Ethyl 7-Methyl-1-(4-nitrophenyl)-5-phenyl-3-(thiophen-2-yl)-1,5-dihydro-[1,2,4]triazolo[4,3-a]pyrimidine-6-carboxylate
Abstract
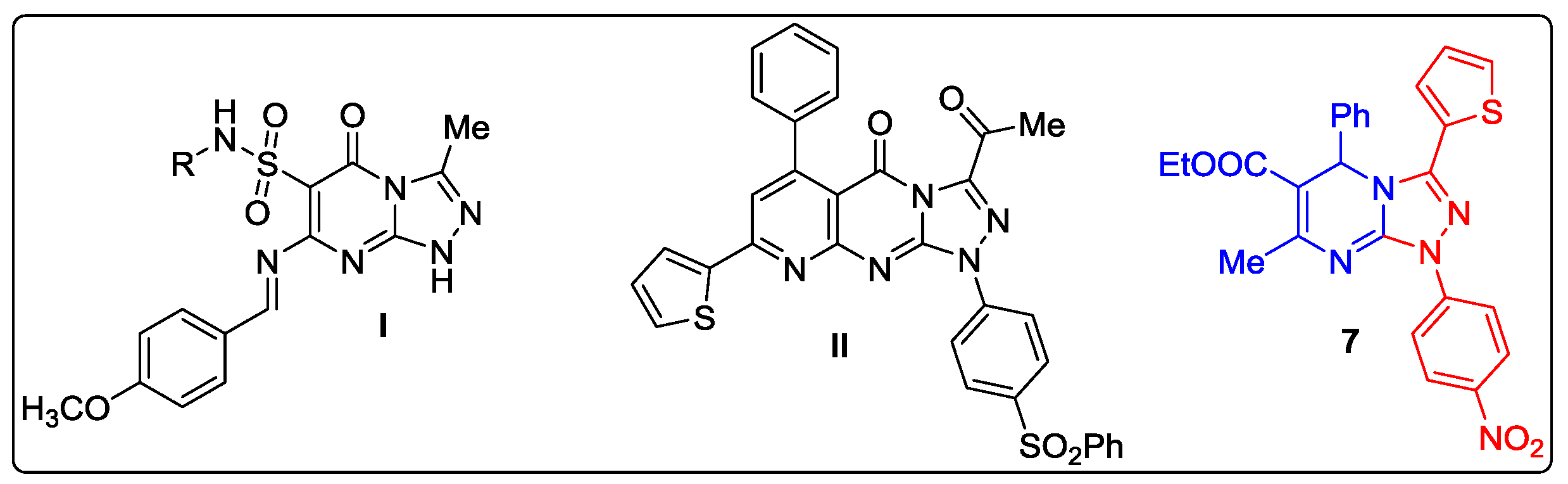
:1. Introduction
2. Results and Discussion
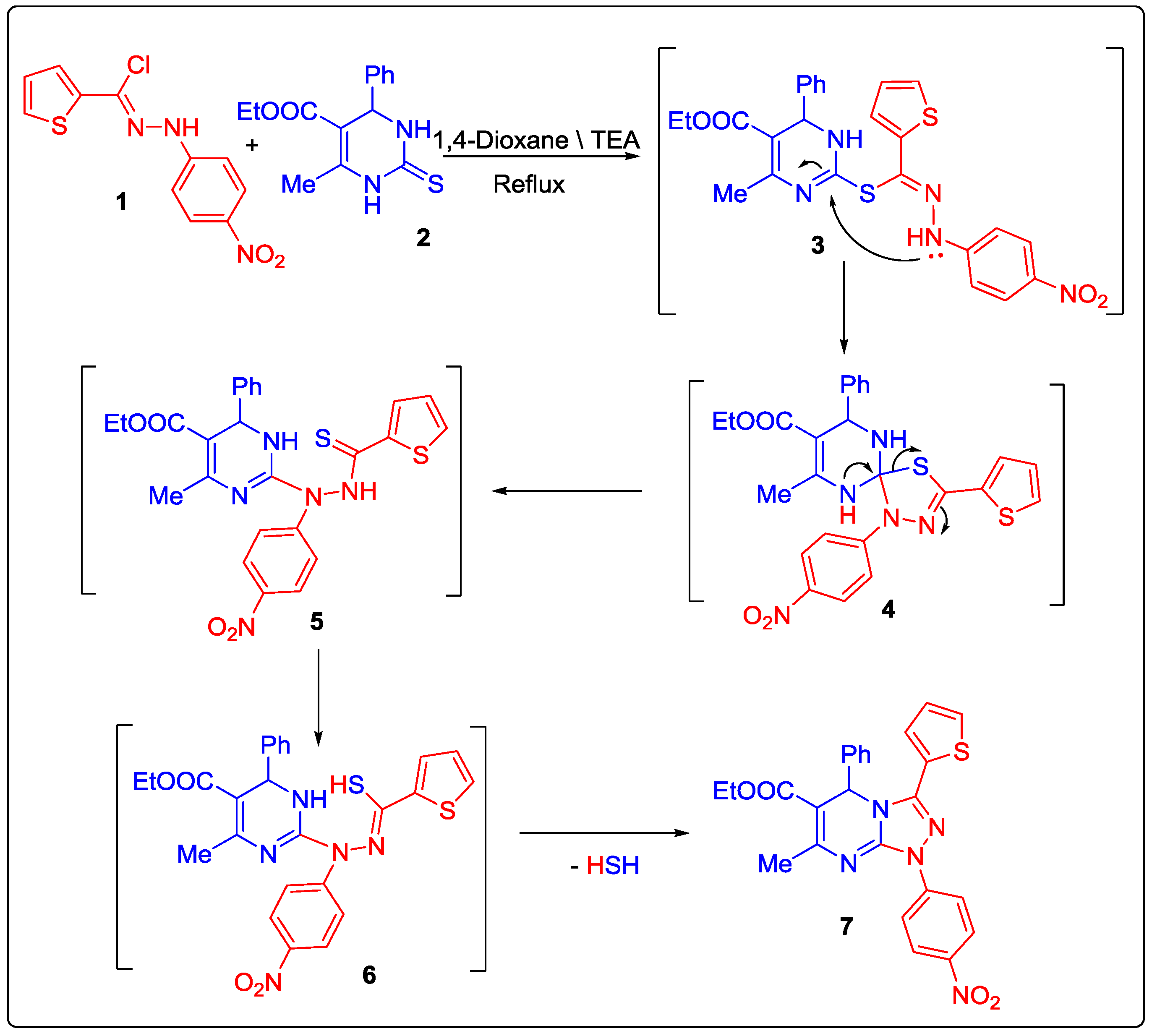
2.1. Chemistry
2.2. Cytotoxic Activity
3. Experimental
3.1. Chemistry
3.2. Synthesis of Ethyl 7-Methyl-1-(4-nitrophenyl)-5-phenyl-3-(thiophen-2-yl)-1,5-dihydro-[1,2,4]triazolo[4,3-a]pyrimidine-6-carboxylate (7)
3.3. Biological Evaluation
Antitumor activity assay:
4. Conclusion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ilies, A.; Vullo, D.; Pastorek, J.; Scozzofava, A.; Ilies, M.; Caproiu, M.T.; Pastorekova, S.; Supuran, C.T. Carbonic anhydrase inhibitors of tumor associated isozyme IX by dihalogenosulfanilamide and halogenophenylamino benzolamide derivatives. Farmaco 2003, 46, 487–496. [Google Scholar]
- Terzioglu, N.; Gursoy, A. Synthesis and anticancer evaluation of some new hydrazone derivatives of 2,6-dimethylimidazo[2,1]-b]-[1,3,4]thiadiazole-5-carbohydrazide. Eur. J. Med. Chem. 1988, 23, 385–389. [Google Scholar] [CrossRef]
- Alagarsamy, V.; Pathak, S.U.; Meena, S.; Rameshu, V.K.; Reyathi, R. Anticancer, antibacterial and antifungal activities of soidc 2-substituted (1,3,4)-thiadiazolothieno(2,3-c)-pyrimidin-5-(4H)-ones. Ind. J. Hetercycl. Chem. 2003, 12, 335–338. [Google Scholar]
- Gomha, S.M.; Edrees, M.M.; Altalbawy, F.M.A. Synthesis and characterization of some new bis-pyrazolyl-thiazoles incorporating the thiophene moiety as potent anti-tumor agents. Int. J. Mol. Sci. 2016, 17, 1499. [Google Scholar] [CrossRef] [PubMed]
- Forsch, R.A.; Wright, J.E.; Rosowsky, A. Synthesis and in vitro antitumor activity of thiophene analogues of 5-chloro-5,8-dideazafolic acid and 2-methyl-2-desamino-5-chloro-5,8-dideazafolic acid. Bioorg. Med. Chem. 2002, 10, 2067–2076. [Google Scholar] [CrossRef]
- Shawali, A.S. Tandem in situ generation and 1,5-electrocyclization of N-hetaryl nitrilimines. A facile methodology for synthesis of annulated 1,2,4-triazoles and their acyclo C-nucleosides. Arkivoc 2010, 1, 33–97. [Google Scholar] [CrossRef]
- Zhang, N.; Ayral-Kaloustian, S.; Nguyen, T.; Afragola, J.; Hernandez, R.; Lucas, J. Synthesis and SAR of [1,2,4]triazolo[1,5-a]pyrimidines, a class of anticancer agents with a unique mechanism of tubulin inhibition. J. Med. Chem. 2007, 50, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.L.; Zhao, Y.F.; Guo, S.C.; Song, H.S.; Wang, D.; Gong, P. Synthesis and anti-tumor activities of novel [1,2,4]triazolo[1,5-a]pyrimidines. Molecules 2007, 12, 1136–1146. [Google Scholar] [CrossRef] [PubMed]
- Gomha, S.M.; Abdelrazek, F.M.; Abdulla, M.M. Synthesis of new functionalised derivatives of [1,2,4]triazolo[4,3-a]pyrimidine and pyrimido[2,1-b][1,3,5]thiadiazine as aromatase inhibitors. J. Chem. Res. 2015, 39, 425–429. [Google Scholar]
- Baraldi, P.G.; Pavani, M.G.; Nunez, M.D.; Brigidi, P.; Vitali, B.; Gambari, R.; Romagnoli, R. Antimicrobial and antitumor activity of N-heteroimmine-1,2,3-dithiazoles and their transformation in triazolo-, imidazo-, and pyrazolopirimidines. Bioorg. Med. Chem. 2002, 10, 449–456. [Google Scholar] [CrossRef]
- Farghaly, T.A.; Gomha, S.M.; Abbas, E.M.H.; Abdalla, M.M. Hydrazonoyl halides as precursors for new fused heterocycles of 5α-reductase inhibitors. Arch. Pharm. Chem. Life Sci. 2012, 345, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Gomha, S.M.; Abbas, I.M.; Elneairy, M.A.A.; Elaasser, M.M.; Mabrouk, B.K.A. Synthesis and biological evaluation of novel fused triazolo[4,3-a]pyrimidinones. Turk. J. Chem. 2015, 39, 510–531. [Google Scholar]
- Gomha, S.M.; Ahmed, S.A.; Abdelhamid, A.O. Synthesis and cytotoxicity evaluation of some novel thiazoles, thiadiazoles, and pyrido[2,3-d][1,2,4]triazolo[4,3-a]pyrimidin-5(1H)-one incorporating triazole moiety. Molecules 2015, 20, 1357–1376. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, A.M.; Al-Qalawi, H.R.M.; El-Sayed, W.A.; Arafa, W.A.A.; Alhumaimess, M.S.; Hassan, A.K. Anticancer activity of newly synthesized triazolopyrimidine derivatives and their nucleoside analogs. Acta Pol. Pharm. Drug Res. 2015, 72, 307–318. [Google Scholar]
- Hafez, H.N.; El-Gazzar, A.R. Synthesis and antitumor activity of substituted triazolo[4,3-a]pyrimidin-6-sulfonamide with an incorporated thiazolidinone moiety. Bioorg. Med. Chem. Lett. 2009, 19, 4143–4147. [Google Scholar] [CrossRef] [PubMed]
- Fares, M.; Abou-Seri, S.M.; Abdel-Aziz, H.A.; Abbas, S.E.S.; Youssef, M.M.; Eladwy, R.A. Synthesis and antitumor activity of pyrido [2,3-d]pyrimidine and pyrido[2,3-d] [1,2,4]triazolo[4,3-a]pyrimidine derivatives that induce apoptosis through G1 cell-cycle arrest. Eur. J. Med. Chem. 2014, 83, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Gomha, S.M.; Salah, T.A.; Abdelhamid, A.O. Synthesis, characterization, and pharmacological evaluation of some novel thiadiazoles and thiazoles incorporating pyrazole moiety as anticancer agents. Monatsh. Chem. 2015, 146, 149–158. [Google Scholar] [CrossRef]
- Dawood, K.M.; Gomha, S.M. Synthesis and anti-cancer activity of 1,3,4-thiadiazole and 1,3-thiazole derivatives having 1,3,4-oxadiazole moiety. J. Heterocycl. Chem. 2015, 52, 1400–1405. [Google Scholar] [CrossRef]
- Gomha, S.M.; Riyadh, S.M.; Abbas, I.M.; Bauomi, M.A. Synthetic utility of ethylidene-thiosemicarbazide: Synthesis and and anti-cancer activity of 1,3-thiazines and thiazoles with imidazole moiety. Heterocycles 2013, 87, 341–356. [Google Scholar]
- Gomha, S.M.; Zaki, Y.H.; Abdelhamid, A.O. Utility of 3-acetyl-6-bromo-2H-chromen-2-one for synthesis of new heterocycles as potential anticancer agents. Molecules 2015, 20, 21826–21839. [Google Scholar] [CrossRef] [PubMed]
- Gomha, S.M.; Salah, T.A.; Hassaneen, H.M.E.; Abdel-Aziz, H.; Khedr, M.A. Synthesis, characterization and molecular docking of novel bioactive thiazolyl-thiazole derivatives as promising cytotoxic antitumor drug. Molecules 2016, 21, 3. [Google Scholar] [CrossRef] [PubMed]
- Hassaneen, H.M.; Mousa, A.H.; Abeed, N.M.; Shawali, A.S. Chemistry of C-Heteroarylhydrazidoyl Halides. Synthesis and Reactions of N-(p-Nitrophenyl)-C-(2-thienyl)-formohydrazidoyl Halides. Heterocycles 1988, 27, 695–706. [Google Scholar] [CrossRef]
- Vijay, K.; Ganapaty, S.; Rao, A.S. Synthesis, characterization and biological evaluation of some dihydropyrimidinones. Asian J. Chem. 2010, 22, 2518–2528. [Google Scholar]
- Fulmer, G.R.; Miller, A.J.M.; Sherden, N.H.; Gottlieb, H.E.; Nudelman, A.; Stoltz, B.M.; Bercaw, J.E.; Goldberg, K.I. NMR Chemical shifts of trace impurities: Common laboratory solvents, organics, and gases in deuterated solvents relevant to the organometallic chemist. Organometallics 2010, 29, 2176–2179. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Gomha, S.M.; Riyadh, S.M.; Mahmmoud, E.A.; Elasser, M.M. Synthesis and anticancer activities of thiazoles, 1,3-thiazines, and thiazolidine using chitosan-grafted-poly(vinylpyridine) as basic catalyst. Heterocycles 2015, 91, 1227–1243. [Google Scholar]
| Tested Compounds | Tumor Cell Lines | |
|---|---|---|
| A-549 | HepG2 | |
| 7 | 3.46 ± 0.5 | 5.54 ± 0.7 |
| Cisplatin | 19.3 ± 0.9 | 18.4 ± 1.1 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gomha, S.M.; Muhammad, Z.A.; Edrees, M.M. Ethyl 7-Methyl-1-(4-nitrophenyl)-5-phenyl-3-(thiophen-2-yl)-1,5-dihydro-[1,2,4]triazolo[4,3-a]pyrimidine-6-carboxylate. Molbank 2017, 2017, M942. https://doi.org/10.3390/M942
Gomha SM, Muhammad ZA, Edrees MM. Ethyl 7-Methyl-1-(4-nitrophenyl)-5-phenyl-3-(thiophen-2-yl)-1,5-dihydro-[1,2,4]triazolo[4,3-a]pyrimidine-6-carboxylate. Molbank. 2017; 2017(2):M942. https://doi.org/10.3390/M942
Chicago/Turabian StyleGomha, Sobhi M., Zeinab A. Muhammad, and Mastoura M. Edrees. 2017. "Ethyl 7-Methyl-1-(4-nitrophenyl)-5-phenyl-3-(thiophen-2-yl)-1,5-dihydro-[1,2,4]triazolo[4,3-a]pyrimidine-6-carboxylate" Molbank 2017, no. 2: M942. https://doi.org/10.3390/M942








