1. Introduction
A paper based product typically contains 90–99% cellulose fibers which are the primary structural element and the most important component influencing end use properties. A network of self-bonding cellulose fibers within network structure affects chemical and physical characteristics of the paper products. However, the chemical structure of cellulose is now well established and consists β-anhydroglucose units with dominant hydroxyl groups, which are appropriate groups for reactions. These are mainly due to the one primary and two secondary hydroxyl groups in each monomer unit in polymer structure [
1,
2]. Each of anhydroglucose units are linked together by β-1-4 glycosidic bonds. Due to specific chemical arrangements of cellulose, it is completely linear homopolysacharide and has the ability to form extensive intra and intermolecular hydrogen bonds [
1].
Because, paper network is composed from randomly laid fibrous (cellulose) and non-fibrous (fillers) materials, it contains a complicated set of cavity pore channels with a variety of capillary dimensions. Hence, it is readily permeable to liquids. However, the structure of paper can be modified during contact of liquids because it disrupts hydrogen bonds, relaxes fibers, and produces dimensional changes in pores and capillaries. If the effect of external pressure is neglected, the Washburn equation could be used to describe the rate of capillary rise of fluids into paper.
Where dl/dt is the rate of penetration of the capillary, l is the distance penetrated into a cylindrical capillary of radius r, in time t, θ angle between liquid and surface, by a liquid with surface tension γ
lv and viscosity η [
3,
4].
A number of studies have been conducted on the reasonably explanation for paper-liquid interactions. However, most studies have shown that the chemical and physical properties of fibers (cellulose) are reduced upon rewetting. Consistently, the decrease of the absorption and strength properties of rewetted fibers because of changes in the basic structure during swelling and reducing fiber bonding potential [
5–
7]. It was speculated that during drying, cellulose lose both free and bound water and start to shrink. It has modified to hard and stiff state (hardening) that have not reswell properly compare to never dried state [
8–
10]. The mechanism of cellulose hardening is complicated and probably related with removal of bound water, lowers the distance between microfibrils and reducing the elasticity of fibrils. As a result of these modifications, the hydrogen bond cross-links could be formed between adjacent microfibrils in cellulose structure. This specific structure is usually called hornification of cellulose fibers [
11].
The hydrophilic natures of the cellulose within paper structure are very important for good fiber-fiber bonding. However, the challenging aspects for wetting of papers understand how to increase and control fibers wetting and bonding. It is believed that repeating wetting and drying brings about irreversible changes in cellulose bonding potential. Therefore, there is a need to understand more clearly the changes that take place in the rewetted cellulose fibers [
6–
9]. These may open the possibility to increase recycling of cellulose based products (e.g. paper and paperboards). Moreover, new modified processes, involve various type of organosolv pulping approaches can also be required more detailed information on the effect of solvent systems on cell wall chemical constituents especially on cellulose.
The objective of this study is to investigate the influence of various solvent formulations over repeating drying and wetting paper, composed only cellulose, in terms of evaluating chemical and physical properties. The experiments were conducted to ascertain the effect of various solvent formulations on liquid absorption, chemical and strength properties as well as capillary pore structure of papers.
2. Experimental Section
Whatman filter paper, composed only from cellulose fibers, has completely free of all additives and fillers, was utilized as paper source under laboratory conditions. The Whatman filter papers (no: 1) and the chemicals with a purity of 97–99%, used in this study were purchased from Yildiz Kimya Ltd (Ankara-Turkey) and utilized as received.
The solvent treatments are designed to evaluate liquid sorption capacity through chemical modification and pore size distribution in paper network after continuously repeating drying and wetting cycles. Four different solvents and their appropriate proportion mixtures were utilized in treatments: water (H
2O), ethanol (EtOH), methanol (MeOH), sodium hydroxide (NaOH). The constituents of solvent mixtures that paper samples treated at atmospheric conditions are presented in
Table 1.
Five repeating wetting-drying stages were carried out to evaluate some chemical and physical paper properties. The sheets were soaked in the solvents at the atmospheric conditions for 8 hours. At the end of each wetting stage, the sheets were dried in an oven at 105°C for 6 hours.
The sorption properties of the samples were determined in conformity with Tappi test method T 441. But longer time was used to calculate liquid sorption as well as strength properties of samples. The tensile strength and stretch properties were conducted with Lloyd-Lrk 5plus strength testing machine following Tappi standards T-494.
Simons stain technique was used for evaluating the capillary pore structure of sheets. This technique was based on the differences between the two type dyes (Direct Blue I and Fast Orange 15) molecular size and affinity for cellulose. The dyes were purchased from Pylam Products Co., Inc., Tepme, AZ, under the commercial names of Pontamine Fast Sky Blue 6BX and Pontamine Fast Orange 6RN, respectively. The dyes were used as received, without further purification. The 1 g Orange and Blue dye were dissolved in 100 ml distilled water then the dye solutions mixed in a ratio of 1:1 by volume. The small samples (1x1 cm) were first soaked in dye solutions for 1 min and then 1 min in water. After that, they were put on a slide and dried at 75°C, examined under a light microscope.
Direct Blue I has a defined chemical structure with a molecular diameter of 1 nm. Fast Orange 15 dye is a condensation product of 5-nitro-o-toluenesulfonic acid in aqueous alkali solution and its formula and structure are not well defined. However, It was proposed that the purified Fast Orange 15 dye has a molecular diameter in the range of 5–36 nm, which is much larger than the Direct Blue I molecular size [
13,
14].
A Shimadzu IR Prestige-21 series IR spectrophotometer was used to evaluate the chemical groups on the untreated and solvent treated papers. The data were collected in the range of 4000–400 cm−1.
3. Results and Discussion
Figure 1 shows liquid sorption properties of papers which are treated in water (A), (anhydrous) ethanol (B), (anhydrous) methanol (C), 1 N sodium hydroxide (D), up to five repeating drying and wetting stages. It can be revealed that the highest sorption occured with sample D (230.5% in 1
st cycle and 179.8% in 5
th cycle) followed by sample A (182.3% in 1
st cycle and 170.6% in 5
th cycle), sample B (109.4% in 1
st cycle and 97.6% in 5
th cycle), and sample C (101.5% in 1
st cycle and 84.3% in 5
th cycle), respectively. However, repeating drying and wetting usually affects on decreasing liquid absorption of sheets. This is not surprising because a number of researchers have already reported a definite correlation between chemical treatments and sorption properties for cellulosic materials [
6–
10].
These reductions can be attributed to a change in the fiber polymer structure, such as hydrogen bonds cross-links or the elimination of H-bonding sites for liquids. Because, all the liquids used in this study have hydrogen bonding ability and can penetrate into the paper network structure, followed by substitution of -OH for cellulose to cellulose hydrogen bonds. Moreover, the closure of larger pores which do not reopen upon rewetting can be affects of high surface tension forces. This subsequent strain-hardening mechanism and forces may be welded the pores and resist against liquid penetration some degree. These are in good agreement with the results reported by Wistara (1999) that dried fibers with higher initial swelling capacity have lower capability to reswell [
6].
However, 1 N sodium hydroxide treated sample (D) has considerably higher absorption value than others. After 5
th repeating wetting-drying cycle, the sample D shows approximately 6% higher absorption from sample A, 85% from sample B and 114% from sample C, respectively (
Figure 1). It can be attributed to reorientation of microfibrils and a better alignment of cellulose chains in sheet network structure that promote additional H-bonding sites for liquid. Das and Chakraborty (2006) was found that during alkali treatment, a lattice transformation from cellulose-I to cellulose-II took place and sodium hydroxide introduces considerable changes in crystallinity and orientation angle within cellulose structure [
16]. These can be affects less crystalline region in cellulose hence further H-bonding place for liquids.
The marked effect of sodium hydroxide addition to ethanol (B) in terms of sorption of paper (E, F, and G) is clearly evident in
Figure 2. The highest absorption was obtained with sample F (EtOH-NaOH 1:3 by vol) after 1
st wetting-drying cycle (185.7%) and further treatments decresing affects on liquid sorption of papers but they are still considerably higher than only EtOH and lower amount NaOH including systems. Sample F showed approximately 40–70% higher liquid absorption compare to sample B (109.4%) at similar wetting-drying stages (
Figure 2). It seemed as if the increase of the liquid absorption was closely related to NaOH content in EtOH system and inversely related to wetting-drying stage.
The sorption properties of papers at proportional mixture of methanol and sodium hydroxide reaction conditions are shown in
Figure 3. Similar to ethanol-alkali system, the highest absorption of 162.3% was also found with sample I (MeOH-NaOH 1:3 by vol) but after 5
th wetting-drying stage. One may also observe that extended drying-wetting usually affects 2–10% increased liquid absorption properties for sample I. However, it can be realized that the liquid absorption was positively correlated with sodium hydroxide content in methanol system.
These comparisons between the treatments and the measured results reveal that the sodium hydroxide response of cellulose can be predicted quite well by proportional mixture with ethanol (
Figure 2) and methanol (
Figure 3).
It was reported by Rowell that the interaction between cellulosics and organic liquids was mainly influenced by the solvent pH, the molar size, and the hydrogen bonding capacity of the liquid [
15]. However, the variable sorption properties of sheets that treated with mixtures of ethanol and methanol with alkali, at similar conditions clearly reveals that the liquid absorption into paper sheet structure closely related to nature of the liquid (pH) and its surface tension as revealed in
Figures 1–
3. Moreover, it has already proposed that swelling and sorption properties of papers in alkali conditions were higher than acidic conditions [
6–
9]. This is probably related to fibril openings, which is influences the swelling capacity of cellulose, is an important factor to improve absorption potential of the fibers.
Watanabe and his group speculated that approximately 3–7 wt % of adsorbed water was responsible for the stabilization of the H-bond network at the cellulose-water surface [
17]. But, all the solvent systems used in this study have hydrogen bonding potential and they can penetrate into the web structure, followed by substitution of -OH for cellulose to cellulose hydrogen bonds. Hence, inter and intrachain H-bonds of cellulose can be formed by solvent molecules during absorption.
Tensile strength of papers have usually used as an indication of the treatment affects on the paper strength derived from factors such as fiber strength, and bonding. The comparative tensile strengths of ten different solvent (A-J) treated papers with comparison to control that have strength of 37.3 N/mm are shown in
Figure 4. All treatment conditions apparently bring about an effect of decreased strength for papers. After 1
st wetting-drying stage, the highest strength loss was observed with sample A (16.8 N/mm, –54.9%) followed by sample B (18.0 N/mm, –51.3%), sample F (18.6 N/mm, –50%), respectively. Generally, continuously wetting-drying favors further strength loss of papers. However, the lowest strength found with sample B and D (13.0 N/mm, –65%) after 5
th repeating wetting-drying stage. It is apparent that the treatment conditions were too severe, thus the strength properties of the papers sharply decreased. Moreover, the addition of alkali to both ethanol and methanol (E-J) seems to have less affect on strength loss on papers.
The porous structure of the sheets has a major influence on the penetration of a liquid into network. When paper contact with liquids, they tend to move from large cavities into smaller ones according to their different capillary pressures. However, solvents may quickly penetrate into fiber and affect the cellulose degradation. It was proposed by Atalla that the dehydration process of cellulose promote molecular mobility could result in enhancing levels of molecular organization. However, this molecular organization limits the capacity of fibers to respond to stress in an elastic mode without failure [
12]. The strength results found for cellulose paper in this study consisted with this information.
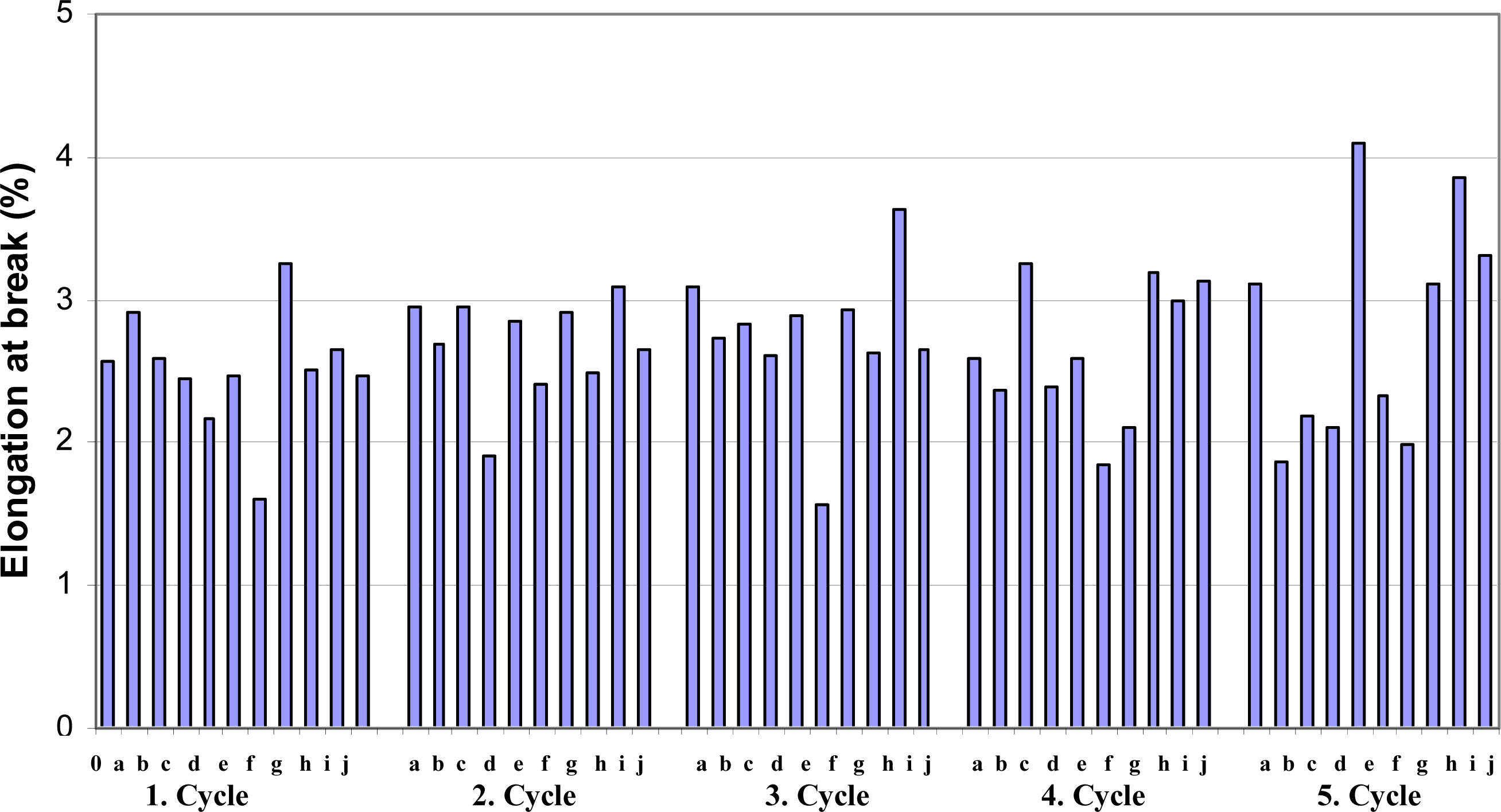
When evaluating the tensile strength, also the stretch of paper at break (elongation %) should be considered. It is indicative of the ability of paper to conform to a desired property under nonuniform tensile stress. The comparative stretch (as reported, elongation at break %) of samples (A-J) are shown in
Figure 5. In general, stretch properties of sheets usually behave contrary to tensile strength. It appears that repeated wetting-drying some level affects stretch in either increase and of decrease depends on solvent type and treatment stage. This is somewhat surprising, considering that the strength properties are considerably decreased with all treatments conditions.
After 1st wetting-drying stage, the highest stretch improvement was found with sample G (26.8%) followed by with sample A (13.2%), and marginally similar stretch improvements with samples B, C, H, I and J (4–8%), in that order. However, after 5th wetting-drying stage, some papers shows considerably higher stretch than untreated and and/or lower treated samples. The highest stretch, hence, improvement was observed with sample E (59.5%) followed by sample I (49.8%), sample J (28.7%), sample H (21%), respectively. Moreover, samples B, C, D and F show approximately 18–27% stretch loss compare to control.
The strength developments as well as physical behavior of papers are very complicated and depend on many factors such as structure of cellulose especially on crystalline region, and H-bonding of randomly laid network elements. But in brief, it is apparent that the strength properties of sheets considerably decrease with liquid treatments especially in acidic environment (B and C) but the addition of alkali (NaOH) to both ethanol and methanol solvents seems to have less effect on strength loss, even some level improvement on stretch of papers realized in various conditions.
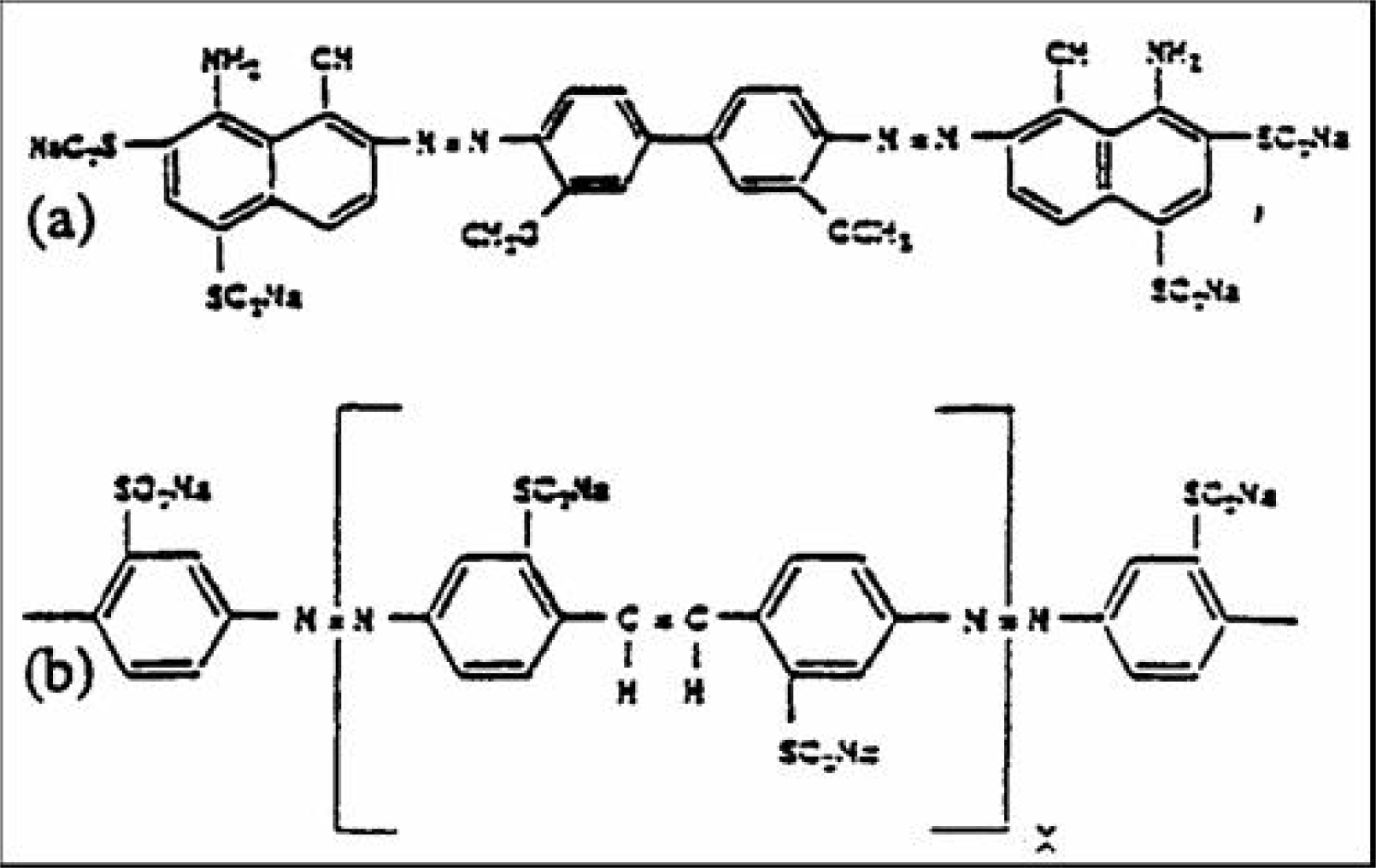
Simons stain is a practical technique for evaluating the pore structure of cellulose fibers, especially in recycling [
6,
9] and pulping processes [
18]. It can provide additional evidence on modification in cellulose macromolecule. When a mixture of two dyes (Direct Blue I and Fast Orange 15) is applied to fibers, the blue and/or orange dye can absorbs to fibers with depending pore size. For fibers with a various pore size distribution range, the color of the stained fiber can depend on the ratio of surface area accessible to both dyes. The chemical structures of both dyes are shown in
Figure 6 [
13,
14].
Table 2 shows a summary of the paper samples obtained from various conditions and their staining properties under light microscope. It is realized that papers obtained from different conditions showed variable intensities of blue and orange stain. However, this rating does not reflect any quantitative measurements and that the comparison should to be made only within each experimental set.
After 1
st wetting-drying stage, except sample B, other single solvent treated samples (A, C, D) shows intensive light blue-orange color. Moreover, paper samples that treated with proportional mixtures of EtOH, MeOH and NaOH (E-J) have various level intermediate orange to dark orange color. This is clear evidence that alkali addition to acidic condition effects on increase pore size of fibers. Because Direct Blue I has a smaller molecular size (approx. 1 nm) and a weaker affinity for cellulose compared to Direct Orange 15, which has a larger molecular size (5–36 nm) and a stronger affinity. But after 5
th wetting-drying stage, most of the samples stained dark blue to light blue-orange color with the exception of sample F (EtOH-NaOH, 1:3 by vol) which the fibers stains intermediate orange and slightly dark orange. This is clear evidence that increasing alkali concentration in acidic solvents may be effects restoration of fiber pore size and effects less damaging with continuous drying and wetting. It has already stated above that, Direct Orange 15 molecules can preferentially be adsorbed on the fiber surface where the pore size is appropriate (e.g. > 5 nm) for accessing Direct Orange 15 molecules. For the small capillary pores (e.g. < 5 nm), where the Direct Orange 15 molecules are not accessible, the Direct Blue I molecules can be adsorbed. Therefore, the ratio of adsorbed Direct Orange 15 to adsorbed Direct Blue I is anticipated to serve as an indicator of the pore structure, specifically, the pore size population distribution of the paper. Simons stain results are usually correlated with strength and sorption properties of papers (
Figures 1–
5).
The FTIR spectra of various level solvent treated paper samples were obtained in range of 400–4000 cm
−1. However, the characteristic spectrum of cellulose structure concentrate in the range of 800–2000 cm
−1 and the major peaks in this range had been identified. The comparative FTIR spectra of various solvent treated papers after 5
th repeating drying-wetting stage are shown in
Figure 7 (a–g).
All spectra exhibit multi-modal absorption in the 600–1000 cm
−1 region due to –OH groups in cellulose. The out-of-plane C-H vibration was assigned at 750 cm
−1. The band at 900–1150 cm
−1 is attributed to C-C out of the plane stretching, C-C-O stretching at 1060 cm
−1; C-O-C symmetric stretching 1150 cm
−1 (
Figure 7a). However, the spectra of solvent treated samples (
Figure 7d–f), exhibit the more absorption in the 800–1200 cm
−1 region relative to the untreated cellulose, where a more complex in-plane OH vibration is dominant. However, the less intense CH
2–CH
2- vibrations (1450–1700 cm
−1) and C-C and C-O-C peak areas (
Figure 7c, d, g) indicates the generation of new surface chemistry which is related to the solvent induced/dried chain modification. Young (1994) and Weise and Paulapuro (1996) found that drying affects lowering distance between microfibrils and cross-links in cellulose structure [
8,
10]. This modification can be effects level of H-bonds in cellulose. The result found with FTIR evaluation of cellulose sheets that treated with various solvents and continuously wetted-dried cycles, support this information. On the basis of FTIR measurements it was concluded that some modification had taken place during solvent diffusion and solvent induced chain modification occurred in cellulose structure.