Photodynamic Effect of Hypericin on the Conformation and Catalytic Activity of Hemoglobin
Abstract
:1. Introduction
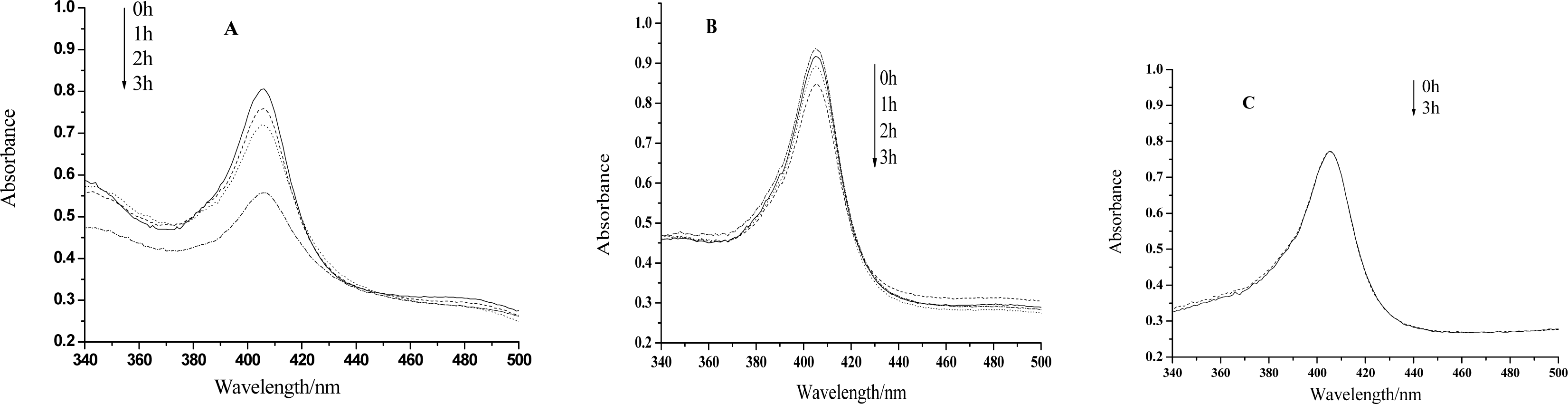
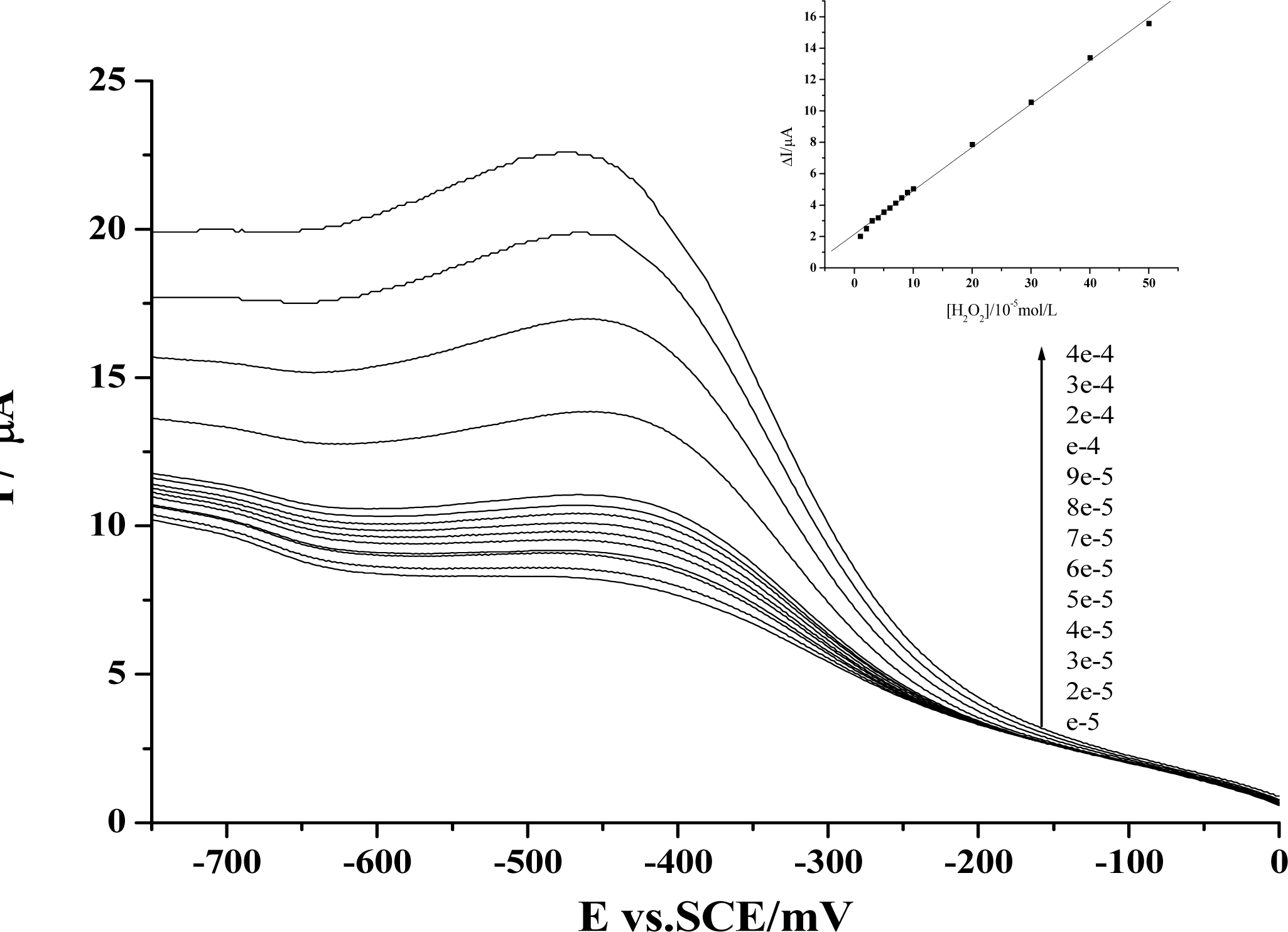
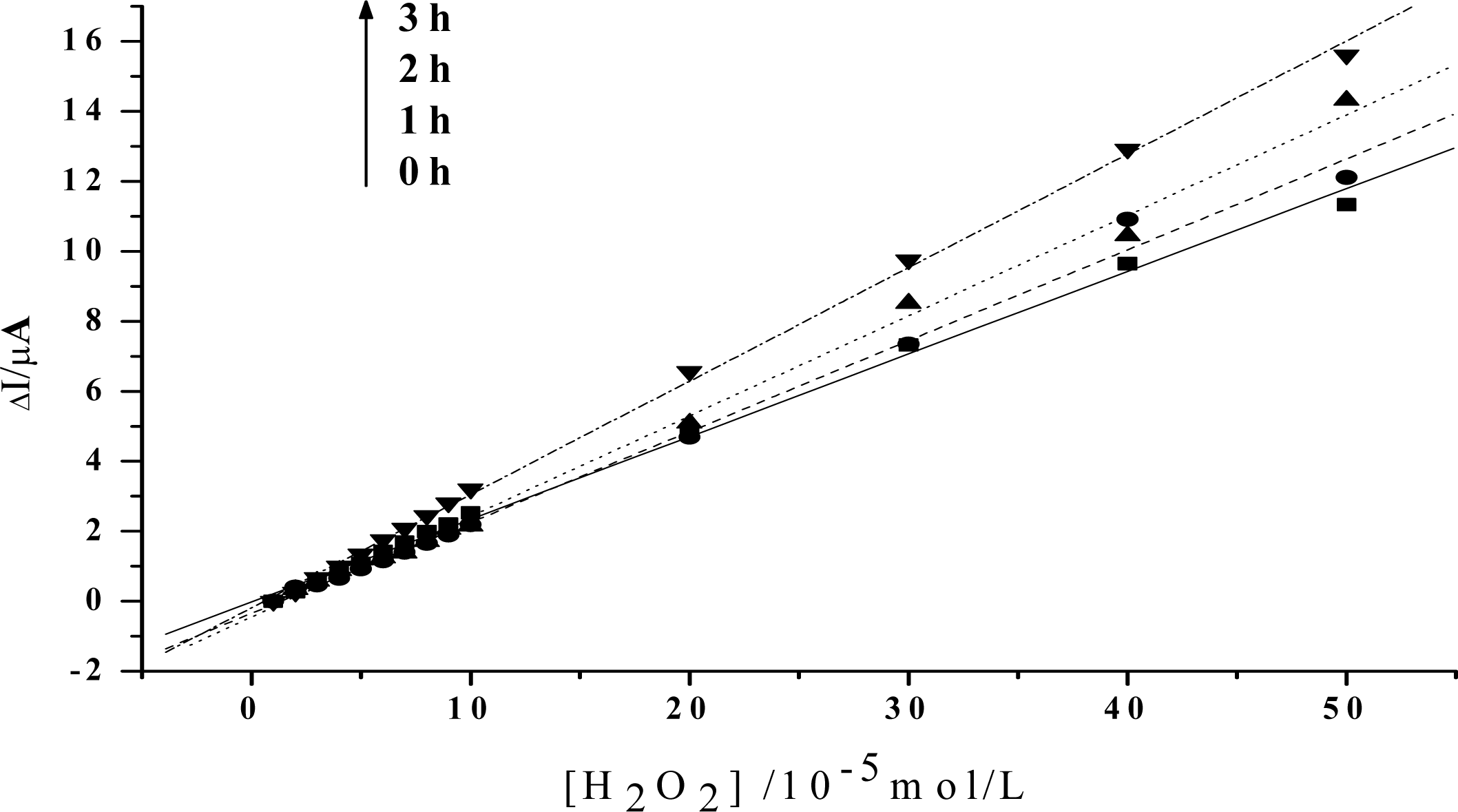
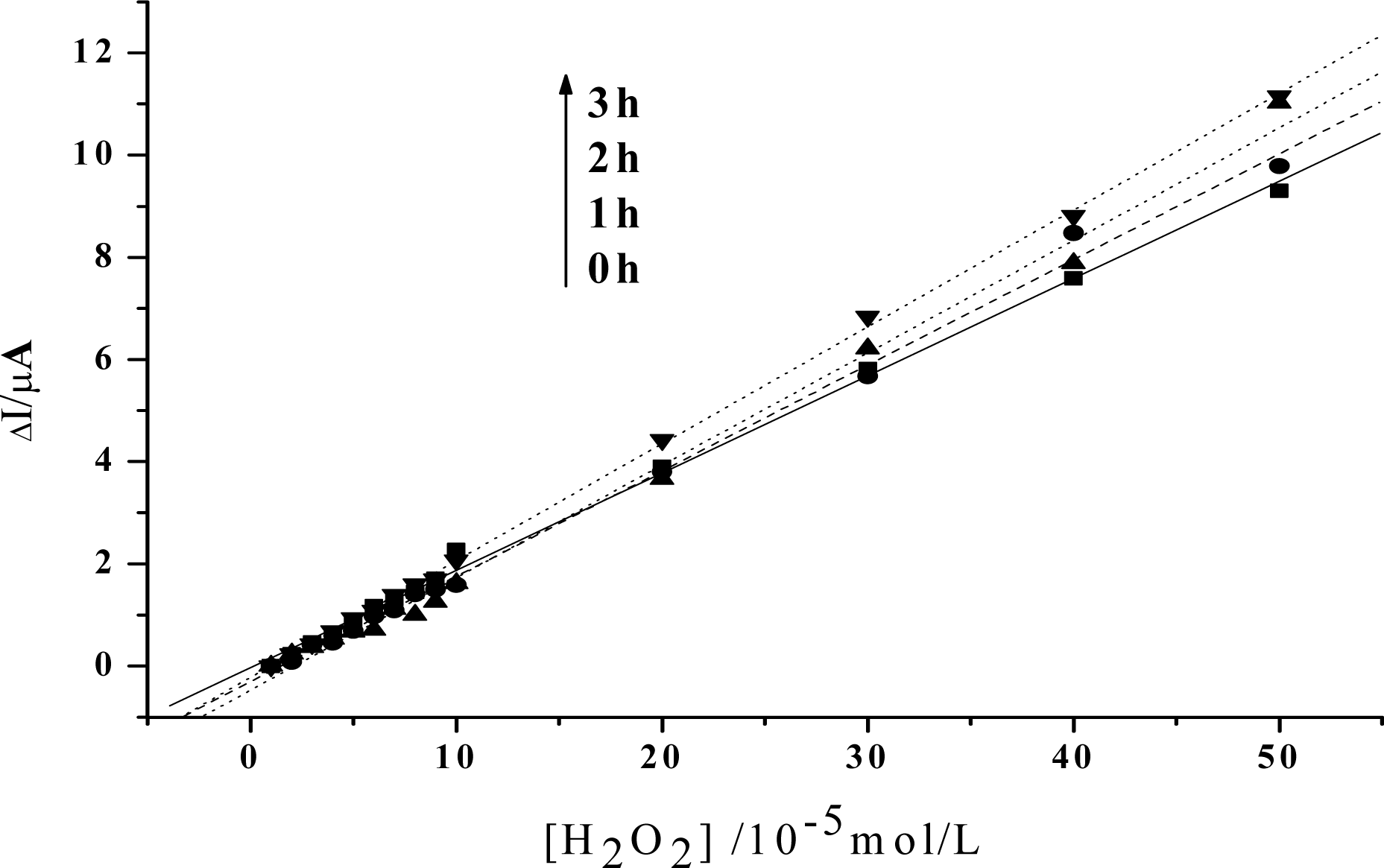
2. Results and Discussion
3. Experimental Section
3.1 Reagents
3.2 Preparation of UV-vis spectroscopic samples and light treatment
- Sample I preparation: 1μL HY solution (0.1 mol/L), 5μL Hb (8mg/ml, pH 7.0) and 4μL Tris-HCl buffer (pH 7.0) were mixed in a microcentrifuge tube.
- Sample II preparation: 0.5μL HY solution (0.1 mol/L), 5μL Hb (8mg/ml, pH 7.0) and 4μL Tris-HCl buffer (pH 7.0) were mixed in a microcentrifuge tube.
- Sample III preparation:1μL HY solution (0.1 mol/L), 5μL Hb (8mg/ml, pH 7.0) and 4μL Tris-HCl buffer (pH 7.0) were mixed in a microcentrifuge tube. This sample was thoroughly deoxygenated by blowing high-purity nitrogen for at least 1 min, and then sealed by parafilm.
3.3 Spectrocopic measurements
3.4 Preparation of the modified electrode
3.5 Electrochemical Apparatus
Acknowledgments
References and Notes
- Pace, N; MacKinney, G. Hypericin. The Photodynamic Pigment from St. John's Wort. J Am Chem Soc 1941, 63, 2570–2574. [Google Scholar]
- Bilia, AR; Gallori, S; Vincieri, FF. St. John's Wort and depression: efficacy, safety and tolerability – an update. Life Sci 2002, 70, 3077–3096. [Google Scholar]
- Greeson, JM; Sanford, B; Monti, DA. St. John's Wort (Hypericum perforatum):a review of the current pharmacological, toxicological, and clinical literaturel. Psychopharmacology (Berl) 2001, 153, 402–414. [Google Scholar]
- Barnes, J; Anderson, LA; Phillipson, JD. St John's Wort (Hypericum perforatum L.): a review of its chemistry, pharmacology and clinical properties. J Pharm Pharmacol 2001, 53, 583–600. [Google Scholar]
- Miskovsky, P; Sureau, F; Chinsky, L; Turpin, PY. Subcellular distribution of hypericin in human cancer cells. Photochem Photobiol 1995, 62, 546–549. [Google Scholar]
- Kamuhabwa, AR; Augustijns, P; De Witte, PA. In vitro transport and uptake of protohypericin and hypericin in the Caco-2 model. Int J Pharm 1999, 188, 81–86. [Google Scholar]
- Chen, B; de Witte, PA. Photodynamic therapy efficacy and tissue distribution of hypericin in a mouse P388 lymphoma tumor model. Cancer Lett 2000, 150, 111–117. [Google Scholar]
- Andreoni, A; Colasanti, A; Colasanti, P; Mastrocinque, M; Riccio, P; Roberti, G. Laser photosensitization of cells by hypericin. Photochem Photobiol 1994, 59, 529–533. [Google Scholar]
- Guedes, RC; Eriksson, LA. Theoretical study of hypericin. J Photochem Photobiol A-Chem 2005, 172, 293–299. [Google Scholar]
- Miskovsky, P. A New Antiviral and Antitumor Photosensitizer: Mechanism of Action and Interaction with Biological Macromolecules. Curr Drug Targets 2002, 3, 55–84. [Google Scholar]
- Wang, S; Liang, J; Yang, L; Cui, Y; Shang, R; Luo, Y. Studies on the inhibitory effect of hypericin on foot-and-mouth virus in vitro. Chin J Vet med 2006, 42, 6–9. [Google Scholar]
- Ulicny, J; Laaksonen, A. Hypericin, an intriguing internally heterogenous molecule, forms a covalent intramolecular hydrogen bond. Chem Phys Lett 2000, 319, 396–402. [Google Scholar]
- Hadjur, C; Richard, MJ; Parat, MO; Jardon, P; Favier, A. Photodynamic effects of hypericin on lipid peroxidation and antioxidant status in melanoma cells. Photochem Photobiol 1996, 64, 375–381. [Google Scholar]
- Theodossiou, T; Spiro, MD; Jacobson, J; Hothersall, JS; Mac-Robert, AJ. Evidence for intracellular aggregation of hypericin and the impact on its photocytotoxicity in PAM 212 murine keratinocytes. Photochem Photobiol 2004, 80, 438–443. [Google Scholar]
- Thomas, C; MacGill, RS; Miller, GC; Pardini, RS. Photoactivation of hypericin generates singlet oxygen in mitochondria and inhibits succinoxidase. Photochem Photobiol 1992, 55, 47–53. [Google Scholar]
- Thomas, C; Pardini, RS. Oxygen dependence of hypericininduced phototoxicity to EMT6 mouse mammary carcinoma cells. Photochem Photobiol 1992, 55, 831–837. [Google Scholar]
- Lu, WD; Atkins, WM. A Novel Antioxidant Role for Ligandin Behavior of Glutathione S-Transferases: Attenuation of the Photodynamic Effects of Hypericin. Biochemistry 2004, 43, 12761–12769. [Google Scholar]
- Theodossiou, TA; Noronha-Dutra, A; Hothersall, JS. Mitochondria are a primary target of hypericin phototoxicity:Synergy of intracellular calcium mobilisation in cell killing. Int J Biochem Cell B 2006, 38, 1946–1956. [Google Scholar]
- Wang, H; Guan, R; Fan, C; Zhu, D; Li, G. A hydrogen peroxide biosensor based on the bioelectrocatalysis of hemoglobin incorporated in a kieselgubr film. Sensors and Actuators B 2002, 84, 214–218. [Google Scholar]
- Fan, C; Gao, Q; Zhu, D; Wagner, G; Li, G. An unmediated hydrogen peroxide biosensor based on hemoglobin incorporated in a montmorillonite membrane. Analyst 2001, 126, 1086–1089. [Google Scholar]
- Zhou, H; Gan, X; Wang, J; Zhu, X; Li, G. Hemoglobin-Based Hydrogen Peroxide Biosensor Tuned by the Photovoltaic Effect of Nano Titanium Dioxide. Anal Chem 2005, 77, 6102–6104. [Google Scholar]
- Geoge, P; Hanania, G. A spectrophotometric study of ionizations in methaemoglobin. Biochem J 1953, 55, 236–243. [Google Scholar]
- Sakai, H; Masada, Y; Onuma, H; Takeoka, S; Tsuchida, E. Reduction of Methemoglobin via Electron Transfer from Photoreduced Flavin: Restoration of O2-Binding of Concentrated Hemoglobin Solution Coencapsulated in Phospholipid Vesicles. Bioconjugate Chem 2004, 15, 1037–1045. [Google Scholar]
- Sakai, H; Onuma, H; Umeyama, M; Takeoka, S; Tsuchida, E. Photoreduction of Methemoglobin by Irradiation in the Near-Ultraviolet Region. Biochemistry 2000, 39, 14595–14602. [Google Scholar]
- Teramura, Y; Kanazawa, H; Sakai, H; Takeoka, S; Tsuchida, E. Prolonged Oxygen-Carrying Ability of Hemoglobin Vesicles by Coencapsulation of Catalase in Vivo. Bioconjugate Chem 2003, 14, 1171–1176. [Google Scholar]
- Brown, WD; Snyder, HE. Nonenzymatic reduction and oxidation of myoglobin and hemoglobin by nicotinamide adenine dinucleotides and flavins. J Boil Chem 1969, 244, 6702–6706. [Google Scholar]
- Li, G. Encyclopedia of Sensor; Grimes, CA, Dickey, EC, Pishko, MV, Eds.; American Scientific Publishers: Valencia, CA, 2006; pp. 301–313. [Google Scholar]
- Sanchez-Cortes, S; Miskovsky, P; Jancura, D; Bertoluzza, A. Specific Interactions of Antiretroviraly Active Drug Hypericin with DNA As Studied by Surface-Enhanced Resonance Raman Spectroscopy. J Phys Chem 1996, 100, 1938–1944. [Google Scholar]


Share and Cite
Zhao, J.; Meng, W.; Miao, P.; Yu, Z.; Li, G. Photodynamic Effect of Hypericin on the Conformation and Catalytic Activity of Hemoglobin. Int. J. Mol. Sci. 2008, 9, 145-153. https://doi.org/10.3390/ijms9020145
Zhao J, Meng W, Miao P, Yu Z, Li G. Photodynamic Effect of Hypericin on the Conformation and Catalytic Activity of Hemoglobin. International Journal of Molecular Sciences. 2008; 9(2):145-153. https://doi.org/10.3390/ijms9020145
Chicago/Turabian StyleZhao, Jing, Wenying Meng, Peng Miao, Zhiguo Yu, and Genxi Li. 2008. "Photodynamic Effect of Hypericin on the Conformation and Catalytic Activity of Hemoglobin" International Journal of Molecular Sciences 9, no. 2: 145-153. https://doi.org/10.3390/ijms9020145



