Predicting Aqueous Solubility of Chlorinated Hydrocarbons by the MCI Approach
Abstract
:1. Introduction
2. Data Set
3. Methods

| Group | δ | δv | Group | δ | δv |
|---|---|---|---|---|---|
| -CH3 | 1 | 1 | =CH2 | 1 | 2 |
| -CH2- | 2 | 2 | =CH- | 2 | 3 |
| >CH- | 3 | 3 | =C< | 3 | 4 |
| >C< | 4 | 4 | -Cl | 1 | 7/9 |












4. Results and Discussion
| n | Descriptor | Coefficient | t-test |
|---|---|---|---|
| 0 | Intercept | 0.3556 | |
| 1 | 0χ | -0.4373 | -7.7127 |
| 2 | 3χc | -3.0857 | -4.7889 |
| 3 | 3χcv | 1.9657 | 4.2359 |

| No | CAS No. | Name | Experimental | Delgado | This work |
|---|---|---|---|---|---|
| 1 | 75-09-2 | Dichloromethane | -0.74 | -0.77 | -0.83 |
| 2 | 67-66-3 | Trichloromethane | -1.19 | -1.32 | -1.34 |
| 3 | 56-23-5 | Tetrachloromethane | -2.26 | -1.20 | -2.05 |
| 4 | 79-34-5 | 1,1,2,2-Tetrachloroethane | -1.76 | -2.42 | -2.27 |
| 5 | 540-59-0 | 1,2-Dichloroethene | -1.07 | -1.54 | -1.14 |
| 6 | 79-01-6 | Trichloroethene | -2.04 | -2.05 | -2.05 |
| 7 | 127-18-4 | Tetrachloroethene | -2.57 | -2.51 | -2.69 |
| Benzene | |||||
| 8 | 108-90-7 | Monochloro | -2.42 | -2.46 | -2.40 |
| 9 | 541-73-1 | 1,3-Dichloro | -3.04 | -3.31 | -3.30 |
| 10 | 95-50-1 | 1,2-Dichloro | -3.02 | -3.17 | -3.07 |
| 11 | 106-46-7 | 1,4-Dichloro | -3.31 | -3.33 | -3.30 |
| 12 | 120-82-1 | 1,2,4-Trichloro | -3.64 | -4.11 | -3.97 |
| 13 | 87-61-6 | 1,2,3-Trichloro | -4.08 | -3.94 | -3.77 |
| 14 | 108-70-3 | 1,3,5-Trichloro | -4.55 | -4.26 | -4.20 |
| 15 | 634-66-2 | 1,2,3,4-Tetrachloro | -4.38 | -4.72 | -4.46 |
| 16 | 95-94-3 | 1,2,4,5-Tetrachloro | -5.19 | -4.94 | -4.64 |
| 17 | 634-90-2 | 1,2,3,5-Tetrachloro | -4.73 | -4.94 | -4.67 |
| 18 | 608-93-5 | Pentachloro | -5.37 | -5.56 | -5.16 |
| Dibenzo-p-dioxin | |||||
| 19 | 39227-53-7 | 1-Chloro | -5.72 | -5.66 | -6.05 |
| 20 | 39227-54-8 | 2-Chloro | -5.86 | -5.92 | -6.24 |
| 21 | 29446-15-9 | 2,3-Dichloro | -7.23 | -6.83 | -6.91 |
| 22 | 33857-26-0 | 2,7-Dichloro | -7.83 | -7.02 | -7.14 |
| 23 | 39227-58-2 | 1,2,4-Trichloro | -7.53 | -7.50 | -8.11 |
| 24 | 30746-58-8 | 1,2,3,4-Tetrachloro | -8.77 | -8.08 | -8.63 |
| Biphenyl | |||||
| 25 | 2051-60-7 | 2-Chloro | -4.63 | -4.55 | -4.66 |
| 26 | 2051-61-8 | 3-Chloro | -4.88 | -5.38 | -4.84 |
| 27 | 2051-62-9 | 4-Chloro | -5.25 | -5.35 | -4.84 |
| 28 | 2050-68-2 | 4,4’-Dichloro | -6.63 | -6.05 | -5.74 |
| 29 | 34883-39-1 | 2,5-Dichloro | -5.27 | -5.52 | -5.56 |
| 30 | 33284-50-3 | 2,4-Dichloro | -5.29 | -6.28 | -5.56 |
| 31 | 33146-45-1 | 2,6-Dichloro | -5.07 | -4.86 | -5.38 |
| 32 | 2050-68-2 | 2,4’-Dichloro | -5.60 | -5.57 | -5.56 |
| 33 | 13029-08-8 | 2,2’-Dichloro | -5.36 | -4.76 | -5.37 |
| 34 | 37680-65-2 | 2,2’,5-Trichloro | -5.65 | -5.60 | -6.27 |
| 35 | 35693-92-6 | 2,4,6-Trichloro | -6.07 | -5.93 | -6.28 |
| 36 | 15862-07-4 | 2,4,5-Trichloro | -6.27 | -6.47 | -6.23 |
| 37 | 32598-13-3 | 3,3’,4,4’-Tetrachloro | -8.68 | -7.90 | -7.09 |
| 38 | 35693-99-3 | 2,2’,5,5’-Tetrachloro | -6.44 | -6.33 | -7.17 |
| 39 | 33284-53-6 | 2,3,4,5-Tetrachloro | -7.26 | -7.04 | -6.74 |
| 40 | 18259-05-7 | 2,3,4,5,6-Pentachloro | -7.78 | -8.40 | -7.29 |
| 41 | 37680-73-2 | 2,2’,4,5,5’-Pentachloro | -7.44 | -7.53 | -7.84 |
| 42 | 55312-69-1 | 2,2’,3,4,5-Pentachloro | -7.10 | -7.21 | -7.46 |
| 43 | 74472-44-9 | 2,3,3’,4’,5,6-Hexachloro | -7.83 | -8.40 | -8.35 |
| 44 | 55215-18-4 | 2,2’,3,3’,4,5-Hexachloro | -8.04 | -9.30 | -8.15 |
| 45 | 33979-03-2 | 2,2’,4,4’,6,6’-Hexachloro | -8.48 | -8.26 | -8.63 |
| 46 | 35065-27-1 | 2,2’,4,4’,5,5’-Hexachloro | -8.57 | -8.61 | -8.51 |
| 47 | 38411-22-2 | 2,2’,3,3’,6,6’-Hexachloro | -7.86 | -7.27 | -8.22 |
| 48 | 38380-07-3 | 2,2’,3,3’,4,4’-Hexachloro | -9.00 | -8.53 | -8.15 |
| 49 | 2136-99-4 | 2,2’,3,3’,5,5’,6,6’-Octachloro | -9.30 | -9.45 | -9.61 |
| 50 | 40186-72-9 | 2,2’,3,3’,4,4’,5,5’,6-Nonachloro | -9.93 | -9.88 | -10.09 |
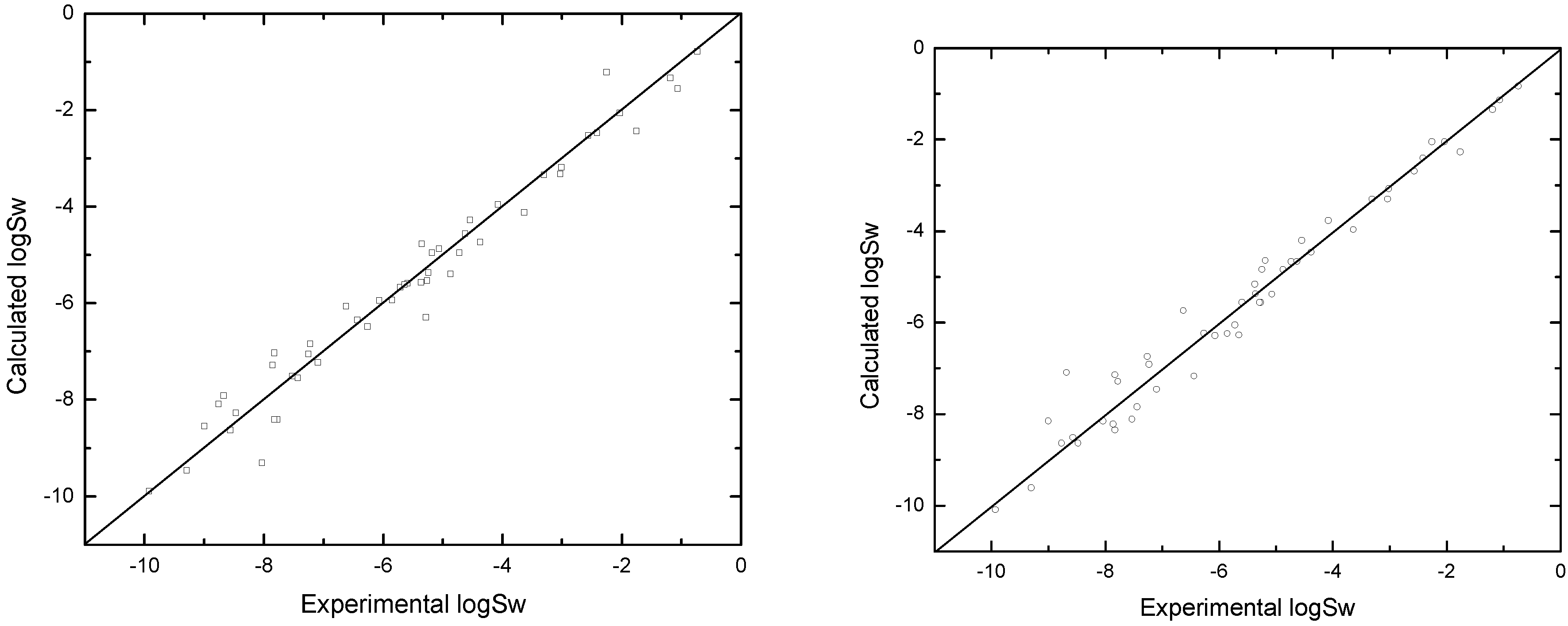
| No. | CAS No. | Name | Experi-mental | Calcula-ted | Residual |
|---|---|---|---|---|---|
| 1 | 58-89-9 | Lindane | -4.59 | -4.77 | 0.18 |
| 2 | 16606-02-3 | 2,4',5-PCB | -6.25 | -6.46 | 0.21 |
| 3 | 31508-00-6 | 2,3',4,4',5-Pentachlorobiphenyl | -7.39 | -7.80 | 0.41 |
| 4 | 32598-11-1 | 2,3',4',5-Tetrachlorobiphenyl | -7.25 | -7.13 | -0.12 |
| 5 | 35065-28-2 | 2,2',3,4,4',5'-Hexachlorobiphenyl | -8.32 | -8.33 | 0.01 |
| 6 | 35694-08-7 | 2,2',3,3',4,4',5,5'-octachlorobiphenyl | -9.16 | -9.54 | 0.38 |
| 7 | 38380-02-8 | 2,2',3,4,5'-Pentachlorodiphenyl | -7.91 | -7.66 | -0.25 |
| 8 | 38380-08-4 | 2,3,3',4,4',5-Hexachlorobiphenyl | -7.82 | -8.32 | 0.50 |
| 9 | 38444-85-8 | 2,3,4'-Trichlorobiphenyl | -6.26 | -6.25 | -0.01 |
| 10 | 41464-39-5 | 2,2',3,5'-Tetrachlorobiphenyl | -6.47 | -6.97 | 0.50 |
| 11 | 52663-63-5 | 2,2',3,5,5',6-Hexachlorobiphenyl | -7.42 | -8.39 | 0.97 |
| 12 | 52663-69-1 | 2,2',3,4,4',5',6-Heptachlorobiphenyl | -7.92 | -9.06 | 1.14 |
| 13 | 52663-77-1 | 2,2',3,3',4,5,5',6,6'-Nonachlorobiphenyl | -10.40 | -10.12 | -0.28 |
| 14 | 52712-04-6 | 2,2',3,4,5,5'-Hexachlorobiphenyl | -7.68 | -8.36 | 0.68 |
| 15 | 52712-05-7 | 2,2',3,4,5,5',6-Heptachlorobiphenyl | -8.94 | -8.90 | -0.04 |
| 16 | 55215-17-3 | 2,2',3,4,6-Pentachlorobiphenyl | -7.43 | -7.49 | 0.06 |
| 17 | 55702-45-9 | 2,3,6-Trichlorobiphenyl | -6.29 | -6.08 | -0.21 |
| 18 | 56558-16-8 | 2,2',4,6,6'-Petachlorobiphenyl | -7.32 | -7.73 | 0.41 |
| 19 | 74472-42-7 | 2,3,3',4,4',6-Hexachlorobiphenyl | -7.66 | -8.35 | 0.69 |
| 20 | 75-09-2 | dichloromethane | -0.63 | -0.83 | 0.20 |
| 21 | 67-66-3 | trichloromethane | -1.17 | -1.34 | 0.17 |
| 22 | 56-23-5 | tetrachloromethane | -2.31 | -2.05 | -0.26 |
| 23 | 75-34-3 | 1,1-dichloroethane | -1.29 | -1.53 | 0.24 |
| 24 | 107-06-2 | 1,2-dichloroethane | -1.06 | -1.14 | 0.08 |
| 25 | 71-55-6 | 1,1,1-trichlorcethane | -2.00 | -2.56 | 0.56 |
| 26 | 79-00-5 | 1,1,2-trichlorcethane | -1.48 | -1.75 | 0.27 |
| 27 | 79-34-5 | 1,1,2,2-tetrachlorcethane | -1.74 | -2.27 | 0.53 |
| 28 | 630-20-6 | 1,1,1,2-tetrachlorcethane | -2.18 | -2.62 | 0.44 |
| 29 | 76-01-7 | pentachlorcethane | -2.60 | -3.06 | 0.46 |
| 30 | 2050-67-1 | 3,3'-PCB | -5.80 | -5.74 | -0.06 |
| 31 | 38444-81-4 | 2,3',5-Trichlorobiphenyl | -6.01 | -6.46 | 0.45 |
| 32 | 7012-37-5 | 2,4,4'-PCB | -6.21 | -6.46 | 0.25 |
| 33 | 38444-86-9 | 2',3,4-Trichlorodiphenyl | -6.29 | -6.23 | -0.06 |
| 34 | 2437-79-8 | 2,2',4,4'-Tetrachlorobiphenyl | -6.51 | -7.17 | 0.66 |
| 35 | 41464-41-9 | 2,2',5,6'-Tetrachlorobiphenyl | -6.80 | -7.00 | 0.20 |
| 36 | 52663-62-4 | 2,2',3,3',4-Pentachlorobiphenyl | -7.05 | -7.46 | 0.41 |
| 37 | 52663-71-5 | 2,2',3,3',4,4',6-Heptachlorobiphenyl | -8.30 | -8.88 | 0.58 |
| 38 | 2051-24-3 | 2,2',3,3',4,4',5,5',6,6'-Decachloro-1,1'-biphenyl | -11.62 | -11.27 | -0.35 |
| 39 | 67-72-1 | hexachloroethane | -3.67 | -3.76 | 0.09 |
| 40 | 540-54-5 | 1-chloropropane | -1.47 | -1.14 | -0.33 |
| 41 | 75-29-6 | 2-chloropropane | -1.41 | -1.70 | 0.29 |
| 42 | 142-28-9 | 1,3-dichloropropane | -1.62 | -1.45 | -0.17 |
| 43 | 78-87-5 | 1,2-dichloropropane | -1.60 | -1.87 | 0.27 |
| 44 | 96-18-4 | 1,2,3-trichloropropane | -1.92 | -2.07 | 0.15 |
| 45 | 109-69-3 | 1-chlorobutane | -2.03 | -1.45 | -0.58 |
| 46 | 78-86-4 | 2-chlorobutane | -1.96 | -1.87 | -0.09 |
| 47 | 513-36-0 | 1-chloro-2-methylpropane | -2.00 | -1.98 | -0.02 |
| 48 | 541-33-3 | 1,1-dichlorobutane | -2.40 | -2.06 | -0.34 |
| 49 | 7581-97-7 | 2,3-dichlombulane | -2.70 | -2.47 | -0.23 |
| 50 | 543-59-9 | 1-chloropentane | -2.73 | -1.76 | -0.97 |
| 51 | 625-29-6 | 2-chloropenlane | -2.63 | -2.18 | -0.45 |
| 52 | 616-20-6 | 3-chloropentane | -2.63 | -2.07 | -0.56 |
| 53 | 594-36-5 | 2-chloro-2-methylbutane | -2.51 | -3.35 | 0.84 |
| 54 | 544-10-5 | 1-chlorohexane | -3.12 | -2.07 | -1.05 |
| 55 | 319-86-8 | δ-hexachlorocyclohexane | -4.51 | -4.77 | 0.26 |
| 56 | 75-35-4 | 1,1-dichlorcethylene | -1.64 | -2.10 | 0.46 |
| 57 | 156-59-2 | 1,2-dichlorcethylene | -1.30 | -1.14 | -0.16 |
| 58 | 107-05-1 | 3-chloropropylene | -1.36 | -1.14 | -0.22 |
| 59 | 87-68-3 | hexachloro-1,3-butadiene | -4.92 | -4.70 | -0.22 |
| 60 | 77-47-4 | hexachlorocyclo-pentadiene | -5.18 | -5.59 | 0.41 |
| 61 | 95-49-8 | 2-chlorotoluene | -3.52 | -3.11 | -0.41 |
| 62 | 100-44-7 | alpha-chlorotoluene | -2.39 | -2.59 | 0.20 |
| 63 | 106-43-4 | p-chlorotoluene | -3.08 | -3.34 | 0.26 |
| 64 | 38444-93-8 | 2,2',3,3'-PCB | -7.28 | -6.76 | -0.52 |
| 65 | 32598-10-0 | 2,3',4,4'-PCB | -7.80 | -7.13 | -0.67 |
| 66 | 41464-40-8 | 2,2',4,5'-PCB | -6.57 | -7.17 | 0.60 |
| 67 | 15968-05-5 | 2,2',6,6'-PCB | -8.03 | -6.83 | -1.20 |
| 68 | 52704-70-8 | 2,2',3,3',5,6-PCB | -8.60 | -8.19 | -0.41 |
| 69 | 72-54-8 | DDD | -7.20 | -7.44 | 0.24 |
| 70 | 50-29-3 | DDT | -7.15 | -8.22 | 1.07 |
| 71 | 72-55-9 | DDE | -6.90 | -7.74 | 0.84 |
| 72 | 91-58-7 | 2-chloronaphthalme | -4.14 | -4.22 | 0.08 |
| 73 | 90-13-1 | 1-chloronaphthalene | -3.93 | -4.04 | 0.11 |
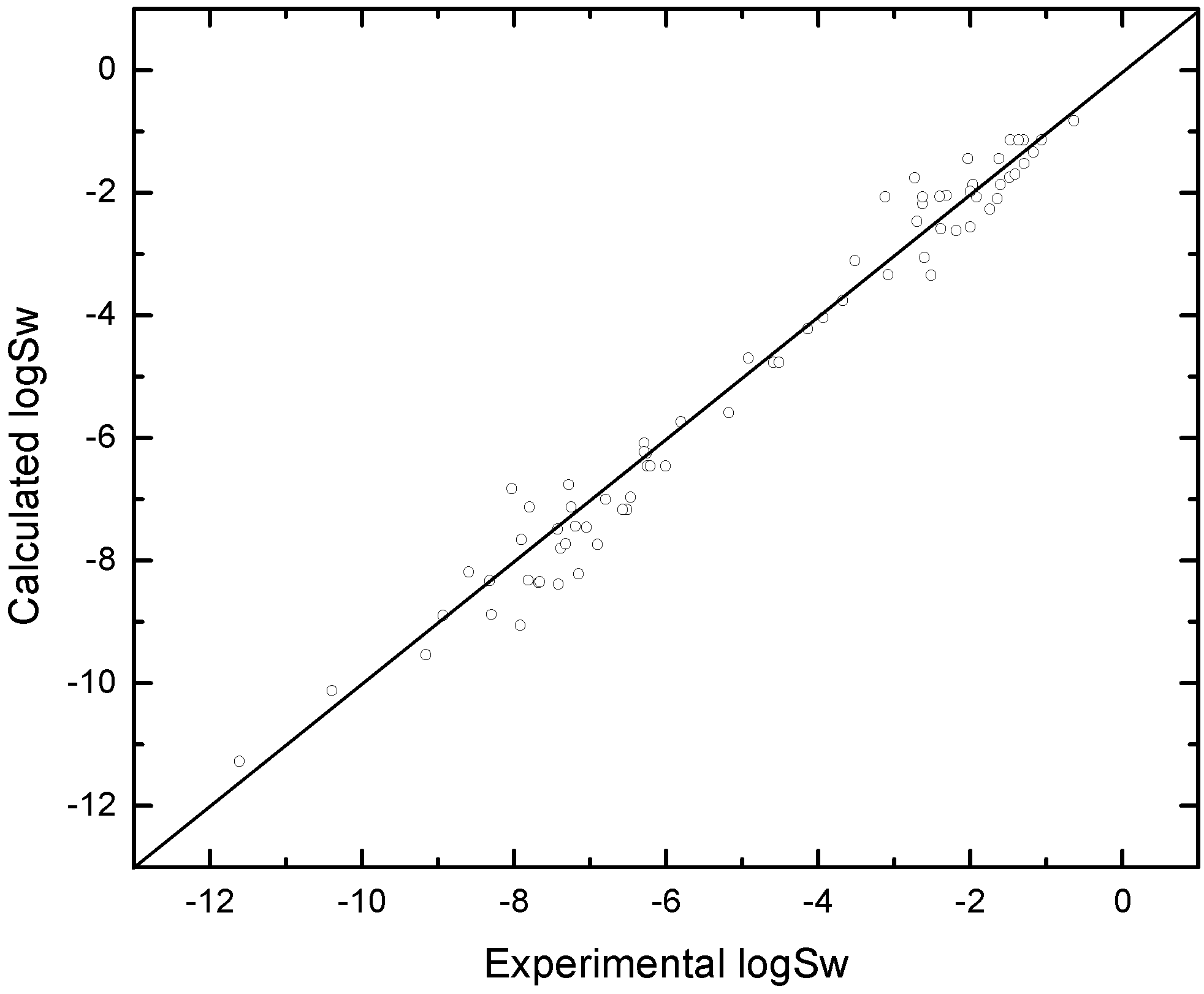
5. Conclusion
Acknowledgements
References and Notes
- Butina, D.; Gola, J. M. R. Modeling Aqueous Solubility. J. Chem. Inf. Model. 2003, 43, 837–841. [Google Scholar] [CrossRef]
- Erös, D.; Kéria, G.; Kövesdi, I.; Szántai-Kis, C.; Mészáros, G.; Örfi, L. Comparison of Predictive Ability of Water Solubility QSPR Models Generated by MLR, PLS and ANN Methods. Mini-Rev. Med. Chem. 2004, 4, 167–177. [Google Scholar] [CrossRef]
- Delaney, J. S. Predicting aqueous solubility from structure. Drug. Discov. Today 2005, 10, 289–295. [Google Scholar] [CrossRef]
- Yalkowsky, S. H.; Pinal, R. Estimation of the aqueous solubility of complex organic molecules. Chemosphere. 1993, 26, 1239–1261. [Google Scholar] [CrossRef]
- Jain, N.; Yalkowsky, S. H. Estimation of the aqueous solubility I: Application to organic nonelectrolytes. J. Pharm. Sci. 2001, 90, 234–252. [Google Scholar] [CrossRef]
- Peterson, D. L.; Yalkowsky, S. H. Comparison of Two Methods for Predicting Aqueous Solubility. J. Chem. Inf. Model. 2001, 41, 1531–1534. [Google Scholar] [CrossRef]
- Tolls, J.; Dijk, J. V.; Verbruggen, E. J. M.; Hermens, J. L. M.; Loeprecht, B.; Schuurmann, G. Aqueous Solubility-Molecular Size Relationships: A Mechanistic Case Study Using C10- to C19-Alkanes. J. Phys. Chem. A 2002, 106, 2760–2765. [Google Scholar] [CrossRef]
- Yang, G.; Ran, Y.; Yalkowsky, S. H. Prediction of the aqueous solubility: Comparison of the general solubility equation and the method using an amended solvation energy relationship. J. Pharm. Sci. 2002, 91, 517–533. [Google Scholar] [CrossRef]
- Klopman, G.; Wang, S.; Balthasar, D. M. Estimation of Aqueous Solubility of Organic Molecules by the Group Contribution Approach. Application to the Study of Biodegradation. J. Chem. Inf. Model. 1992, 32, 474–482. [Google Scholar] [CrossRef]
- Kühne, R.; Ebert, R.-U.; Kleint, F.; Schmidt, G.; Schüürmann, G. Group Contribution Methods to Estimate Water Solubility of Organic Chemicals. Chemosphere. 1995, 30, 2061–2077. [Google Scholar] [CrossRef]
- Klopman, G.; Zhu, H. Estimation of the Aqueous Solubility of Organic Molecules by the Group Contribution Approach. J. Chem. Inf. Model. 2001, 41, 439–445. [Google Scholar] [CrossRef]
- Hou, T. J.; Xia, K.; Zhang, W.; Xu, X. J. ADME Evaluation in Drug Discovery. 4. Prediction of Aqueous Solubility Based on Atom Contribution Approach. J. Chem. Inf. Model. 2004, 44, 266–275. [Google Scholar] [CrossRef]
- Nirmalakhandan, N. N.; Speece, R. E. Prediction of aqueous solubility of organic chemicals based on molecular structure. Environ. Sci. Technol. 1988, 22, 328–338. [Google Scholar] [CrossRef]
- Nelson, T. M.; Jurs, P. C. Prediction of Aqueous Solubility of Organic Compounds. J. Chem. Inf. Model. 1994, 34, 601–609. [Google Scholar] [CrossRef]
- Huibers, P. D. T.; Katritzky, A. R. Correlation of the Aqueous Solubility of Hydrocarbons and Halogenated Hydrocarbons with Molecular Structure. J. Chem. Inf. Model. 1998, 38, 283–292. [Google Scholar] [CrossRef]
- Huuskonen, J.; Salo, M.; Taskinen, J. Aqueous solubility prediction of drugs based on molecular topology and neural network modeling. J. Chem. Inf. Model. 1998, 38, 450–456. [Google Scholar] [CrossRef]
- Mitchell, B. E.; Jurs, P. C. Prediction of Aqueous Solubility of Organic Compounds from Molecular Structure. J. Chem. Inf. Model. 1998, 38, 489–496. [Google Scholar] [CrossRef]
- Makino, M. Prediction of aqueous solubility coefficients of polychlorinated biphenyls by use of computer-calculated molecular properties. Environ. Int. 1998, 24, 653–663. [Google Scholar] [CrossRef]
- Huuskonen, J. Estimation of Aqueous Solubility for a Diverse Set of Organic Compounds Based on Molecular Topology. J. Chem. Inf. Model. 2000, 40, 773–777. [Google Scholar] [CrossRef]
- Yaffe, D.; Cohen, Y.; Espinosa, G.; Arenas, A.; Giralt, F. A Fuzzy ARTMAP Based on Quantitative Structure-Property Relationships (QSPRs) for Predicting Aqueous Solubility of Organic Compounds. J. Chem. Inf. Model. 2001, 41, 1177–1207. [Google Scholar] [CrossRef] [Green Version]
- Liu, R.; So, S. S. Development of Quantitative Structure-Property Relationship Models for Early ADME Evaluation in Drug Discovery. 1. Aqueous Solubility. J. Chem. Inf. Model. 2001, 41, 1633–1639. [Google Scholar] [CrossRef]
- Delgado, E. J. Predicting aqueous solubility of chlorinated hydrocarbons from molecular structure. Fluid. Phase. Equilibr. 2002, 199, 101–107. [Google Scholar] [CrossRef]
- Hua, G.; Veerabahu, S.; Pil, L. Estimation of Aqueous Solubility of Organic Compounds with QSPR Approach. Pharm. Res. 2002, 19, 497–503. [Google Scholar] [CrossRef]
- Engkvist, O.; Wrede, P. High-Throughput, In Silico Prediction of Aqueous Solubility Based on One- and Two-Dimensional Descriptors. J. Chem. Inf. Model. 2002, 42, 1247–1249. [Google Scholar] [CrossRef]
- Chen, X.-Q.; Cho, S. J.; Li, Y.; Venkatesh, S. Prediction of aqueous solubility of organic compounds using a quantitative structure-property relationship. J. Pharm. Sci. 2002, 91, 1838–1852. [Google Scholar] [CrossRef]
- Yan, A.; Gasteiger, J. Prediction of Aqueous Solubility of Organic Compounds by Topological Descriptors. Qsar. Comb. Sci 2003, 22, 821–829. [Google Scholar] [CrossRef]
- Zhong, C.; Hu, Q. Estimation of the aqueous solubility of organic compounds using molecular connectivity indices. J. Pharm. Sci. 2003, 92, 2284–2294. [Google Scholar] [CrossRef]
- Delaney, J. S. ESOL: Estimating Aqueous Solubility Directly from Molecular Structure. J. Chem. Inf. Model. 2004, 44, 1000–1005. [Google Scholar] [CrossRef]
- Bergstrom, C. A. S.; Wassvik, C. M.; Norinder, U.; Luthman, K.; Artursson, P. Global and Local Computational Models for Aqueous Solubility Prediction of Drug-Like Molecules. J. Chem. Inf. Model. 2004, 44, 1477–1488. [Google Scholar] [CrossRef]
- Votano, J. R.; Parham, M.; Hall, L. H.; Kier, L. B.; Hall, L. M. Prediction of Aqueous Solubility Based on Large Datasets Using Several QSPR Models Utilizing Topological Structure Representation. Chem. Biodivers. 2004, 1, 1829–1841. [Google Scholar] [CrossRef]
- Yan, A.; Gasteiger, J.; Krug, M.; Anzali, S. Linear and nonlinear functions on modeling of aqueous solubility of organic compounds by two structure representation methods. J. Comput. Aid. Mol. Des. 2004, 15, 75–87. [Google Scholar]
- Catana, C.; Gao, H.; Orrenius, C.; Stouten, P. F. W. Linear and Nonlinear Methods in Modeling the Aqueous Solubility of Organic Compounds. J. Chem. Inf. Model. 2005, 45, 170–176. [Google Scholar] [CrossRef]
- Bicerano, J. Prediction of Polymer Properties; Marcel Dekker: New York, 1996. [Google Scholar]
- Randic, M. The connectivity index 25 years after. J. Mol. Graph. Model. 2001, 20, 19–35. [Google Scholar] [CrossRef]
- Randic, M.; Pompe, M.; Mills, D.; Basak, S. C. Variable connectivity index as a tool for modeling structure-property relationships. Molecules. 2004, 9, 1177–1193. [Google Scholar] [CrossRef]
- Camarda, K. V.; Sunderesan, P. An Optimization Approach to the Design of Value-Added Soybean Oil Products. Ind. Eng. Chem. Res. 2005, 44, 4361–4367. [Google Scholar] [CrossRef]
- Randic, M. On Characterization of Molecular Branching. J. Am. Chem. Soc. 1975, 97, 6609–6615. [Google Scholar] [CrossRef]
- Randic, M.; Hansen, P. J.; Jurs, P. C. Search for usefule graph theoretical invariants of molecular structure. J. Chem. Inf. Model. 1988, 28, 60–68. [Google Scholar] [CrossRef]
- Randic, M.; Plavsic, D.; Lers, N. Variable Connectivity Index for Cycle-Containing Structures. J. Chem. Inf. Model. 2001, 41, 657–662. [Google Scholar] [CrossRef]
- Kier, L. B.; Hall, L. H.; Murray, W. J.; Randic, M. Molecular connectivity. I: Relationship to nonspecific local anesthesia. J Pharm Sci 1975, 64, 1971–1974. [Google Scholar] [CrossRef]
- Kier, L. B.; Hall, L. H. Molecular connectivity in chemistry and drug research; Academic Press: New York, 1976. [Google Scholar]
- Kier, L. B.; Hall, L. H. Molecular connectivity in structure-activity analysis; Wiley: New York, 1986. [Google Scholar]
- Hall, L. H.; Kier, L. B. Structure-activity studies using valence molecular connectivity. J. Pharm. Sci. 1977, 66, 642–644. [Google Scholar] [CrossRef]
- Hall, L. H.; Kier, L. B. Molecular connectivity and substructure analysis. J. Pharm. Sci. 1978, 67, 1743–1747. [Google Scholar] [CrossRef]
- Martinez, W. L.; Martinez, A. R. Computational Statistics Handbook with MATLAB; Chapman & Hall/CRC: Boca Raton London New York Washington, D.C., 2002. [Google Scholar]
© 2006 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Wang, Y.-L.; Hu, Y.-D.; Wu, L.-Y.; An, W.-Z. Predicting Aqueous Solubility of Chlorinated Hydrocarbons by the MCI Approach. Int. J. Mol. Sci. 2006, 7, 47-58. https://doi.org/10.3390/i7020047
Wang Y-L, Hu Y-D, Wu L-Y, An W-Z. Predicting Aqueous Solubility of Chlorinated Hydrocarbons by the MCI Approach. International Journal of Molecular Sciences. 2006; 7(2):47-58. https://doi.org/10.3390/i7020047
Chicago/Turabian StyleWang, Ying-Long, Yang-Dong Hu, Lian-Ying Wu, and Wei-Zhong An. 2006. "Predicting Aqueous Solubility of Chlorinated Hydrocarbons by the MCI Approach" International Journal of Molecular Sciences 7, no. 2: 47-58. https://doi.org/10.3390/i7020047




