Relation of Certain Quantum Chemical Parameters to Lubrication Behavior of Solid Oxides
Abstract
:Introduction
| Mori et al. (1987) - Describes adsorption activity of organics on fresh steel surfaces by hard and soft acid and base concepts and the hard-soft acid-base (HSAB) principle [2]. |
| Lee (1989-1991) – Applies the HSAB principle to solid adhesion and surface tribointeractions [6,7,8]. |
| Kajdas (1995) - Models tribochemical reactions based on low-energy electron emission from tribosurfaces and formulates a generalized Negative Ion Radical-HSAB action mechanism [4]. |
| Fischer et al. (1995) - Characterizes solid tribosurfaces with Lewis acid and base theory and frontier molecular orbital theory [9]. |
| Mansuy (1995) - Investigates effect of Lewis acid-base interactions between ZDTP and n-dodecylamine on the composition of ZDTP tribochemical films [10]. |
| Martin et al (1995-2000) - Illustrates interactions in binary additive system, formation of ZDTP tribochemical films, synergism in MoDTC/ZDTP and MoDTC/calcium borates, and transfer of tribochemical films by Lewis acid and base concept and the HSAB principle, chemical hardness, and maximum hardness principle [3,11]. |
| Jiang et al. (1996-1997) - Develops antiwear model of ZDTP based on its local charges calculated by ab initio quantum chemistry [12,13]. |
| Bhatia et al. (1999) - Elucidates tribochemical reactions of PFPE on magnetic head/disk interface by catalysis of Lewis acids [14]. |
| Li (2000) - Attempts bond valence matching principle and Saville’s rule for understanding formation thermodynamics of inorganic and organic species yielded from ZDTP on rubbing steel surfaces [15]. |
| Li et al (2001) - Employs electronegativity, electron affinity, and ionization potential of functional antiwear additive elements (S, P) from ZDTP to account for their preferential residence on rubbing coating surfaces of varied mechanical and chemical nature [16]. |
Electronegativity and Chemical Hardness
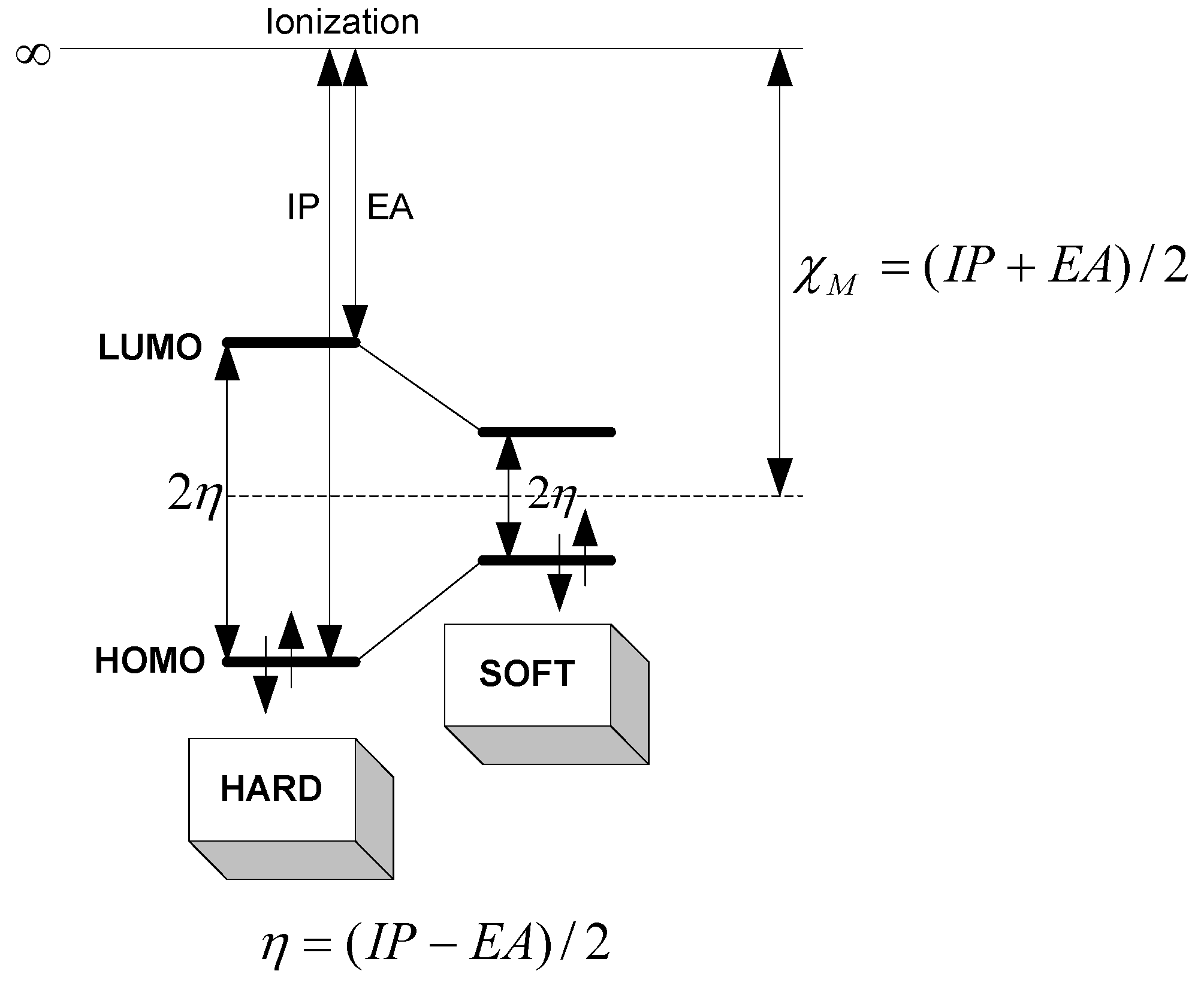
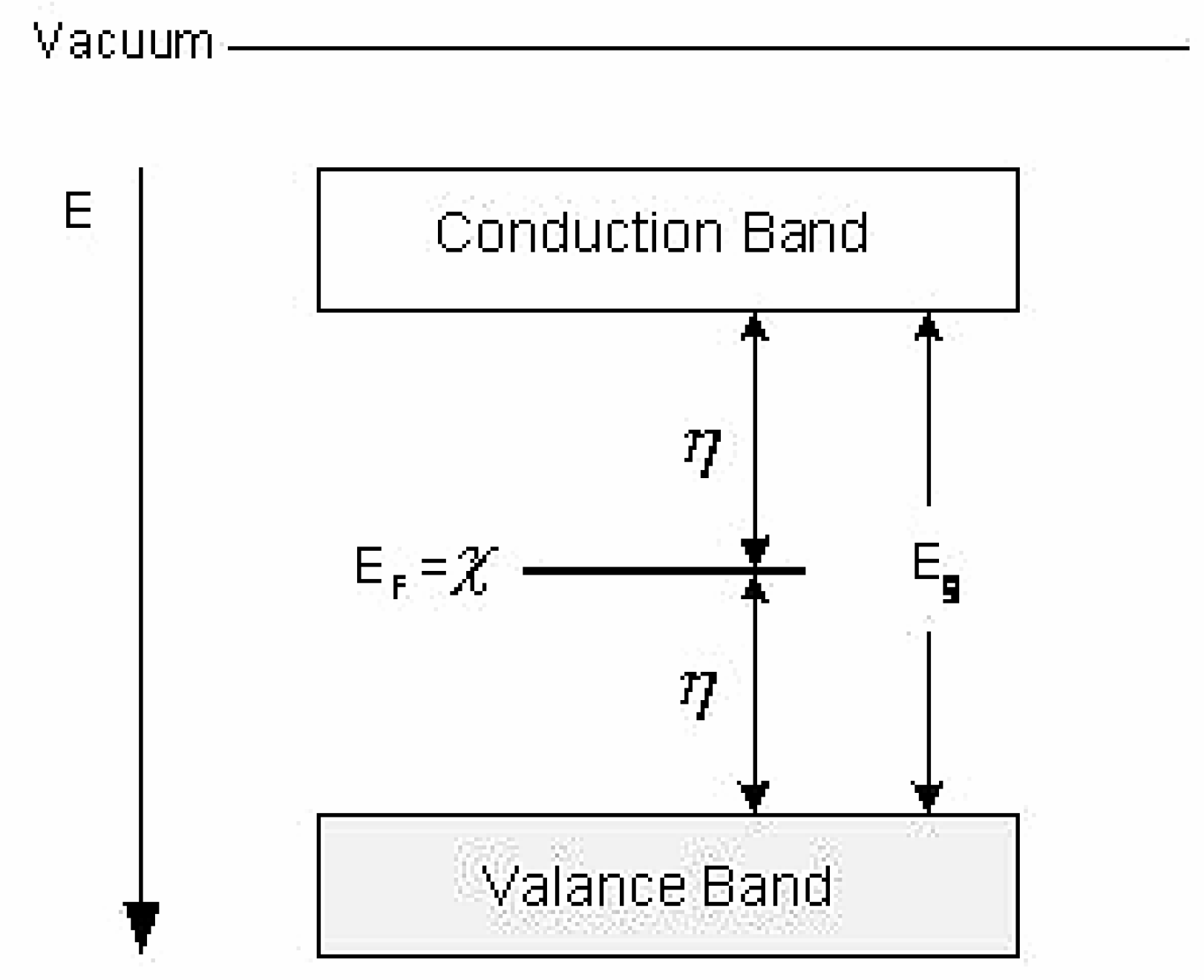
Rationale
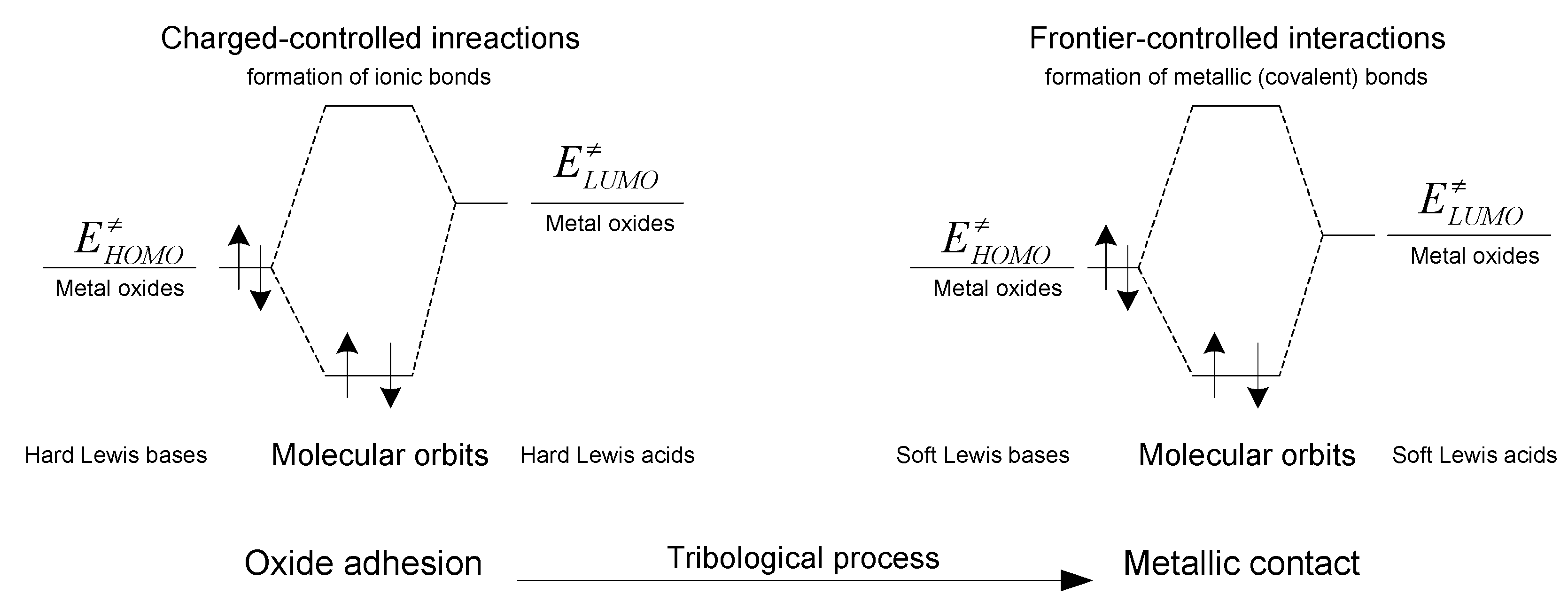
Relation of Electronegativity and Chemical Hardness to Lubricity
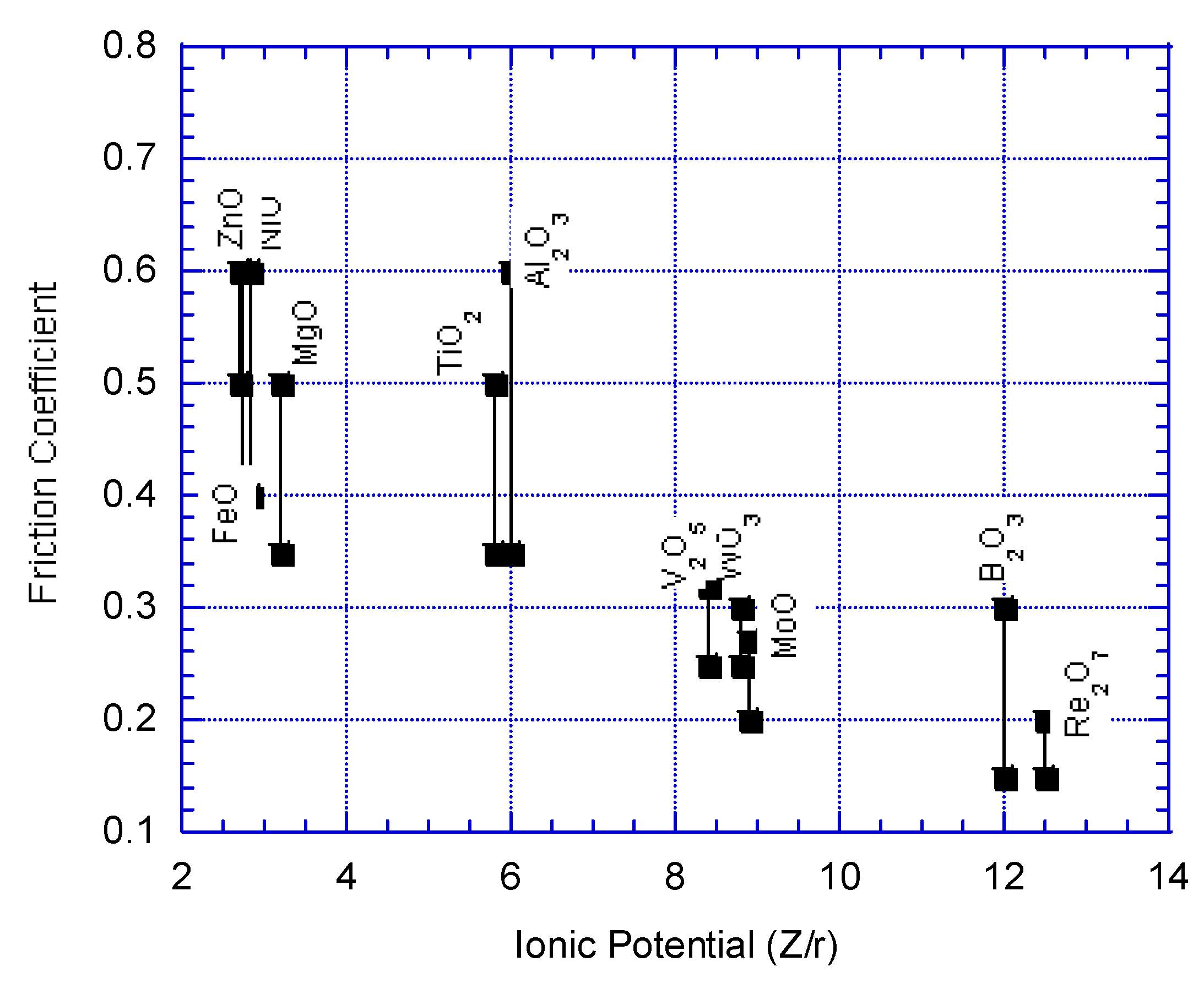
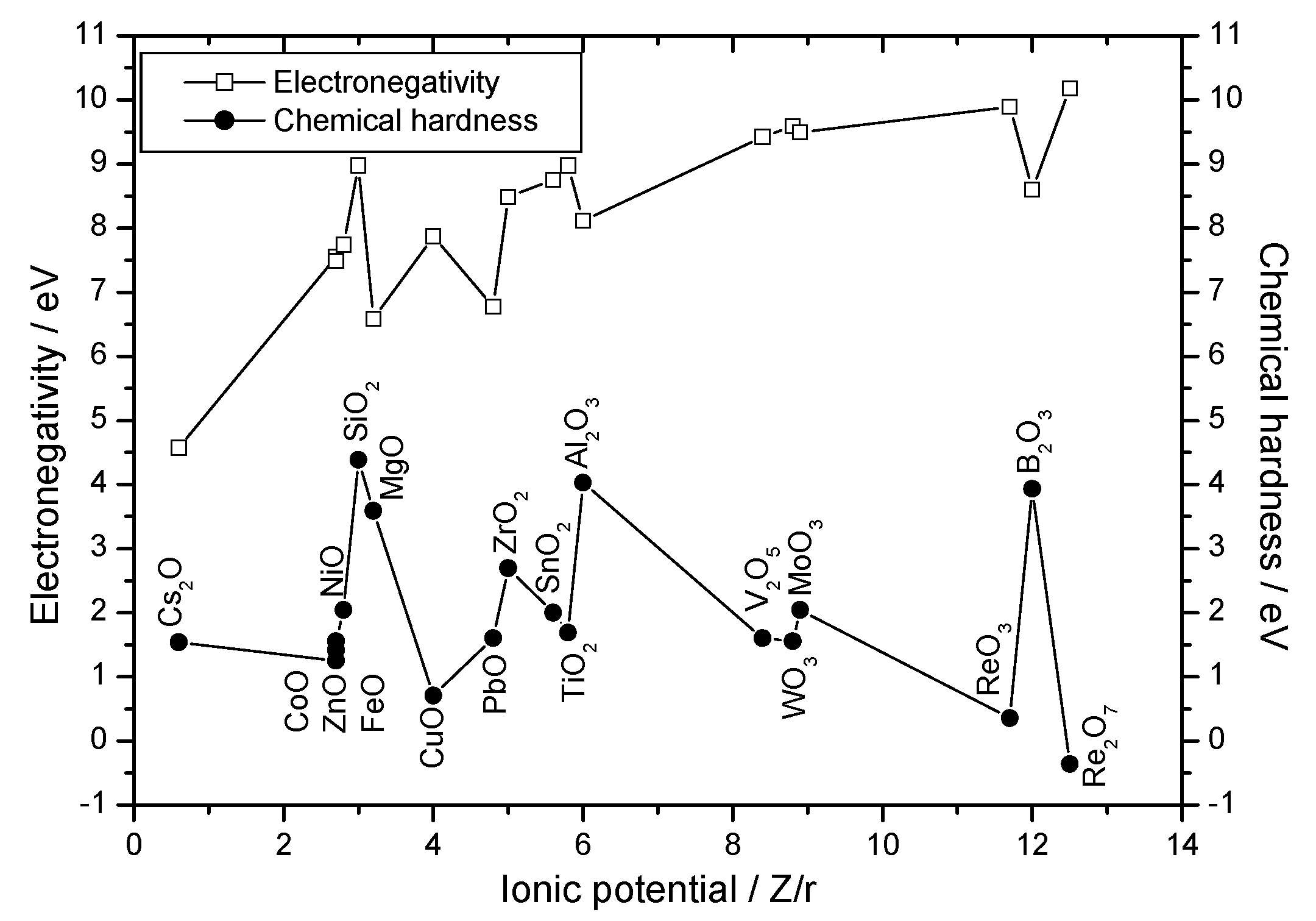
| Oxides | φ / Z/r | χ / eV | η / eV |
| ReO3 | 11.7 | 9.895(6a) | 0.356(6) |
| Re2O7 | 12.5 | 10.311(4), 10.181(6) | -0.101(4), -0.358(6) |
| B2O3 | 12 | 8.801(3), 8.724(4), 8.598(6) | 13.857(3), 6.432(4), 3.936(6) |
| V2O5 | 8.4 | 9.592(4), 9.496(5), 9.422(6) | 2.230(4), 1.807(5), 1.603(6) |
| MoO3 | 8.9 | 9.674(4), 9.585(5) 9.493(6), 9.344(7) | 2.337(4), 2.158(5) 2.044(6), 2.009(7) |
| WO3 | 8.8 | 9.764(4), 9.678(5), 9.588(6) | 1.868(4), 1.663(5), 1.558(6) |
| TiO2 | 5.8 | 9.138(4), 9.058 (5), 8.973 (6), 8.849(8) | 2.285(4), 1.943(5), 1.691(6), 1.465(8) |
| Al2O3 | 6 | 8.116(6) | 4.031(6) |
| SnO2 | 5.6 | 8.885(4), 8.820(5), 8.753(6), 8.695(7), 8.637(8) | 2.292(4), 2.125(5), 2.000(6), 1.919(7), 1.859(8) |
| ZrO2 | 5 | 8.618(4), 8.548(5), 8.487(6) | 2.930(4), 2.788(5), 2.695(6) |
| MgO | 3.2 | 6.702(4), 6.632(5), 6.585(6), 6.449(8) | 4.100(4), 3.773(5), 3.590(6), 3.180(8) |
| NiO | 2.8 | 7.476(4), 7.421(5), 7.739(6) | 2.549(4), 2.240(5), 2.044(6) |
| CoO | 2.7 | 7.598(4), 7.536(5), 7.485(6) | 2.099(4), 1.779(5), 1.558(6) |
| ZnO | 2.7 | 7.650(4), 7.596(5), 7.556(6), 7.446(8) | 1.867(4), 1.593(5), 1.418(6), 1.044(8) |
| FeO | 2.7 | 7.650(4), 7.556(6), 7.460(8) | 1.661(4), 1.249(6), 0.942(8) |
| PbO | 4.8 | 6.933(4), 6.774(6), 6.744(7), 6.697(8) | 1.893(4), 1.603(6), 1.560(7), 1.502(8) |
| CuO | 4.0 | 7.977(4), 7.925(5), 7.874(6) | 1.249(4), 0.955(5), 0.710(6) |
| SiO2 | 3.0 | 8.974(4/6) | 4.384(4/6) |
| Cs2O | 0.6 | 4.571(6), 4.535(8), 4.514(9), 4.499(10), 4.478(11), 4.463(12) | 1.539(6), 1.464(8), 1.424(9), 1.395(10), 1.358(11), 1.331(12) |
Single Oxides
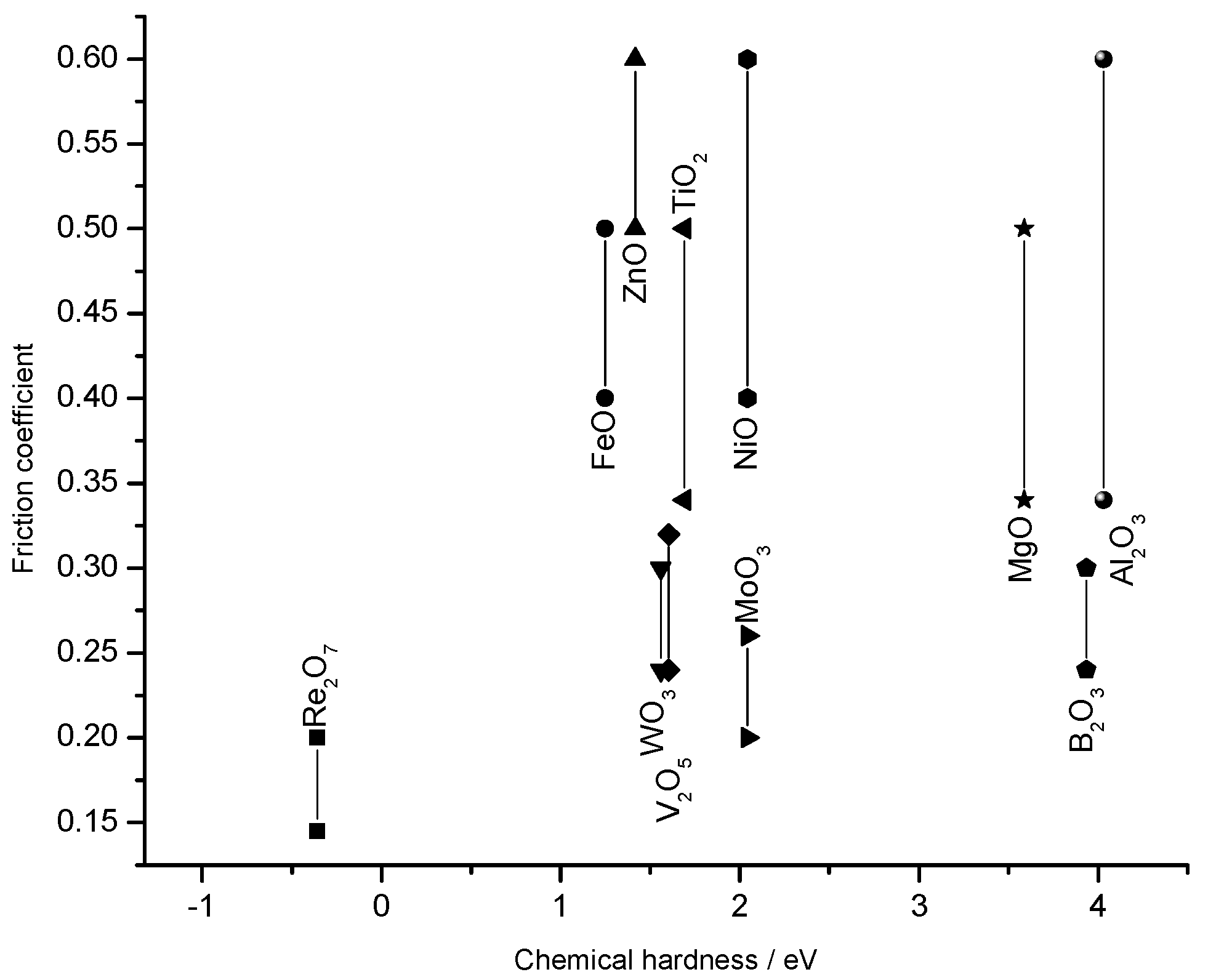
Electronegativity vs friction coefficient
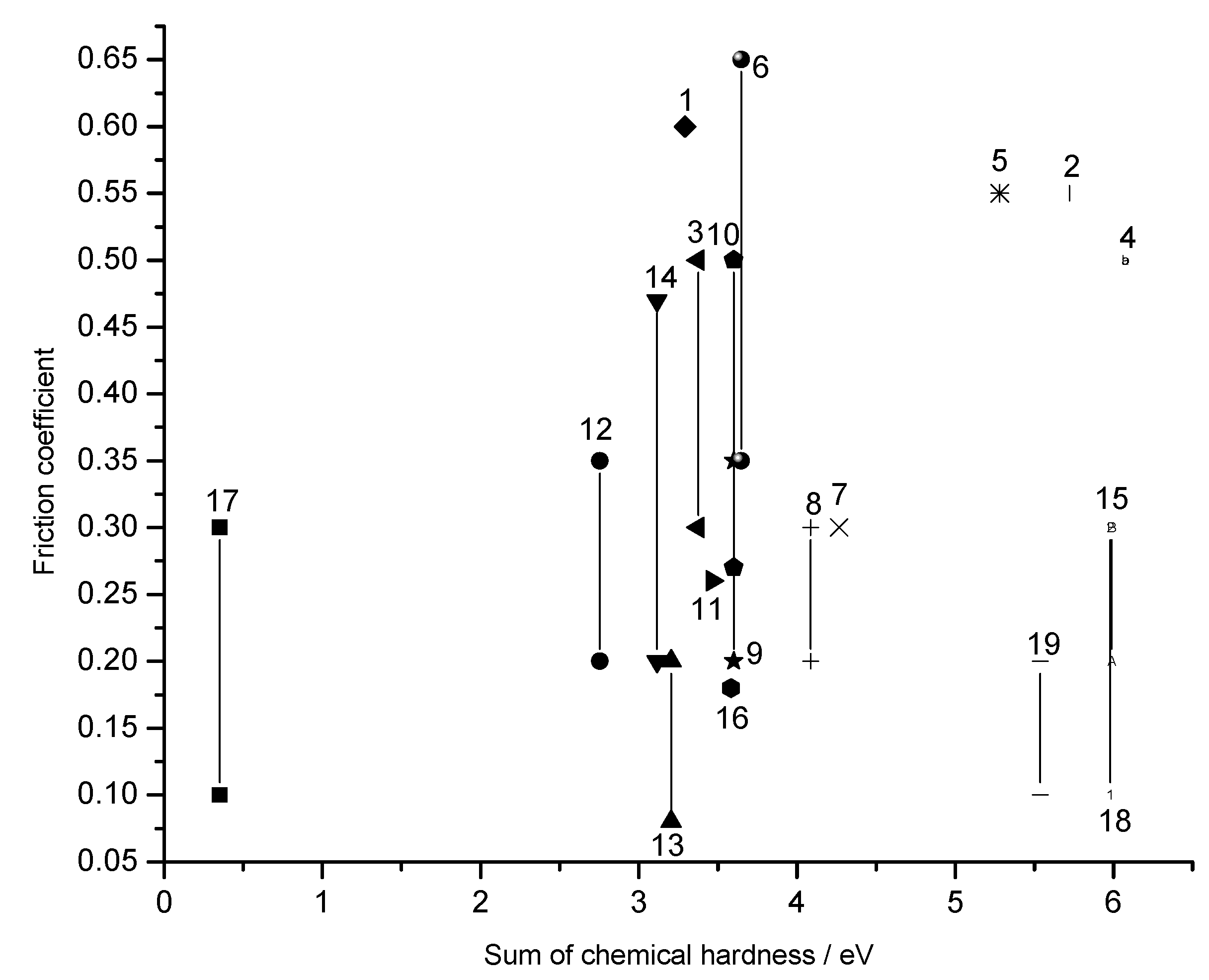
Chemical hardness vs friction coefficient

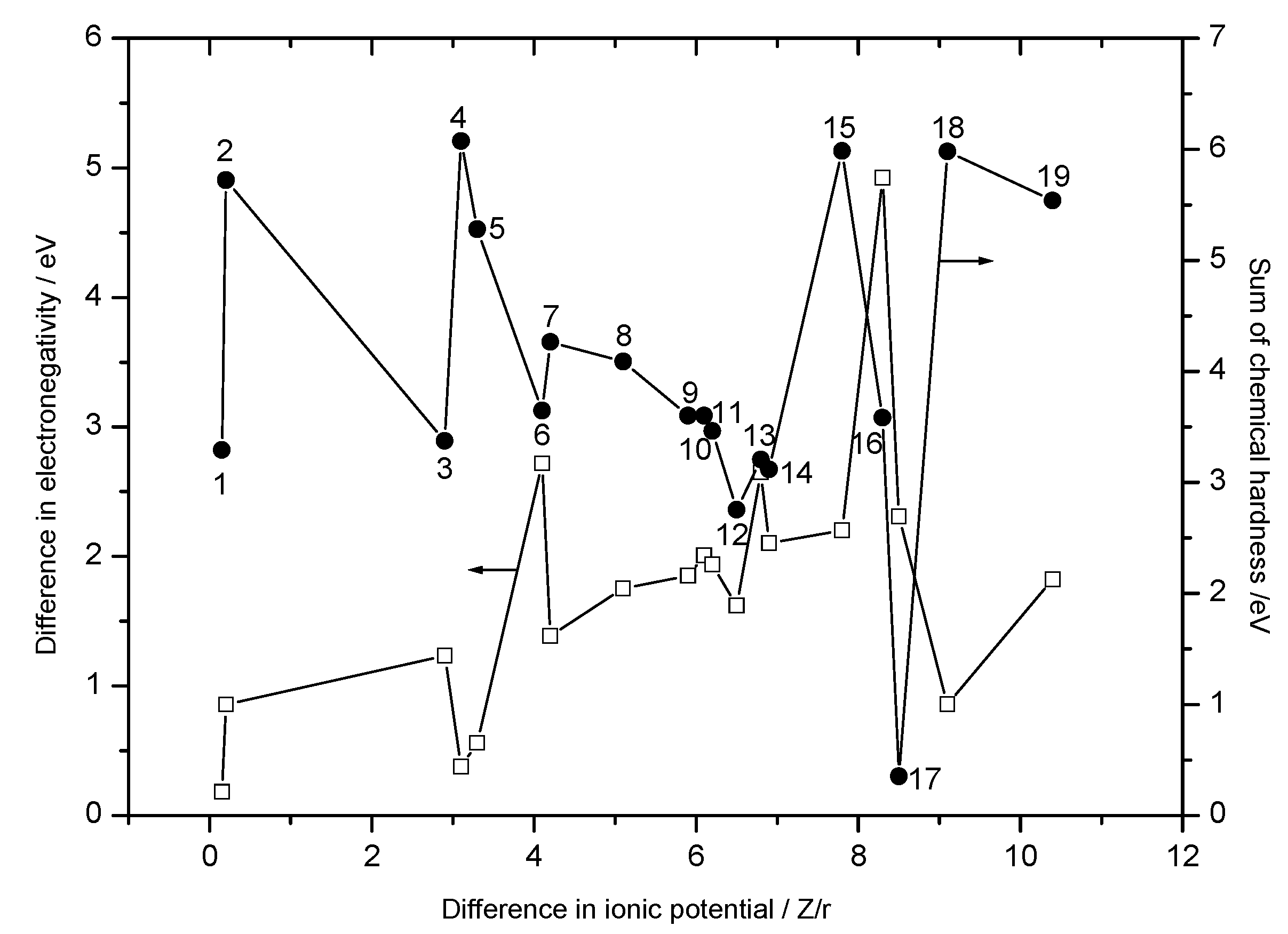
Double Oxides
| No. | Oxide pairs | Δϕ | Δχ | Δη | ΔN |
| 1 | NiO-FeO | 0.15 | 0.183 | 3.293 | 0.0278 |
| 2 | Al2O3-TiO2 | 0.2 | 0.857 | 5.722 | 0.0749 |
| 3 | NiO-TiO2 | 2.9 | 1.234 | 3.375 | 0.183 |
| 4 | Al2O3-NiO | 3.1 | 0.377 | 6.075 | 0.0310 |
| 5 | Al2O3-FeO | 3.3 | 0.560 | 5.280 | 0.0530 |
| 6 | PbO-MoO3 | 4.1 | 2.719 | 3.647 | 0.373 |
| 7 | NiO-Ta2O5 | 4.2 | 1.387 | 4.265 | 0.163 |
| 8 | NiO-MoO3 | 5.1 | 1.754 | 4.088 | 0.215 |
| 9 | NiO-WO3 | 5.9 | 1.849 | 3.602 | 0.257 |
| 10 | CoO-MoO3 | 6.1 | 2.008 | 3.602 | 0.279 |
| 11 | ZnO-MoO3 | 6.2 | 1.937 | 3.462 | 0.280 |
| 12 | CuO-MoO3 | 6.5 | 1.619 | 2.754 | 0.294 |
| 13 | PbO-V2O5 | 6.8 | 2.648 | 3.206 | 0.413 |
| 14 | CoO-WO3 | 6.9 | 2.103 | 3.116 | 0.337 |
| 15 | SiO2-PbO | 7.8 | 2.200 | 5.987 | 0.184 |
| 16 | Cs2O-MoO3 | 8.3 | 4.922 | 3.583 | 0.687 |
| 17 | CuO-Re2O7 | 8.5 | 2.307 | 0.352 | 3.277 |
| 18 | NiO-B2O3 | 9.1 | 0.859 | 5.980 | 0.0718 |
| 19 | PbO-B2O3 | 10.4 | 1.824 | 5.539 | 0.165 |
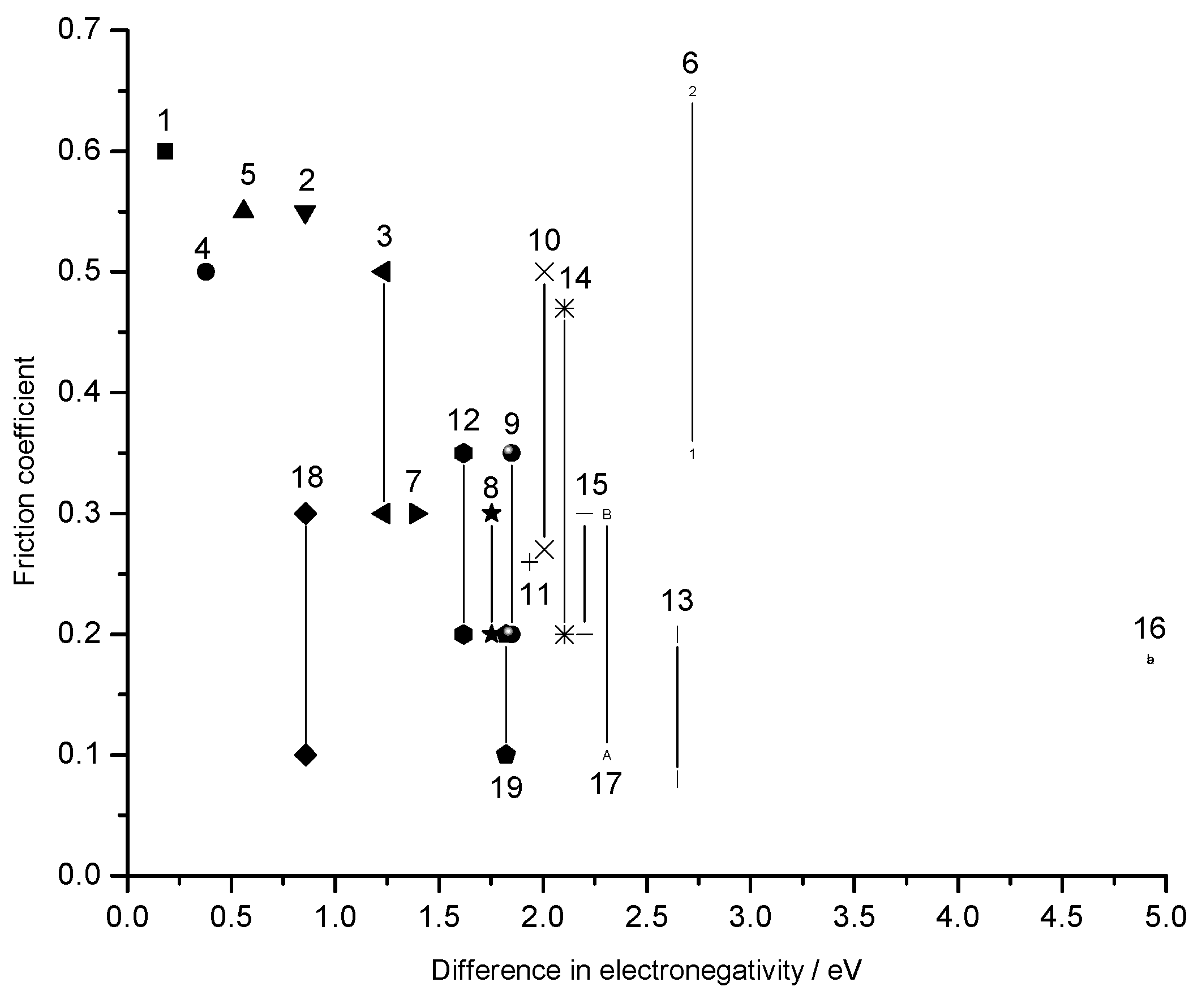
Conclusions
Acknowledgment
References and Notes
- Erdemir, A. Trib. Lett. 2000, 8, 97–102.
- Mori, S. Tribochemical activity of nascent metal surfaces. In Proc. Int. Trib. Conf., Yokohama, Japan; Satellite Forum on Tribochemistry. 1995; pp. 37–42. [Google Scholar]
- Martin, J.M. Trib. Lett. 1999, 6, 1–8.
- Kajdas, C. A novel approach to tribochemical reactions: Generalized NIRAM-HSAB action mechanism. In Proc. Int. Trib. Conf., Yokohama, Japan; Satellite Forum on Tribochemistry. 1995; pp. 31–35. [Google Scholar]
- Singer, I.L.; Mogne, T.L.; Donnet, C.; Martin, J.M. J. Vac. Sci. Technol. A 1996, 14, 38–45. [CrossRef]
- Lee, L.H. Molecular dynamics and energetics of metal-polymer friction and wear. I: Application of the hard-soft acid-base (HSAB) principle to solid adhesion and surface tribointeractions. In Proc. 5th International Congress on Tribology; (eds. K. Holmberg and I. Nieminen), Finnish Society for Tribology, Espoo, Finland. 1989; Vol. 3, pp. 308–313. [Google Scholar]
- Lee, L.H. Hard-soft acid-base (HSAB) principle for solid adhesion and surface interactions. In Fundamentals of Adhesion; (ed. L. H. Lee), Plenum Press, New York, Chapter 12; 1990; pp. 349–362. [Google Scholar]
- Lee, L.H. Relevance of the density-functional theory to acid-base interactions and adhesion in solids. In Acid-Base Interactions; (Eds. K.L. Mittal and H.R. Anderson, Jr.); VSP: Netherlands, 1991; pp. 25–46. [Google Scholar]
- Fischer, T.E.; Mullins, W.M. J. Phys. Chem. 1992, 96, 5690–5701. [CrossRef]
- Mansuy, H. Study of interactions between anti-wear additives and surfactants: Influence on the limited films formation. NTIS No. DE96753703/HDM. 1995. [Google Scholar]
- Martin, J. M.; Grossiord, C.; Mogne, T.L.; Bec, S.; Tonck, A. Tribol. Int. 2001, 34, 523–530.
- Jiang, S.Y.; Dasgupta, S.; Blanco, M.; Frazier, R.; Yamaguchi, E. S.; Tang, Y. C.; Goddard, W. A. J. Phys. Chem. 1996, 100, 15760–15769. [CrossRef]
- Jiang, S.Y.; Frazier, R.; Yamaguchi, E. S.; Blanco, M.; Dasgupta, S.; Zhou, Y.H.; Cagin, T.; Tang, Y.C.; Goddard, W. A. J. Phys. Chem. B 1997, 101, 7702–7709. [CrossRef]
- Bhatia, C.S.; Fong, W.; Chen, C.Y.; Wei, J.J.; Bogy, D.B.; Andres, S.; Stammler, T.; Stohr, J. IEEE Trans. on Magnetics 1999, 35, 910–915. [CrossRef]
- Li, S.H. Study on tribochemodesign based on tribochemical case analyses. In Postdoctoral Report, Tsinghua University, December 2000.
- Li, S.H.; Zhang, R.J.; Jin; Wang, Y.C.; Tung, S.C. Tribology Transactions, 2003; 46, 200–205.
- Parr, R.G.; Pearson, R.G. J. Amer. Chem. Soc. 1983, 105, 7512–7516. [CrossRef]
- Pearson, R.G. Proc. Natl. Acad. Sci. 1986, 83, 8440–8441. [CrossRef]
- Lee, L.H. Relevance of the density-functional theory to acid-base interactions and adhesion in solids. In Acid-Base Interactions; (Eds. K.L. Mittal and H.R. Anderson, Jr.); VSP: Netherlands, 1991; pp. 25–46. [Google Scholar]
- Lee, L.H. Molecular dynamics and energetics of metal-polymer friction and wear. I: Application of the hard-soft acid-base (HSAB) principle to solid adhesion and surface tribointeractions. In Proc. 5th International Congress on Tribology; (eds. K. Holmberg and I. Nieminen), Finnish Society for Tribology, Espoo, Finland. 1989; 3, pp. 308–313. [Google Scholar]
- Lee, L.H. Hard-soft acid-base (HSAB) principle for solid adhesion and surface interactions. In Fundamentals of Adhesion; (ed. L. H. Lee); Plenum Press: New York, Chapter 12; 1990; pp. 349–362. [Google Scholar]
- Klopman, G. J. Amer. Chem. Soc. 1968, 90, 223–234. [CrossRef]
- Quinn, T. F. J. Tribol. Int., 1983, 16, 257–271.
- Pearson, R.G. J. Chem. Edu. 1999, 76, 267–275. [CrossRef]
- Shannon, R.D. Acta Crystallogr. A 1976, 32, 751–767. (Also see: Shannon, R.D. Electronic properties of cations and corresponding oxides. See: http://www.icmcb.u-bordeaux.fr/pub/elecprop/ions.htm.)
- Guo, Q.Z.; Chen, A.Q. Principles of Organic Acids and Bases. Xiamen University Press: Xiamen, 1991; 120–130. [Google Scholar]
- Lugscheider, E.; Knotek, O.; Barwulf, S.; Bobzin, K. Surf. Coat. Technol. 2001, 142-144, 137–142. [CrossRef]
- Mayrhofer, P.H.; Hovsepian, P.E.; Mitterer, C.; Munz, W.D. Surf. Coat. Technol. 2004, 177, 341–347. [CrossRef]
- Gulbinski, W.; Suszko, T.; Sienicki, W.; Warcholinski, B. Wear, 2003; 254, 129–135.
- Strong, K.L.; Zabinski, J.S. Thin Solid Films, 2002; 406, 174–184.
- Rosado, L.; Forster, N.H.; Trivedi, H.K.; King, J.P. Tribol. Trans. 2000, 43, 489–497.
© 2005 by MDPI (http://www.mdpi.org).
Share and Cite
Erdemir, A.; Li, S.; Jin, Y. Relation of Certain Quantum Chemical Parameters to Lubrication Behavior of Solid Oxides. Int. J. Mol. Sci. 2005, 6, 203-218. https://doi.org/10.3390/i6060203
Erdemir A, Li S, Jin Y. Relation of Certain Quantum Chemical Parameters to Lubrication Behavior of Solid Oxides. International Journal of Molecular Sciences. 2005; 6(6):203-218. https://doi.org/10.3390/i6060203
Chicago/Turabian StyleErdemir, Ali, Shenghua Li, and Yuansheng Jin. 2005. "Relation of Certain Quantum Chemical Parameters to Lubrication Behavior of Solid Oxides" International Journal of Molecular Sciences 6, no. 6: 203-218. https://doi.org/10.3390/i6060203




