Responsive Hydrogels for Label-Free Signal Transduction within Biosensors
Abstract
:1. Introduction
2. Biospecific Response of Hydrogels – What Can Be Achieved?
3. Biospecific Response of Hydrogels – How Can It Be Achieved in Practice?
3.1. Osmotically Induced Hydrogel Response
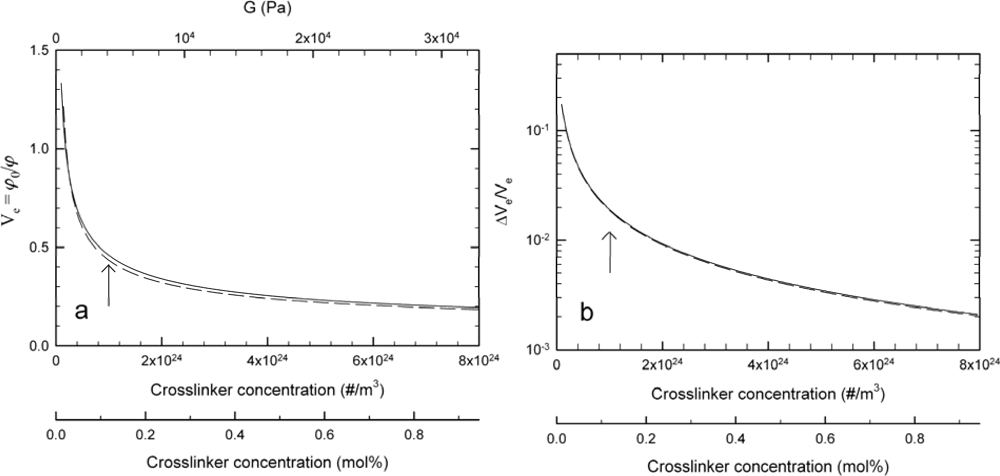
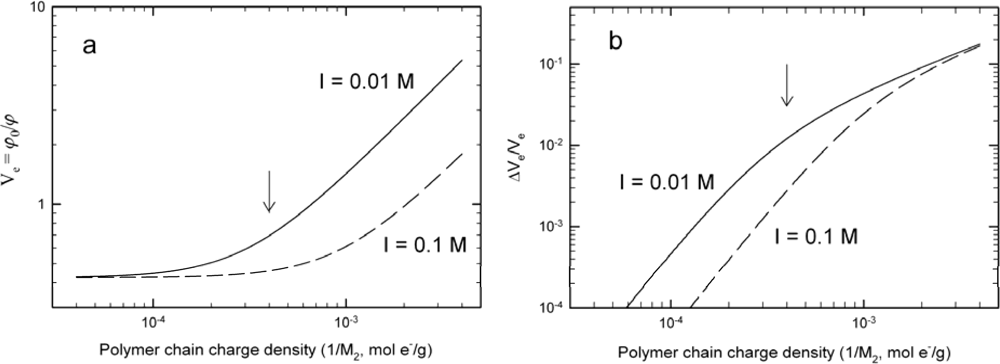
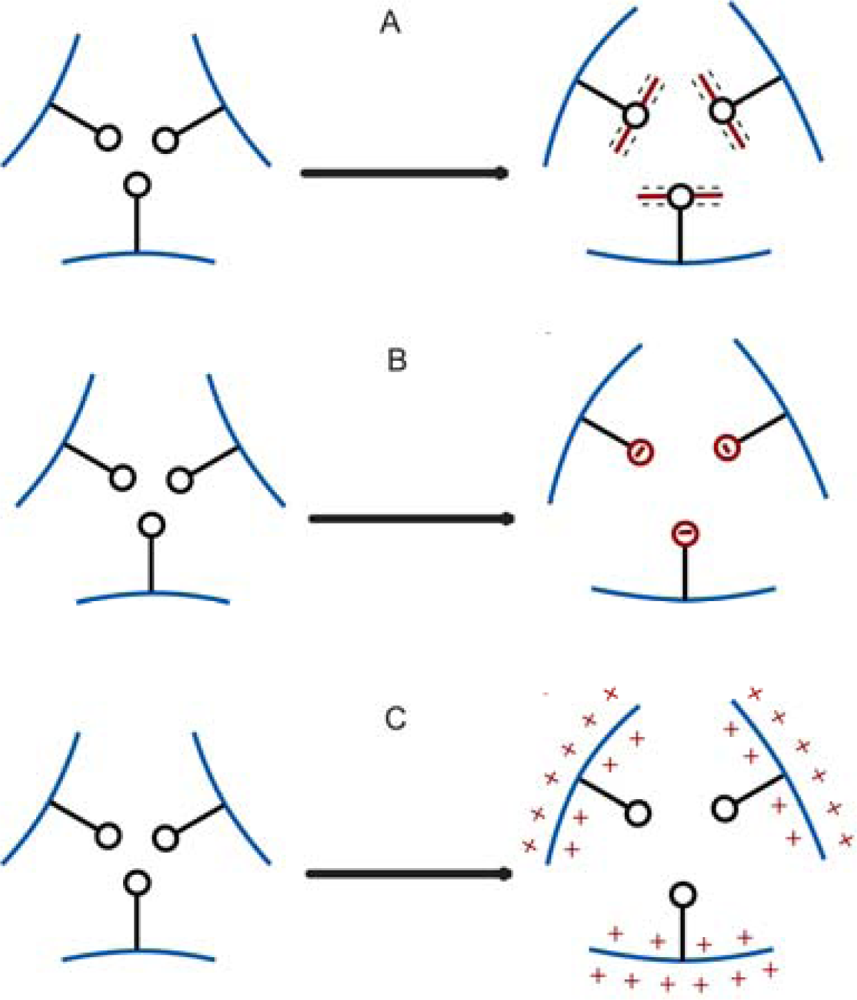
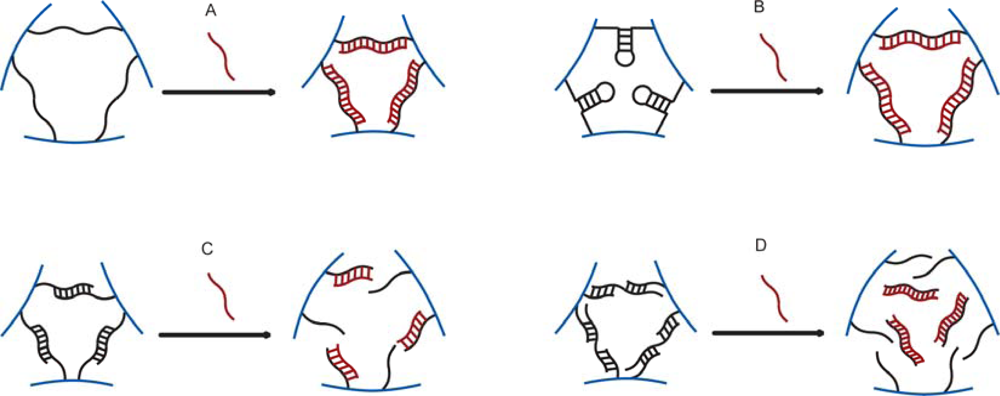
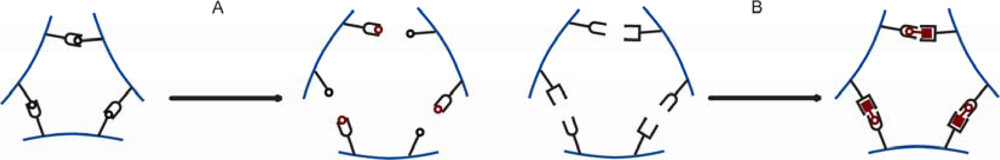
3.2. Changes in Hydrogel Swelling Volume by Biospecific Changes in Crosslink Density
4. Principles of Readout Platforms Supporting Bioresponsive Hydrogels in Biosensing
4.1. Electrochemical Transduction
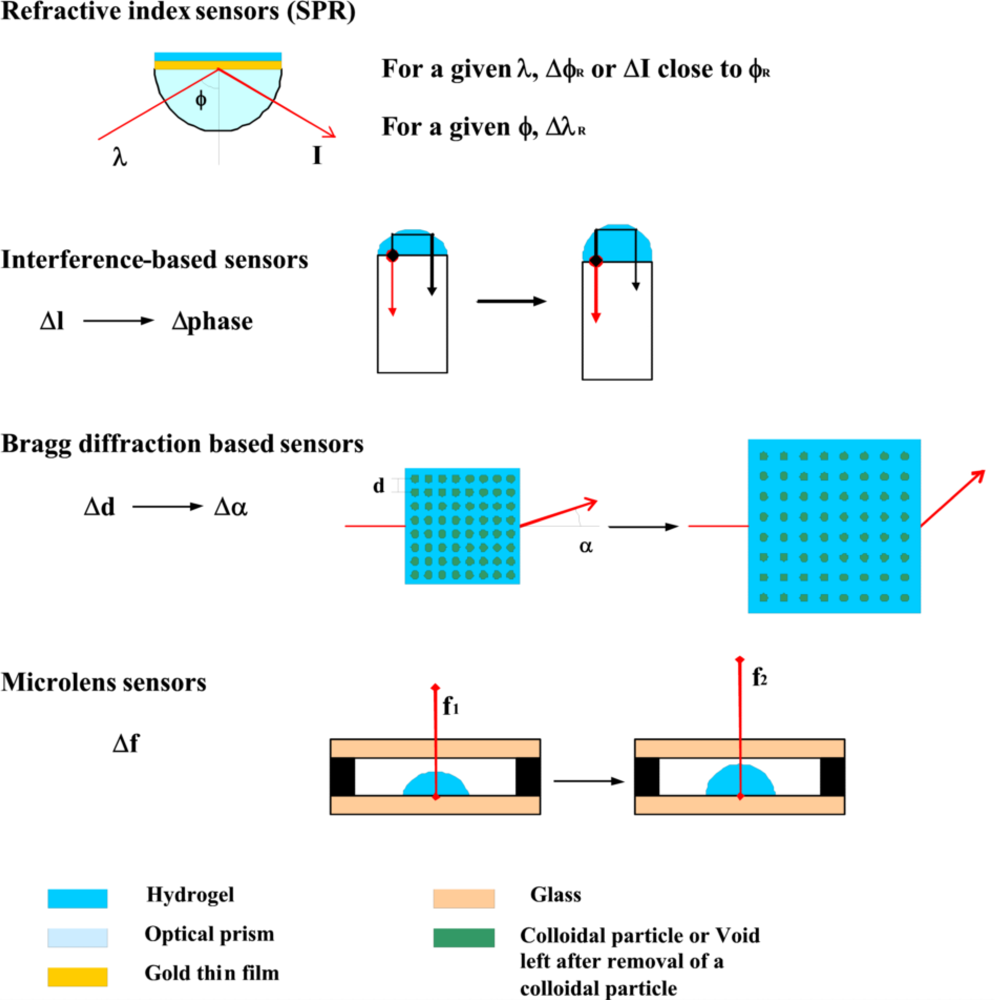
4.2. Optical Transduction (Figure 6)
4.3. Detection of Refractive Index Changes Using Surface Plasmon Resonance (SPR)
4.4. Optical Transducers Using Light Interference and Diffraction Principles
Interference Based Sensors
Bragg Diffraction Based Sensors
Microlens Sensors
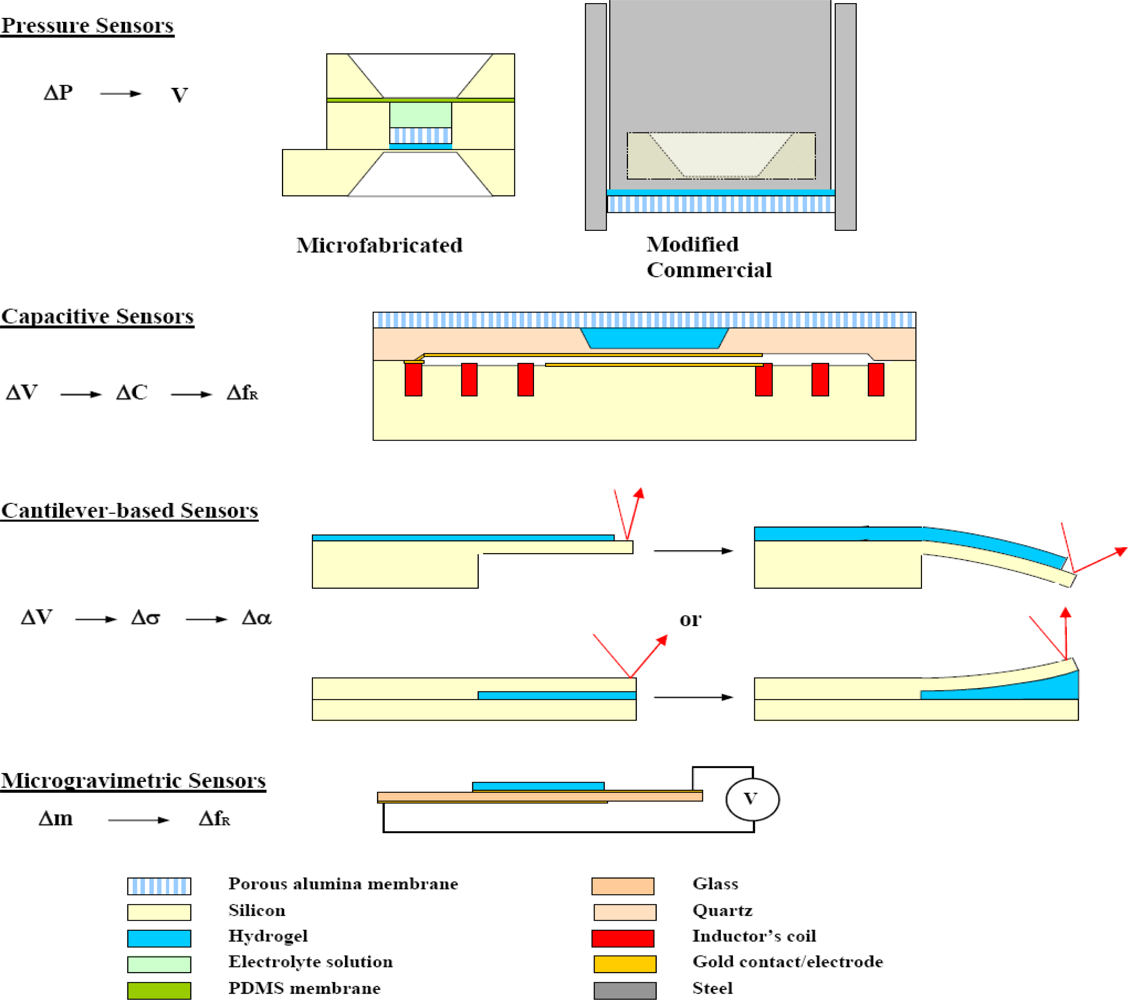
4.5. Mechanical Transduction (Figure 7)
Pressure Sensors
4.6. Capacitive Sensors
4.7. Cantilever Based Sensors
4.8. Microgravimetric Sensors
5. Conclusions
Acknowledgments
References and Notes
- Brannon-Peppas, L.; Peppas, N.A. Time-dependent response of ionic polymer networks to pH and ionic-strength changes. Int. J. Pharm 1991, 70, 53–57. [Google Scholar]
- Ricka, J.; Tanaka, T. Swelling of ionic gels - quantitative performance of the donnan theory. Macromolecules 1984, 17, 2916–2921. [Google Scholar]
- Miyata, T.; Onakamae, K.; Hoffman, A.S.; Kanzaki, Y. Stimuli-sensitivities of hydrogels containing phosphate groups. Macromol. Chem. Phys 1994, 195, 1111–1120. [Google Scholar]
- Lee, W.F.; Yuan, W.Y. Thermoreversible hydrogels X: Synthesis and swelling behavior of the (N-isopropylacrylamide-co-sodium 2-acrylamido-2-methylpropyl sulfonate) copolymeric hydrogels. J. Appl. Polym. Sci 2000, 77, 1760–1768. [Google Scholar]
- Park, T.G.; Hoffman, A.S. Sodium chloride-induced phase-transition in nonionic poly(n-isopropylacrylamide) gel. Macromolecules 1993, 26, 5045–5048. [Google Scholar]
- Schröder, U.P.; Oppermann, W. Properties of polyelectrolyte gels. In Physical Properties of Polymeric Gels; Cohen Addad, J.P., Ed.; John Wiley and Sons: Chichester, UK, 1996; pp. 19–38. [Google Scholar]
- Grimshaw, P.E.; Nussbaum, J.H.; Grodzinsky, A.J.; Yarmush, M.L. Kinetics of electrically and chemically-induced swelling in polyelectrolyte gels. J. Chem. Phys 1990, 93, 4462–4472. [Google Scholar]
- Eeckman, F.; Moes, A.J.; Amighi, K. Surfactant induced drug delivery based on the use of thermosensitive polymers. J. Controlled Release 2003, 88, 105–116. [Google Scholar]
- Ulijn, R.V.; Bib, N.; Jayawarna, V.; Thornton, P.D.; Todd, S.J.; Mart, R.J.; Smith, A.M.; Gough, J.E. Bioresponsive hydrogels. Materials Today 2007, 10, 40–48. [Google Scholar]
- Ulijn, R.V. Enzyme-responsive materials: A new class of smart biomaterials. J. Mater. Chem 2006, 16, 2217–2225. [Google Scholar]
- Miyata, T.; Uragami, T.; Nakamae, K. Biomolecule-sensitive hydrogels. Adv. Drug Del. Rev 2002, 54, 79–98. [Google Scholar]
- Fernández-Barberos, A.; Suárez, I.J.; Sierra-Martín, B.; Fernández-Nieves, A.; de las Nieves, F.J.; Marquez, M.; Rubio-Retama, J.; López-Cabarcos, E. Gels and microgels for nanotechnological applications. Adv. Colloid Int. Sci 2009, (147–148), 88–108. [Google Scholar]
- Hendrickson, G .R.; Lyon, L A. Bioresponsive hydrogels for sensing applications. Soft Matter 2009, 5, 29–35. [Google Scholar]
- Treloar, L.R.G. The Physics of Rubber Elasticity; Clarendon Press: Oxford, UK, 1975. [Google Scholar]
- Shibayama, M.; Tanaka, T. Volume phase-transition and related phenomena of polymer gels. Adv. Polym. Sci 1993, 109, 1–62. [Google Scholar]
- Brannon-Peppas, L.; Peppas, N.A. Equilibrium swelling behavior of pH-sensitve hydrogels. Chem. Eng. Sci 1991, 46, 715–722. [Google Scholar]
- Eichenbaum, G.M.; Kiser, P.F.; Dobrynin, A.V.; Simon, S.A; Needham, D. Investigation of the swelling response and loading of ionic microgels with drugs and proteins: Dependence on cross-link density. Macromolecules 1999, 32, 4867–4878. [Google Scholar]
- Gerlach, G.; Guenther, M.; Suchaneck, G.; Sorber, J.; Arndt, K.F.; Richter, A. Application of sensitive hydrogels in chemical and pH sensors. Macromol. Symp 2004, 210, 403–410. [Google Scholar]
- Suzuki, H.; Kumagai, A. A disposable biosensor employing a glucose-sensitive biochemomechanical gel. Biosens. Bioelectron 2003, 18, 1289–1297. [Google Scholar]
- Lee, Y.J.; Pruzinsky, S.A.; Braun, P.V. Glucose-sensitive inverse opal hydrogels: Analysis of optical diffraction response. Langmuir 2004, 20, 3096–3106. [Google Scholar]
- Marshall, A.J.; Young, D.S.; Kabilan, S.; Hussain, A.; Blyth, J.; Lowe, C.R. Holographic sensors for the determination of ionic strength. Anal. Chim. Acta 2004, 527, 13–20. [Google Scholar]
- Arregui, F.J.; Ciaurriz, Z.; Oneca, M.; Matias, I.R. An experimental study about hydrogels for the fabrication of optical fiber humidity sensors. Sens. Actuators, B 2003, 96, 165–172. [Google Scholar]
- Tierney, S.; Hjelme, D.R.; Stokke, B.T. Determination of swelling of responsive gels with nanometer resolution. Fiber-optic based platform for hydrogels as signal transducers. Anal. Chem 2008, 80, 5086–5093. [Google Scholar]
- Wong, J.E.; Gaharwar, A.K.; Muller-Schulte, D.; Bahadur, D.; Richtering, W. Layer-by-layer assembly of a magnetic nanoparticle shell on a thermoresponsive microgel core. J. Magn. Magn. Mater 2007, 311, 219–223. [Google Scholar]
- Wong, J.E.; Richtering, W. Layer-by-layer assembly on stimuli-responsive microgels. Curr. Opin. Colloid Interface Sci 2008, 13, 403–412. [Google Scholar]
- Wu, Z.; Hu, X.B.; Tao, C.A.; Li, Y.; Liu, J.; Yang, C.D.; Shen, D.Z.; Li, G.T. Direct and label-free detection of cholic acid based on molecularly imprinted photonic hydrogels. J. Mater. Chem 2008, 18, 5452–5458. [Google Scholar]
- Riccardi, C.D.S.; Kranz, C.; Kowalik, J.; Yamanaka, H.; Mizaikoff, B.; Josowicz, M. Label-free DNA detection of hepatitis C virus based on modified conducting polypyrrole films at microelectrodes and atomic force microscopy tip-integrated electrodes. Anal. Chem 2008, 80, 237–245. [Google Scholar]
- Tanford, C. Physical Chemistry of Macrmolecules; John Wiley and Sons Inc: New York, NY, USA, 1961. [Google Scholar]
- Guo, B.; Elgsaeter, A.; Christensen, B.E.; Stokke, B.T. Sclerox-chitosan co-gels: Effects of charge density on swelling of gels in ionic aqueous solution and in poor solvents, and on the rehydration of dried state. Polym. Gels Networks 1998, 6, 471–492. [Google Scholar]
- Kokufata, E.; Zhang, Y.Q.; Tanaka, T. Saccharide-sensitive phase-transition of a lectin-loaded gel. Nature 1991, 351, 302–304. [Google Scholar]
- Ballerstadt, R.; Evans, C.; McNichols, R.; Gowda, A. Concanavalin A for in vivo glucose sensing: A biotoxicity review. Biosens. Bioelectron 2006, 22, 275–284. [Google Scholar]
- Brownlee, M.; Cerami, A. Glycosylated insulin complexed to Concanavalin A. Biochemical basis for a closed-loop insulin delivery system. Diabetes 1983, 32, 499–504. [Google Scholar]
- Holtz, J.H.; Asher, S.A. Polymerized colloidal crystal hydrogel films as intelligent chemical sensing materials. Nature 1997, 389, 829–832. [Google Scholar]
- Holtz, J.H.; Holtz, J.S.W.; Munro, C.H.; Asher, S.A. Intelligent polymerized crystalline colloidal arrays: Novel chemical sensor materials. Anal. Chem 1998, 70, 780–791. [Google Scholar]
- Kamenjicki, M.; Asher, S.A. Epoxide functionalized polymerized crystalline colloidal arrays. Sens. Actuators, B 2005, 106, 373–377. [Google Scholar]
- Raba, J.; Mottola, H.A. Glucose-oxidase as an analytical reagent. Crit. Rev. Anal. Chem 1995, 25, 1–42. [Google Scholar]
- Ishihara, K.; Kobayashi, M.; Ishimaru, N.; Shinohara, I. Glucose-induced permeation control of insulin through a complex membrane consisting of immobilized glucose-oxidase and a poly(amine). Polym. J 1984, 16, 625–631. [Google Scholar]
- Guiseppi-Elie, A.; Brahim, S.I.; Narinesingh, D. A chemically synthesized artificial pancreas: Release of insulin from glucose–responsive hydrogels. Adv. Mater 2002, 14, 743–746. [Google Scholar]
- Albin, G.; Horbett, T.A.; Ratner, B.D. Glucose sensitive membranes for controlled delivery of insulin: Insulin transport studies. J. Controlled Release 1985, 2, 153–164. [Google Scholar]
- Asher, S.A.; Alexeev, V.L.; Goponenko, A.V.; Sharma, A.C.; Lednev, I.K.; Wilcox, C.S.; Finegold, D.N. Photonic crystal carbohydrate sensors: Low ionic strength sugar sensing. J. Am. Chem. Soc 2003, 125, 3322–3329. [Google Scholar]
- Sharma, A.C.; Jana, T.; Kesavamoorthy, R.; Shi, L.J.; Virji, M.A.; Finegold, D.N.; Asher, S.A. A general photonic crystal sensing motif: Creatinine in bodily fluids. J. Am. Chem. Soc 2004, 126, 2971–2977. [Google Scholar]
- Fossati, P.; Ponti, M.; Passoni, G.; Tarenghi, G.; Deril, G.V.M.; Prencipe, L. A step forward in enzymatic measurement of creatinine. Clin. Chem 1994, 40, 130–137. [Google Scholar]
- Soldatkin, A.P.; Montoriol, J.; Sant, W.; Martelet, C.; Jaffrezic-Renault, N. Development of potentiometric creatinine-sensitive biosensor based on ISFET and creatinine deiminase immobilised in PVA/SbQ photopolymeric membrane. Mater. Sci. Eng., C 2002, 21, 75–79. [Google Scholar]
- Soldatkin, A.P.; Montoriol, J.; Sant, W.; Martelet, C.; Jaffrezic-Renault, N. Creatinine sensitive biosensor based on ISFETs and creatinine deiminase immobilised in BSA membrane. Talanta 2002, 58, 351–357. [Google Scholar]
- Kenney, M.; Ray, S.; Boles, T.C. Mutation typing using electrophoresis and gel-immobilized acrydite (TM) probes. Biotechniques 1998, 25, 516–521. [Google Scholar]
- Nelson, C.; Hendy, S.; Reid, K.; Cavanagh, J. Acrylamide capture of DNA-bound complexes: Electrophoretic purification of transcription factors. Biotechniques 2002, 32, 808–815. [Google Scholar]
- Chan, A.; Krull, U.J. Capillary electrophoresis for capture and concentrating of target nucleic acids by affinity gels modified to contain single-stranded nucleic acid probes. Anal. Chim. Acta 2006, 578, 31–42. [Google Scholar]
- Reynaldo, L.P.; Vologodskii, A.V.; Neri, B.P.; Lyamichev, V.I. The kinetics of oligonucleotide replacements. J. Mol. Biol 2000, 297, 511–520. [Google Scholar]
- Lin, D.C.; Yurke, B.; Langrana, N.A. Mechanical properties of a reversible, DNA-crosslinked polyacrylamide hydrogel. J. Biomech. Eng 2004, 126, 104–110. [Google Scholar]
- Murakami, Y.; Maeda, M. DNA-responsive hydrogels that can shrink or swell. Biomacromolecules 2005, 6, 2927–2929. [Google Scholar]
- Tierney, S.; Stokke, B.T. Development of an Oligonucleotide Functionalized Hydrogel Integrated on a High Resolution Interferometric Readout Platform as a Label-Free Macromolecule Sensing Device. Biomacromolecules 2009, 10, 1619–1626. [Google Scholar]
- Liedl, T.; Dietz, H.; Yurke, B.; Simmel, F. Controlled trapping and release of quantum dots in a DNA-Switchable hydrogel. Small 2007, 3, 1688–1693. [Google Scholar]
- Yurke, B.; Lin, D.C.; Langrana, N.A. Use of DNA nanodevices in modulating the mechanical properties of polyacrylamide gels. In Springer Berlin: Heidelberg, Germany, DNA Computing, Proceedings of 11th International Workshop on DNA Computing, London, Canada, June 6–9, 2005; Carbone, A., Pierce, N.A., Eds.; 2006; 3892, pp. 417–426. [Google Scholar]
- Yuan, W.W.; Yang, J.Y.; Kopeckova, P.; Kopecek, J. Smart Hydrogels Containing Adenylate Kinase: Translating Substrate Recognition into Macroscopic Motion. J. Am. Chem. Soc 2008, 130, 15760–15761. [Google Scholar]
- Miyata, T.; Asami, N.; Uragami, T. A reversibly antigen-responsive hydrogel. Nature 1999, 399, 766–769. [Google Scholar]
- Miyata, T.; Asami, N.; Uragami, T. Structural Design of Stimuli-Responsive Bioconjugated Hydrogels That Respond to a Target Antigen. J. Polym. Sci., Part B: Polym. Phys 2009, 47, 2144–2157. [Google Scholar]
- Miyata, T.; Asami, N.; Uragami, T. Preparation of an antigen-sensitive hydrogel using antigen-antibody bindings. Macromolecules 1999, 32, 2082–2084. [Google Scholar]
- Miyata, T.; Jige, M.; Nakaminami, T.; Uragami, T. Tumor marker-responsive behavior of gels prepared by biomolecular imprinting. Proc. Natl. Acad. Sci. USA 2006, 103, 1190–1193. [Google Scholar]
- Hu, X.B.; An, Q.; Li, G.T.; Tao, S.Y.; Liu, B. Imprinted photonic polymers for chiral recognition. Angew. Chem., Int. Ed 2006, 45, 8145–8148. [Google Scholar]
- Ehrbar, M.; Schoenmakers, R.; Christen, E.H.; Fussenegger, M.; Weber, W. Drug-sensing hydrogels for the inducible release of biopharmaceuticals. Nat. Mater 2008, 7, 800–804. [Google Scholar]
- Brownlee, M.; Cerami, A. Glucose-controlled insulin-delivery system - semi-synthetic insulin bound to lectin. Science 1979, 206, 1190–1191. [Google Scholar]
- Seminoff, L.A.; Gleeson, J.M.; Zheng, J.; Olsen, G.B.; Holmberg, D.; Mohammad, S.F.; Wilson, D.; Kim, S.W. A self-regulating insulin delivery system. 2. In vivo characteristics of a synthetic glycosylated insulin. Int. J. Pharm 1989, 54, 251–257. [Google Scholar]
- Seminoff, L.A.; Olsen, G.B.; Kim, S.W. A self-regulating insulin delivery system. 1. Characterization of a synthetic glycosylated insulin derivative. Int. J. Pharm 1989, 54, 241–249. [Google Scholar]
- Makino, K.; Mack, E.J.; Okano, T.; Kim, S.W. A microcapsule self-regulating delivery system for insulin. J. Controlled Release 1990, 12, 235–239. [Google Scholar]
- Lee, S.J.; Park, K. Synthesis and characterization of sol-gel phase-reversible hydrogels sensitive to glucose. J. Mol. Recognit 1996, 9, 549–557. [Google Scholar]
- Obaidat, A.A.; Park, K. Characterization of glucose dependent gel-sol phase transition of the polymeric glucose-concanavalin A hydrogel system. Pharm. Res 1996, 13, 989–995. [Google Scholar]
- Obaidat, A.A.; Park, K. Characterization of protein release through glucose-sensitive hydrogel membranes. Biomaterials 1997, 18, 801–806. [Google Scholar]
- Miyata, T.; Jikihara, A.; Nakamae, K.; Hoffman, A.S. Preparation of poly(2-glucosyloxyethyl methacrylate)concanavalin A complex hydrogel and its glucose-sensitivity. Macromol. Chem. Phys 1996, 197, 1135–1146. [Google Scholar]
- Alexeev, V.L.; Sharma, A.C.; Goponenko, A.V.; Das, S.; Lednev, I.K.; Wilcox, C.S.; Finegold, D.N.; Asher, S.A. High ionic strength glucose-sensing photonic crystal. Anal. Chem 2003, 75, 2316–2323. [Google Scholar]
- Cui, Q.; Muscatello, M.M.W.; Asher, S.A. Photonic crystal borax competitive binding carbohydrate sensing motif. Analyst 2009, 134, 875–880. [Google Scholar]
- Muscatello, M.M.W.; Stunja, L.E.; Asher, S.A. Polymerized Crystalline Colloidal Array Sensing of High Glucose Concentrations. Anal. Chem 2009, 81, 4978–4986. [Google Scholar]
- Ben-Moshe, M.; Alexeev, V.L.; Asher, S.A. Fast responsive crystalline colloidal array photonic crystal glucose sensors. Anal. Chem 2006, 78, 5149–5157. [Google Scholar]
- Alexeev, V.L.; Das, S.; Finegold, D.N.; Asher, S.A. Photonic crystal glucose-sensing material for noninvasive monitoring of glucose in tear fluid. Clin. Chem 2004, 50, 2353–2360. [Google Scholar]
- Tierney, S.; Volden, S.; Stokke, B.T. Glucose sensors based on a responsive gel incorporated as a Fabry-Perot cavity on a fiber-optic readout platform. Biosens. Bioelectron 2009, 24, 2034–2039. [Google Scholar]
- Tierney, S.; Falch, B.M.H.; Hjelme, D.R.; Stokke, B.T. Determination of glucose levels using a functionalized hydrogel-optical fiber biosensor: toward continuous monitoring of blood glucose in vivo. Anal. Chem 2009, 81, 3630–3636. [Google Scholar]
- Hydrogel Sensors and Actuators : Engineering and Technology; Gerlach, G.; Arndt, K.F. (Eds.) Springer-Verlag Berlin: Heidelberg, Germany, 2009.
- Heller, A. Amperometric biosensors. Curr. Opin. Biotechnol 1996, 7, 50–54. [Google Scholar]
- Heller, A. Electron-conducting redox hydrogels: design, characteristics and synthesis. Curr. Opin. Chem. Biol 2006, 10, 664–672. [Google Scholar]
- Sirkar, K.; Revzin, A.; Pishko, M.V. Glucose and lactate biosensors based on redox polymer/oxidoreductase nanocomposite thin films. Anal. Chem 2000, 72, 2930–2936. [Google Scholar]
- Peppas, N.A.; Ali Khademhosseini, J.Z.H; Langer, R. Hydrogels in Biology and Medicine: From Molecular Principles to Bionanotechnology. Adv. Mater 2006, 18, 1345–1360. [Google Scholar]
- Mannbuxbaum, E.; Pittner, F.; Schalkhammer, T.; Jachimowicz, A.; Jobst, G.; Olcaytug, F.; Urban, G. New microminiaturized glucose sensors using covalent immobilization techniques. Sens. Actuators B-Chem 1990, 1, 518–522. [Google Scholar]
- Koudelka, M.; Rohnerjeanrenaud, F.; Terrettaz, J.; Bobbioniharsch, E.; Derooij, N.F.; Jeanrenaud, B. In vivo behaviour of hypodermically implanted microfabricated glucose sensors. Biosens. Bioelectron 1991, 6, 31–36. [Google Scholar]
- Guiseppi-Elie, A.; Brahim, S.; Slaughter, G.; Ward, K.R. Design of a subcutaneous implantable biochip for monitoring of glucose and lactate. IEEE Sens. J 2005, 5, 345–355. [Google Scholar]
- Abraham, S.; Brahim, S.; Ishihara, K.; Guiseppi-Elie, A. Molecularly engineered p(HEMA)-based hydrogels for implant biochip biocompatibility. Biomaterials 2005, 26, 4767–4778. [Google Scholar]
- Sheppard, N.F.; Lesho, M.J.; McNally, P. Microfabricated conductimetric pH sensor. Sens. Actuators, B 1995, 28, 95–102. [Google Scholar]
- Brahim, S.; Narinesingh, D.; Guiseppi-Elie, A. Bio-smart hydrogels: co-joined molecular recognition and signal transduction in biosensor fabrication and drug delivery. Biosens. Bioelectron 2002, 17, 973–981. [Google Scholar]
- Pogorelova, S.P.; Bourenko, T.; Kharitonov, A.B.; Willner, I. Selective sensing of triazine herbicides in imprinted membranes using ion-sensitive field-effect transistors and microgravimetric quartz crystal microbalance measurements. Analyst 2002, 127, 1484–1491. [Google Scholar]
- Sallacan, N.; Zayats, M.; Bourenko, T.; Kharitonov, A.B.; Willner, I. Imprinting of nucleotide and monosaccharide recognition sites in acrylamidephenylboronic acid-acrylamide copolymer membranes associated with electronic transducers. Anal. Chem 2002, 74, 702–712. [Google Scholar]
- Dempsey, E.; Diamond, D.; Smyth, M.R.; Urban, G.; Jobst, G.; Moser, I.; Verpoorte, E.M.J.; Manz, A.; Widmer, H.M.; Rabenstein, K.; Freaney, R. Design and development of a miniaturised total chemical analysis system for on-line lactate and glucose monitoring in biological samples. Anal. Chim. Acta 1997, 346, 341–349. [Google Scholar]
- Moser, I.; Jobst, G.; Urban, G.A. Biosensor arrays for simultaneous measurement of glucose, lactate, glutamate, and glutamine. Biosens. Bioelectron 2002, 17, 297–302. [Google Scholar]
- Justin, G.; Finley, S.; Rahman, A.R.; Guiseppi-Elie, A. Biomimetic hydrogels for biosensor implant biocompatibility: electrochemical characterization using micro-disc electrode arrays (MDEAs). Biomed. Microdevices 2009, 11, 103–115. [Google Scholar]
- Justin, G.; Rahman, A.R.A.; Guiseppi-Elie, A. Bioactive hydrogel layers on microdisk electrode arrays: cyclic voltammetry experiments and simulations. Electroanalysis 2009, 21, 1125–1134. [Google Scholar]
- Guiseppi-Elie, A. Electroconductive hydrogels: synthesis, characterization and biomedical applications. Biomaterials 2010, 31, 2701–2716. [Google Scholar]
- Ma, W.M.J.; Morais, M.P.P.; D’Hooge, F.; van den Elsen, J.M.H.; Cox, J.P.L.; James, T.D.; Fossey, J.S. Dye displacement assay for saccharide detection with boronate hydrogels. Chem. Commun 2009, 5, 532–534. [Google Scholar]
- Johnsson, B.; Lofas, S.; Lindquist, G. Immobilization of proteins to a carboxymethyldextran-modified gold surface for biospecific interaction analysis in surface-plasmon resonance sensors. Anal. Biochem 1991, 198, 268–277. [Google Scholar]
- Rothenhausler, B.; Knoll, W. Surface-plasmon microscopy. Nature 1988, 332, 615–617. [Google Scholar]
- Andersson, O.; Larsson, A.; Ekblad, T.; Liedberg, B. Gradient hydrogel matrix for microarray and biosensor applications: an imaging SPR study. Biomacromolecules 2009, 10, 142–148. [Google Scholar]
- Aulasevich, A.; Roskamp, R.F.; Jonas, U.; Menges, B.; Dostalek, J.; Knoll, W. Optical waveguide spectroscopy for the investigation of protein-functionalized hydrogel films. Macromol. Rapid Commun 2009, 30, 872–877. [Google Scholar]
- Hjelme, D.R.; Berg, A.; Ellingsen, R.; Falch, B.; Bjorkoy, A.; Ostling, D. Optical sensing of measurands. United States Patent 2003. 20030112443. [Google Scholar]
- Kimble, K.W.; Walker, J.P.; Finegold, D.N.; Asher, S.A. Progress toward the development of a point-of-care photonic crystal ammonia sensor. Anal. Bioanal. Chem 2006, 385, 678–685. [Google Scholar]
- Hiltner, P.A.; Krieger, I.M. Diffraction of light by ordered suspensions. J. Phys. Chem 1969, 73, 2386–2389. [Google Scholar]
- Hu, X.B.; Li, G.T.; Li, M.H.; Huang, J.; Li, Y.; Gao, Y.B.; Zhang, Y.H. Ultrasensitive specific stimulant assay based on molecularly imprinted photonic hydrogels. Adv. Funct. Mater 2008, 18, 575–583. [Google Scholar]
- Marshall, A.J.; Blyth, J.; Davidson, C.A.B.; Lowe, C.R. PH-sensitive holographic sensors. Anal. Chem 2003, 75, 4423–4431. [Google Scholar]
- Marshall, A.J.; Young, D.S.; Blyth, J.; Kabilan, S.; Lowe, C.R. Metabolite-sensitive holographic biosensors. Anal. Chem 2004, 76, 1518–1523. [Google Scholar]
- Lee, M.C.; Kabilan, S.; Hussain, A.; Yang, X.P.; Blyth, J.; Lowe, C.R. Glucose-sensitive holographic sensors for monitoring bacterial growth. Anal. Chem 2004, 76, 5748–5755. [Google Scholar]
- Tan, E.V.; Lowe, C.R. Holographic Enzyme Inhibition Assays for Drug Discovery. Anal. Chem 2009, 81, 7579–7589. [Google Scholar]
- Kim, J.S.; Singh, N.; Lyon, L.A. Label-free biosensing with hydrogel microlenses. Angew. Chem., Int. Ed 2006, 45, 1446–1449. [Google Scholar]
- Kim, J.S.; Singh, N.; Lyon, L.A. Displacement-induced switching rates of bioresponsive hydrogel microlenses. Chem. Mater 2007, 19, 2527–2532. [Google Scholar]
- Dong, L.; Agarwal, A.K.; Beebe, D.J.; Jiang, H.R. Adaptive liquid microlenses activated by stimuli-responsive hydrogels. Nature 2006, 442, 551–554. [Google Scholar]
- Chinowsky, T.M.; Soelberg, S.D.; Baker, P.; Swanson, N.R.; Kauffman, P.; Mactutis, A.; Grow, M.S.; Atmar, R.; Yee, S.S.; Furlong, C.E. Portable 24-analyte surface plasmon resonance instruments for rapid, versatile biodetection. Biosens. Bioelectron 2007, 22, 2268–2275. [Google Scholar]
- Han, I.S.; Han, M.H.; Kim, J.; Lew, S.; Lee, Y.J.; Horkay, F.; Magda, J.J. Constant-volume hydrogel osmometer: A new device concept for miniature biosensors. Biomacromolecules 2002, 3, 1271–1275. [Google Scholar]
- Lin, G.; Chang, S.; Kuo, C.H.; Magda, J.; Solzbacher, F. Free swelling and confined smart hydrogels for applications in chemomechanical sensors for physiological monitoring. Sens. Actuators B 2009, 136, 186–195. [Google Scholar]
- Herber, S.; Eijkel, J.; Olthuis, W.; Bergveld, P.; van den Berg, A. Study of chemically induced pressure generation of hydrogels under isochoric conditions using a microfabricated device. J. Chem. Phys 2004, 121, 2746–2751. [Google Scholar]
- Lin, G.; Chang, S.; Hao, H.; Tathireddy, P.; Orthner, M.; Magda, J.; Solzbacher, F. Osmotic swelling pressure response of smart hydrogels suitable for chronically implantable glucose sensors. Sens. Actuators B 2010, 144, 332–336. [Google Scholar]
- Herber, S.; Bomer, J.; Olthuis, W.; Bergveld, P.; van den Berg, A. A miniaturized carbon dioxide gas sensor based on sensing of pH-sensitive hydrogel swelling with a pressure sensor. Biom. Microdev 2005, 7, 197–204. [Google Scholar]
- Strong, Z.A.; Wang, A.W.; McConaghy, C.F. Hydrogel-actuated capacitive transducer for wireless biosensors. Biom. Microdev 2002, 4, 97–103. [Google Scholar]
- Ziaie, B.; Baldi, A.; Lei, M.; Gu, Y.D.; Siegel, R.A. Hard and soft micromachining for BioMEMS: review of techniques and examples of applications in microfluidics and drug delivery. Adv. Drug Delivery Rev 2004, 56, 145–172. [Google Scholar]
- Lei, M.; Baldi, A.; Nuxoll, E.; Siegel, R.A.; Ziaie, B. A hydrogel-based implantable micromachined transponder for wireless glucose measurement. Diab. Tech. Ther 2006, 8, 112–122. [Google Scholar]
- Hilt, J.Z.; Gupta, A.K.; Bashir, R.; Peppas, N.A. Ultrasensitive biomems sensors based on microcantilevers patterned with environmentally responsive hydrogels. Biom. Microd 2003, 5, 177–184. [Google Scholar]
- Carrascosa, L.G.; Moreno, M.; Alvarez, M.; Lechuga, L.M. Nanomechanical biosensors: a new sensing tool. TrAC, Trends Anal. Chem 2006, 25, 196–206. [Google Scholar]
- Liu, K.; Ji, H.F. Detection of Pb2+ using a hydrogel swelling microcantilever sensor. Anal. Sci 2004, 20, 9–11. [Google Scholar]
- Bashir, R.; Hilt, J.Z.; Elibol, O.; Gupta, A.; Peppas, N.A. Micromechanical cantilever as an ultrasensitive pH microsensor. Appl. Phys. Lett 2002, 81, 3091–3093. [Google Scholar]
- Lei, M.; Ziaie, B.; Nuxoll, E.; Ivan, K.; Noszticzius, Z.; Siegel, R.A. Integration of hydrogels with hard and soft microstructures. J. Nanosci. Nanotechnol 2007, 7, 780–789. [Google Scholar]
- Singamaneni, S.; LeMieux, M.C.; Lang, H.P.; Gerber, C.; Lam, Y.; Zauscher, S.; Datskos, P.G.; Lavrik, N.V.; Jiang, H.; Naik, R.R.; Bunning, T.J.; Tsukruk, V.V. Bimaterial microcantilevers as a hybrid sensing platform. Adv. Mater 2008, 20, 653–680. [Google Scholar]
- Hook, F.; Kasemo, B.; Nylander, T.; Fant, C.; Sott, K.; Elwing, H. Variations in coupled water, viscoelastic properties, and film thickness of a Mefp-1 protein film during adsorption and cross-linking: A quartz crystal microbalance with dissipation monitoring, ellipsometry, and surface plasmon resonance study. Anal. Chem 2001, 73, 5796–5804. [Google Scholar]
- Chen, H.M.; Huang, T.H.; Tsai, R.M. A biotin-hydrogel-coated quartz crystal microbalance biosensor and applications in immunoassay and peptide-displaying cell detection. Anal. Biochem 2009, 392, 1–7. [Google Scholar]
- Asberg, P.; Bjork, P.; Hook, F.; Inganas, O. Hydrogels from a water-soluble zwitterionic polythiophene: Dynamics under pH change and biomolecular interactions observed using quartz crystal microbalance with dissipation monitoring. Langmuir 2005, 21, 7292–7298. [Google Scholar]
- Carrigan, S.D.; Scott, G.; Tabrizian, M. Real-time QCM-D immunoassay through oriented antibody immobilization using cross-linked hydrogel biointerfaces. Langmuir 2005, 21, 5966–5973. [Google Scholar]
- Stair, J.L.; Watkinson, M.; Krause, S. Sensor materials for the detection of proteases. Biosens. Bioelectron 2009, 24, 2113–2118. [Google Scholar]
- Malitesta, C.; Losito, I.; Zambonin, P.G. Molecularly imprinted electrosynthesized polymers: New materials for biomimetic sensors. Anal. Chem 1999, 71, 1366–1370. [Google Scholar]
- Muscatello, M.M.W.; Stunja, L.E.; Thareja, P.; Wang, L.L.; Bohn, J.J.; Velankar, S.S.; Asher, S.A. Dependence of Photonic Crystal Nanocomposite Elasticity on Crystalline Colloidal Array Particle Size. Macromolecules 2009, 42, 4403–4406. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gawel, K.; Barriet, D.; Sletmoen, M.; Stokke, B.T. Responsive Hydrogels for Label-Free Signal Transduction within Biosensors. Sensors 2010, 10, 4381-4409. https://doi.org/10.3390/s100504381
Gawel K, Barriet D, Sletmoen M, Stokke BT. Responsive Hydrogels for Label-Free Signal Transduction within Biosensors. Sensors. 2010; 10(5):4381-4409. https://doi.org/10.3390/s100504381
Chicago/Turabian StyleGawel, Kamila, David Barriet, Marit Sletmoen, and Bjørn Torger Stokke. 2010. "Responsive Hydrogels for Label-Free Signal Transduction within Biosensors" Sensors 10, no. 5: 4381-4409. https://doi.org/10.3390/s100504381
APA StyleGawel, K., Barriet, D., Sletmoen, M., & Stokke, B. T. (2010). Responsive Hydrogels for Label-Free Signal Transduction within Biosensors. Sensors, 10(5), 4381-4409. https://doi.org/10.3390/s100504381




