The Influence of Carbon-Carbon Multiple Bonds on the Solvolyses of Tertiary Alkyl Halides: a Grunwald-Winstein Analysis
Abstract
:1. Introduction
2. Results and Discussion
2.1. Correlations of log k with solvent ionizing power as measured by YX
2.2. Correlations of log k with G-W parameters
3. Experimental Section
4. Conclusions
Acknowledgments
References
- Abraham, MH; Doherty, RM; Kamlet, MJ; Harris, JM; Taft, RW. Linear solvation energy relationships. 37. An analysis of contributions of dipolarity polarizability, nucleophilic assistance, electrophilic assistance, and cavity terms to solvent effects on tert-butyl halide solvolysis rates. J. Chem. Soc. Perkin 1987, 2, 913–920. [Google Scholar]
- Bentley, TW; Carter, GE. The SN2-SN1 spectrum. 4. The SN2 (intermediate) mechanism for solvolyses of tert-butyl chloride-a revised Y-scale of solvent ionizing power based on solvolyses of 1-adamantyl chloride. J. Am. Chem. Soc 1982, 104, 5741–5747. [Google Scholar]
- Martins, F; Leitao, RE; Moreira, L. Solvation effects in the heterolyses of 3-X-3-methylpentanes (X = Cl, Br, I). J. Phys. Org. Chem 2004, 17, 1061–1066. [Google Scholar]
- Grunwald, E; Winstein, S. The correlation of solvolysis rates. J. Am. Chem. Soc 1948, 70, 846–859. [Google Scholar]
- Kevill, DN; D’Souza, MJ. Additional Y(Cl) values and the correlation of the specific rates of solvolysis of tert-butyl chloride in terms of N(T) and Y(Cl) scales. J. Chem. Res.-S 1993, 174–175. [Google Scholar]
- Bentley, TW; Llewellyn, G; Ryu, ZH. Solvolytic reactions in fluorinated alcohols. Role of nucleophilic and other solvation effects. J. Org. Chem 1998, 63, 4654–4659. [Google Scholar]
- Kevill, DN; D’Souza, MJ. Correlation of the rates of solvolysis of benzoyl chloride and derivatives using extended forms of the Grunwald-Winstein equation. J. Phys. Org. Chem 2002, 15, 881–888. [Google Scholar]
- Bentley, TW; Llewellyn, G. Yx scales of solvent ionizing power. Prog. Phys. Org. Chem 1990, 17, 121–158. [Google Scholar]
- Kevill, DN; D’Souza, MJ. Sixty years of the Grunwald-Winstein equation: development and recent applications. J. Chem. Res. 2008, 61–66. [Google Scholar]
- Winstein, S; Grunwald, E; Jones, HW. The correlation of solvolysis rates and the classification of solvolysis reactions into mechanistic categories. J. Am. Chem. Soc 1951, 73, 2700–2707. [Google Scholar]
- Winstein, S; Fainberg, AH; Grunwald, E. Correlation of solvolysis rates .8. Benzhydryl chloride and bromide - comparison of mY and Swain’s correlations. J. Am. Chem. Soc 1957, 79, 4146–4155. [Google Scholar]
- Bentley, TW; Schleyer, PvR. Medium effects on the rates and mechanisms of solvolytic reactions. Adv. Phys. Org. Chem 1977, 14, 1–67. [Google Scholar]
- Bentley, TW; Schadt, FL; Schleyer, PvR. Correlation of solvolysis rates with 3-parameter and 4-parameter relationships - scale of solvent nucleophilicities. J. Am. Chem. Soc 1972, 94, 992–995. [Google Scholar]
- Minegishi, S; Kobayashi, S; Mayr, H. Solvent nucleophilicity. J. Am. Chem. Soc 2004, 126, 5174–5181. [Google Scholar]
- Kevill, DN; D’Souza, MJ. Considerably improved Grunwald–Winstein correlations for solvolyses of several secondary and tertiary benzylic derivatives upon inclusion of a term governed by the aromatic ring parameter (I). J. Chem. Soc. Perkin 1995, 2, 973–980. [Google Scholar]
- Kevill, DN; Anderson, SW. An improved scale of solvent nucleophilicity based on the solvolysis of the s-methyldibenzothiophenium ion. J. Org. Chem 1991, 56, 1845–1850. [Google Scholar]
- Gajewski, JJ. Is the tert-butyl chloride solvolysis the most misunderstood reaction in organic chemistry? Evidence against nucleophilic solvent participation in the tert-butyl chloride transition state and for increased hydrogen bond donation to the 1-adamantyl chloride solvolysis transition state. J. Am. Chem. Soc 2001, 123, 10877–10883. [Google Scholar]
- Farcasiu, D; Jahme, J; Ruchardt, C. Relative reactivity of bridgehead adamantyl and homoadamantyl substrates from solvolyses with heptafluorobutyrate as a highly reactive carboxylate leaving group - absence of SN2 character of solvolysis of tert-butyl derivatives. J. Am. Chem. Soc 1985, 107, 5717–5722. [Google Scholar]
- Richard, JP; Toteva, MM; Amyes, TL. What is the stabilizing interaction with nucleophilic solvents in the transition state for solvolysis of tertiary derivatives: Nucleophilic solvent participation or nucleophilic solvation? Org. Lett 2001, 3, 2225–2228. [Google Scholar]
- Liu, KT; Hou, SJ; Tsao, ML. B-strain and solvolytic reactivity revisited. Nucleophilic solvent participation and abnormal rate ratios for tertiary chloroalkanes. J. Org. Chem 1998, 63, 1360–1362. [Google Scholar]
- Takeuchi, K; Ohga, Y; Ushino, T; Takasuka, M. Structural effects on the Grunwald-Winstein correlations in the solvolysis of some simple tertiary alkyl chlorides. J. Phys. Org. Chem 1997, 10, 717–724. [Google Scholar]
- Lee, I; Koh, HJ; Chang, BD. Solvent effects on localized and delocalized cationic charges in solvolysis. Bull. Kor. Chem. Soc 1995, 16, 1104–1108. [Google Scholar]
- Kevill, DN; Ismail, NHJ; D’Souza, MJ. Solvolysis of the (p-methoxybenzyl)dimethylsulfonium ion–development and use of a scale to correct for dispersion in Grunwald-Winstein plots. J. Org. Chem 1994, 59, 6303–6312. [Google Scholar]
- Kevill, DN; D’Souza, MJ. Application of the aromatic ring parameter (I) to solvolyses of extremely crowded alkyl derivatives. Tetrahedron Lett 1998, 39, 3973–3976. [Google Scholar]
- Kevill, DN; D’Souza, MJ. Incorporation of a term governed by the aromatic ring parameter (I) into Grunwald-Winstein correlations of the solvolyses of diarylmethyl, naphthylmethyl and arylmethyl bromides.
- Liu, KT. Comments on the utility of aromatic ring parameters in the correlation analysis of solvolytic reactivities for benzylic substrates. J. Chem. Soc. Perkin 1996, 2, 327–328. [Google Scholar]
- Bentley, TW; Dauschmidt, JP; Llewellyn, G; Mayr, H. Solvation effects adjacent to the reaction site - differences in solvation between alkyl, alkenyl, or alkynyl and aryl groups in binary aqueous mixtures. J. Org. Chem 1992, 57, 2387–2392. [Google Scholar]
- Moreira, L; Leitao, RE; Martins, F. Solvent and temperature effects on ion association and mobility of 2,6-lutidinium chloride in non-aqueous solvents. Mol. Phys 2006, 104, 1905–1913. [Google Scholar]
- Taft, RW; Abboud, JLM; Kamlet, MJ; Abraham, MH. Linear solvation energy relations. J. Solution Chem 1985, 14, 153–186. [Google Scholar]
- Moreira, L; Martins, F; Leitao, RE. Comparative studies of solvent effects in heterolyses of tertiary alkyl halides: the Grunwald-Winstein plots vs. enlarged TAKA equation. In Communication to ESORX; Rome, 2005. [Google Scholar]
- Liu, KT; Chen, PS; Hu, CR; Sheu, HC. Nucleophilic solvent intervention in the solvolysis of 2-aryl-2-chloropropanes - an example to demonstrate the advantage of using the YBnCl scale in correlation-analysis. J. Phys. Org. Chem 1993, 6, 122–125. [Google Scholar]
- Hwang, YH; Kim, SH; Gee, JG; Yoh, SD. Solvent effects on the solvolysis of 2-aryl-1,1-dimethylethyl bromides. Bull. Kor. Chem. Soc 1998, 19, 349–353. [Google Scholar]
- Kyong, JB; Won, H; Kevill, DN. Application of the extended Grunwald-Winstein equation to solvolyses of n-propyl chloroformate. Int. J. Mol. Sci 2005, 6, 87–96. [Google Scholar]
- Masada, H; Murotani, Y. A convenient method for the preparation of highly pure tert-alkyl bromides and iodides. Bull. Chem. Soc. Jpn 1980, 53, 1181–1182. [Google Scholar]
- Moreira, L; Martins, F; Leitao, RE. Design of an excel spreadsheet to estimate rate constants, determine associated errors, and choose curve’s extent. J. Chem. Educ 2006, 83, 1879–1883. [Google Scholar]
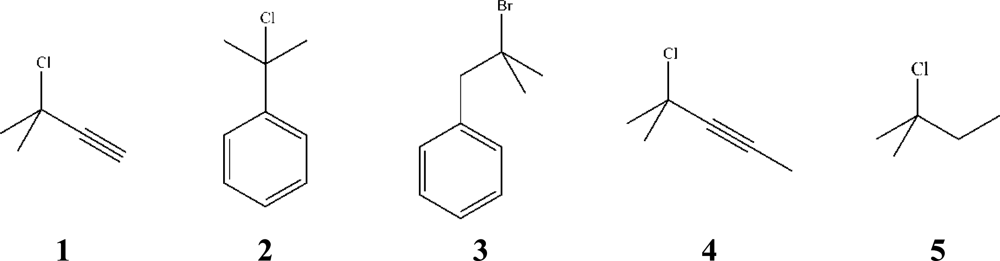
 ), 2 (
), 2 (
 ), 4 (
), 4 (
 ) and 5 (
) and 5 (
 ) at 25,00 °C and of log k vs. YBr for the solvolyses of 3 (
) at 25,00 °C and of log k vs. YBr for the solvolyses of 3 (
 ) at 25,00 °C. The filled symbols correspond to the solvolyses of 1 (
) at 25,00 °C. The filled symbols correspond to the solvolyses of 1 (
 ), 2 (
), 2 (
 ), 3 (
), 3 (
 ), 4 (
), 4 (
 ) and 5 (
) and 5 (
 ) in TFE and its mixtures. The points for 3 and 5 are shifted downwards by 0.5 and 4 units, respectively, for clarity.
) in TFE and its mixtures. The points for 3 and 5 are shifted downwards by 0.5 and 4 units, respectively, for clarity.
 ), 2 (
), 2 (
 ), 4 (
), 4 (
 ) and 5 (
) and 5 (
 ) at 25,00 °C and of log k vs. YBr for the solvolyses of 3 (
) at 25,00 °C and of log k vs. YBr for the solvolyses of 3 (
 ) at 25,00 °C. The filled symbols correspond to the solvolyses of 1 (
) at 25,00 °C. The filled symbols correspond to the solvolyses of 1 (
 ), 2 (
), 2 (
 ), 3 (
), 3 (
 ), 4 (
), 4 (
 ) and 5 (
) and 5 (
 ) in TFE and its mixtures. The points for 3 and 5 are shifted downwards by 0.5 and 4 units, respectively, for clarity.
) in TFE and its mixtures. The points for 3 and 5 are shifted downwards by 0.5 and 4 units, respectively, for clarity.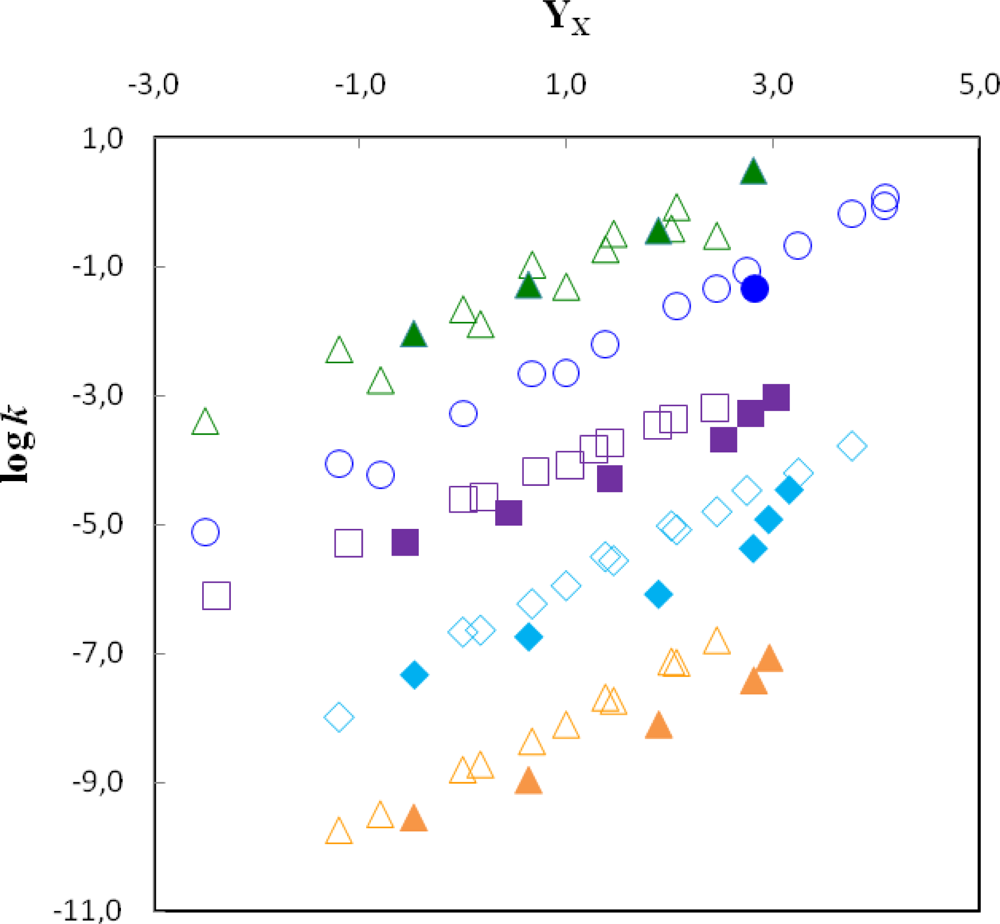
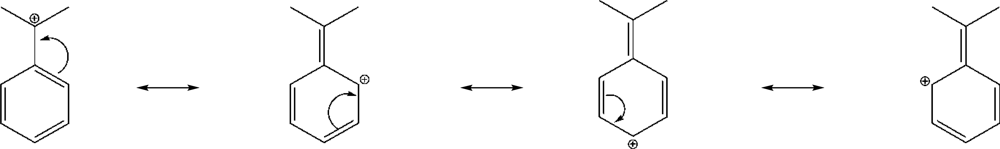
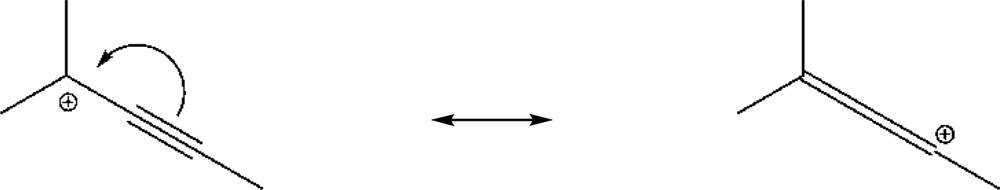
| Solventa | YBrb | YClb | NOTsb | Ic | log k | ||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 2d | 3 | 4e | 5f | |||||
| 80%Ac | −0.7 | −0.8 | −0.42 | −0.23 | — | −2.75g | — | −4.22 | −5.49 |
| 70%Ac | 0.2 | 0.17 | −0.42 | −0.29 | −6.63 | −1.88 | − 4.08 | — | −4.72 |
| 60%Ac | 1.03 | 1.00 | −0.41 | −0.28 | −5.94 | −1.30 | −3.58 | −2.64 | −4.10 |
| 40%Ac | 2.44 | 2.46 | −0.38 | −0.35 | −4.79 | −0.51 | −2.69 | −1.34 | −2.79 |
| 20%Ac | 3.66 | 3.77 | −0.38 | −0.40 | −3.77 | — | — | −0.17 | — |
| 100%Me | −1.12 | −1.20 | −0.04 | 0.41 | −7.98 | −2.27 | −4.78 | −4.06 | −5.74 |
| 80%Me | 0.70 | 0.67 | −0.05 | 0.14 | −6.22 | −0.96 | −3.68 | −2.66 | −4.36 |
| 70%Me | 1.42 | 1.46 | −0.08 | 0.05 | −5.55 | −0.48 | −3.24 | — | −3.73 |
| 60%Me | 2.04 | 2.07 | −0.13 | −0.02 | −5.07 | −0.07 | −2.86 | −1.61 | −3.14 |
| 40%Me | 3.14 | 3.25 | −0.21 | −0.13 | −4.19 | — | — | −0.67 | — |
| 20%Me | 3.94 | 4.10 | −0.35 | −0.26 | — | — | — | 0.08 | — |
| 100%Et | −2.40 | −2.50 | 0.00 | 0.20 | — | −3.38 | −5.60 | −5.12 | — |
| 80%Et | 0.00 | 0.00 | 0.00 | 0.00 | −6.66 | −1.65 | −4.11 | −3.27 | −4.80 |
| 60%Et | 1.26 | 1.38 | −0.08 | −0.15 | −5.49 | −0.72 | −3.33 | −2.20 | −3.68 |
| 50%Et | 1.88 | 2.02 | −0.09 | −0.20 | −5.01 | −0.40 | −2.96 | — | −3.12 |
| 40%Et | 2.62 | 2.75 | −0.23 | −0.24 | −4.46 | — | — | −1.06 | — |
| 20%Et | 3.92 | 4.09 | −0.34 | −0.33 | — | — | — | −0.04 | — |
| TFE | 2.53 | 2.81h | −3.07 | 0.37 | −5.37 | 0.50 | −3.18 | — | −3.42 |
| 97% TFE | 2.53 | 2.83 | −2.79 | 0.49 | — | — | — | −1.33 | — |
| 70% TFE | 2.79 | 2.96 | −1.2 | 0.25 | −4.92 | — | −2.78 | — | −3.07 |
| 50% TFE | 3.04 | 3.16 | −0.93 | 0.09 | −4.46 | — | −2.52 | — | — |
| 80TFE20Et | 1.42i | 1.89h | −1.72 | 0.52 | −6.08 | −0.43 | −3.79 | — | −4.10 |
| 60TFE40Et | 0.44i | 0.63h | −1.01 | 0.59 | −6.74 | −1.27 | −4.33 | — | −4.96 |
| 40TFE60Et | −0.57 | −0.48h | −0.55 | 0.43 | −7.33 | −2.03 | −4.79j | — | −5.54 |
| 97% HFIP | 4.51 | 5.08 | −4.27 | 0.73 | — | — | — | — | −1.88 |
| Water | 4.44 | 4.57 | −0.44 | −0.45 | −3.09 | — | — | — | — |
| Substrate | m±s(m) (%SL)a | l±s(l) (%SL)a | h±s(h) (%SL)a | c±s(c) (%SL)a | sdfitb | R2adjc | Fd | ne | eq. |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 0.79 ± 0.05
100% | — | — | −6.88 ± 0.11
100% | 0.315 | 0.934 | 271 | 1 | |
| 1 | 0.84 ± 0.02
100% | 0.39 ± 0.04
100% | — | −6.75 ± 0.05
100% | 0.129 | 0.989 | 854 | 20 | 2 |
| 1 | 0.70 ± 0.03
100% | — | −0.92 ± 0.13
100% | −6.71 ± 0.06 | 0.164 | 0.982 | 521 | 4 | |
| 1
| 0.78 ± 0.03 100% | 0.26 ± 0.04 100% | −0.45 ± 0.10 100% | −6.71 ± 0.03 100% | 0.088 | 0.995 | 1240 | 3 | |
| 2 | 0.69 ± 0.04
100% | — | — | −1.72 ± 0.07
100% | 0.264 | 0.936 | 219 | 1 | |
| 2 | 0.68 ± 0.05
100% | −0.04 ± 0.10
33% | — | −1.74 ± 0.08
100% | 0.272 | 0.932 | 104 | 16 | 2 |
| 2 | 0.71 ± 0.04 100% | — | 0.52 ± 0.18 99% | −1.78 ± 0.06 100% | 0.214 | 0.958 | 172 | 4 | |
| 2
| 0.75 ± 0.05
100% | 0.14±0.09
86% | 0.72 ± 0.21
99% | −1.75 ± 0.06
100% | 0.203
| 0.962
| 128
| 3 | |
| 3 | 0.57 ± 0.04
100% | — | — | −4.22 ± 0.06
100% | 0.217 | 0.936 | 236 | 1 | |
| 3 | 0.63 ± 0.02
100% | 0.26 ± 0.03
100% | — | −4.14 ± 0.03
100% | 0.094 | 0.988 | 654 | 17 | 2 |
| 3 | 0.54 ± 0.02
100% | — | −0.57 ± 0.12
100% | −4.15 ± 0.04
100% | 0.139 | 0.974 | 298 | 4 | |
| 3
| 0.60 ± 0.02 100% | 0.20 ± 0.04 100% | −0.24 ± 0.09 97% | −4.13 ± 0.03 100% | 0.080 | 0.991 | 607 | 3 | |
| 4 | 0.79 ± 0.02
100% | — | — | −3.28 ± 0.05
100% | 0.150 | 0.991 | 1622 | 1 | |
| 4 | 0.80 ± 0.02
100% | — | — | −3.27 ± 0.04
100% | 0.128 | 0.994 | 2173f | 1 | |
| 4 | 0.81 ± 0.02
100% | 0.15 ± 0.05
99% | — | −3.25 ± 0.04
100% | 0.116 | 0.995 | 1357 | 2 | |
| 4 | 0.82 ± 0.01
100% | 0.63 ± 0.20
99% | — | −3.17 ± 0.04
100% | 0.097 | 0.997 | 1920f | 15 | 2 |
| 4 | 0.79 ± 0.02
100% | — | −0.01 ± 0.17
6% | −3.28 ± 0.05
100% | 0.156 | 0.991 | 749 | 4 | |
| 4 | 0.85 ± 0.02 100% | — | 0.58 ± 0.15 100% | −3.27 ± 0.03 100% | 0.087 | 0.997 | 2356f | 4 | |
| 4 | 0.84 ± 0.02
100% | 0.25 ± 0.05
100% | 0.42 ± 0.12
99% | −3.24 ± 0.03
100% | 0.084 | 0.997 | 1709 | 3 | |
| 4
| 0.84 ± 0.05
100% | 0.24 ± 0.28
58% | 0.42 ± 0.24
89% | −3.24 ± 0.05
100% | 0.088
| 0.997
| 1532f | 3 | |
| 5 | 0.65 ±0.05
100% | — | — | −4.89 ± 0.10
100% | 0.324 | 0.910 | 163 | 1 | |
| 5 | 0.83 ±0.04
100% | 0.32 ± 0.05
100% | — | −4.85 ± 0.05
100% | 0.165 | 0.977 | 335 | 17 | 2 |
| 5 | 0.69 ± 0.03
100% | — | −0.79 ± 0.14
100% | −4.85 ± 0.06
100% | 0.180 | 0.972 | 279 | 4 | |
| 5 | 0.79 ± 0.03 100% | 0.20 ± 0.05 100% | −0.44 ± 0.13 100% | −4.84 ± 0.04 100% | 0.124 | 0.987 | 403 | 3 |
© 2008 by MDPI This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Reis, M.C.; Elvas-Leitão, R.; Martins, F. The Influence of Carbon-Carbon Multiple Bonds on the Solvolyses of Tertiary Alkyl Halides: a Grunwald-Winstein Analysis. Int. J. Mol. Sci. 2008, 9, 1704-1716. https://doi.org/10.3390/ijms9091704
Reis MC, Elvas-Leitão R, Martins F. The Influence of Carbon-Carbon Multiple Bonds on the Solvolyses of Tertiary Alkyl Halides: a Grunwald-Winstein Analysis. International Journal of Molecular Sciences. 2008; 9(9):1704-1716. https://doi.org/10.3390/ijms9091704
Chicago/Turabian StyleReis, Marina C., Ruben Elvas-Leitão, and Filomena Martins. 2008. "The Influence of Carbon-Carbon Multiple Bonds on the Solvolyses of Tertiary Alkyl Halides: a Grunwald-Winstein Analysis" International Journal of Molecular Sciences 9, no. 9: 1704-1716. https://doi.org/10.3390/ijms9091704




