State-of-the-Art of (Bio)Chemical Sensor Developments in Analytical Spanish Groups
Abstract
:1. Introduction
2. Materials and Technologies in Chemical and Biochemical Sensors
2.1. Nanomaterials and nanotechnologies
2.1.1. Carbon nanotubes
2.1.2. Noble metal nanoparticles
2.1.3. Quantum dots
2.1.4. Magnetic beads
2.1.5. Metal nanoclusters
2.1.6. Sensor nanofilms
Self-assembled monolayers
Langmuir-Blodgett films
2.2. Materials for chemical sensing
2.2.1. Molecular imprinted polymers
2.2.2. Metal complexes
2.2.3. Sol-gel materials
2.2.4. Organic ligands
2.2.5. Other materials
3. Electrochemical Sensors
3.1. Electronic tongues
3.2. Ion-selective electrodes
3.3. Modified electrodes
3.4. Electrochemical flow-through sensors
4. Optical Sensors
4.1. Optical fibers
4.2. Optical flow injetion sensors
4.3. Surface plasmon resonance
4.4. Mach-Zehnder interferometric (MZI) biosensor
4.5. Attenuated total reflection Fourier sensors
4.6. Slot-waveguide based refractometric sensors
4.7. Cantilever sensors
4.8. Commercial optical biosensors
5. Piezoelectric Sensors
5.1. Quartz crystal microbalance
5.2. Cantilever
6. Commercial Spanish Devices
7. Conclusions
Acknowledgments
References
- Rivera, L.; Izquierdo, D.; Garcés, I.; Salinas, I.; Alonso, J.; Puyol, M. Simple dip-probe fluorescence setup sensor for in situ environmental determinations. Sens. Actuat. B 2009, 137, 420–425. [Google Scholar]
- Martínez-Cisneros, C.S.; Ibáñez-García, N.; Valdés, F.; Alonso, J. LTCC microflow analyzers with monolithic integration of thermal control. Sens. Actuat. A 2007, 138, 63–70. [Google Scholar]
- Gallardo, J.; Alegret, S.; Valle, M. Flow-injection electronic tongue based on potentiometric sensors for the determination of nitrate in the presence of chloride. Sens. Actuat. B 2004, 101, 72–80. [Google Scholar]
- Bosch-Ojeda, C.; Sánchez-Rojas, F. Recent development in optical chemical sensors coupling with flow injection analysis. Sensors 2006, 6, 1245–1307. [Google Scholar]
- Farré, M.; Kantiani, L.; Pérez, S.; Barcelo, D. Sensors and biosensors in support of EU Directives. Trends Anal. Chem 2009, 28, 170–185. [Google Scholar]
- Goeders, K.M.; Colton, J.S.; Bottomley, L.A. Amperometric gas sensors. A review. Chem. Rev 2008, 108, 522–542. [Google Scholar]
- Alegret, S. Integrated Analytical Systems; Elsevier: Amsterdam, The Netherlands, 2003; pp. 13–16. [Google Scholar]
- Rosi, N.L.; Mirkin, C.A. Nanostructures and biodiagnosics. Chem. Rev 2005, 105, 1547–1562. [Google Scholar]
- Merkoçi, A. Nanobiomaterials in electroanalysis. Electroanalysis 2007, 19, 739–741. [Google Scholar]
- Lavrik, N.V.; Sepaniak, M.J.; Datskos, P.G. Cantilever transducers as a platform for chemical and biological sensors. Rev. Sci. Instrum 2004, 75, 2229–2253. [Google Scholar]
- Puyol, M.; Encinas, C.; Rivera, L.; Miltsov, S.; Alonso, J. Nortricarbocyanine dyes as suitable long wavelength pH indicators for chemical sensing. Sens. Actuat. B 2007, 122, 53–59. [Google Scholar]
- Shtykov, S.N.; Rusanova, T.Y. Nanomaterials and nanotechnologies in chemical and biochemical sensors: capabilities and applications. Russian J. Gen. Chem 2008, 78, 2521–2531. [Google Scholar]
- Riu, J.; Maroto, A.; Rius, F.X. Nanosensors in environmental analysis. Talanta 2006, 69, 288–301. [Google Scholar]
- Dresselhaus, M.S.; Dresselhaus, G.; Eklund, P.C. Science of Fullerenes and Carbon Nanotubes; Academic Press: New York, NY, USA, 1996. [Google Scholar]
- Treachy, M.M.; Ebbesen, T.W.; Gibson, J.M. Exceptionally high youngs modulus observed for individual carbon nanotubes. Nature 1996, 381, 678–680. [Google Scholar]
- Sánchez-Acevedo, Z.C.; Riu, J.; Rius, F.X. Fast picomolar selective detection of bisphenol A in water using a carbon nanotube field effect transistor functionalized with estrogen receptor-α. Biosens. Bioelectron 2009, 24, 2842–2846. [Google Scholar]
- Villamizar, R.A.; Maroto, A.; Rius, F.X. Improved detection of Candida albicans with carbon nanotube field-effect transistors. Sens. Actuat. B 2009, 136, 451–457. [Google Scholar]
- Cid, C.C.; Riu, J.; Maroto, A.; Rius, F.X. Detection of human immunoglobulin G at physiological conditions with chemically functionalizated carbon nanotube field effect transistors. Curr. Nanosci 2008, 4, 314–317. [Google Scholar]
- Cid, C.C.; Riu, J.; Maroto, A.; Rius, F.X. Carbon nanotube field effect transistors for the fast and selective detection of human immunoglobulin G. Analyst 2008, 133, 1005–1008. [Google Scholar]
- Alarcón-Angeles, G.; Pérez-López, B.; Palomar-Pardave, M.; Ramírez-Silva, M.T.; Alegret, S.; Merkoçi, A. Enhanced host-guest electrochemical recognition of dopamine using cyclodextrin in the presence of carbon nanotubes. Carbon 2008, 46, 898–906. [Google Scholar]
- Alarcón-Angeles, G.; Corona-Avendano, S.; Palomar-Pardave, M.E.; Merkoci, A.; Romero-Romo, M.A.; Rojas-Hernández, A.; Ramirez-Silva, M.T. Electrochemical study of dopamine and ascorbic acid by means of supramolecular systems. ECS Trans 2008, 15, 325–334. [Google Scholar]
- Agui, L.; Eguilaz, M.; Pena-Farfal, C.; Yanez-Sedeno, P.; Pingarrón, J.M. Lactate dehydrogenase biosensor based on an hybrid carbon nanotube-conducting polymer modified electrode. Electroanalysis 2009, 21, 386–391. [Google Scholar]
- Pumera, M.; Merkoçi, A.; Alegret, S. Carbon nanotube-epoxy composites for electrochemical sensing. Sens. Actuat. B 2006, 113, 617–622. [Google Scholar]
- Perez, B.; Pumera, M.; del Valle, M.; Merkoci, A.; Alegret, S. Glucose biosensor based on carbon nanotube epoxy composites. J. Nanosci. Nanotechnol 2005, 5, 1694–1698. [Google Scholar]
- Kirgoz, U.A.; Timur, S.; Odaci, D.; Perez, B.; Alegret, S.; Merkoci, S. Carbon nanotube composite as novel platform for microbial biosensor. Electroanalysis 2007, 19, 893–898. [Google Scholar]
- López, B.P.; Merkoci, A. Improvement of the electrochemical detection of catechol by the use of a carbon nanotube based biosensor. Analyst 2009, 134, 60–64. [Google Scholar]
- Mendoza, E.; Orozco, J.; Jiménez-Jorquera, C.; González-Guerrero, A.B.; Calle, A.; Lechuga, L.M.; Fernández-Sánchez, C. Scalable fabrication of immunosensors based on carbon nanotube polymer composites. Nanotechnology 2008, 19, 075102/1–075102/6. [Google Scholar]
- Hernández-Santos, D.; González-García, M.B.; Costa-García, A. Metal-nanoparticles based electroanalysis. Electroanalysis 2002, 14, 1225–1235. [Google Scholar]
- Penn, S.G.; He, L.; Natan, M. Nanoparticles for bioanalysis. Curr. Opin. Chem. Biol 2003, 7, 609–615. [Google Scholar]
- Gooding, J.J. Electrochemical DNA Hybridization Biosensors. Electroanalysis 2002, 14, 1149–1156. [Google Scholar]
- Zamora, P.; Narváez, A.; Domínguez, E. Enzyme-modified nanoparticles using biomimetically synthesized silica. Bioelectrochemistry 2009, 76, 100–106. [Google Scholar]
- Jiménez, J.; Sheparovych, R.; Pita, M.; Narvaez García, A.; Domínguez, E.; Minko, S.; Katz, E. Magneto-induced self-assembling of conductive nanowires for biosensor applications. J. Phys. Chem. C 2008, 112, 7337–7344. [Google Scholar]
- Lucarelli, F.; Marrazza, G.; Turner, A.P.F.; Mascini, M. Carbon and gold electrodes as electrochemical transducers for DNA hybridisation sensors. Biosens. Bioelectron 2004, 19, 515–530. [Google Scholar]
- Merkoçi, A.; Aldavert, M.; Marín, S.; Alegret, S. New materials for electrochemical sensing V: Nanoparticles for DNA labelling. Trends Anal. Chem 2005, 24, 341–349. [Google Scholar]
- Pumera, M.; Aldavert, M.; Miles, C.; Merkoçi, A.; Alegret, S. Direct voltammetric determination of gold nanoparticles using graphite-epoxy composite electrode. Electrochim. Acta 2005, 50, 3702–3707. [Google Scholar]
- González-García, M.B.; Costa-García, A. Adsorptive stripping voltammetric behaviour of colloidal gold and immunogold on carbon paste electrode. Bioelectrochem. Bioenerg 1995, 38, 389–395. [Google Scholar]
- Pumera, M.; Castañeda, M.T.; Pividori, M.I.; Eritja, R.; Merkoçi, A.; Alegret, S. Magnetically trigged direct electrochemical detection of DNA hybridization using Au-67 quantum dot as electrical tracer. Langmuir 2005, 21, 9625–9629. [Google Scholar]
- Castañeda, M.T.; Merkoçi, A.; Pumera, M.; Alegret, S. Electrochemical genosensors for biomedical applications based on gold nanoparticles. Biosens. Bioelectron 2007, 22, 1961–1967. [Google Scholar]
- García, T.; Revenga-Parra, M.; Abruna, H.D.; Pariente, F.; Lorenzo, E. Single-Mismatch Position-Sensitive Detection of DNA Based on a Bifunctional Ruthenium Complex. Anal. Chem 2008, 80, 77–84. [Google Scholar]
- García, T.; Casero, E.; Revenga-Parra, M.; Martín-Benito, J.; Pariente, F.; Vazquez, L.; Lorenzo, E. Architectures based on the use of gold nanoparticles and ruthenium complexes as a new route to improve genosensor sensitivity. Biosens. Bioelectron 2008, 24, 184–190. [Google Scholar]
- García, T.; Casero, E.; Revenga-Parra, M.; Pariente, F.; Lorenzo, E. Dual-Stage DNA Sensing: Recognition and Detection. Anal. Chem 2008, 80, 9443–9449. [Google Scholar]
- Agui, L.; Manso, J.; Yañez-Sendeño, P.; Pingarrón, J.M. Amperometric biosensor for hypoxanthine based on immobilized xanthine oxidase on nanocrystal gold-carbon paste electrodes. Sens. Actuat. B 2006, 113, 272–280. [Google Scholar]
- Renedo, O.D.; Arcos Martínez, M.J. A novel method for the anodic stripping voltammetry determination of Sb(III) using silver nanoparticle-modified screen-printed electrodes. Electrochem. Commun 2007, 9, 820–826. [Google Scholar]
- Burgoa-Calvo, M.E.; Domínguez-Renedo, O.; Arcos-Martínez, M.J. Determination of lamotrigine by adsorptive stripping voltammetry using silver nanoparticle modified carbon screen-printed electrodes. Talanta 2007, 74, 59–64. [Google Scholar]
- Domínguez-Renedo, O.; Arcos-Martínez, M.J. Anodic stripping voltammetry of antimony using gold nanoparticle-modified carbon screenprinted electrodes. Anal. Chim. Acta 2007, 589, 255–260. [Google Scholar]
- Sanllorente-Mendez, S.; Domínguez-Renedo, O.; Arcos-Martínez, M.J. Determination of arsenic(III) using platinum nanoparticle-modified screen-printed carbon based electrodes. Electroanalysis 2009, 21, 635–639. [Google Scholar]
- Domínguez-Renedo, O.; Ruiz-Espelt, L.; García-Astorgano, N.; Arcos-Martínez, M.J. Electrochemical determination of chromium(VI) using metallic nanoparticle-modified carbon screen-printed electrodes. Talanta 2008, 76, 854–858. [Google Scholar]
- Domínguez-Renedo, O.; Alonso-Lomillo, M.A.; Ferreira-Goncalves, L.; Arcos-Martínez, M.J. Development of urease based amperometric biosensors for the inhibitive determination of Hg (II). Talanta 2009, 79, 1306–1310. [Google Scholar]
- Alonso-Lomillo, M.A.; Yardimci, C.; Domínguez-Renedo, O.; Arcos-Martínez, M.J. CYP450 2B4 covalently attached to carbon and gold screen printed electrodes by diazonium salt and thiols monolayers. Anal. Chim. Acta 2009, 633, 51–56. [Google Scholar]
- Muraviev, D.N.; Macanás, J.; Farré, M.; Muñoz, M.; Alegret, S. Novel routes for inter-matrix synthesis and characterization of polymer stabilized metal nanoparticles for molecular recognition devices. Sens. Actuat. B 2006, 118, 408–417. [Google Scholar]
- Muraviev, D.N.; Pividory, M.L.; Soto, J.L.M.; Alegret, S. Extractant assisted synthesis of polymer stabilized platinum and palladium metal nanoparticles for sensor applications. Solvent Extr. Ion Exch 2006, 24, 731–745. [Google Scholar]
- Zhong, C.; Maye, M.M. Core-Shell Assembled Nanoparticles as Catalysts. Adv. Mater 2001, 13, 1507–1511. [Google Scholar]
- Son, S.U.; Jang, Y.; Park, J.; Na, H.B.; Park, H.M.; Yun, H.J.; Lee, J.; Hyeon, T. Designed synthesis of atom-economical Pd/Ni bimetallic nanoparticle-based catalysts for sonogashira coupling reactions. J. Am. Chem. Soc 2004, 126, 5026–5027. [Google Scholar]
- Sao-Joao, S.; Giorgio, S.; Penisson, J.M.; Chapon, C.; Bourgeois, S.; Henry, C. Structure and deformation of Pd-Ni-Core-shell nanoparticle. J. Phys. Chem. B 2005, 109, 342–347. [Google Scholar]
- Zhou, S.; Varughese, B.; Eichhorn, B.; Jackson, G.; McIlwrath, K. Pt-Cu Core-Shell and Alloy Nanoparticles for Heterogeneous NOx Reduction: Anomalous Stability and Reactivity of a Core-Shell Nanostructure. Angew. Chem., Int. Ed 2005, 44, 4539–4543. [Google Scholar]
- Muraviev, D.N.; Macanas, J.; Esplandiu, M.J.; Farré, M.; Munoz, M.; Alegret, S. Simple route for intermatrix synthesis of polymer stabilized core-shell metal nanoparticles for sensor applications. Phys. Status Solidi A 2007, 204, 1686–1692. [Google Scholar]
- Muraviev, D.N.; Macanas, J.; Larrondo, J.; Munoz, M.; Alonso, A.; Alegret, S.; Ortueta, M.; Mijangos, F. Cation-exchange membrane as nanoreactor: Intermatrix synthesis of platinum-copper coreshell nanoparticles. React. Funct. Polym 2007, 67, 1612–1621. [Google Scholar]
- Douglas, F.; Yanez, R.; Ros, J.; Marin, S.; de la Escosura-Muníz, A.; Alegret, S.; Merkoci, A. Silver, gold and the corresponding core shell nanoparticles: synthesis and characterization. J. Nanopart. Res 2008, 10, 97–106. [Google Scholar]
- Murphy, C.J.; Coffer, J.L. Quantum dots: a primer. Appl. Spectrosc 2002, 56, 16–27. [Google Scholar]
- Alivisatos, A.P. Semiconductor Clusters, Nanocrystals, and Quantum Dots. Science 1996, 271, 933–937. [Google Scholar]
- Costa-Fernández, J.M.; Pereiro, R.; Sanz-Medel, A. The use of luminescent quantum dots for optical sensing. Trends Anal. Chem 2006, 25, 207–218. [Google Scholar]
- Fernández-Arguelles, M.T.; Jin, W.J.; Costa-Fernández, J.M.; Pereiro, R.; Sanz-Medel, A. Surface-modified CdSe quantum dots for the sensitive and selective determination of Cu(II) in aqueous solutions by luminescent measurements. Anal. Chim. Acta 2005, 549, 20–25. [Google Scholar]
- Jin, W.J.; Fernández-Arguelles, M.T.; Costa-Fernández, J.M.; Pereiro, R.; Sanz-Medel, A. Photoactivated luminescent CdSe quantum dots as sensitive cyanide probes in aqueous solutions. Chem. Commun 2005, 7, 883–885. [Google Scholar]
- Richardson, J.; Hawkins, P.; Luxton, R. The use of coated paramagnetic particles as a physical label in a magneto-immunoassay. Biosens. Bioelectron 2001, 16, 989–993. [Google Scholar]
- Pividori, M.I.; Lermo, A.; Hernández, S.; Barbe, J.; Alegret, S.; Campoy, S. Rapid electrochemical DNA biosensing strategy for the detection of food pathogens based on enzyme-DNA-magnetic bead conjugate. Afinidad 2006, 63, 13–18. [Google Scholar]
- Lermo, A.; Campoy, S.; Barbe, J.; Hernández, S.; Alegret, S.; Pividori, M.I. In situ DNA amplification with magnetic primers for the electrochemical detection of food pathogens. Biosens. Bioelectron 2007, 22, 2010–2017. [Google Scholar]
- Castaneda, M.T.; Alegret, S.; Merkoci, A. Electrochemcial detection of DNA hybridization using micro and nanoparticles. Methods Mol. Biol 2009, 504, 127–143. [Google Scholar]
- Zacco, E.; Adrian, J.; Galve, R.; Marco, M.P.; Alegret, S.; Pividori, M.I. Electrochemical magneto immunosensing of antibiotic residues in milk. Biosens. Bioelectron 2007, 22, 2184–2191. [Google Scholar]
- Lermo, A.; Fabiano, S.; Hernández, S.; Galve, R.; Marco, M.P.; Alegret, S.; Pividori, M.I. Immunoassay for folic acid detection in vitamin-fortified milk based on electrochemical magneto sensors. Biosens. Bioelectron 2009, 24, 2057–2063. [Google Scholar]
- De la Escosura-Muníz, A.; Costa, M.M.D.; Merkoci, A. Controlling the electrochemical deposition of silver onto gold nanoparticles: Reducing interferences and increasing the sensitivity of magnetoimmuno assays. Biosens. Bioelectron 2009, 24, 2475–2482. [Google Scholar]
- Cosnier, S.; Ding, S.; Pellissier, A.; Gorgy, K.; Holzinger, M.; López, B.P.; Merkoci, A. Permeability Improvement of Electropolymerized Polypyrrole Films in Water Using Magnetic Hydrophilic Microbeads. Electroanalysis 2009, 21, 887–890. [Google Scholar]
- Macanas, J.; Farré, M.; Munoz, M.; Alegret, S.; Muraviev, D.N. Preparation and characterization of polymer-stabilized metal nanoparticles for sensor applications. Phys. Status Solidi A 2006, 203, 1194–1200. [Google Scholar]
- Campuzano, S.; Pedrero, M.; Pingarrón, J.M. A peroxidase-tetrathiafulvalene biosensor based on self-assembled monolayer modified Au electrodes for the flow-injection determination of hydrogen peroxide. Talanta 2005, 66, 1310–1319. [Google Scholar]
- Loaiza, O.A.; Campuzano, S.; López-Berlanga, M.; Pedrero, M.; Pingarrón, J.M. Development of a DNA sensor based on alkanethiol self-assembled monolayer-modified electrodes. Sensors 2005, 5, 344–363. [Google Scholar]
- Loaiza, O.A.; Campuzano, S.; Pedrero, M.; Pingarrón, J.M. DNA sensor based on an Escherichia coli lac Z gene probe immobilization at self-assembled monolayers-modified gold electrodes. Talanta 2007, 73, 838–844. [Google Scholar]
- Gamella, M.; Campuzano, S.; Reviejo, A.J.; Pingarrón, J.M. Integrated multienzyme electrochemical biosensors for the determination of glycerol in wines. Anal. Chim. Acta 2008, 609, 201–209. [Google Scholar]
- Manso, J.; Mena, M.L.; Yanez-Sedeno, P.; Pingarrón, J.M. Bienzyme amperometric biosensor using gold nanoparticle-modified electrodes for the determination of inulin in foods. Anal. Biochem 2008, 375, 345–353. [Google Scholar]
- Campuzano, S.; Escamilla-Gómez, V.; Herranz, M.A.; Pedrero, M.; Pingarrón, J.M. Development of amperometric biosensors using thiolated tetrathiafulvalene-derivatised self assembled monolayer modified electrodes. Sens. Actuat. B 2008, 134, 974–980. [Google Scholar]
- Escamilla-Gómez, V.; Campuzano, S.; Pedrero, M.; Pingarrón, J.M. Immunosensor for the determination of Staphylococcus aureus using a tyrosinasemercaptopropionic acid modified electrode as an amperometric transducer. Anal. Bioanal. Chem 2008, 391, 837–845. [Google Scholar]
- Escamilla-Gómez, V.; Campuzano, S.; Pedrero, M.; Pingarrón, J.M. Electrochemical immunosensor designs for the determination of Staphylococcus aureus using 3,3-dithiodipropionic acid di(N-succinimidyl ester)-modified gold electrodes. Talanta 2008, 77, 876–881. [Google Scholar]
- Jiménez, C.; Rochefeuille, S.; Berjoan, R.; Seta, P.; Desfours, J.P.; Domínguez, C. Nanostructures for chemical recognition using ISFET sensors. Microelectron. J 2004, 35, 69–71. [Google Scholar]
- Moreno-Bondi, M.C.; Urraca, J.L.; Benito-Pena, E.; Navarro-Villoslada, F.; Martins, S.A.; Orellana, G.; Sellergren, B. Molecularly imprinted polymers as biomimetic receptors for fluorescence-based optical sensors. Proc. SPIE 2007, 6619, 66190C/1–66190C/4. [Google Scholar]
- Urraca, J.L.; Moreno-Bondi, M.C.; Orellana, G.; Sellergren, B.; Hall, A.J. Molecularly Imprinted Polymers as Antibody Mimics in Automated On-Line Fluorescent Competitive Assays. Anal. Chem 2007, 79, 4915–4923. [Google Scholar]
- Benito-Pena, E.; Moreno-Bondi, M.C.; Aparicio, S.; Orellana, G.; Cederfur, J.; Kempe, M. Molecular engineering of fluorescent penicillins for molecularly imprinted polymer assays. Anal. Chem 2006, 78, 2019–2027. [Google Scholar]
- Navarro-Villoslada, F.; Urraca, J.L.; Moreno-Bondi, M.C.; Orellana, G. Zearalenone sensing with molecularly imprinted polymers and tailored fluorescent probes. Sens. Actuat. B 2007, 121, 67–73. [Google Scholar]
- González, G.P.; Hernando, P.F.; Alegría, J.S.D. A morphological study of molecularly imprinted polymers using the scanning electron microscope. Anal. Chim. Acta 2006, 557, 179–183. [Google Scholar]
- González, G.P.; Hernando, P.F.; Alegría, J.S.D. Determination of digoxin in serum samples using a flow-through fluorosensor based on a molecularly imprinted polymer. Biosens. Bioelectron 2008, 23, 1754–1758. [Google Scholar]
- González, G.P.; Hernando, P.F.; Durand-Alegría, J.S. A MIP-based flow-through fluoroimmunosensor as an alternative to immunosensors for the determination of digoxin in serum samples. Anal. Bioanal. Chem 2009, 394, 963–970. [Google Scholar]
- González, G.P.; Hernando, P.F.; Alegría, J.S.D. An optical sensor for the determination of digoxin in serum samples based on a molecularly imprinted polymer membrane. Anal. Chim. Acta 2009, 638, 209–212. [Google Scholar]
- Salinas-Castillo, A.; Sánchez-Barragán, I.; Costa-Fernández, J.M.; Pereiro, R.; Ballesteros, A.; González, J.M.; Segura-Carretero, A.; Fernández-Gutiérrez, A.; Sanz-Medel, A. Iodinated molecularly imprinted polymer for room temperature phosphorescence optosensing of fluoranthene. Chem. Commun 2005, 25, 3224–3226. [Google Scholar]
- Sánchez-Barragán, I.; Costa-Fernández, J.M.; Pereiro, R.; Sanz-Medel, A.; Salinas, A.; Segura, A.; Fernández-Gutiérrez, A.; Ballesteros, A.; González, J.M. Molecularly Imprinted Polymers Based on Iodinated Monomers for Selective Room-Temperature Phosphorescence Optosensing of Fluoranthene in Water. Anal. Chem 2005, 77, 7005–7011. [Google Scholar]
- Valero-Navarro, A.; Damiani, P.C.; Fernández-Sánchez, J.F.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Chemometric-assisted MIP-optosensing system for the simultaneous determination of monoamine naphthalenes in drinking waters. Talanta 2009, 78, 57–65. [Google Scholar]
- Valero-Navarro, A.; Salinas-Castillo, A.; Fernández-Sánchez, J.F.; Segura-Carretero, A.; Mallavia, R.; Fernández-Gutiérrez, A. The development of a MIP-optosensor for the detection of monoamine naphthalenes in drinking water. Biosens. Bioelectron 2009, 24, 2305–2311. [Google Scholar]
- Medina-Castillo, A.L.; Fernández-Sánchez, J.F.; Segura-Carretero, A.; Fernández-Gutiérrez, A. A semi-empirical model to simplify the synthesis of homogeneous and transparent crosslinked polymers and their application in the preparation of optical sensing films. Biosens. Bioelectron 2009, 25, 442–449. [Google Scholar]
- Dineiro, Y.; Menendez, M.I.; Blanco-López, M.C.; Lobo-Castanon, M.J.; Miranda-Ordieres, A.J.; Tunon-Blanco, P. Computational approach to the rational design of molecularly imprinted polymers for voltammetric sensing of homovanillic acid. Anal. Chem 2005, 77, 6741–6746. [Google Scholar]
- Gómez-Caballero, A.; Unceta, N.; Aranzazu Goicolea, M.; Barrio, R.J. Evaluation of the selective detection of 4,6-dinitro-o-cresol by a molecularly imprinted polymer based microsensor electrosynthesized in a semiorganic media. Sens. Actuat. B 2008, 130, 713–722. [Google Scholar]
- Gómez-Caballero, A.; Unceta, N.; Goicolea, M.A.; Barrio, R.J. Voltammetric determination of metamitron with an electrogenerated molecularly imprinted polymer microsensor. Electroanalysis 2007, 19, 356–363. [Google Scholar]
- Valero-Navarro, A.; Fernández-Sánchez, J.F.; Segura-Carretero, A.; Spichiger-Keller, U.E.; Fernández-Gutiérrez, A.; Ona, P.; Fernández, I. Iron-phthalocyanine complexes immobilized in nanostructured metal oxide as optical sensors of NOx and CO: NMR and photophysical studies. J. Porphyrins Phthalocyanines 2009, 13, 616–623. [Google Scholar]
- Medina-Castillo, A.L.; Fernández-Sánchez, J.F.; Klein, C.; Nazeeruddin, M.K.; Segura-Carretero, A.; Fernández-Gutiérrez, A.; Graetzel, M.; Spichiger-Keller, U.E. Engineering of efficient phosphorescent iridium cationic complex for developing oxygen sensitive polymeric and nanostructured films. Analyst 2007, 132, 929–936. [Google Scholar]
- Casado-Terrones, S.; Elosua-Aguado, C.; Bariain, C.; Segura-Carretero, A.; Matias-Maestro, I.R.; Fernández-Gutiérrez, A.; Luquin, A.; Garrido, J.; Laguna, M. Volatile-organic-compound optic fiber sensor using a gold-silver vapochromic complex. Opt. Eng 2006, 45, 044401/1–044401/7. [Google Scholar]
- García, E.A.; Fernández, R.G.; Díaz-García, M.E. Tris(bipyridine)ruthenium(II) doped sol-gel materials for oxygen recognition in organic solvents. Microporous Mesoporous Mater 2005, 77, 235–239. [Google Scholar]
- Basabe-Desmonts, L.; Reinhoudt, D.N.; Crego-Calama, M. Design of fluorescent materials for chemical sensing. Chem. Soc. Rev 2007, 36, 993–1017. [Google Scholar]
- Llobera, A.; Cadarso, V.J.; Darder, M.; Domínguez, C.; Fernández-Sánchez, C. Full-field photonic biosensors based on tunable bio-doped sol-gel glasses. Lab Chip 2008, 8, 1185–1190. [Google Scholar]
- Fernández-Sánchez, C.; Cadarso, V.J.; Darder, M.; Domínguez, C.; Llobera, A. Patterning high-aspect-ratio sol-gel structures by microtransfer molding. Chem. Mater 2008, 20, 2662–2668. [Google Scholar]
- Costero, A.M.; Llaosa, U.; Gil, S.; Parra, M.; Colera, M. Enantioselective sensing of dicarboxylates. Influence of the stoichiometry of the complexes on the sensing mechanism. Tetrahedron: Asymmetry 2009, 20, 1468–1471. [Google Scholar]
- Costero, A.M.; Colomer, J.V.; Gil, S.; Parra, M. Fluorescent cyclohexyl-based chemosensors for selective sensing of TMA malonate in DMSO/water. Eur. J. Org. Chem 2009, 22, 3673–3677. [Google Scholar]
- Costero, A.M.; Colera, M.; Gavina, P.; Gil, S.; Kubinyi, M.; Pal, K.; Kallay, M. Chiral cyclohexane based fluorescent chemosensors for enantiomeric discrimination of aspartate. Tetrahedron 2008, 64, 3217–3224. [Google Scholar]
- Costero, A.M.; Colera, M.; Gavina, P.; Gil, S. Fluorescent sensing of maleate versus fumarate by a neutral cyclohexane based thiourea receptor. Chem. Commun 2006, 7, 761–763. [Google Scholar]
- Costero, A.M.; Colera, M.; Gavina, P.; Gil, S.; Llaosa, U. Fluorescent chemosensors based on cyclohexane: selective sensing of succinate and malonate versus their longer or shorter homologues. Tetrahedron 2008, 64, 7252–7257. [Google Scholar]
- Costero, A.M.; Gil, S.; Parra, M.; Allouni, Z.; Lakhmiri, R.; Atlamsani, A. Complexation of α,ω-dicarboxylates by 3,3′-bis(5-phenyl-1,4-dioxo-2,3,5-triaza)-2,2′-bipyridine. J. Inclusion Phenom. Macrocyclic Chem 2008, 62, 203–207. [Google Scholar]
- Costero, A.M.; Gil, S.; Parra, M.; Huguet, N.; Allouni, Z.; Lakmiri, R.; Atlamsani, A. 3,3′-Disubstituted 2,2′-bipyridines as carboxylate receptors: conformational regulation of the bipyridine moiety. Eur. J. Org. Chem 2008, 6, 1079–1084. [Google Scholar]
- Costero, A.M.; Gavina, P.; Rodríguez-Muníz, G.M.; Gil, S. Relationship between ligand conformations and complexation properties in ditopic biphenyl thioureas. Tetrahedron 2007, 63, 7899–7905. [Google Scholar]
- Costero, A.M.; Peransi, S. Colorimetric sensing of anions by a neutral biphenyl based amide receptor. Arkivoc 2007, 4, 92–101. [Google Scholar]
- Costero, A.M.; Gavina, P.; Rodríguez-Muníz, G.M.; Gil, S. N-Biphenyl thioureas as carboxylate receptors. Effect of the ligand substituents on the geometry of the complexes. Tetrahedron 2006, 62, 8571–8577. [Google Scholar]
- Costero, A.M.; Sanchís, J.; Gil, S.; Sanz, V. Influence of Cation Size on the Fluorescent Properties of Bis-coronand Biphenyl-derived Complexes. Supramol. Chem 2007, 19, 151–158. [Google Scholar]
- Costero, A.M.; Banuls, M.J.; Aurell, M.J.; Domenech, A. 4,4′-Substituted biphenyl coronands. Preparation of a new selective fluorescent sensor for mercury salts. Tetrahedron 2006, 62, 11972–11978. [Google Scholar]
- Costero, A.M.; Banuls, M.J.; Aurell, M.J.; De Arellano, M.C.R. Biphenyl Macrolactams as Colorimetric Sensors for Anions through Displacement Reactions. J. Inclusion Phenom. Macrocyclic Chem 2006, 54, 61–66. [Google Scholar]
- Costero, A.M.; Banuls, M.J.; Aurell, M.J.; Ochando, L.E.; Domenech, A. Cation and anion fluorescent and electrochemical sensors derived from 4,4′-substituted biphenyl. Tetrahedron 2005, 61, 10309–10320. [Google Scholar]
- Costero, A.M.; Sanchís, J.; Gil, S.; Sanz, V.; Williams, J.A.G. Poly(amine) biphenyl derivatives as fluorescent sensors for anions and cations. J. Mater. Chem 2005, 15, 2848–2853. [Google Scholar]
- Costero, A.M.; Sanchís, J.; Gil, S.; Sanz, V.; Ramírez de Arellano, M.C.; Gareth Williams, J.A. Polyazapodands derived from biphenyl. Study of their behavior as conformationally regulated fluorescent sensors. Supramol. Chem 2004, 16, 435–446. [Google Scholar]
- Rivero, I.A.; González, T.; Pina-Luis, G.; Díaz-García, M.E. Library preparation of derivatives of 1,4,10,13-tetraoxa-7,16-diaza-cycloctadecane and their fluorescence behavior for signaling purposes. J. Comb.Chem 2005, 7, 46–53. [Google Scholar]
- Rivero, I.A.; González, T.; Díaz-García, M.E. Synthesis of metallothionein-mimic decapeptides with heavy atom signaling properties. Comb. Chem. High Throughput Screen 2006, 9, 535–544. [Google Scholar]
- Tan, J.; Bergantin, J.H.; Merkoci, A.; Alegret, S.; Sevilla, F. Oil dispersion of AgI/Ag2S salts as a new electroactive material for potentiometric sensing of iodide and cyanide. Sens. Actuat. B 2004, 101, 57–62. [Google Scholar]
- Bratov, A.; Abramova, N.; Domínguez, C. Investigation of chloride sensitive ISFETs with different membrane compositions suitable for medical applications. Anal. Chim. Acta 2004, 514, 99–106. [Google Scholar]
- Barbadillo, M.; Casero, E.; Petit-Domínguez, M. D.; Vázquez, L.; Pariente, F.; Lorenzo, E. Gold nanoparticles-induced enhancement of the analytical response of an electrochemical biosensor based on an organic-inorganic hybrid composite material. Talanta 2009, 80, 797–802. [Google Scholar]
- Granado Rico, M.A.; Olivares-Marín, M.; Pinilla-Gil, E. Modification of carbon screen-printed electrodes by adsorption of chemically synthesized Bi nanoparticles for the voltammetric stripping detection of Zn(II), Cd(II) and Pb(II). Talanta 2009, 80, 631–635. [Google Scholar]
- Díaz-García, M.E.; Noval-Gutiérrez, B.; Badia, R. Tailoring room-temperature phosphorescent ormosil particles for oxygen recognition in organic solvents. Sens. Actuat. B 2005, 110, 66–72. [Google Scholar]
- Castillo, M.; Pina-Luis, G.; Díaz-García, M.E.; Rivero, I.A. Solid-phase organic synthesis of sensing sorbent materials for copper and lead recovery. J. Braz. Chem. Soc 2005, 16, 412–417. [Google Scholar]
- Joo, S.; Brown, R.B. Chemical Sensors with Integrated Electronics. Chem. Rev 2008, 108, 638–651. [Google Scholar]
- Abad-Valle, P.; Fernández-Abedul, M.T.; Costa-García, A. DNA single-base mismatch study with an electrochemical enzymatic genosensor. Biosens. Bioelectron 2007, 22, 1642–1650. [Google Scholar]
- Bratov, A.; Abramova, N.; Ramón-Azcón, J.; Merlos, A.; Sánchez-Baeza, F.; Marco, M.P.; Domínguez, C. Characterisation of the interdigitated electrode array with tantalum silicide electrodes separated by insulating barriers. Electrochem. Commun 2008, 10, 1621–1624. [Google Scholar]
- Bratov, A.; Ramón-Azcón, J.; Abramova, N.; Merlos, A.; Adrián, J.; Sánchez-Baeza, F.; Marco, M.P.; Domínguez, C. Three-dimensional interdigitated electrode array as a transducer for label-free biosensors. Biosens. Bioelectron 2008, 24, 729–735. [Google Scholar]
- De-los-Santos-Alvarez, N.; Lobo-Castanón, M.J.; Miranda-Ordieres, A.J.; Tunón-Blanco, P. Modified-RNA Aptamer-Based Sensor for Competitive Impedimetric Assay of Neomycin B. J. Am. Chem. Soc 2007, 129, 3808–3809. [Google Scholar]
- Ipatov, A.; Abramova, N.; Bratov, A.; Domínguez, C. Integrated multisensor chip with sequential injection technique as a base for “electronic tongue” devices. Sens. Actuat. B 2008, 131, 48–52. [Google Scholar]
- Alonso, J.; Baró, J.; Bartrolí, J.; Sánchez, J.; del Valle, M. Flow-through tubular ion-selective electrodes responsive to anionic surfactants for flow-injection analysis. Anal. Chim. Acta 1995, 308, 115–121. [Google Scholar]
- Durán, A.; Cortina, M.; Velasco, L.; Rodríguez, J.A.; Alegret, S.; del Valle, M. Virtual instrument for an automated potentiometric e-tongue employing the SIA technique. Sensors 2006, 6, 19–29. [Google Scholar]
- Gallardo, J.; Alegret, S.; Munoz, R.; Leija, L.; Hernández, P. Ro.; Del Valle, M. Use of an electronic tongue based on all-solid-state potentiometric sensors for the quantitation of alkaline ions. Electroanalysis 2005, 17, 348–355. [Google Scholar]
- Gutes, A.; Ibanez, A.B.; Céspedes, F.; Alegret, S.; Valle, M. Simultaneous determination of phenolic compounds by means of an automated voltammetric “electronic tongue”. Anal. Bioanal. Chem 2005, 382, 471–476. [Google Scholar]
- Gutes, A.; Céspedes, F.; Alegret, S.; del Valle, M. Sequential injection system with higher dimensional electrochemical sensor signals Part 1. Voltammetric e-tongue for the determination of oxidizable compounds. Talanta 2005, 66, 1187–1196. [Google Scholar]
- Cortina, M.; Gutes, A.; Alegret, S.; del Valle, M. Sequential injection system with higher dimensional electrochemical sensor signals. Talanta 2005, 66, 1197–1206. [Google Scholar]
- Cortina, M.; Duran, A.; Alegret, S.; del Valle, M. A sequential injection electronic tongue employing the transient response from potentiometric sensors for anion multidetermination. Anal. Bioanal. Chem 2006, 385, 1186–1194. [Google Scholar]
- Gallardo, J.; Alegret, S.; del Valle, M. Application of a potentiometric electronic tongue as a classification tool in food análisis. Talanta 2005, 66, 1303–1309. [Google Scholar]
- Moreno-Barón, L.; Cartas, R.; Merkoci, A.; Alegret, S.; Gutiérrez, J.M.; Leija, L.; Hernández, P.R.; Munóz, R.; del Valle, M. Data Compression for a Voltammetric Electronic Tongue Modelled with Artificial Neural Networks. Anal. Lett 2005, 38, 2189–2206. [Google Scholar]
- Moreno-Barón, L.; Cartas, R.; Merkoci, A.; Alegret, S.; del Valle, M.; Leija, L.; Hernández, P.R.; Munóz, R. Application of the wavelet transform coupled with artificial neural networks for quantification purposes in a voltammetric electronic tongue. Sens. Actuat. B 2006, 113, 487–499. [Google Scholar]
- Gutiérrez, M.; Alegret, S.; Cáceres, R.; Casadesus, J.; Marfa, O.; del Valle, M. Nutrient Solution Monitoring in Greenhouse Cultivation Employing a Potentiometric Electronic Tongue. J. Agric. Food Chem 2008, 56, 1810–1817. [Google Scholar]
- Gutiérrez, M.; Alegret, S.; del Valle, M. Bioelectronic tongue for the simultaneous determination of urea, creatinine and alkaline ions in clinical samples. Biosens. Bioelectron 2008, 23, 795–802. [Google Scholar]
- Gutiérrez, M.; Moo, V.M.; Alegret, S.; Leija, L.; Hernández, P.R.; Munóz, R.; del Valle, M. Electronic tongue for the determination of alkaline ions using a screen-printed potentiometric sensor array. Microchip. Acta 2008, 163, 81–88. [Google Scholar]
- Gutiérrez, M.; Gutiérrez, J.M.; Alegret, S.; Leija, L.; Hernández, P.R.; Favari, L.; Munóz, R.; del Valle, M. Remote environmental monitoring employing a potentiometric electronic tongue. Int. J. Environ. Anal. Chem 2008, 88, 103–117. [Google Scholar]
- Mimendia, A.; Legin, A.; Merkoci, A.; del Valle, M. Use of Sequential Injection Analysis to construct a Potentiometric Electronic Tongue: Application to the Multidetermination of Heavy Metals. AIP Conf. Proc 2009, 1137, 239–242. [Google Scholar]
- Lemos, S.G.; Nogueira, A.R.A.; Torre-Neto, A.; Parra, A.; Artigas, J.; Alonso, J. In-soil potassium sensor system. J. Agric. Food Chem 2004, 52, 5810–5815. [Google Scholar]
- Lemos, S.G.; Nogueira, A.R.A.; Torre-Neto, A.; Parra, A.; Alonso, J. Soil calcium and pH monitoring sensor system. J. Agric. Food Chem 2007, 55, 4658–4663. [Google Scholar]
- Ortuno, J.A.; Serna, C.; Molina, A.; Gil, A. Differential pulse voltammetry and additive differential pulse voltammetry with solvent polymeric membrane ion sensors. Anal. Chem 2006, 78, 8129–8133. [Google Scholar]
- Molina, A.; Serna, C.; Ortuno, J.A.; González, J.; Torralba, E.; Gil, A. Differential Pulse Voltammetry for Ion Transfer at Liquid Membranes with Two Polarized Interfaces. Anal. Chem 2009, 81, 4220–4225. [Google Scholar]
- Vidal, J.C.; Esteban, S.; Gil, J.; Castillo, J.R. A comparative study of immobilization methods of a tyrosinase enzyme on electrodes and their application to the detection of dichlorvos organophosphorus insecticide. Talanta 2006, 68, 791–799. [Google Scholar]
- Manso, J.; Agui, L.; Yanez-Sedeno, P.; Pingarrón, J.M. Development and characterization of colloidal gold- cysteamine-carbon paste electrodes. Anal. Lett 2004, 37, 887–902. [Google Scholar]
- Sanz, V.C.; Mena, M.L.; González-Cortes, A.; Yanez-Sedeno, P.; Pingarrón, J.M. Development of a tyrosinase biosensor based on gold nanoparticles-modified glassy carbon electrodes—Application to the measurement of a bioelectrochemical polyphenols index in wines. Anal. Chim. Acta 2005, 528, 1–8. [Google Scholar]
- Serra, B.; Morales, M.D.; Zhang, J.B.; Reviejo, A.J.; Hall, E.H.; Pingarrón, J.M. In-a-Day Electrochemical Detection of Coliforms in Drinking Water Using a Tyrosinase Composite Biosensor. Anal. Chem 2005, 77, 8115–8121. [Google Scholar]
- Carralero, V.; Mena, M.L.; González-Cortes, A.; Yanez-Sedeno, P.; Pingarrón, J.M. Development of a high analytical performance-tyrosinase biosensor based on a composite graphite—Teflon electrode modified with gold nanoparticles. Biosens. Bioelectron 2006, 22, 730–736. [Google Scholar]
- Morales, M.D.; Serra, B.; Guzmán-Vázquez de Prada, A.; Reviejo, A.J.; Pingarrón, J.M. An electrochemical method for simultaneous detection and identification of Escherichia coli, Staphylococcus aureus and Salmonella choleraesuis using a glucose oxidase-peroxidase composite biosensor. Analyst 2007, 132, 572–578. [Google Scholar]
- Serra, B.; Zhang, J.; Morales, M.D.; Guzmán-Vázquez de Prada, A.; Reviejo, A.J.; Pingarrón, J.M. A rapid method for detection of catalase-positive and catalase-negative bacteria based on monitoring of hydrogen peroxide evolution at a composite peroxidase biosensor. Talanta 2008, 75, 1134–1139. [Google Scholar]
- Herrero, A.M.; Requena, T.; Reviejo, A.J.; Pingarrón, J.M. Determination of L-lactic acid in yoghurt by a bienzyme amperometric graphite-Teflon composite biosensor. Eur. Food Res. Technol 2004, 219, 556–559. [Google Scholar]
- Cano, M.; Ávila, J.L.; Mayen, M.; Mena, M.L.; Pingarrón, J.M.; Rodríguez-Amaro, R. A new, third generation, PVC/TTF-TCNQ composite amperometric biosensor for glucose determination. J. Electroanal. Chem 2008, 615, 69–74. [Google Scholar]
- Revenga-Parra, M.; Lorenzo, E.; Pariente, F. Synthesis and electrocatalytic activity towards oxidation of hydrazine of a new family of hydroquinone salophen derivatives: application to the construction of hydrazine sensors. Sens. Actuat. B 2005, 107, 678–687. [Google Scholar]
- Revenga-Parra, M.; García, T.; Lorenzo, E.; Pariente, F. Electrocatalytic oxidation of methanol and other short chain aliphatic alcohols on glassy carbon electrodes modified with conductive films derived from Ni-II-(N,N’-bis(2,5-dihydroxybenzylidene)-1,2-diaminobenzene). Sens. Actuat. B 2008, 130, 730–738. [Google Scholar]
- García, T.; Casero, E.; Lorenzo, E.; Pariente, F. Electrochemical sensor for sulfite determination based on iron hexacyanoferrate film modified electrodes. Sens. Actuator B 2005, 106, 803–809. [Google Scholar]
- Domínguez-Renedo, O.; Alonso-Lomillo, M.A.; Arcos-Martínez, M.J. Optimisation procedure for the inhibitive determination of chromium(III) using an amperometric tyrosinase biosensor. Anal. Chim. Acta 2004, 521, 215–221. [Google Scholar]
- Rodríguez-Gutiérrez, J.A.; Petit-Domínguez, M.D.; Pinilla-Macias, J.M. Development of ionoselective electrochemical sensors by using the sol-gel process. Anal. Chim. Acta 2004, 524, 339–346. [Google Scholar]
- Cabello-Carramolino, G.; Petit-Domínguez, M.D. Application of new sol-gel electrochemical sensors to the determination of trace mercury. Anal. Chim. Acta 2008, 614, 103–111. [Google Scholar]
- Cabello-Carramolino, G.; Petit-Domínguez, M.D. Development of new sol-gel carbon composite electrodes and their application as electrochemical sensors. Microchim. Acta 2009, 164, 405–410. [Google Scholar]
- Díaz-González, M.; Hernández-Santos, D.; González-García, M.B.; Costa-García, A. Development of an immunosensor for the determination of rabbit IgG using streptavidin modified screen-printed carbon electrodes. Talanta 2005, 65, 565–573. [Google Scholar]
- Kreuzer, M.P.; O’Sullivan, C.K.; Pravda, M.; Guilbault, G.G. Development of an immunosensor for the determination of allergy antibody (IgE) in blood samples. Anal. Chim. Acta 2001, 442, 45–53. [Google Scholar]
- Wang, J.; Pamidi, P.V.A.; Rogers, K.R. Sol-gel-derived thick-film amperometric immunosensors. Anal. Chem 1998, 70, 1171–1175. [Google Scholar]
- Fernández-Romero, J.M.; Stiene, M.; Kast, R.; Luque de Castro, M.D.; Bilitewski, U. Application of screen-printed electrodes as transducers in affinity flow-through sensor systems. Biosens. Bioelectron 1998, 13, 1107–1115. [Google Scholar]
- Valat, C.; Limoges, B.; Huet, D.; Romette, J.L. A disposable Protein A-based immunosensor for flow-injection assay with electrochemical detection. Anal. Chim. Acta 2000, 404, 187–194. [Google Scholar]
- Hernández-Santos, D.; Díaz-González, M.; González-García, M.B.; Costa-García, A. Enzymatic genosensor on streptavidin-modified screen-printed carbon electrodes. Anal. Chem 2004, 76, 6887–6893. [Google Scholar]
- Fanjul-Bolado, P.; González-García, M.B.; Costa-García, A. 3-indoxyl phosphate as an electrochemical substrate for horseradish peroxidase. Electroanalysis 2004, 16, 988–993. [Google Scholar]
- Hernández-Santos, D.; González-García, M.B.; Costa-García, A. Genosensor based on a Platinum(II) complex as electrocatalytic label. Anal. Chem 2005, 77, 2868–2874. [Google Scholar]
- Fanjul-Bolado, P.; González-García, M.B.; Costa-García, A. Amperometric detection in TMB/HRP-based assays. Anal. Bioanal. Chem 2005, 382, 297–302. [Google Scholar]
- Fanjul-Bolado, P.; González-García, M.B.; Costa-García, A. Flow screen-printed amperometric detection of p-nitrophenol in alkaline phosphatasebased assays. Anal. Bioanal. Chem 2006, 385, 1202–1208. [Google Scholar]
- De la Escosura-Muníz, A.; González-García, M.B.; Costa-García, A. DNA hybridization sensor based on aurothiomalate electroactive label on glassy carbon electrodes. Biosens. Bioelectron 2007, 22, 1048–1054. [Google Scholar]
- Abad-Valle, P.; Fernández-Abedul, M.T.; Costa-García, A. Genosensor on gold films with enzymatic electrochemical detection of SARS virus sequence. Biosens. Bioelectron 2005, 20, 2251–2260. [Google Scholar]
- Fanjul-Bolado, P.; Hernández-Santos, D.; González-García, M.B.; Costa-García, A. Alkaline Phosphatase-Catalyzed Silver Deposition for Electrochemical Detection. Anal. Chem 2007, 79, 5272–5277. [Google Scholar]
- García-González, R.; Fernández-Abedul, M.T.; Pernia, A.; Costa-García, A. Electrochemical characterization of different screen-printed gold electrodes. Electrochim. Acta 2008, 53, 3242–3249. [Google Scholar]
- Lamas-Ardisana, P.J.; Queipo, P.; Fanjul-Bolado, P.; Costa-García, A. Multiwalled carbon nanotube modified screen-printed electrodes for the detection of paminophenol: Optimisation and application in alkaline phosphatase-based assays. Anal. Chim. Acta 2008, 615, 30–38. [Google Scholar]
- Martínez-Paredes, G.; González-García, M.B.; Costa-García, A. In situ electrochemical generation of gold nanostructured screen-printed carbon electrodes. Application to the detection of lead underpotential deposition. Electrochim. Acta 2009, 54, 4801–4808. [Google Scholar]
- Martínez-Paredes, G.; González-García, M.B.; Costa-García, A. Genosensor for SARS virus detection based on gold nanostructured screen-printed carbon electrodes. Electroanalysis 2009, 21, 379–385. [Google Scholar]
- Escamilla-Gómez, V.; Hernández-Santos, D.; González-García, M.B.; Pingarrón-Carrazón, J.M.; Costa-García, A. Simultaneous detection of free and total prostate specific antigen on a screen-printed electrochemical dual sensor. Biosens. Bioelectron 2009, 24, 2678–2683. [Google Scholar]
- Martínez-Paredes, G; González-García, M.B.; Costa-García, A. Lead sensor using gold nanostructured screen-printed carbon electrodes as transducers. Electroanalysis 2009, 21, 925–930. [Google Scholar]
- Gamella, M.; Campuzano, S.; Parrado, C.; Reviejo, A.J.; Pingarrón, J.M. Microorganisms recognition and quantification by lectin adsorptive affinity impedance. Talanta 2009, 78, 1303–1309. [Google Scholar]
- Domínguez-Renedo, O.; Calvo, M.E.B.; Arcos-Martínez, M.J. Determination of lamotrigine in pharmaceutical preparations by adsorptive stripping voltammetry using screen printed electrodes. Sensors 2008, 8, 4201–4212. [Google Scholar]
- Domínguez-Renedo, O.; González, M.J.G.; Arcos-Martínez, M.J. Determination of antimony (III) in real samples by anodic stripping voltammetry using a mercury film screen-printed electrode. Sensors 2009, 9, 219–231. [Google Scholar]
- Alonso-Lomillo, M.A.; Domínguez-Renedo, O.; Matos, P.; Arcos-Martínez, M.J. Electrochemical determination of levetiracetam by screen-printed based biosensors. Bioelectrochemistry 2009, 74, 306–309. [Google Scholar]
- Alonso-Lomillo, M.A.; Domínguez-Renedo, O.; Hernández-Martín, A.; Arcos-Martínez, M.J. Horseradish peroxidase covalent grafting onto screen-printed carbon electrodes for levetiracetam chronoamperometric determination. Anal. Biochem 2009, 395, 86–90. [Google Scholar]
- Alonso-Lomillo, M.A.; Gonzalo-Ruíz, J.; Domínguez-Renedo, O.; Munóz, F.J.; Arcos-Martínez, M.J. CYP450 biosensors based on gold chips for antiepileptic drugs determination. Biosens. Bioelectron 2008, 23, 1733–1737. [Google Scholar]
- Rico, M.A.G.; Olivares-Marín, M.; Gil, E.P. A novel cell design for the improved stripping voltammetric detection of Zn (II), Cd (II), and Pb (II) on commercial screen-printed strips by bismuth codeposition in stirred solutions. Electroanalysis 2008, 20, 2608–2613. [Google Scholar]
- Domínguez, E.; Rincón, O.; Narváez, A. Electrochemical DNA sensors based on enzyme dendritic architectures: an approach for enhanced sensitivity. Anal. Chem 2004, 76, 3132–3138. [Google Scholar]
- Del Pozo, M.V.; Alonso, C.; Pariente, F.; Lorenzo, E. Electrochemical DNA sensing using osmium complexes as hybridization indicators. Biosens. Bioelectron 2005, 20, 1549–1558. [Google Scholar]
- Del Pozo, M.V.; Alonso, C.; Pariente, F.; Lorenzo, E. DNA Biosensor for Detection of Helicobacter pylori Using Phen-dione as the Electrochemically Active Ligand in Osmium Complexes. Anal. Chem 2005, 77, 2550–2557. [Google Scholar]
- Revenga-Parra, M.; García, T.; Lorenzo, E.; Pariente, F. Comprehensive study of interactions between DNA and new electroactive Schiff base ligands. Biosens. Bioelectron 2007, 22, 2675–2681. [Google Scholar]
- Parra, A.; Casero, E.; Vázquez, L.; Jin, J.; Pariente, F.; Lorenzo, E. Microscopic and Voltammetric Characterization of Bioanalytical Platforms Based on Lactate Oxidase. Langmuir 2006, 22, 5443–5450. [Google Scholar]
- Parra, A.; Casero, E.; Vázquez, L.; Pariente, F.; Lorenzo, E. Design and characterization of a lactate biosensor based on immobilized lactate oxidase onto gold surfaces. Anal. Chim. Acta 2006, 555, 308–315. [Google Scholar]
- Parra, A; Casero, E; Pariente, F; Vázquez, L; Lorenzo, E. Bioanalytical device based on cholesterol oxidase-bonded SAM-modified electrodes. Anal. Bioanal. Chem 2007, 388, 1059–1067. [Google Scholar]
- Parra, A.; Casero, E.; Pariente, F.; Vázquez, L.; Lorenzo, E. Cholesterol oxidase modified gold electrodes as bioanalytical devices. Sens. Actuat. B 2007, 124, 30–37. [Google Scholar]
- Miranda-Castro, R.; de-los-Santos-Álvarez, N.; Lobo-Castanón, M.J.; Miranda-Ordieres, A.J.; Tunón-Blanco, P. PCR-coupled electrochemical sensing of Legionella pneumophila. Biosens. Bioelectron 2009, 24, 2390–2396. [Google Scholar]
- Alonso-Lomillo, M.A.; Domínguez-Renedo, O.; Arcos-Martínez, M.J. Optimization of a cyclodextrin-based sensor for rifampicin monitoring. Electrochim. Acta 2005, 50, 1807–1811. [Google Scholar]
- Vidal, J.C.; Espuelas, J.; García-Ruíz, E.; Castillo, J.R. Amperometric cholesterol biosensors based on the electropolymerization of pyrrole and the electrocatalytic effect of Prussian-Blue layers helped with self-assembled monolayers. Talanta 2004, 64, 655–664. [Google Scholar]
- Vidal, J.C.; Espuelas, J.; Castillo, J.R. Amperometric cholesterol biosensor based on in situ reconstituted cholesterol oxidase on an immobilized monolayer of flavin adenine dinucleotide cofactor. Anal. Biochem 2004, 333, 88–98. [Google Scholar]
- Miranda-Castro, R.; De-los-Santos-Álvarez, P.; Lobo-Castanón, M.J.; Miranda-Ordieres, A.J.; Tunón-Blanco, P. Hairpin-DNA Probe for Enzyme-Amplified Electrochemical Detection of Legionella pneumophila. Anal. Chem 2007, 79, 4050–4055. [Google Scholar]
- Miranda-Castro, R.; Lobo-Castanon, M.J.; Miranda-Ordieres, A.J.; Tunon-Blanco, P. Stem-loop DNA probes for the voltammetric determination of Legionella pneumophila on disposable screen-printed gold electrodes. Electroanalysis 2009, 21, 267–273. [Google Scholar]
- Miranda-Castro, R.; de-los-Santos-Álvarez, N.; Lobo-Castanón, M.J.; Miranda-Ordieres, A.J.; Tunón-Blanco, P. Structured Nucleic Acid Probes for Electrochemical Devices. Electroanalysis 2009, 21, 2077–2090. [Google Scholar]
- Maestre, E.; Katakis, I.; Narváez, A.; Domínguez, E. A multianalyte flow electrochemical cell: application to the simultaneous determination of carbohydrates based on bioelectrocatalytic detection. Biosens. Bioelectron 2005, 21, 774–781. [Google Scholar]
- Ortuno, J.A.; Hernández, J.; Sánchez-Pedreno, C. Flow-injection amperometric detection with solvent polymeric membrane ion sensors. Electroanalysis 2004, 16, 827–831. [Google Scholar]
- Ortuno, J.A.; Rueda, C. Flow-injection amperometric determination of tacrine based on ion transfer across a water plasticized polymeric membrane interface. Sensors 2007, 7, 1185–1192. [Google Scholar]
- Ortuno, J.A.; Rodenas, V.; García, M.S.; Albero, M.I.; Sánchez-Pedreno, C. A new tiapride selective electrode and its clinical application. Sensors 2007, 7, 400–409. [Google Scholar]
- García, M.S.; Ortuno, J.A.; Albero, M.I.; Abuherba, M.S. Development of membrane selective electrode for determination of the antipsychotic sulpiride in pharmaceuticals and urine. Sensors 2009, 9, 4309–4322. [Google Scholar]
- Ortuno, J.A.; Gil, A.; Serna, C.; Molina, A. Voltammetry of some catamphiphilic drugs with solvent polymeric membrane ion sensors. J. Electroanal. Chem 2007, 605, 157–161. [Google Scholar]
- Ortuno, J.A.; Serna, C.; Molina, A.; Torralba, E. Ion Transfer Square Wave Voltammetry of Ionic Liquid Cations with a Solvent Polymeric Membrane Ion Sensor. Electroanalysis 2009, 21, 2297–2302. [Google Scholar]
- Ballesta-Claver, J.; Valencia-Mirón, M.C.; Capitán-Vallvey, L.F. Disposable electro-chemiluminescent biosensor for lactate determination in saliva. Analyst 2009, 134, 1423–1432. [Google Scholar]
- Ballesta-Claver, J.; Valencia-Mirón, M.C.; Capitán-Vallvey, L.F. One-shot lactate chemiluminescent biosensor. Anal. Chim. Acta 2008, 629, 136–144. [Google Scholar]
- Martínez-Olmos, A.; Ballesta-Claver, J.; Palma, A.J.; Valencia-Mirón, M.C.; Capitán-Vallvey, L.F. A portable luminometer with a disposable electrochemiluminescent biosensor for lactate determination. Sensors 2009, 9, 7694–7701. [Google Scholar]
- Capel-Cuevas, S.; de Orbe-Payá, I.; Santoyo-González, F.; Capitán-Vallvey, L.F. Double-armed crown ethers for calcium optical sensors. Talanta 2009, 78, 1484–1488. [Google Scholar]
- Capitán-Vallvey, L.F.; Fernández-Ramos, M.D.; Al-Natsheh, M. A disposable single use optical sensor for potassium determination based on neutral ionophore. Sens. Actuat. B 2003, 88, 217–222. [Google Scholar]
- Lapresta-Fernández, A.; Capitán-Vallvey, L.F. Scanometric potassium determination with ionophore-based disposable sensors. Sens. Actuat. B 2008, 134, 694–701. [Google Scholar]
- de Orbe-Paya, I.; Erenas, M.M.; Capitán-Vallvey, L.F. Potassium disposable optical sensor based on transflectance and cromaticity measuremets. Sens. Actuat. B 2007, 127, 586–592. [Google Scholar]
- Palma, A.; Lapresta-Fernández, A.; Ortigosa-Moreno, J.M.; Fernández-Ramos, M.D.; Carvajal, M.A.; Capitán-Vallvey, L.F. A simplified measurement procedure and portable electronic photometer for disposable sensors based on ionophore-chromoionophore chemistry for potassium determination. Anal. Bioanal. Chem 2006, 386, 1215–1224. [Google Scholar]
- Lapresta-Fernández, A.; Huertas, R.; Melgosa, M.; Capitán-Vallvey, L.F. Colourimetric characterisation of disposable optical sensors from spectroradiometric measurements. Anal. Bioanal. Chem 2009, 393, 1361–1366. [Google Scholar]
- Lapresta-Fernández, A.; Huertas, R.; Melgosa, M.; Capitán-Vallvey, L.F. Multianalyte imaging in one-shot format sensors for natural waters. Anal. Chim. Acta 2009, 636, 210–217. [Google Scholar]
- Palma, A.J.; López-González, J.; Asensio, L.J.; Fernández-Ramos, M.D.; Capitán-Vallvey, L.F. Microcontroller-based portable instrument for stabilised chemistry. Sens. Actuat. B 2007, 121, 629–638. [Google Scholar]
- Capitán-Vallvey, L.F.; Asensio, L.J.; López-González, J.; Fernández-Ramos, M.D.; Palma, A.J. Oxygen-sensing film coated photodetectors for portable instrumentation. Anal. Chim. Acta 2007, 583, 166–173. [Google Scholar]
- Palma, A.J.; López-González, J.; Asensio, L.J.; Fernández-Ramos, M.D.; Capitán-Vallvey, L.F. Open air calibration with temperature compensation of a luminescence quenching-based oxygen sensor for portable instrumentation. Anal. Chem 2007, 79, 3173–3179. [Google Scholar]
- Capitán-Vallvey, L.F.; Raya, C.C.; López, E.L.; Ramos, M.D.F. Irreversible optical test strip for mercury determination based on neutral ionophore. Anal. Chim. Acta 2004, 524, 365–372. [Google Scholar]
- Cano-Raya, C.; Fernández-Ramos, M.D.; Gómez-Sánchez, J.; Capitán-Vallvey, L.F. Irreversible optical sensor for mercury determination based on tetraarylborate decomposition. Sens. Actuat. B 2006, 117, 135–142. [Google Scholar]
- Fernández-Ramos, M.D.; Greluk, M.; Palma, A.J.; Arroyo-Guerrero, E.; Gómez-Sánchez, J.; Capitán-Vallvey, L.F. The use of one-shot sensors with a dedicated portable electronic radiometer for nitrate measurements in aqueous solutions. Meas. Sci. Technol 2008, 19(095204). [Google Scholar]
- Sanz, V.; de Marcos, S.; Galbán, J. A reagentless optical biosensor based on the intrinsic absorption properties of peroxidase. Biosens. Bioelectron 2007, 22, 956–964. [Google Scholar]
- Sanz, V.; de Marcos, S.; Galbán, J. Direct glucose determination in blood using a reagentless optical biosensor. Biosens. Bioelectron 2007, 22, 2876–2883. [Google Scholar]
- Sanz, V.; de Marcos, S.; Galbán, J. A blood-assisted optical biosensor for automatic glucose determination. Talanta 2009, 78, 846–851. [Google Scholar]
- Sanz, V.; de Marcos, S.; Galbán, J. Hydrogen peroxide and peracetic acid determination in waste water using a reversible reagentless biosensor. Anal. Chim. Acta 2007, 583, 332–339. [Google Scholar]
- Cadarso, V.J.; Fernández-Sánchez, C.; Llobera, A.; Darder, M.; Domínguez, C. Optical Biosensor Based On Hollow Integrated Waveguides. Anal. Chem 2008, 80, 3498–3501. [Google Scholar]
- Wolfbeis, O.S. Fiber-optic chemical sensors and biosensors. Anal. Chem 2006, 78, 3859–3873. [Google Scholar]
- Wolfbeis, O.S. Fiber-Optic Chemical Sensors and Biosensors. Anal. Chem 2008, 80, 4269–4283. [Google Scholar]
- Lemos, S.G.; Menezes, E.A.; Chaves, F.S.; Nogueira, A.R.A.; Torre-Neto, A.; Parra, A.; Alonso, J. In Situ Soil Phosphorus Monitoring Probe Compared with Conventional Extraction Procedures. Commun. Soil Sci. Plant Anal 2009, 40, 1282–1294. [Google Scholar]
- De Marcos, S.; Wolfbeis, O.S. Characterization of polypirrole fimls for use in optical sensing. Sens. Mater. 1997, 9, 253–285. [Google Scholar]
- de Marcos, S.; Wolfbeis, O.S. Optical Sensing of pH Based on Polypyrrole Films. Anal. Chim. Acta 1996, 334, 149–153. [Google Scholar]
- de Marcos, S.; Alcubierre, N.; Galbán, J.; Castillo, J.R. Reagents system for sulphite determination based on polyaniline. Anal. Chim. Acta 2004, 502, 7–13. [Google Scholar]
- Andreu, Y.; de Marcos, S.; Castillo, J.R.; Galbán, J. Sensor film for Vitamin C determination based on absorption properties of polyaniline. Talanta 2005, 65, 1045–1051. [Google Scholar]
- Puyol, M.; Villuendas, F.; Domínguez, C.; Cadarso, V.; Llobera, A.; Salinas, I.; Garcés, I.; Alonso, J.; Orellana, G.; Moreno-Bondi, M.C. Frontiers in Chemical Sensors: Novel Principles and Techniques; Springer-Verlag: Heidelberg, Germany, 2005; Volume 3, pp. 1–44. [Google Scholar]
- Rivera, L.; Puyol, M.; Villuendas, F.; Alonso, J. Miniaturized setup for fluorescence sensing with optodes: Characterization of a new hemicyanine ion-selective-based membrane. Sens. Actuat. B 2008, 134, 863–868. [Google Scholar]
- Izquierdo, D.; Puyol, M.; Salinas, I.; Alonso, R.; Alonso, J.; Domínguez, C.; Garcés, I. Dual-wavelength measurement system for absorbance chemical sensing. Meas. Sci. Tech 2007, 18, 3443–3450. [Google Scholar]
- Miltsov, S.; Encinas, C.; Alonso, J. Nortricarbocyanines: New Near-Infrared pH-Indicators. Tetrahedron Lett 1998, 39, 9253–9254. [Google Scholar]
- Miltsov, S.; Encinas, C.; Alonso, J. New cyanine dyes: norindosquarocyanines. Tetrahedron Lett 1999, 40, 4067–4068. [Google Scholar]
- Miltsov, S.; Encinas, C.; Alonso, J. Novel synthesis of ketocyanine dyes. Tetrahedron Lett 2001, 42, 6129–6131. [Google Scholar]
- Encinas, C.; Miltsov, S.; Otazo, E.; Alonso, J. Croconines: new acidochromic dyes for the near infrared region. Tetrahedron Lett 2002, 43, 8391–8393. [Google Scholar]
- Encinas, C.; Miltsov, S.; Otazo, E.; Rivera, L.; Puyol, M.; Alonso, J. Synthesis and spectroscopic characterization of heptamethine cyanine NIR dyes for their use in optochemical sensors. Dyes Pigm 2005, 71, 28–36. [Google Scholar]
- Rivera, L.; Puyol, M.; Miltsov, S.; Villuendas, F.; Alonso, J. New ketocyanine-based fluorescent optodes for the development of integrated waveguide. Sens. Actuat. B 2006, 114, 705–712. [Google Scholar]
- Rivera, L.; Puyol, M.; Miltsov, S.; Alonso, J. New hexamethine-hemicyanine dyes for the development of integrated optochemical. Anal. Bioanal. Chem 2007, 387, 2111–2119. [Google Scholar]
- Puyol, M.; Encinas, C.; Rivera, L.; Miltsov, S.; Alonso, J. Characterisation of new norcyanine dyes and their application as pH chromoionophores in optical sensors. Dyes Pigm 2006, 73, 383–389. [Google Scholar]
- Puyol, M.; Encinas, C.; Rivera, L.; Miltsov, S.; Alonso, J. Synthesis of new ketocyanine dyes for the development of optical sensors. Sens. Actuat. B 2006, 115, 287–296. [Google Scholar]
- Martín, F.J.F.; Rodríguez, J.C.C.; Antón, J.C.A.; Pérez, J.C.V.; Sánchez-Barragán, I.; Costa-Fernández, J.M.; Sanz-Medel, A. Design of a low-cost optical instrument for pH fluorescence measurements. IEEE Trans. Instrum. Meas 2006, 55, 1215–1221. [Google Scholar]
- Sánchez-Barragán, I.; Costa-Fernández, J.M.; Sanz-Medel, A. Tailoring the pH response range of fluorescent-based pH sensing phases by sol-gel surfactants co-immobilization. Sens. Actuat. B 2005, 107, 69–76. [Google Scholar]
- Urraca, J.L.; Benito-Pena, E.; Pérez-Conde, C.; Moreno-Bondi, M.C.; Pestka, J.J. Analysis of Zearalenone in Cereal and Swine Feed Samples Using an Automated Flow-Through Immunosensor. J. Agric. Food Chem 2005, 53, 3338–3344. [Google Scholar]
- Traviesa-Álvarez, J.M.; Costa-Fernández, J.M.; Pereiro, R.; Sanz-Medel, A. Flow-through solid-phase energy transfer-room temperature phosphorescence for orthophosphate determinations at trace levels. Talanta 2004, 62, 827–833. [Google Scholar]
- Rojas-Durán, T.; Sánchez-Barragán, I.; Costa-Fernández, J.M.; Sanz-Medel, A. Direct and rapid discrimination of aflatoxigenic strains based on fibre-optic room temperature phosphorescence detection. Analyst 2007, 132, 307–313. [Google Scholar]
- Guardia, L.; Badia, R.; Díaz-García, M.E. Molecularly Imprinted Sol-Gels for Nafcillin Determination in Milk-Based Products. J. Agric. Food Chem 2007, 55, 566–570. [Google Scholar]
- García, M.S.; Ortuno, J.A.; Sánchez-Pedreno, C.; Albero, M.I.; Fernández, M.J. Flow-through bulk optode for spectrophotometric determination of thiocyanate and its application to water and saliva analysis. Sensors 2006, 6, 1224–1233. [Google Scholar]
- Fernández-Sánchez, J.F.; Segura-Carretero, A.; Cruces-Blanco, C.; Fernández-Gutiérrez, A. Highly sensitive and selective fluorescence optosensor to detect and quantify benzo[a]pyrene in water samples. Anal. Chim. Acta 2004, 506, 1–7. [Google Scholar]
- Salinas-Castillo, A.; Fernández-Sánchez, J.F.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Solid-surface phosphorescence characterization of polycyclic aromatic hydrocarbons and selective determination of benzo(a)pyrene in water samples. Anal. Chim. Acta 2005, 550, 53–60. [Google Scholar]
- Fernández-Sánchez, J.F.; Segura-Carretero, A.; Benitez-Sánchez, J.M.; Cruces-Blanco, C.; Fernández-Gutiérrez, A. Fluorescence optosensor using an artificial neural network for screening of polycyclic aromatic hydrocarbons. Anal. Chim. Acta 2004, 510, 183–187. [Google Scholar]
- Casado-Terrones, S.; Fernández-Sánchez, J.F.; Cañabate-Díaz, B.; Segura-Carretero, A.; Fernández-Gutiérrez, A. A fluorescence optosensor for analyzing naphazoline in pharmaceutical preparations. J. Pharm. Biomed. Anal 2005, 38, 785–789. [Google Scholar]
- Salinas-Castillo, A.; Fernández-Sánchez, J.F.; Segura-Carretero, A.; Fernández-Gutiérrez, A. A facile flow-through phosphorimetric sensing device for simultaneous determination of naptalam and its metabolite 1-naphthylamine. Anal. Chim. Acta 2004, 522, 19–24. [Google Scholar]
- Casado-Terrones, S.; Fernández-Sánchez, J.F.; Segura-Carretero, A.; Fernández-Gutiérrez, A. The development and comparison of a fluorescence and a phosphorescence optosensors for determining the plant growth regulator 2-naphthoxyacetic acid. Sens. Actuat. B 2005, 107, 929–935. [Google Scholar]
- Garcés-García, M.; Morais, S.; González-Martínez, M.A.; Puchades, R.; Maquieira, A. Rapid immunoanalytical method for the determination of atrazine residues in olive oil. Anal. Bioanal. Chem 2004, 378, 484–489. [Google Scholar]
- González-Martínez, M.A.; Brun, E.M.; Puchades, R.; Maquieira, A.; Ramsey, K.; Rubio, F. Glyphosate Immunosensor. Application for Water and Soil Analysis. Anal. Chem 2005, 77, 4219–4227. [Google Scholar]
- González-Martínez, M.A.; Penalva, J.; Rodríguez-Urbis, J.C.; Brunet, E.; Maquieira, A.; Puchades, R. Immunosensors for pollutants working in organic media. Study of performances of different tracers with luminescent detection. Anal. Bioanal. Chem 2006, 384, 1540–1547. [Google Scholar]
- Jornet, D.; González-Martínez, M.A.; Maquieira, A.; Puchades, R. Advanced homogeneous-heterogeneous immunosensing format employing restricted access supports. Anal. Chem 2007, 79, 9331–9339. [Google Scholar]
- Banuls, M.J.; González-Pedro, V.; Puchades, R.; Maquieira, A. PMMA Isocyanate-Modified Digital Discs as a Support for Oligonucleotide-Based Assays. Bioconjugate Chem 2007, 18, 1408–1414. [Google Scholar]
- Morais, S.; Tamarit-López, J.; Carrascosa, J.; Puchades, R.; Maquieira, A. Analytical prospect of compact disk technology in immunosensing. Anal. Bioanal. Chem 2008, 391, 2837–2844. [Google Scholar]
- Ruedas-Rama, M.J.; Ruíz-Medina, A.; Molina-Díaz, A. Implementation of flow-through multi-sensors with bead injection spectroscopy: fluorimetric renewable surface biparameter sensor for determination of beryllium and aluminum. Talanta 2004, 62, 879–886. [Google Scholar]
- Reguera, I.P.; Rubio, M.G.; Díaz, A.M. Native fluorescence flow-through optosensor for the fast determination of diphenhydramine in pharmaceuticals. Anal. Sci 2004, 20, 799–803. [Google Scholar]
- Pascual-Reguera, M.I.; Perez-Parras, G.; Molina-Díaz, A. A single spectroscopic flow-through sensing device for determination of ciprofloxacin. J. Pharm. Biomed. Anal 2004, 35, 689–695. [Google Scholar]
- Ortega-Algar, S.; Ramos-Martos, N.; Molina-Díaz, A. Fluorimetric Flow-Through Sensing of Quinine and Quinidine. Microchim. Acta 2004, 147, 211–217. [Google Scholar]
- Ruedas-Rama, M.J.; Ruiz-Medina, A.; Molina-Díaz, A. Bead injection spectroscopy-flow injection analysis (BIS-FIA): an interesting tool applicable to pharmaceutical analysis. Determination of promethazine and trifluoperazine. J. Pharm. Biomed. Anal 2004, 35, 1027–1034. [Google Scholar]
- Ruedas-Rama, M.J.; Ruíz-Medina, A.; Molina-Díaz, A. A Prussian blue-based flow-through renewable surface optosensor for analysis of ascorbic acid. Microchem. J 2004, 78, 157–162. [Google Scholar]
- Llorent-Martínez, E.J.; García-Reyes, J.F.; Ortega-Barrales, P.; Molina-Díaz, A. Solid-phase ultraviolet sensing system for determination of methylxanthines. Anal. Bioanal. Chem 2005, 382, 158–163. [Google Scholar]
- Llorent-Martínez, E.J.; García-Reyes, J.F.; Ortega-Barrales, P.; Molina-Díaz, A. Flow-through fluorescence-based optosensor with on-line solid-phase separation for the simultaneous determination of a ternary pesticide mixture. J. AOAC Inter 2005, 88, 860–865. [Google Scholar]
- Ruedas-Rama, M.J.; Ruíz-Medina, A.; Molina-Díaz, A. A flow-injection renewable surface sensor for the fluorometric determination of vanadium (V) with Alizarin Red S. Talanta 2005, 66, 1333–1339. [Google Scholar]
- Llorent-Martínez, E.J.; Domínguez-Vidal, A.; Ortega-Barrales, P.; de la Guardia, M.; Molina-Díaz, A. Implementation of multicommutation principle with flow-through multioptosensors. Anal. Chim. Acta 2005, 545, 113–118. [Google Scholar]
- Llorent-Martínez, E.J.; Ortega-Barrales, P.; Molina-Díaz, A. Multicommuted flow-through fluorescence optosensor for determination of furosemide and triamterene. Anal. Bioanal. Chem 2005, 383, 797–803. [Google Scholar]
- Ruedas-Rama, M.J.; López-Sánchez, M.; Ruíz-Medina, A.; Molina-Díaz, A.; Ayora-Canada, M.J. Flow-through sensor with Fourier transform Raman detection for determination of sulfonamides. Analyst 2005, 130, 1617–1623. [Google Scholar]
- Llorent-Martínez, E.J.; Ortega-Barrales, P.; Molina-Díaz, A. Multicommuted optosensor for the determination of pipemidic acid in biological fluids. Anal. Biochem 2005, 347, 330–332. [Google Scholar]
- Ruedas-Rama, M.J.; Ruíz-Medina, A.; Molina-Díaz, A. Resolution of biparametric mixtures using bead injection spectroscopic flow-through renewable surface sensors. Anal. Sci 2005, 21, 1079–1084. [Google Scholar]
- Llorent-Martínez, E.J.; Ortega-Barrales, P.; Molina-Díaz, A. Chemiluminescence optosensing implemented with multicommutation: Determination of salicylic acid. Anal. Chim. Acta 2006, 580, 149–154. [Google Scholar]
- Gilbert-López, B.; Llorent-Martínez, E.J.; Ortega-Barrales, P.; Molina-Díaz, A. Development of a multicommuted flow-through optosensor for the determination of a ternary pharmaceutical mixture. J. Pharm. Biomed. Anal 2007, 43, 515–521. [Google Scholar]
- López-Flores, J.; Fernández-de Cordova, M.L.; Molina-Díaz, A. Multicommutated flow-through optosensors implemented with photochemically induced fluorescence: determination of flufenamic acid. Anal. Biochem 2007, 361, 280–286. [Google Scholar]
- López-Flores, J.; Molina-Díaz, A.; Fernández-de Cordova, M.L. Determination of azoxystrobin residues in grapes, musts and wines with a multicommuted flow-through optosensor implemented with photochemically induced fluorescente. Anal. Chim. Acta 2007, 585, 185–191. [Google Scholar]
- López-Flores, J.; Molina-Díaz, A.; Fernández-de Cordova, M.L. Development of a photochemically induced fluorescence-based optosensor for the determination of imidacloprid in peppers and environmental waters. Talanta 2007, 72, 991–997. [Google Scholar]
- Domínguez-Vidal, A.; Ortega-Barrales, P.; Molina-Díaz, A. Environmental water samples analysis of pesticides by means of chemometrics combined with fluorimetric multioptosensing. J. Fluoresc 2007, 17, 271–277. [Google Scholar]
- García-Reyes, J.F.; Ortega-Barrales, P.; Molina-Díaz, A. Multicommuted fluorometric multiparameter sensor for simultaneous determination of naproxen and salicylic acid in biological fluids. Anal. Sci 2007, 23, 423–428. [Google Scholar]
- García-Reyes, J.F.; Ortega-Barrales, P.; Molina-Díaz, A. Flow-through fluorescence-based optosensor for the screening of zinc in drinking water. Anal. Sci 2007, 23, 1179–1183. [Google Scholar]
- Ortega-Algar, S.; Ramos-Martos, N.; Molina-Díaz, A. Flow-injection solid surface lanthanide-sensitized luminescence sensor for determination of p-aminobenzoic acid. Anal. Bioanal. Chem 2008, 391, 715–719. [Google Scholar]
- Llorent-Martínez, E.J.; Ortega-Barrales, P.; Molina-Díaz, A. Sequential injection multi-optosensor based on a dual-luminescence system using two sensing zones: application to multivitamin determination. Microchim. Acta 2008, 162, 199–204. [Google Scholar]
- Teles, F.R.R.; Fonseca, L.P. Trends in DNA biosensor. Talanta 2008, 77, 606–623. [Google Scholar]
- Farré, M.; Martínez, E.; Ramón, J.; Navarro, A.; Radjenovic, J.; Mauriz, E.; Lechuga, L.M.; Marco, M.P.; Barceló, D. Part per trillion determination of atrazine in natural water samples by a surface plasmon resonance immunosensor. Anal. Bioanal. Chem 2007, 388, 207–214. [Google Scholar]
- Mauriz, E.; Calle, A.; Manclús, J.J.; Montoya, A.; Hildebrandt, A.; Barceló, D.; Lechuga, L.M. Optical immunosensor for fast and sensitive detection of DDT and related compounds in river water simples. Biosens. Bioelectron 2007, 22, 1410–1418. [Google Scholar]
- Mauriz, E.; Calle, A.; Abad, A.; Montoya, A.; Hildebrandt, A.; Barceló, D.; Lechuga, L.M. Determination of carbaryl in natural water samples by a surface plasmon resonance flow-through immunosensor. Biosens. Bioelectron 2006, 21, 2129–2136. [Google Scholar]
- Mauriz, E.; Calle, A.; Lechuga, L.M.; Quintana, J.; Montoya, A.; Manclus, J.J. Real-time detection of chlorpyrifos at part per trillion levels in ground, surface and drinking water samples by a portable surface plasmon resonance immunosensor. Anal. Chim. Acta 2006, 561, 40–47. [Google Scholar]
- Mauriz, E.; Calle, A.; Montoya, A.; Lechuga, L.M. Determination of environmental organic pollutants with a portable optical immunosensor. Talanta 2006, 69, 359–364. [Google Scholar]
- Mauriz, E.; Calle, A.; Manclus, J.J.; Montoya, A.; Escuela, A.M.; Sendra, J.R.; Lechuga, L.M. Single and multi-analyte surface plasmon resonance assays for simultaneous detection of cholinesterase inhibiting pesticidas. Sens. Actuat. B 2006, 118, 399–407. [Google Scholar]
- Mauriz, E.; Calle, A.; Manclus, J.J.; Montoya, A.; Lechuga, L.M. On-line determination of 3,5,6-trichloro-2-pyridinol in human urine samples by surface plasmon resonance immunosensing. Anal. Bioanal. Chem 2007, 387, 2757–2765. [Google Scholar]
- Trevino, J.; Calle, A.; Rodríguez-Frade, J.M.; Mellado, M.; Lechuga, L.M. Determination of human growth hormone in human serum samples by surface plasmon resonance immunoassay. Talanta 2009, 78, 1011–1016. [Google Scholar]
- Mauriz, E.; Calle, A.; Manclus, J.J.; Montoya, A.; Lechuga, L.M. Multi-analyte SPR immunoassays for environmental biosensing of pesticides. Anal. Bioanal. Chem 2007, 387, 1449–1458. [Google Scholar]
- Aguilar, M.R.; Gallardo, A.; Lechuga, L.M.; Calle, A.; San-Roman, J. Modulation of proteins adsorption onto the surface of chitosan complexed with anionic copolymers. Real time analysis by surface plasmon resonance. Macromol. Biosci 2004, 4, 631–638. [Google Scholar]
- Sepúlveda, B.; Calle, A.; Lechuga, L.M.; Armelles, G. Highly sensitive detection of biomolecules with the magneto-optic surface-plasmonresonance sensor. Opt. Lett 2006, 31, 1085–1087. [Google Scholar]
- Brian, B.; Sepúlveda, B.; Alaverdyan, Y.; Lechuga, L.M.; Kaell, M. Sensitivity enhancement of nanoplasmonic sensors in low refractive index substrates. Opt. Express 2009, 17, 2015–2023. [Google Scholar]
- Lechuga, L.M.; Sepúlveda, B.; Sánchez-del Río, J.; Blanco, F.; Calle, A.; Domínguez, C. Integrated micro- and nano-optical biosensor silicon devices CMOS compatible. Proc. SPIE 2004, 5357, 96–110. [Google Scholar]
- Blanco, F.J.; Agirregabiria, M.; Berganzo, J.; Mayora, K.; Elizalde, J; Calle, A; Domínguez, C.; Lechuga, L.M. Microfluidic-optical integrated CMOS compatible devices for label-free biochemical sensing. J. Micromech. Microeng 2006, 16, 1006–1016. [Google Scholar]
- Sepúlveda, B.; del Río, J.S.; Moreno, M.; Blanco, F.J.; Mayora, K.; Domínguez, C.; Lechuga, L.M. Optical biosensor microsystems based on the integration of highly sensitive Mach-Zehnder interferometer devices. J. Opt. A: Pure Appl. Opt 2006, 8, S561–S566. [Google Scholar]
- Del Río, J.S.; Carrascosa, L.G.; Blanco, F.; Moreno, M.; Berganzo, J.; Calle, A; Domínguez, C.; Lechuga, L.M. Lab-on-a-chip platforms based on highly sensitive nanophotonic Si biosensors for single nucleotide DNA testing. Silicon Photonics II 2007, 6477, 64771B/1–64771B/9. [Google Scholar]
- Lucena, R.; Cárdenas, S.; Gallego, M.; Valcárcel, M. ATR-FT-IR membrane-based sensor for integrated microliquid-liquid extraction and detection. Anal. Chem 2005, 77, 7472–7477. [Google Scholar]
- Lucena, R.; Cárdenas, S.; Gallego, M.; Valcárcel, M. ATR-FTIR membrane-based sensor for the simultaneous determination of surfactant and oil total indexes in industrial degreasing baths. Analyst 2006, 131, 415–421. [Google Scholar]
- Alcudia-León, M.C.; Lucena, R.; Cárdenas, S.; Valcárcel, M. Characterization of an attenuated total reflection-based sensor for integrated solid-phase extraction and infrared detection. Anal. Chem 2008, 80, 1146–1151. [Google Scholar]
- Barrios, C.A.; Banuls, M.J.; González-Pedro, V.; Gylfason, K.B.; Sánchez, B.; Griol, A.; Maquieira, A.; Sohlstrom, H.; Holgado, M.; Casquel, R. Label-free optical biosensing with slot-waveguides. Opt. Lett 2008, 33, 708–710. [Google Scholar]
- Fritz, J. Cantilever biosensors. Analyst 2008, 133, 855–863. [Google Scholar]
- Jiménez-Cadena, G.; Riu, J.; Rius, F.X. Gas sensors based on nanostructured materials. Analyst 2007, 132, 1083–1099. [Google Scholar]
- Ono, M.; Lange, D.; Brand, O.; Hagleitner, C.; Baltes, H. A complementary-metal-oxide-semiconductor-field-effect-transistor-compatible atomic force microscopy tip fabrication process and integrated atomic force microscopy cantilevers fabricated with this process. Ultramicroscopy 2002, 91, 9–20. [Google Scholar]
- Chow, E.M.; Yaralioglu, G.G.; Quate, C.F.; Kenny, T.W. Characterization of a two-dimensional cantilever array with through-wafer electrical interconnects. Appl. Phys. Lett 2002, 80, 664–666. [Google Scholar]
- Ziegler, C. Cantilever-based biosensors. Anal. Bioanal. Chem 2004, 379, 946–959. [Google Scholar]
- Carrascosa, L.G.; Moreno, M.; Álvarez, M.; Lechuga, L.M. Nanomechanical biosensors: a new sensing tool. Trends Anal. Chem 2006, 25, 196–206. [Google Scholar]
- Zinoviev, K.; Plaza, J.A.; Cadarso, V; Domínguez, C.; Lechuga, L.M. Optical biosensor based on arrays of waveguide microcantilevers. Proc. SPIE 2007, 6477, 64771A/1–64771A/13. [Google Scholar]
- Calleja, M.; Nordstrom, M.; Álvarez, M.; Tamayo, J.; Lechuga, L.M.; Boisen, A. Highly sensitive polymer-based cantilever-sensors for DNA detection. Ultramicroscopy 2005, 105, 215–222. [Google Scholar]
- Plaza, J.A.; Zinoviev, K.; Villanueva, G.; Álvarez, M.; Tamayo, J.; Domínguez, C.; Lechuga, L.M. T-shaped microcantilever sensor with reduced deflection offset. Appl. Phys. Lett 2006, 89, 094109/1–094109/3. [Google Scholar]
- Álvarez, M.; Carrascosa, L.G.; Moreno, M.; Calle, A.; Zaballos, A.; Lechuga, L.M.; Martínez, C.; Tamayo, J. Nanomechanics of the formation of DNA self-assembled monolayers and hybridization on microcantilevers. Langmuir 2004, 20, 9663–9668. [Google Scholar]
- Klotz, A.; Brecht, A.; Barzen, C.; Gauglitz, G.; Harris, R.D.; Quigley, G.R.; Wilkinson, J.S.; Abuknesha, R.A. Optical immunoprobe development for multiresidue monitoring in water. Sens. Actuat. B 1998, 51, 181–187. [Google Scholar]
- Rodríguez-Mozaz, S.; López de Alda, M.J.; Barceló, D. Analysis of bisphenol A in natural waters by means of an optical immunosensor. Water Res 2005, 39, 5071–5079. [Google Scholar]
- Rodríguez-Mozaz, S.; Reder, S.; López de Alda, M.J.; Gauglitz, G.; Barceló, D. Simultaneous multi-analyte determination of estrone, isoproturon and atrazine in natural waters by the River ANAlyser (RIANA), an optical immunosensor. Biosens. Bioelectron 2004, 19, 633–640. [Google Scholar]
- Rodríguez-Mozaz, S.; López de Alda, M.J.; Barceló, D. An optical immunosensor for pesticide determination in natural waters. Methods in Biotechnol 2006, 19, 481–489. [Google Scholar]
- Grate, J.W. Acoustic wave microsensor arrays for vapor sensing. Chem. Rev 2000, 100, 2627–2647. [Google Scholar]
- Verd, J.; Abadal, G.; Teva, J.; Gaudo, M.V.; Uranga, A.; Borrisé, X.; Campabadal, F.; Esteve, J.; Costa, E.F.; Pérez-Murano, F.; Davis, Z.J.; Forsén, E.; Boisen, A.; Barniol, N. Design, fabrication, and characterization of a submicroelectromechanical resonator with monolithically integrated CMOS readout circuit. J. Microelectromech. Syst 2005, 14, 508–519. [Google Scholar]
- Li, Y.C.; Ho, M.H.; Hung, S.J.; Chen, M.H.; Lu, M. CMOS micromachined capacitive cantilevers for mass sensing. J. Micromech. Microeng 2006, 16, 2659–2665. [Google Scholar]
- Palenzuela, B.; Simonet, B.M.; Ríos, A.; Valcárcel, M. Determination of free and total sulphur dioxide in wine by use of an amalgamated piezoelectric sensor. Anal. Chim. Acta 2005, 535, 65–72. [Google Scholar]
- Zougagh, M.; Ríos, A.; Valcárcel, M. Direct determination of total carbonate salts in soil samples by continuous-flow piezoelectric detection. Talanta 2005, 65, 29–35. [Google Scholar]
- Zougagh, M.; Ríos, A.; Valcárcel, M. Automatic selective determination of caffeine in coffee and tea samples by using a supported liquidmembrane-modified piezoelectric flow sensor with molecularly imprinted polymer. Anal. Chim. Acta 2005, 539, 117–124. [Google Scholar]
- Ávila, M.; Zougagh, M.; Escarpa, A.; Ríos, A. Supported liquid membrane-modified piezoelectric flow sensor with molecularly imprinted polymer for the determination of vanillin in food simples. Talanta 2007, 72, 1362–1369. [Google Scholar]
- Vidal, J.C.; Duato, P.; Bonel, L.; Castillo, J.R. Use of polyclonal antibodies to ochratoxin A with a quartz-crystal microbalance for developing real-time mycotoxin piezoelectric immunosensors. Anal. Bioanal. Chem 2009, 394, 575–582. [Google Scholar]
- Plata, M.R.; Hernando, J.; Zougagh, M.; Contento, A.M.; Villaseñor, M.J.; Sánchez-Rojas, J.L.; Ríos, A. Characterization and analytical validation of a microcantilever-based sensor for the determination of total carbonate in soil samples. Sens. Actuat. B 2008, 134, 245–251. [Google Scholar]
- Sensia. Homepage. http://www.sensia.es (accessed on 1 December 2009).
- DropSens. Homepage. http://www.dropsens.com (accessed on 1 December 2009).
- Escamilla-Gómez, V.; Campuzano, S.; Pedrero, M.; Pingarrón, J.M. Gold screen-printed-based impedimetric immunobiosensors for direct and sensitive Escherichia coli quantisation. Biosens. Bioelectron 2009, 24, 3365–3371. [Google Scholar]
- Loaiza, O.A.; Campuzano, S.; Pedrero, M.; García, P.; Pingarrón, J.M. Ultrasensitive detection of coliforms by means of direct asymmetric PCR combined with disposable magnetic amperometric genosensors. Analyst 2009, 134, 34–37. [Google Scholar]
- Loaiza, O.A.; Campuzano, S.; Pedrero, M.; Pingarrón, J.M. Designs of enterobacteriaceae Lac Z gene DNA gold screen printed biosensors. Electroanalysis 2008, 20, 1397. [Google Scholar]
- Fanjul-Bolado, P.; Lamas-Ardisana, P.J.; Hernández-Santos, D.; Costa-García, A. Electrochemical study and flow injection analysis of paracetamol in pharmaceutical formulations based on screen-printed electrodes and carbon nanotubes. Anal. Chim. Acta 2009, 638, 133–138. [Google Scholar]
- InBea. Homepage. http://www.inbea.com (accessed on 1 December 2009).
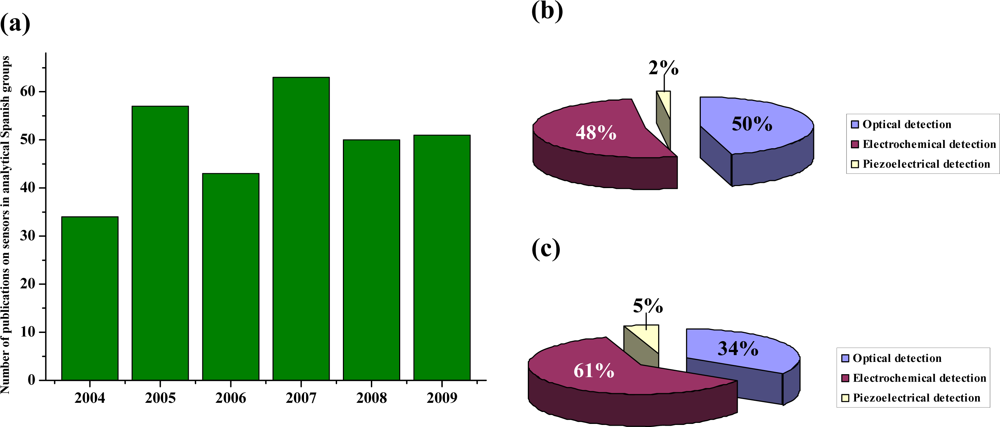
| Investigation group | Responsible | Developed Sensors | Links |
|---|---|---|---|
| Sensors and biosensors group (University Autonoma Barcelona) | Salvador Alegret |
| http://webs2002.uab.es/_c_gr_gsb/index.html |
| Sensors and biosensors group (University Autonoma Barcelona) | Julián Alonso |
| http://einstein.uab.es/_c_gr_gsb/GSB/julian/index.htm |
| Electroanalysis group (University of Pais Vasco) | Ramón J. Barrio |
| -- |
| Electroanalysis group (University of Burgos) | María J. Arcos-Domínguez |
| http://www.ubu.es/paginas/grupos_investigacion/cien_biotec/elan/index.htm |
| Environmental chemistry group (IDAEA-CSIC, Barcelona) | Damiá Barceló |
| http://grac.org/Barcelo.pdf |
| Analytical spectroscopy & sensors group (University of Zaragoza) | Juan R. Castillo |
| http://www.unizar.es/departamentos/quimica_analitica/personal/jr_castillo.html |
| Solid-phase spectrometry group (University of Granada) | Luis F. Capitán-Vallvey |
| http://www.ugr.es/~efasesol/inicial.htm |
| Inmunoeletroanalysis group (University of Oviedo) | Agustín Costa |
| http://inmunoweb.iespana.es/proyectos.htm |
| Organic chemistry of metal complexes group (University of Valencia) | Ana M. Costero |
| http://www.uv.es/supraorg |
| Optical sensors and bioanalysis group (University of Oviedo) | Marta E. Díaz-García |
| http://www.uniovi.es/vicinves/web_vieja/portal/ot/activos/075.pdf |
| Microelectronic institute of Barcelona | Carlos Domínguez |
| http://www.imb-cnm.csic.es/index.php?lang=en |
| Bioelectrochemistry and biosensors group (University of Alcalá) | Elena Domínguez |
| http://www.uah.es/INVESTIGACION/INVESTIGACION/docs/Grupos/CC_Experimentales.pdf |
| Techniques and methods of chemical analysis group (UNED) | Jesús S. Durand |
| http://www.uned.es/gtymaq/1.htm |
| Environmental, biochemical and foodstuffs analytical control research group (University of Granada) | Alberto Fernández-Gutiérrez |
| http://feugr.ugr.es/ProyectoConecta/php/contenidogrupo.php3?id=5 |
| Analytical spectroscopy & sensors group (University of Zaragoza) | Javier Galban |
| http://www.unizar.es/departamentos/quimica_analitica/JavierGalban.html |
| Nano biosensors and molecular biophysics group (Nanoscience & Nanotechnology Investigation Centre, CSIC, Barcelona) | Laura M. Lechuga |
| http://www.cin2.es/biosensores |
| Biosensors group (University of Autonoma of Madrid) | M. Encarnación Lorenzo |
| http://www.uam.es/gruposinv/biosens/presentacion.html |
| Institute of applied molecular chemistry (University of Valencia) | A. Maquieira |
| http://iqma.webs.upv.es/eng/index.php |
| Nanobioelectronics & biosensors group (Catalonian Institut of Nanotecnology, Barcelona) | Arben Merkoçi |
| http://nanocat.uab.cat/dataeng/recerca/biopriv/bio_home.php |
| Analytical chemistry group (University of Jaen) | Antonio Molina |
| http://www.ujaen.es/serv/vicinv/verGrupo.php?grupo=42 |
| Laboratory of optical sensors (University Complutense of Madrid) | María C. Moreno-Bondi |
| http://www.ucm.es/info/gsolfa |
| Automatic analytical method and chemical sensors group (University of Murcia) | Joaquín A. Ortuño |
| https://curie.um.es/curie/catalogo-grupo-investigacion.du?cods=E044*02 |
| Chemical sensors and biosensors (University of Autonoma of Madrid) | María. D. Petit-Domínguez |
| http://www.uam.es/gruposinv/biosens |
| Electroanalysis and electrochemical (bio)sensors group. (University of Complutense of Madrid) | José M. Pingarrón |
| http://www.ucm.es/info/analitic |
| Chemical analysis of environmental (University of Extremadura) | Eduardo Pinilla-Gil |
| http://www.unex.es/quianaelec/aquima |
| Automatization, simplification and miniaturization of analytical processes (University of Castilla La Mancha) | Ángel Ríos |
| http://www.uclm.es/organos/vic_investigacion/GruposUCLM/grupos.aspx?gr=191&inf=per |
| Chemometrics, qualimetrics and nanosensors group (Universitat Rovira i Virgili) | Xavier Rius |
| http://www.quimica.urv.es/quimio/ang/maincat.html |
| Analytical spectrometry (University of Oviedo) | Alfredo Sanz-Medel |
| www12.uniovi.es/spectrometry |
| Electroanalysis group (University of Oviedo) | Paulino Tuñon-Blanco |
| http://www.uniovi.es/electroanalisis/englishversion.htm |
| Automation, simplification, miniaturization and quality of the (bio)chemical measurement processes (University of Cordoba) | Miguel Válcarcel |
| http://www.uco.es/grupos/FQM-215/index.htm |
| System | Indicator electrode | No. of electrodes | Detection technique | Analyte | Sample | Detection mode | Reference |
|---|---|---|---|---|---|---|---|
| e-tongue, ANN and FI | Ion-selective PVC polymer membrane | 4 | Potentiometric sensor | Nitrate, Chloride | Water | Direct detection | [3] |
| e-tongue, ANN and FI | Ion-selective PVC polymer membrane | 8 | Potentiometric sensor | NH4+, K+ and Na+ ions. | Synthetic and river water, waste water and fertilizer | Simultaneous multi-determination | [137] |
| e-tongue, ANN and SIA | Home-made epoxy-graphite electrode | -- | Voltammetric sensor | o-cresol, pchlorophenol, 4-chloro-3-methylphenol | -- | Direct and simultaneous multi-determination | [138] |
| e-tongue, ANN and SIA | Ion-selective PVC polymer membrane | 5 | Potentiometric sensor | Cl−, NO3− and HCO3− | Water | Direct and simultaneous multi-determination | [139–141] |
| e-tongue, ANN and FI | Ion-selective PVC polymer membrane | -- | Potentiometric sensor | -- | Commercial waters, orange-based drinks, tea samples and natural juice | Direct detection | [142] |
| e-tongue, ANN | -- | -- | Voltammetric sensor | Tryptophan, cysteine, and tyrosine | Animal “feed” | Direct detection | [143,144] |
| e-tongue, ANN | Ion-selective PVC polymer membrane | 8 | Potentiometric sensor | Ammonium, potassium, sodium, chloride, phosphate and nitrate ions | Soils | Direct and simultaneous multide-termination | [145] |
| Bioe-tongue and ANN | Urease and creatinine deiminase covalently immobilized onto ammonium selective electrodes and polymeric membranes | -- | -- | Urea, creatitine, ammonium, potassium and sodium. | Clinical samples | Direct and simultaneous multi-determination | [146] |
| e-tongue, ANN | Screen printed on polymeric substrate | 5 | Potentiometric sensor | Ammonium, potassium, sodium, chloride and nitrate ions | Surface waters | Direct and simultaneous multi-determination | [147,148] |
| e-tongue, ANN and SIA | Two based on chalcogenide glasses Cd and Cu sensor, and the rest on PVC membranes Pb and Zn sensor. | 4 | Potentiometric sensor | Cd, Cu, Pb and Zn | -- | Direct and simultaneous multi-determination | [149] |
| Indicator electrode | Functionalization | Detection technique | Analyte | Reference | ||
|---|---|---|---|---|---|---|
| Type of immobilization | Enzimatic label | Substrate | ||||
| SPCEs | Streptavidin/Biotin reaction | Alkaline phosphatase (AP) | 3-indoxyl phosphate (3-IP) | Voltametric sensor | Virulence nucleic acid in Pneumolysin and autolysin genes of the human pathogen Streptococcus pneumoniae | [175] |
| SPCEs and flow system | -- | Alkaline phosphatase (AP) and Horseradish peroxidase (HRP) | 3-indoxyl phosphate (3-IP) | Voltametric sensor | -- | [176] |
| SPCEs | Streptavidin/Biotin reaction | Platinum (II) complex | -- | Voltametric sensor | Virulence nucleic acid in Pneumolysin and autolysin genes of the human pathogen Streptococcus pneumoniae | [177] |
| SPCEs | Streptavidin/Biotin reaction | Alkaline phosphatase (AP) | 3-indoxyl phosphate (3-IP) | Voltametric sensor | Rabbit IgG in direct determination Competitive immunoassay | [170] |
| Comercial SPCEs Flow cell | Horseradish peroxidase (HRP) | -- | 3,3′,5,5′-Tetramethylbenzidine (TMB) | Amperometric sensor | Interleukin 6 | [178] |
| SPCEs Flow cell | Alkaline phosphatase | -- | P-Nitrophenyl phosphate | Amperometric sensor | p-nitrophenol | [179] |
| Glassy carbon electrodes | -- | Gold complex | -- | Voltametric sensor | SARS virus | [180] |
| -- | Streptavidin/Biotin reaction | Alkaline phosphatase (AP) | 3-indoxyl phosphate (3-IP) | Voltametric sensor | DNA | [130,181] |
| Carbon SPCEs | Streptavidin/Biotin reaction | Alkaline phosphatase (AP) | 3-indoxyl phosphate (3-IP) and silver ions | Voltametric sensor | Virulence nucleic acid in autolysin gene of the human pathogen Streptococcus pneumoniae | [182] |
| Gold SPCEs | -- | -- | Polycarbonate and alumina | Voltametric sensor | Potassium ferricyanide, p-aminophenol, indigo carmine, silver nitrate and ferrocene | [183] |
| SPCEs | p-aminophenol- phosphatase | Alkaline phosphatase (AP) | MWCNT-COOH | Voltametric sensor | p-aminophenol | [184] |
| SPCEs | -- | Gold nanoparticles | -- | Voltametric sensor | Lead | [185] |
| SPCEs | -- | Alkaline phosphatase (AP) and gold nanoparticles | 3-indoxyl phosphate (3-IP) | Voltametric sensor | SARS (severe acute respiratory syndrome) virus | [186] |
| SPCEs carbon, gold or carbon nanotubes | -- | Alkaline phosphatase (AP) | 3-indoxyl phosphate (3-IP) | Voltammetric sensor | Prostate specific antigen (fPSA and tPSA) | [187] |
| Gold nanostructured SPCEs | -- | -- | -- | Voltammetric sensor | Lead in blood | [188] |
| System | Detection technique | Active sorbent substrate | Reagents | Analyte | Reference |
|---|---|---|---|---|---|
| Bead injection spectroscopy-flow injection analysis (BIS-FIA) | Spectrofluorometry | Sephadex QAE A-25 | Morin (2′,3,4′,5,7-pentahydroxyflavone) | Berillium and Aluminum | [277] |
| Flow injection (FI) manifold | Spectrofluorometry | Sephadex SP C-25 cation-exchange gel beads | -- | Diphenhydramine in pharmaceutical samples | [278] |
| Flow injection (FI) manifold | UV spectrophotometry | Sephadex SP G-15 sorption gel | -- | Ciprofloxacin | [279] |
| Flow injection (FI) manifold | Spectrofluorometry | -- | Quinine (QN) and quinidine (QD) | [280] | |
| Bead injection spectroscopy-flow injection analysis (BIS-FIA) | Spectrophotometry | Sephadex QAE A-25 resin | Ferrozine (FeFz3) 4− | Promethazine and trifluoperazine in pharmaceutical samples | [281] |
| Bead injection spectroscopy-flow injection analysis (BIS-FIA) | Spectrophotometry | Sephadex QAE A-25 resin | Prussian blue (PB) | Ascorbic acid | [282] |
| Flow injection (FI) manifold | UV spectrophotometry | Octadecyl silane C18 gel | -- | Methylxanthines: caffeine (CF) and theophylline (TP) in pharmaceuticals samples and CF and theobromine (TB) in food and beverages. | [283] |
| Flow injection (FI) manifold | Spectrofluorometry | C18 silica gel | -- | Pesticides: carbendazim (CBZ), carbofuran (CF) and benomyl (BNM) | [284] |
| Bead injection spectroscopy-flow injection analysis (BIS-FIA) | Spectrofluorometry | Sephadex QAE A-25 resin | 1,2-dihydroxyanthrquinone-3-sulfonic acid (Alazarin Red S) | Vanadium (V) | [285] |
| Flow injection (FI) manifold and multioptosensors | UV spectrophotometry | -- | Salicylamide (SLC) and caffeine (CF) | [286] | |
| Flow injection (FI) manifold | Spectrofluorometry | Sephadex SPC-25 microbeads | -- | Furosemide and triamterene in human urine and blood serum. | [287] |
| Flow injection (FI) manifold | Fourier transform (FT) Raman spectroscopy | Sephadex QAE A-25 resin | -- | Sulfonamides: sulfathiazole and sulfamethoxazole | [288] |
| Flow injection (FI) manifold | Luminescence technique | -- | A luminiscent TB chelate | Pipemidic acid and quinolone antibacterial agents (norfloxacin, ciprofloxacin, enoxacin, trovafloxacin) in biological fluids | [289] |
| Bead injection spectroscopy-flow injection analysis (BIS-FIA) | UV spectrophotometry | Sephadex QAE A-25 anion exchange gel | 2-carboxyl-2-hydroxy-5-sulfoformazylbenzene (Zincon) | Biparametric mixtures (copper (II) and zinc (II)) | [290] |
| Flow injection (FI) manifold | Chemiluminiscency | -- | -- | Salicylic acid | [291] |
| Flow injection (FI) manifold | UV spectrophotometry | -- | -- | Ternary pharmaceutical mixture | [292] |
| Flow injection (FI) manifold | UV spectrophotometry | C18 silica gel | -- | Flufenamic acid (FFA) | [293] |
| Flow injection (FI) manifold | UV spectrophotometry | C18 silica gel | -- | Azoxystrobin residues in grapes, musas and wines | [294] |
| Flow injection (FI) manifold | Spectrofluorometry | C18 silica gel | -- | Imidacloprid in peppers and environmental waters | [295] |
| Flow injection (FI) manifold | Spectrofluorometry | C18 silica gel | -- | Pesticides: a-naphthol, o-phenylphenol and thiabendazole in water samples | [296] |
| Flow injection (FI) manifold | Spectrofluorometry | C18 silica gel | -- | Naproxen and salicylic acid in biological samples | [297] |
| Flow injection (FI) manifold | Spectrofluorometry | C18 silica gel | p-(tosylamino) quinoline | Zinc (II) in drinking water | [298] |
| Flow injection (FI) manifold | Luminescency | Sephadex QAE A-25 anion exchange gel | Tb (III) and lanthanide-sensitized luminiscence | p-aminobenzoic acid (PABA) | [299] |
| Sequential injection análisis (SIA) | Spectrofluorometry | Vitamins B2, B6 and C | [300] |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Plata, M.R.; Contento, A.M.; Ríos, A. State-of-the-Art of (Bio)Chemical Sensor Developments in Analytical Spanish Groups. Sensors 2010, 10, 2511-2576. https://doi.org/10.3390/s100402511
Plata MR, Contento AM, Ríos A. State-of-the-Art of (Bio)Chemical Sensor Developments in Analytical Spanish Groups. Sensors. 2010; 10(4):2511-2576. https://doi.org/10.3390/s100402511
Chicago/Turabian StylePlata, María Reyes, Ana María Contento, and Angel Ríos. 2010. "State-of-the-Art of (Bio)Chemical Sensor Developments in Analytical Spanish Groups" Sensors 10, no. 4: 2511-2576. https://doi.org/10.3390/s100402511
APA StylePlata, M. R., Contento, A. M., & Ríos, A. (2010). State-of-the-Art of (Bio)Chemical Sensor Developments in Analytical Spanish Groups. Sensors, 10(4), 2511-2576. https://doi.org/10.3390/s100402511




