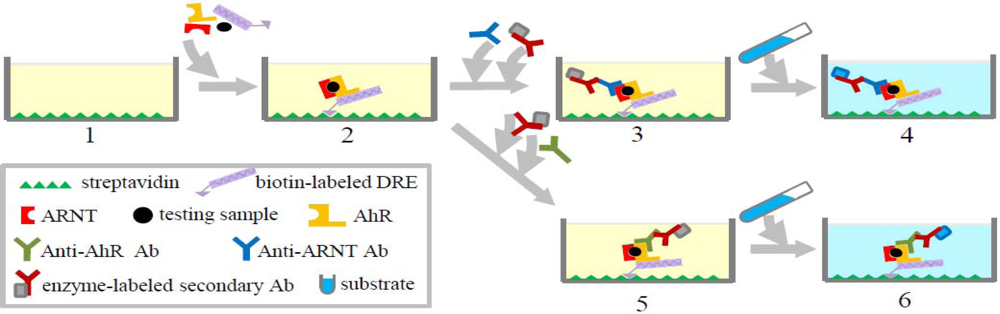
Ah-I is another common method to detect total toxicity potential of dioxins based on the ability of these compounds to bind to and activate AhR followed by downstream signaling. For determining TEQ concentrations of dioxins, reporter gene assays, such as CALUX, are currently regarded as the best method, especially for food and feed [30]. However, this still has some drawbacks, including the need for cell culture, which requires skilled personnel and elaborate equipment, and licensing. Ah-I, which is an ELISA-based AhR binding assay, is a simpler alternative to obtain TEQ directly that does not require cell culture. It is a hybrid of an immunoassay and an in vitro AhR-binding assay. The principle of this method is as follows: activation of AhR by dioxin or dioxin-like compounds leads to conformational changes, allowing the formation of a heterodimer with ARNT which then binds to dioxin receptor elements (DREs) in DNA. This AhR:ARNT heterodimer can be detected by an immunoassay-based color reaction using an enzyme-conjugated antibody that binds to the ARNT or AhR. The assay, which converts into TCDD equivalents using a TCDD standard curve, can detect the total TEQ concentrations of the dioxin-like compounds. The detailed procedure of this method is shown in Figure 2. The sensitivity and specificity of Ah-I are determined by sample preparation methods as well as by the features of the antibody used in the assay.
Like all of the bioassays based on antibodies, Ah-I also requires expensive antibodies, limiting its applications. One of the main advantages of this method is that it is less likely to produce false-negative results than cell-based assays, such as the CALUX, because Ah-I is based on very specific antigen-antibody recognition. For example, PCB 118, which is a good example of mono-ortho PCBs in certain kind of fish, was reported to be relatively less responsive in the CALUX assay [111–113], which may lead to underestimation [114]. However, according to studies of Tsutumi et al.[45], amounts of PCB 118 detected by Ah-I were consistent with the results of GC/MS analysis. On the other hand, sample cytotoxicity resulting from toxic matrices or solutions, may generate false negative results in CALUX, but in are less likely to interfere with the cell-free Ah-I assay. So far the detection limit of Ah-I assay for PCBs was 10 fmol.
2.3. Multi-Analyte Immunoassay
ELISAs have been generally considered to be rapid, simple and low-cost analytical procedures and used to detect dioxins for years. However, the sensitivity of these methods is limited. Using modified extraction and cleanup methods, the limit of detection was 28 ± 6 pg TMDD·mL−1 DMSO [2,3,7-trichloro-8-methyl-dibenzo-p-dioxin (TMDD)] [63]. For CALUX, the limit of detection was 50 fg of TCDD [112]. Therefore it is difficult to use ELISA assays for the evaluation of biological samples containing dioxins at extremely low concentrations, such as in human milk and blood. Since improvements in the ELISA assay itself may be not enough to make the assay suitable to detect trace amount of dioxins, novel detection systems consisting of other bio-analytical methods and ELISA have been developed for the purpose.
Real-time immuno-polymerase chain reaction (Rt-IPCR) is a versatile and robust technique whereby the exponential amplification ability of PCR is coupled to the detection of proteins by antibodies in an ELISA format. Briefly the antigen is coated on PCR tubes, and then anti-dioxin antibodies and samples containing dioxins are added. The antigen competes with dioxins in the sample for binding to the antibodies. Biotinylated secondary antibody is added to bind to anti-dioxin antibodies immobilized on the tube. After addition of avidin and biotinylated DNA tag, a complex is formed between avidin and the biotinylated secondary antibody and DNA. The DNA tag is then amplified and quantified by Rt-PCR. Rt-IPCR has been widely used in clinical diagnosis and biochemical analysis for detection of pathological proteins and various viral antigens [115–117]. Recently, it has been introduced to detect environmental contaminants, such as dioxin. For example, Chen et al.[118] first developed a fluorescent quantitative IPCR and assessed its potential for the detection of antibodies recognizing PCBs in soil samples, in which the PCB concentrations were as low as 10 fg·mL−1. Recovery rates were 95.0–105.0% and the Rt-IPCR results correlated well with the concentration of PCBs obtained by GC/MS (r = 0.99, n = 6). These data indicate that this highly specific, sensitive, and robust assay could serve as a potential tool for detecting PCB compounds in the environment. However, there are several disadvantages of Rt-IPCR that needed to be mentioned. Above all, it is hard to avoid laborious preparation to stably couple the DNA tag to the antibody. In addition incubation and washing steps are laborious compared to other competitive immunoassays. Finally, the sensitivity of Rt-IPCR is limited by antibody-antigen interaction.
Although ELISAs are rapid, these methods still require an hour at least for a single measurement and need enzyme-labeled reagents. Surface Plasmon Resonance (SPR)-based biosensor immunoassays (BIAs) using Biacore instruments can eliminate the need for enzyme-labeled reagents and are mostly highly automated. BIA is mainly used to detect the residues of veterinary drugs in foodstuffs, but recently it was introduced to screen dioxins. Using this method, Shimomura et al.[119] detected TCDD and PCB with respective detection limits of 0.1 and 2.5 ng·mL−1, requiring 15 min for a single sample measurement. The first food monitoring program using BIA was reported by Tsutsumi et al.[70]. In this study, an SPR-based BIA was developed using a monoclonal antibody that is specific for PCB 118, by which TEQ concentrations of dioxin-like PCBs in retail fish was obtained. The quantitative limit of the assay was 1 ng PCB 118 per gram of test samples and the assay results correlated well with GC/MS (r = 0.89). Thus, the BIA will be useful for the preliminary screening of large numbers of fish samples before GC/MS. However, BIA is not appropriate for detecting high concentrations of dioxins as it takes too long to complete dissociation of magnetic particles from the gold film which acts as a SPR tag. Another drawback is that this method requires expensive equipment and supplies.
3. Conclusions
Compared to GC/MS detection of dioxins, bioassays based on antibodies such as ELISA and Ah-I are more rapid, time-saving, cost-effective and practical. The cost-effective features of the bioassays are not only due to the relative low cost of the establishment of the detection system, but also because of the low cost for the maintenance of the instruments and sample preparation and detection. However, their drawbacks in sensitivity and specificity preclude them as a replacement for the instrumental method. In order to improve sensitivity and specificity, efforts have been made to optimize the sample preparation, hapten design and development of the antibodies. Combinations of other techniques with the antibody-based bioassays have been proposed to improve the performance in screening dioxins. For sample extractions, MAE, ASE, SPE and SPLE methods have been proposed, among which SPE and SPLE can save the cleanup step for the extracted samples; MAE and ASE are fast and use little solvent. For the cleanup of the extracted samples, multilayered columns and an activated carbon or IP are easy and effective methods. In order to obtain suitable antibodies, a well-designed hapten is essential. In general, polyclonal antibodies are easier to obtain, but monoclonal antibodies are more specific. ELISA assays using a mixture of various monoclonal antibodies can be employed to screen different classes of dioxin congeners. In addition, recombinant monoclonal antibodies can be employed in ELISA to improve the feasibility of the assay. Compared to single-analytic antibody-based analysis, multi-analytic analyses are able to not only further improve the sensitivity and specificity of the detections, such as Rt-IPCR; but also make the assays more rapid and automatic, such as BIAs. With the cell-free, simple and rapid features, these antibody-based bioassays could be feasible for direct on-site screening for dioxins. To make good use of all the optimizations, for example, dioxins in soils and sediments could be screened on-site by using MAE extraction and IA cleanup to prepare samples, followed by Ah-I to estimate the total TEQ. If the TEQ is relative high, ELISA can be adopted to determine different classes of the dioxins. If the TEQ is relative low, it is better to adopt Rt-IPCR. Alternative sample preparations using SPE or SPLE combined with BIAs can be used to screen a sufficient number of samples in a short time.
Ah-I can be used for specific applications. Although Ah-I can provide TEQ directly in a cell-free system, it has an obvious drawback in its specificity. Recently, more and more AhR ligands have been found which have diverse structures. The emerging evidence suggests that various ligands of AhR may bind to AhR in different ways [120] and these new findings suggest a possibility that once AhR is activated by different ligands, it may undergo distinct conformational changes. If there is an antibody specifically recognizing a certain conformational change induced by certain ligand, it would be possible to increase the specificity of Ah-I. The question is how to obtain such antibodies. So far antibodies used in Ah-I assays are mainly raised against short peptides derived from the AhR protein. Using this method to generate antibodies will result in a smaller probability of obtaining antibodies against special conformational changes compared to using full-length AhR as an antigen. With the further optimization, it may be possible to make these immune-analysis methods more sensitive and feasible to meet the requirements for on-site fast detection and relative quantification of dioxins in the environment.
Acknowledgments
This study was supported by Chinese Academy of Sciences Key Program of Knowledge Innovation (KZCX2-EW-411), National Natural Science Foundation of China (21177150), and National 863 Project (2012AA062804).
References
- Sakai, S.; Hiraoka, M.; Takeda, N.; Shiozaki, K. Coplanar PCBs and PCDDs/PCDFs in municipal waste incineration. Chemosphere 1993, 27, 233–240. [Google Scholar]
- Weber, R.; Takasuga, T.; Nagai, K.; Shiraishi, H.; Sakurai, T.; Matuda, T.; Hiraoka, M. Dechlorination and destruction of PCDD, PCDF and PCB on selected fly ash from municipal waste incineration. Chemosphere 2002, 46, 1255–1262. [Google Scholar]
- Aries, E.; Anderson, D.R.; Ordsmith, N.; Hall, K.; Fisher, R. Development and validation of a method for analysis of “dioxin-like” PCBs in environmental samples from the steel industry. Chemosphere 2004, 54, 23–31. [Google Scholar]
- Wang, J.B.; Hung, C.H.; Hung, C.H.; Chang-Chien, G.P. Polychlorinated dibenzo-p-dioxin and dibenzofuran emissions from an industrial park clustered with metallurgical industries. J. Hazard. Mater 2009, 161, 800–807. [Google Scholar]
- Wang, Z.F.; Ding, Q.; Wang, K.X.; Wu, C.M.; Qu, Y.H.; Zhao, X.D. Study on dioxin emission for typical non-wood pulp making in China. Huan Jing Ke Xue 2012, 33, 574–579. [Google Scholar]
- Bogillo, V.; Bazylevska, M. Variations of organochlorine contaminants in Antarctica. NATO Sci. Peace Secur 2008, 3, 251–267. [Google Scholar]
- Caserini, S.; Cernuschi, S.; Giugliano, M.; Grosso, M.; Lonati, G.; Mattaini, P. Air and soil dioxin levels at three sites in Italy in proximity to MSW incineration plants. Chemosphere 2004, 54, 1279–1287. [Google Scholar]
- Kumagai, S.; Koda, S.; Miyakita, T.; Ueno, M. Polychlorinated dibenzo-p-dioxin and dibenzofuran concentrations in serum samples of workers at intermittently burning municipal waste incinerators in Japan. J. Occup. Environ. Med 2002, 59, 362–368. [Google Scholar]
- Bertazzi, P.A.; Consonni, D.; Bachetti, S.; Rubagotti, M.; Baccarelli, A.; Zocchetti, C.; Pesatori, A.C. Health effects of dioxin exposure: A 20-year mortality study. Am. J. Epidemiol 2001, 153, 1031–1044. [Google Scholar]
- McGregor, D.B.; Partensky, C.; Wilbourn, J.; Rice, J.M. An IARC evaluation of polychlorinated dibenzo-p-dioxins and polychlorinated dibenzofurans as risk factors in human carcinogenesis. Environ. Health Persp 1998, 106, 755–760. [Google Scholar]
- Johnson, J.E.; Heckathorn, W.D.; Thompson, A.L. Dispersal and persistence of 2,3,7,8-tetrachlor-odibenzo-p-dioxin (TCDD) in a contaminated aquatic ecosystem, Bayou Meto, Arkansas. Transact. Am. Fish. Soc 1996, 125, 450–457. [Google Scholar]
- Bertazzi, P.A.; Bernucci, L.; Brambilla, G.; Consonni, D.; Pesatori, A.C. The Seveso studies on early and long-term effects of dioxin exposure: A review. Environ. Health Persp 1998, 106, 625–633. [Google Scholar]
- Koopman-Esseboom, C.; Morse, D.C.; Weisglas-kuperus, N.; Lutkeschipholt, I.J.; Van Der Paauw, C.G.; Tuinstra, L.G.M.T.; Brouwer, A.; Sauer, P.J.J. Effects of dioxins and polychlorinated biphenyls on thyroid hormone status of pregnant women and their infants. Pediatr. Res 1994, 36, 468–473. [Google Scholar]
- Weisglas-Kuperus, N.; Patandin, S.; Berbers, G.A.M.; Sas, T.C.J.; Mulder, P.G.H.; Sauer, P.J.J.; Hooijkaas, H. Immunologic effects of background exposure to polychlorinated biphenyls and dioxins in Dutch preschool children. Environ. Health Persp 2000, 108, 1203–1207. [Google Scholar]
- Van Leeuwen, F.X.R.; Feeley, M.; Schrenk, D.; Larsen, J.C.; Farland, W.; Younes, M. Dioxins: WHO’s tolerable daily intake (TDI) revisited. Chemosphere 2000, 40, 1095–1101. [Google Scholar]
- Eppe, G.; Focant, J.F.; Pirard, C.; De Pauw, E. PTV-LV-GC/MS/MS as screening and complementary method to HRMS for the monitoring of dioxin levels in food and feed. Talanta 2004, 63, 1135–1146. [Google Scholar]
- Garrison, P.M.; Tullis, K.; Aarts, J.M.M.J.; Brouwer, A.; Giesy, J.P.; Denison, M.S. Species-specific recombinant cell lines as bioassay systems for the detection of 2,3,7,8-tetrachlorodibenzo-p-dioxin-like chemicals. Toxicol. Sci 1996, 30, 194–203. [Google Scholar]
- Vandenheuvel, J.P.; Lucier, G. Environmental toxicology of polychlorinated dibenzo-p-dioxins and polychlorinated dibenzofurans. Environ. Health Persp 1993, 100, 189–200. [Google Scholar]
- Behnisch, P.A.; Hosoe, K.; Sakai, S. Bioanalytical screening methods for dioxins and dioxin-like compounds—A review of bioassay/biomarker technology. Environ. Int 2001, 27, 413–439. [Google Scholar]
- Sahlberg, C.; Peltonen, E.; Lukinmaa, P.L.; Alaluusua, S. Dioxin alters gene expression in mouse embryonic tooth explants. J. Dent. Res 2007, 86, 600–605. [Google Scholar]
- Cariou, R.; McAlister, D.R.; Marchand, P.; Fern, M.J.; Antignac, J.P.; Le Bizec, B. Influence of the solvent quality on the AhR mediated Procept® assay measurement of dioxin and dioxin-like compounds. Talanta 2010, 80, 2063–2067. [Google Scholar]
- Orrego, R.; Jimenez, B.; Bordajandi, L.R.; Gavilan, J.F.; Inzunza, B.; Abad, E.; Gonzalez, M.J.; Rivera, J.; Barra, R. EROD induction and PCDD/F levels in fish liver from the Biobio River in Chile. Chemosphere 2005, 60, 829–835. [Google Scholar]
- Jonsson, M.E.; Brunstrom, B.; Brandt, I. The zebrafish gill model: Induction of CYP1A, EROD and PAH adduct formation. Aquat. Toxicol 2009, 91, 62–70. [Google Scholar]
- Pastor, D.; Ruiz, X.; Barcelo, D.; Albaiges, J. Dioxins, furans and AHH-active PCB congeners in eggs of two gull species from the western Mediterranean. Chemosphere 1995, 31, 3397–3411. [Google Scholar]
- Schulz, T.G.; Neubert, D.; Davies, D.S.; Edwards, R.J. Inducibility of cytochromes P-450 by dioxin in liver and extrahepatic tissues of the marmoset monkey (Callithrix jacchus). BBA-Protein Struct. Mol. Enzymol 1996, 1298, 131–140. [Google Scholar]
- Schoeters, G.; Goyvaerts, M.P.; Ooms, D.L.; van Cleuvenbergen, R. The evaluation of dioxin and dioxin-like contaminants in selected food samples obtained from the Belgian market: Comparison of TEQ measurements obtained through the CALUX bioassay with congener specific chemical analyses. Chemosphere 2004, 54, 1289–1297. [Google Scholar]
- Besselink, H.T.; Schipper, C.; Klamer, H.; Leonards, P.; Verhaar, H.; Felzel, E.; Murk, A.J.; Thain, J.; Hosoe, K.; Schoeters, G.; et al. Intra- and interlaboratory calibration of the DR CALUX® bioassay for the analysis of dioxins and dioxin-like chemicals in sediments. Environ. Toxicol. Chem 2004, 23, 2781–2789. [Google Scholar]
- Windal, I.; van Wouwe, N.; Eppe, G.; Xhrouet, C.; Debacker, V.; Baeyens, W.; De Pauw, E.; Goeyens, L. Validation and interpretation of CALUX as a tool for the estimation of dioxin-like activity in marine biological matrixes. Environ. Sci. Technol 2005, 39, 1741–1748. [Google Scholar]
- Hoogenboom, L.; Goeyens, L.; Carbonnelle, S.; Van Loco, J.; Beernaert, H.; Baeyens, W.; Traag, W.; Bovee, T.; Jacobs, G.; Schoeters, G. The CALUX bioassay: Current status of its application to screening food and feed. Trac-Trend Anal. Chem 2006, 25, 410–420. [Google Scholar]
- Nording, M.; Sporring, S.; Wiberg, K.; Bjorklund, E.; Haglund, P. Monitoring dioxins in food and feedstuffs using accelerated solvent extraction with a novel integrated carbon fractionation cell in combination with a CAFLUX bioassay. Anal. Bioanal. Chem 2005, 381, 1472–1475. [Google Scholar]
- Nording, M.; Denison, M.S.; Baston, D.; Persson, Y.; Spinnel, E.; Haglund, P. Analysis of dioxins in contaminated soils with the CALUX and CAFLUX bioassays, an immunoassay, and gas chromato-graphy/high-resolution mass spectrometry. Environ. Toxicol. Chem 2007, 26, 1122–1129. [Google Scholar]
- Zhao, B.; Baston, D.S.; Khan, E.; Sorrentino, C.; Denison, M.S. Enhancing the response of CALUX and CAFLUX cell bioassays for quantitative detection of dioxin-like compounds. Sci. China Chem 2010, 53, 1010–1016. [Google Scholar]
- Pandini, A.; Soshilov, A.A.; Song, Y.J.; Zhao, J.; Bonati, L.; Denison, M.S. Detection of the TCDD binding-fingerprint within the ah receptor ligand binding domain by structurally driven mutagenesis and functional analysis. Biochemistry 2009, 48, 5972–5983. [Google Scholar]
- He, G.C.; Zhao, B.; Denison, M.S. Identification of benzothiazole derivatives and polycyclic aromatic hydrocarbons as aryl hydrocarbon receptor agonists present in tire extracts. Environ. Toxicol. Chem 2011, 30, 1915–1925. [Google Scholar]
- Senthilkumar, P.K.; Robertson, L.W.; Ludewig, G. PCB153 reduces telomerase activity and telomere length in immortalized human skin keratinocytes (HaCaT) but not in human foreskin keratinocytes (NFK). Toxicol. Appl. Pharm 2012, 259, 115–123. [Google Scholar]
- Yang, J.H.; Lee, H.G.; Park, K.Y. Development of human dermal epithelial cell-based bioassay for the dioxins. Chemosphere 2008, 72, 1188–1192. [Google Scholar]
- Farrell, K.; Safe, L.; Safe, S. Synthesis an daryl hydrocarbon receptor binding properties of radiolabeled polychlorinated dibenzofuran congeners. Arch. Biochem. Biophys 1987, 259, 185–195. [Google Scholar]
- Okuyama, M.; Kobayashi, N.; Takeda, W.; Anjo, T.; Matsuki, Y.; Goto, J.; Kambegaw, A.; Hori, S. Enzyme-linked immunosorbent assay for monitoring toxic dioxin congeners in milk based on a newly generated monoclonal anti-dioxin antibody. Anal. Chem 2004, 76, 1948–1956. [Google Scholar]
- Saito, K.; Takekuma, M.; Ogawa, M.; Kobayashi, S.; Sugawara, Y.; Ishizuka, M.; Nakazawa, H.; Matsuki, Y. Enzyme-linked immunosorbent assay toxicity evaluation method for dioxins in human milk. Bull. Environ. Contam. Toxicol 2003, 70, 636–643. [Google Scholar]
- Albro, P.W.; Luster, M.I.; Chae, K.; Chaudhary, S.K.; Clark, G.; Lawson, L.D.; Corbett, J.T.; McKinney, J.D.A. Radioimmunoassay for chlorinated dibenzo-p-dioxins. Toxicol. Appl. Pharmacol 1979, 50, 137–146. [Google Scholar]
- Bradfield, C.A.; Poland, A. A competitive binding assay for 2,3,7,8-tetrachlorodibenzo-p-dioxin and related ligands of the Ah receptor. Mol. Pharmacol 1988, 34, 682–688. [Google Scholar]
- Wei, X.; Ching, L.Y.; Cheng, S.H.; Wong, M.H.; Wong, C.K.C. The detection of dioxin- and estrogen-like pollutants in marine and freshwater fishes cultivated in Pearl River Delta, China. Environ. Pollut 2010, 158, 2302–2309. [Google Scholar]
- Jaborek-Hugo, S.; Von Holst, C.; Allen, R.; Stewart, T.; Willey, J.; Anklam, E. Use of an immunoassay as a means to detect polychlorinated biphenyls in animal fat. Food Addit. Contam 2001, 18, 121–127. [Google Scholar]
- Uchiyama, K.; Yang, M.; Sawazaki, T.; Shimizu, H.; Ito, S. Development of a Bio-MEMS for evaluation of dioxin toxicity by immunoassay method. Sens. Actuators B Chem 2004, 103, 200–205. [Google Scholar]
- Tsutsumi, T.; Amakura, Y.; Ashieda, K.; Okuyama, A.; Tanioka, Y.; Sakata, K.; Kobayashi, Y.; Sasaki, K.; Maitanit, T. PCB 118 and aryl hydrocarbon receptor immunoassays for screening Dioxins in retail fish. J. Agric. Food Chem 2008, 56, 2867–2874. [Google Scholar]
- Harrison, R.O.; Melnychuk, N. Rapid analysis of PCBs in Soil by enzyme-immunoassay. Int. J. Environ. Anal. Chem 1995, 59, 179–185. [Google Scholar]
- Johnson, J.C.; Van Emon, J.M. Quantitative enzyme-linked immunosorbent assay for determination of polychlorinated biphenyls in environmental soil and sediment samples. Anal. Chem 1996, 68, 162–169. [Google Scholar]
- Lawruk, T.S.; Lachman, C.E.; Jourdan, S.W.; Fleeker, J.R.; Hayes, M.C.; Herzog, D.P.; Rubio, F.M. Quantitative determination of PCBs in soil and water by a magnetic particle-based immunoassay. Environ. Sci. Technol 1996, 30, 695–700. [Google Scholar]
- Pullen, S.; Haiber, G.; Scholer, H.F.; Hock, B. Evaluation of an immunochemical test kit for polychlorinated biphenyls in soils and comparison with gas chromatographic analysis. Int. J. Environ. Anal. Chem 1996, 65, 127–138. [Google Scholar]
- Chuang, J.C.; Miller, L.S.; Davis, D.B.; Peven, C.S.; Johnson, J.C.; Emon, J.M.V. Analysis of soil and dust samples for polychlorinated biphenyls by enzyme-linked immunosorbent assay (ELISA). Anal. Chim. Acta 1998, 376, 67–75. [Google Scholar]
- Johnson, J.C.; Emon, J.M.V.; Clarke, A.N.; Wamsley, B.N. Quantitative ELISA of polychlorinated biphenyls in an oily soil matrix using supercritical fluid extraction. Anal. Chim. Acta 2001, 428, 191–199. [Google Scholar]
- Plaza, G.; Ulfig, K.; Tien, A.J. Immunoassays and environmental studies. Pol. J. Environ. Stud 2000, 9, 231–236. [Google Scholar]
- Emon, J.M.V.; Gerlach, C.L.; Bowman, K. Bioseparation and bioanalytical techniques in environmental monitoring. J. Chromatogr. B 1998, 715, 211–228. [Google Scholar]
- Mapes, J.P.; Mckenzie, K.D.; Stewart, T.N.; Mcclelland, L.R.; Studabaker, W.B.; Manning, W.B.; Friedman, S.B. An on-site immunoassay for detecting PCB in soil. Bull. Environ. Contam. Toxicol 1993, 50, 219–225. [Google Scholar]
- Deng, A.P.; Kolar, V.; Ulrich, R.; Franek, M. Direct competitive ELISA for the determination of polychlorinated biphenyls in soil samples. Anal. Bioanal. Chem 2002, 373, 685–690. [Google Scholar]
- Nichkova, M.; Park, E.K.; Koivunen, M.E.; Kamita, S.G.; Gee, S.J.; Chuang, J.; Emon, J.M.V.; Hammock, B.D. Immunochemical determination of dioxins in sediment and serum samples. Talanta 2004, 63, 1213–1223. [Google Scholar]
- Lopez-Avila, V.; Benedicto, J.; Charan, C.; Young, R.; Beckert, W.F. Determination of PCBs in soils/sediments by microwave-assisted extraction and GC/ECD or ELISA. Environ. Sci. Technol. 1995, 2709–2712. [Google Scholar]
- David, F.; Verschuere, M.; Sandra, P. Off-line supercritical fluid extraction-capillary GC applications in environmental-analysis. Fresen. J. Anal. Chem 1992, 344, 479–485. [Google Scholar]
- Bøwadt, S.; Johansson, B.; Pelusio, F.; Larsen, B.R.; Rovida, C. Solid-phase trapping of polychlorinated-biphenyls in supercritical-fluid extraction. J. Chromatogr. A 1994, 662, 424–433. [Google Scholar]
- Bøwadt, S.; Johansson, B.; Fruekilde, P.; Hansen, M.; Zilli, D.; Larsen, B.; Deboer, J. Supercritical-fluid extraction of polychlorinated-biphenyls from lyophilized fish tissue. J. Chromatogr. A 1994, 675, 189–204. [Google Scholar]
- Mannila, M.; Koistinen, J.; Vartiainen, T. Development of supercritical fluid extraction with a solid-phase trapping for fast estimation of toxic load of polychlorinated dibenzo-p-dioxins-di-benzofurans in sawmill soil. J. Chromatogr. A 2002, 975, 189–198. [Google Scholar]
- Bøwadt, S.; Mazeas, L.; Miller, D.J.; Hawthorne, S.B. Field-portable determination of polychlorinated biphenyls and polynuclear aromatic hydrocarbons in soil using supercritical fluid extraction. J. Chromatogr. A 1997, 785, 205–217. [Google Scholar]
- Nording, M.; Nichkova, M.; Spinnel, E.; Persson, Y.; Gee, S.J.; Hammock, B.D.; Haglund, P. Rapid screening of dioxin-contaminated soil by accelerated solvent extraction/purification followed by immunochemical detection. Anal. Bioanal. Chem 2006, 385, 357–366. [Google Scholar]
- Schrock, M.; Dindal, A.; Billets, S. Evaluation of alternative approaches for screening contaminated sediments and soils for polychlorinated dibenzo-p-dioxins and polychlorinated dibenzofurans. J. Environ. Manag 2009, 90, 1289–1295. [Google Scholar]
- Chuang, J.C.; Emon, J.M.V.; Schrock, M.E. High-throughput screening of dioxins in sediment and soil using selective pressurized liquid extraction with immunochemical detection. Chemosphere 2009, 77, 1217–1223. [Google Scholar]
- Samara, F.; Wyrzykowska, B.; Tabor, D.; Touati, D.; Gullett, B.K. Toxicity comparison of chlorinated and brominated dibenzo-p-dioxins and dibenzofurans in industrial source samples by HRGC/HRMS and enzyme immunoassay. Environ. Int 2010, 36, 247–253. [Google Scholar]
- Glass, T.R.; Ohmura, N.; Taemi, Y.; Joh, T. Simple immunoassay for detection of PCBs in transformer oil. Environ. Sci. Technol 2005, 39, 5005–5009. [Google Scholar]
- Lambert, N.; Fan, T.S.; Pilette, J.F. Analysis of PCBs in waste oil by enzyme immunoassay. Sci. Total Environ 1997, 196, 57–61. [Google Scholar]
- Wiberg, K.; Sporring, S.; Haglund, P.; Bjorklund, E. Selective pressurized liquid extraction of polychlorinated dibenzo-p-dioxins, dibenzofurans and dioxin-like polychlorinated biphenyls from food and feed samples. J. Chromatogr. A 2007, 1138, 55–64. [Google Scholar]
- Zajicek, J.L.; Tillitt, D.E.; Schwartz, T.R.; Schmitt, C.J.; Harrison, R.O. Comparison of an enzyme-linked immunosorbent assay (ELISA) to gas chromatography (GC)-measurement of polychlorinated biphenyls (PCBs) in selected US fish extracts. Chemosphere 2000, 40, 539–548. [Google Scholar]
- Tsutsumi, T.; Amakura, Y.; Okuyama, A.; Tanioka, Y.; Sakata, K.; Sasaki, K.; Maitani, T. Application of an ELISA for PCB 118 to the screening of dioxin-like PCBs in retail fish. Chemosphere 2006, 65, 467–473. [Google Scholar]
- Fillmann, G.; Galloway, T.S.; Sanger Ross, C.; Depledge, M.H.; Readman, J.W. Relative performance of immunochemical (enzyme-linked immunosorbent assay) and gas chromatography-electron-capture detection techniques to quantify polychlorinated biphenyls in mussel tissues. Anal. Chim. Acta 2002, 461, 75–84. [Google Scholar]
- Tsutsumi, T.; Miyoshi, N.; Sasaki, K.; Maitani, T. Biosensor immunoassay for the screening of dioxin-like polychlorinated biphenyls in retail fish. Anal. Chim. Acta 2008, 617, 177–183. [Google Scholar]
- Shan, G.; Leeman, W.R.; Gee, S.J.; Sanborn, J.R.; Jones, A.D.; Chang, D.P.Y.; Hammock, B.D. Highly sensitive dioxin immunoassay and its application to soil and biota samples. Anal. Chim. Acta 2001, 444, 169–178. [Google Scholar]
- Van Emon, J.M.; Chuang, J.C.; Lordo, R.A.; Schrock, M.E.; Nichkova, M.; Gee, S.J.; Hammock, B.D. An enzyme-linked immunosorbent assay for the determination of dioxins in contaminated sediment and soil samples. Chemosphere 2008, 72, 95–103. [Google Scholar]
- Kim, I.S.; Setford, S.J.; Saini, S. Determination of polychlorinated biphenyl compounds in electrical insulating oils by enzyme immunoassay. Anal. Chim. Acta 2000, 422, 167–177. [Google Scholar]
- Sugawara, Y.; Saito, K.; Ogawa, M.; Kobayashi, S.; Shan, G.M.; Sanborn, J.R.; Hammock, B.D.; Nakazawa, H.; Matsuki, Y. Development of dioxin toxicity evaluation method in human milk by enzyme-linked immunosorbent assay-assay validation for human milk. Chemosphere 2002, 46, 1471–1476. [Google Scholar]
- Delaunay-Bertoncini, N.; Hennion, M.C. Immunoaffinity solid-phase extraction for pharmaceu-tical and biomedical trace-analysis-coupling with HPLC and CE-perspectives. J. Pharmaceut. Biomed 2004, 34, 717–736. [Google Scholar]
- Hennion, M.C.; Pichon, V. Immuno-based sample preparation for trace analysis. J. Chromatogr. A 2003, 1000, 29–52. [Google Scholar]
- Weller, M.G. Immunochromatographic techniques–A critical review. Fresen. J. Anal. Chem 2000, 366, 635–645. [Google Scholar]
- Kaware, M.; Bronshtein, A.; Safi, J.; Van Emon, J.M.; Chuang, J.C.; Hock, B.; Kramer, K.; Altstein, M. Enzyme-linked immunosorbent assay (ELISA) and sol-gel-based immunoaffinity purification (IAP) of the pyrethroid bioallethrin in food and environmental samples. J. Agric. Food Chem 2006, 54, 6482–6492. [Google Scholar]
- Van Emon, J.M. Bioanalytical methods for food contaminant analysis. J. AOAC Int 2010, 93, 1681–1691. [Google Scholar]
- Altstein, M.; Ben Aziz, O.; Skalka, N.; Bronshtein, A.; Chuang, J.C.; Van Emon, J.M. Development of an immunoassay and a sol-gel-based immunoaffinity cleanup method for coplanar polychlorinated biphenyls from soil and sediment samples. Anal. Chim. Acta 2010, 675, 138–147. [Google Scholar]
- Shelver, W.L.; Larsen, G.L.; Huwe, J.K. Use of an immunoaffinity column for tetrachlorodi-benzo-p-dioxinserum sample cleanup. J. Chromatogr. B 1998, 705, 261–268. [Google Scholar]
- Jung, F.; Gee, S.J.; Harrison, R.O.; Goodrow, M.H.; Karu, A.E.; Braun, A.L.; Li, Q.X.; Hammock, B.D. Use of immunochemical techniques for the analysis of pesticides. Pestic. Sci 1989, 26, 303–317. [Google Scholar]
- Goodrow, M.H.; Harrison, R.O.; Hammock, B.D. Hapten synthesis, antibody development, and competitive-inhibition enzyme-immunoassay for s-triazine herbicides. J. Agric. Food Chem 1990, 38, 990–996. [Google Scholar]
- Harrison, R.O.; Goodrow, M.H.; Hammock, B.D. Competitive-inhibition ELISA for the s-triazine herbicides-assay optimization and antibody characterization. J. Agric. Food Chem 1991, 39, 122–128. [Google Scholar]
- Marco, M.P.; Hammock, B.D.; Kurth, M.J. Hapten design and development of an ELISA (enzyme-linked-immunosorbent-assay) for the detection of the mercapturic acid conjugates of naphthalene. J. Org. Chem 1993, 58, 7548–7556. [Google Scholar]
- Chiu, Y.W.; Carlson, R.E.; Marcus, K.L.; Karu, A.E. A monoclonal immunoassay for the coplanar polychlorinated-biphenyls. Anal. Chem 1995, 67, 3829–3839. [Google Scholar]
- Fránek, M.; Deng, A.P.; Kolar, V.; Socha, J. Direct competitive immunoassays for the coplanar polychlorinated biphenyls. Anal. Chim. Acta 2001, 444, 131–142. [Google Scholar]
- Fránek, M.; Pouzar, V.; Kolar, V. Enzyme-immunoassays for polychlorinated biphenyls: Structural aspects of hapten-antibody binding. Anal. Chim. Acta 1997, 347, 163–176. [Google Scholar]
- Inui, H.; Takeuchi, T.; Uesugi, A.; Doi, F.; Takai, M.; Nishi, K.; Miyake, S.; Ohkawa, H. Enzyme-linked immunosorbent assay with monoclonal and single chain variable fragment antibodies selective to coplanar polychlorinated biphenyls. J. Agric. Food Chem 2012, 7, 1605–1612. [Google Scholar]
- Sherry, J.P. Environmental chemistry the immunoassay option. Crit. Rev. Anal. Chem 1992, 23, 217–300. [Google Scholar]
- Harrison, R.O.; Eduljee, G.H. Immunochemical analysis for dioxins-progress and prospects. Sci. Total Environ 1999, 239, 1–18. [Google Scholar]
- Carlson, R.E.; Harrison, R.O. Measurement of PCDD/F TEQ by immunoassay: Concept development and validation. Organohalogen Compounds 1998, 35, 97–100. [Google Scholar]
- Harrison, R.O.; Carlson, R.E. An immunoassay for TEQ screening of dioxin/furan samples: Current status of assay and applications development. Chemosphere 1997, 34, 915–928. [Google Scholar]
- Fránek, M.; Diblikova, I.; Cernoch, I.; Vass, M.; Hruska, K. Broad-specificity immunoassays for sulfonamide detection: Immunochemical strategy for generic antibodies and competitors. Anal. Chem 2006, 78, 1559–1567. [Google Scholar]
- Fránek, M.; Kolar, V.; Deng, A.; Crooks, S. Determination of sulphadimidine (sulfamethazine) residues in milk, plasma, urine and edible tissues by sensitive ELISA. Food Agric. Immunol 1999, 11, 339–349. [Google Scholar]
- Yakovleve, J.; Zeravik, J.; Michura, I.V.; Formanovsky, A.A.; Franek, M.; Eremin, S.A. Hapten design and development of polarization fluoroimmunoassay for nonylphenol. Int. J. Environ. Anal. Chem 2003, 83, 597–607. [Google Scholar]
- Ohmura, N.; Tsukidate, Y.; Shinozaki, H.; Lackie, S.J.; Saiki, H. Combinational use of antibody affinities in an immunoassay for extension of dynamic range and detection of multiple analytes. Anal. Chem 2003, 75, 104–110. [Google Scholar]
- Kramer, P.M.; Goodrow, M.H.; Kremmer, E. Enzyme-linked immunosorbent assays based on rabbit polyclonal and rat monoclonal antibodies against isoproturon. J. Agric. Food Chem 2004, 52, 2462–2471. [Google Scholar]
- Glass, T.R.; Ohmura, N.; Morita, K.; Sasaki, K.; Saiki, H.; Takagi, Y.; Kataoka, C.; Ando, A. Improving an immunoassay response to related polychlorinated biphenyl analytes by mixing antibodies. Anal. Chem 2006, 78, 7240–7247. [Google Scholar]
- Neuberger, M.S.; Williams, G.T.; Fox, R.O. Recombinant antibodies possessing novel effector functions. Nature 1984, 312, 604–608. [Google Scholar]
- George, A.J.T.; Spooner, R.A.; Epenetos, A.A. Applications of monoclonal antibodies in clinical oncology. Immunol. Today 1994, 15, 559–561. [Google Scholar]
- Pack, P.; Pluckthun, A. Miniantibodies: Use of amphipathic helices to produce functional, flexibly linked dimeric FV fragments with high avidity in Escherichia coli. Biochemistry 1992, 31, 1579–1584. [Google Scholar]
- Li, Y.; Cockburn, W.; Kilpatrick, J.; Whitelam, G.C. Selection of rabbit single-chain Fv fragments against the herbicide atrazine using a new phage display system. Food Agric. Immunol 1999, 11, 5–17. [Google Scholar]
- Nishi, K.; Ishiuchi, M.; Morimune, K.; Ohkawa, H. Molecular and immunochemical characteristics of monoclonal and recombinant antibodies selective for the triazine herbicide simetryn and application to environmental analysis. J. Agric. Food Chem 2005, 53, 5096–5104. [Google Scholar]
- Nishi, K.; Takai, M.; Morimune, K.; Ohkawa, H. Molecular and immunochemical characteristics of monoclonal and recombinant antibodies specific to bisphenol A. Biosci. Biotechnol. Biochem 2003, 67, 1358–1367. [Google Scholar]
- Hock, B.; Dankwardt, A.; Kramer, K.; Marx, A. Immunochemical techniques: Antibody production for pesticide analysis. A review. Anal. Chim. Acta 1995, 311, 393–405. [Google Scholar]
- Shelver, W.L.; Parrotta, C.D.; Slawecki, R.; Li, Q.X.; Ikonomou, M.G. Development of a magnetic particle immunoassay for polybrominated diphenyl ethers and application to environmental and food matrices. Chemosphere 2008, 73, 518–523. [Google Scholar]
- Behnisch, P.A.; Hosoe, K.; Sakai, S. Brominated dioxin-like compounds: In vitro assessment in comparison to classical dioxin-like compounds and other polyaromatic compounds. Environ. Int 2003, 29, 861–877. [Google Scholar]
- Bovee, T.F.H.; Hoogenboom, L.A.P.; Hamers, A.R.M.; Traag, W.A.; Zuidema, T.; Aarts, J.M.M.J.G.; Brouwer, A.; Kuiper, H.A. Validation and use of the CALUX-bioassay for the determination of dioxins and PCBs in bovine milk. Food Addit. Contam 1998, 15, 863–875. [Google Scholar]
- Carbonnelle, S.; Van Loco, J.; Van Overmeire, I.; Windal, I.; Van Wouwe, N.; Van Leeuwen, S.; Goeyens, L. Importance of REP values when comparing the CALUX bioassay results with chemoanalyses results-Example with spiked vegetable oils. Talanta 2004, 63, 1255–1259. [Google Scholar]
- Hoogenboom, R.; Bovee, T.; Traag, W.; Hoogerbrugge, R.; Baumann, B.; Portier, L.; Van De Weg, G.; De Vries, J. The use of the DR CALUX® bioassay and indicator polychlorinated biphenyls for screening of elevated levels of dioxins and dioxin-like polychlorinated biphenyls in eel. Mol. Nutr. Food Res 2006, 50, 945–957. [Google Scholar]
- Barletta, J.M.; Edelman, D.C.; Constantine, N.T. Lowering the detection limits of HIV-1 viral load using real-time immuno-PCR for HIV-1 p24 antigen. Am. J. Clin. Pathol 2004, 122, 20–27. [Google Scholar]
- Sugawara, K.; Kobayashi, D.; Saito, K.; Furuya, D.; Araake, H.; Yagihashi, A.; Yajima, T.; Hosoda, K.; Kamimura, T.; Watanabe, N. A highly sensitive immuno-polymerase chain reaction assay for human angiotensinogen using the identical first and second polyclonal antibodies. Clin. Chim. Acta 2000, 299, 45–54. [Google Scholar]
- Barletta, J.M.; Edelman, D.C.; Highsmith, W.E.; Constantine, N.T. Detection of ultra-low levels of pathologic prion protein in scrapie infected hamster brain homogenates using real-time immuno-PCR. J. Virol. Methods 2005, 127, 154–164. [Google Scholar]
- Chen, H.Y.; Zhuang, H.S. Real-time immuno-PCR assay for detecting PCBs in soil samples. Anal. Bioanal. Chem 2009, 394, 1205–1211. [Google Scholar]
- Shimomura, M.; Nomura, Y.; Zhang, W.; Sakino, M.; Lee, K.H.; Ikebukuro, K.; Karube, I. Simple and rapid detection method using surface plasmon resonance for dioxins, polychlorinated biphenylx and atrazine. Anal. Chim. Acta 2001, 434, 223–230. [Google Scholar]
- Zhao, B.; DeGroot, D.E.; Hayashi, A.; He, G.C.; Denison, M.S. CH223191 is a ligand-selective antagonist of the Ah (dioxin) receptor. Toxicol. Sci 2010, 117, 393–403. [Google Scholar]
| Method | Principle | Reference |
|---|---|---|
| (I) AhR signal pathway | When cell lines are exposed to dioxins, cytoplasmic AhR is activated, enters the nucleus and makes a complex with ARNT (aryl hydrocarbon receptor nuclear translocator). The complex binds to genomic DRE elements inducing the expression of CYP1A1 gene transcripts, measured by one of the assays below (a–e). | |
| (a) Q-PCR | By detecting the increase in CYP1A1 mRNA levels gene, the TEQ of dioxins is deduced, compared with a TCDD standard. | [20,21] |
| (b) AHH/EROD | Enzymatic activity of CYP1A1 is determined using the substrates 7-ethoxyresorufin or benzo[a]pyrene. The TEQ of dioxins is deduced, compared to a TCDD standard. | [22–25] |
| (c) CALUX | Recombinant cell lines, stably transfected with dioxin responsive firefly luciferase reporter genes, are exposed to dioxin-containing samples. When dioxins activate AhR, the AhR-ARNT (aryl hydrocarbon receptor nuclear translocator) complex will be formed which binds to DREs and induces expression of the luciferase reporter gene. Luciferase is easily assayed in a luminometer after addition of luciferin substrate. | [26–29] |
| (d) CAFLUX | The assay is similar to CALUX except enhanced green fluorescent protein is used as the reporter gene. The TEQ of dioxins is determined by measuring the intensity of the fluorescence emitted by the reporter. | [30–32] |
| (e) GRAB | Samples are incubated with hepatic cytosol extracts containing AhR and ARNT. Dioxins in the samples induce the formation of activated AhR/ARNT complexes. When the 32P isotope labeled DNA probes containing DRE consensus sequence bind with these complexes, the migration rate of the DNA probes in polyacrylamide gels will be slowed down compared to unbound probe. | [33,34] |
| (II) Cell proliferation-based assay | Dioxins are potent inducers of chloracne in humans, which is characterized by hyperkeratinization. By detecting the abnormal terminal differentiation of skin cells exposed to the sample, the TEQ of dioxins can be obtained compared with a TCDD standard. | [35,36] |
| (III) AhR ligand binding | Dioxins in samples compete with isotope labeled TCDD for binding to AhR. Therefore, the concentration of the dioxins in the sample is proportional to the decrease of radioactivity of AhR. | [37] |
| (IV) Immunoassay (a) ELISA | This assay is based on binding of dioxin to anti-dioxins antibodies. Dioxins in the samples compete with standard congener immobilized on the plate for binding to anti-dioxin antibodies. After washing away free antibody-antigen complexes, the antibodies bound to immobilized TCDD are detected by binding of secondary antibodies conjugated to an enzyme, followed by enzyme assays using a substrate which is easily measured. Alternatively, dioxins in the sample compete with standard congeners conjugated to an enzyme for binding to anti-dioxin antibodies immobilized on the plate followed by washing and enzyme assays. By using a standard curve generated by known concentrations of dioxin standards, the TEQ of dioxins can be deduced. | [38,39] |
| (b) RIA | Radiolabelled standard antigens (*Ag) and unlabeled sample antigens (Ag) compete with inadequate specific antibodies (Ab) and form *Ag-Ab or Ag-Ab complexes. When the binding reaction to achieve dynamic balance, if Ag quantity increase, Ag-Ab quantity increase, while *Ag-Ab relatively reduce and free Ag increase. Namely the mass of Ag is inversely proportional to the mass of radiolabelled complexes. Complexes and free antigens are effectively separated, and then measure the radioactivity to get the content of samples antigen. | [40,41] |
| (c) DEFIA | Similar to competitive ELISA, except using Eu to label the antigen or antibody. | [42,43] |
| (d) Ah-I | According to AhR signaling pathway, the amount of AhR:ARNT heterodimer is correlated to the concentration of the dioxins. First to immobilize AhR:ARNT complex by DNA fragments containing DRE consensus sequences which are linked on the microplates. Then the immobilized AhR:ARNT complex is detected by using anti-AhR or anti-ARNT antibody. | [44,45] |
| Standard number | Method | Sample | Detection chemicals |
|---|---|---|---|
| US EPA 4020 | Immunoassay | Soils, non-aqueous waste liquids | PCBs |
| US EPA 4025 | Enzyme immunoassay | Soils | PCDD/Fs |
| US EPA 4425 | A reporter gene on a human cell line | Soils, sediments tissues, water | PCBs, PCDD/Fs |
| US EPA 4430 | AhR-PCR assay | Soils, sediments | PCDD/Fs |
| US EPA 4435 | CALUX bioassay | Soils, sediments | Dioxin-like compounds |
| USEPA/600/R-01/052 | DELFIA™ | Soils, solvent extract | PCBs |
| 2002/69/EC | Cell-based bioassays | Foodstuffs | PCDD/Fs, Dioxin-like PCBs |
© 2012 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tian, W.; Xie, H.Q.; Fu, H.; Pei, X.; Zhao, B. Immunoanalysis Methods for the Detection of Dioxins and Related Chemicals. Sensors 2012, 12, 16710-16731. https://doi.org/10.3390/s121216710
Tian W, Xie HQ, Fu H, Pei X, Zhao B. Immunoanalysis Methods for the Detection of Dioxins and Related Chemicals. Sensors. 2012; 12(12):16710-16731. https://doi.org/10.3390/s121216710
Chicago/Turabian StyleTian, Wenjing, Heidi Qunhui Xie, Hualing Fu, Xinhui Pei, and Bin Zhao. 2012. "Immunoanalysis Methods for the Detection of Dioxins and Related Chemicals" Sensors 12, no. 12: 16710-16731. https://doi.org/10.3390/s121216710
APA StyleTian, W., Xie, H. Q., Fu, H., Pei, X., & Zhao, B. (2012). Immunoanalysis Methods for the Detection of Dioxins and Related Chemicals. Sensors, 12(12), 16710-16731. https://doi.org/10.3390/s121216710




