Cichorium intybus L. Extract Suppresses Experimental Gout by Inhibiting the NF-κB and NLRP3 Signaling Pathways
Abstract
:1. Introduction
2. Results
2.1. Animal Experiments
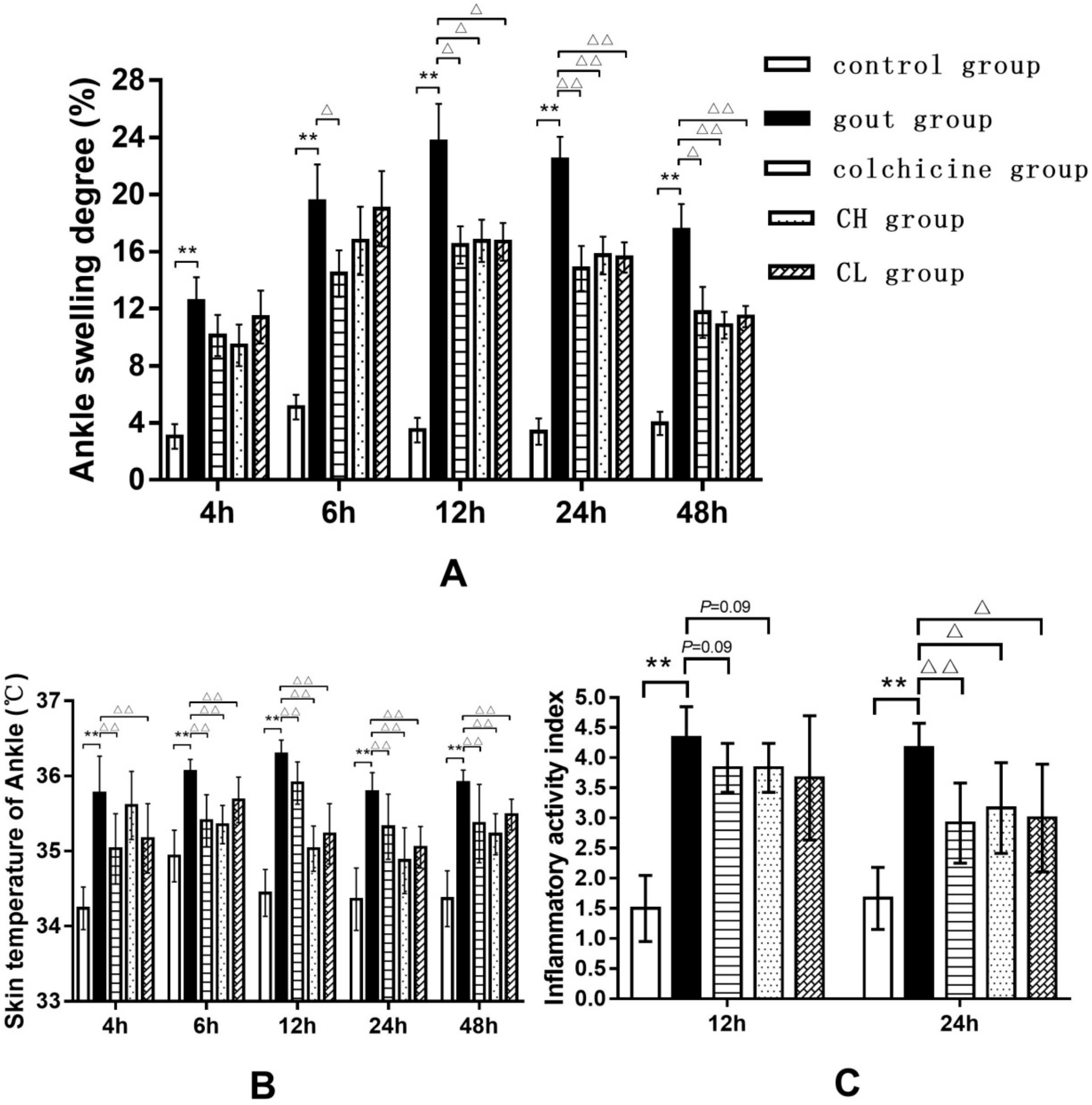
2.1.1. Effect of Chicory Extract on MSU-Induced Ankle Edema and Inflammatory Activity Index
2.1.2. Effect of Chicory Extract on MSU-Induced Synovial Inflammation by Hematoxylin and Eosin (H&E) Staining
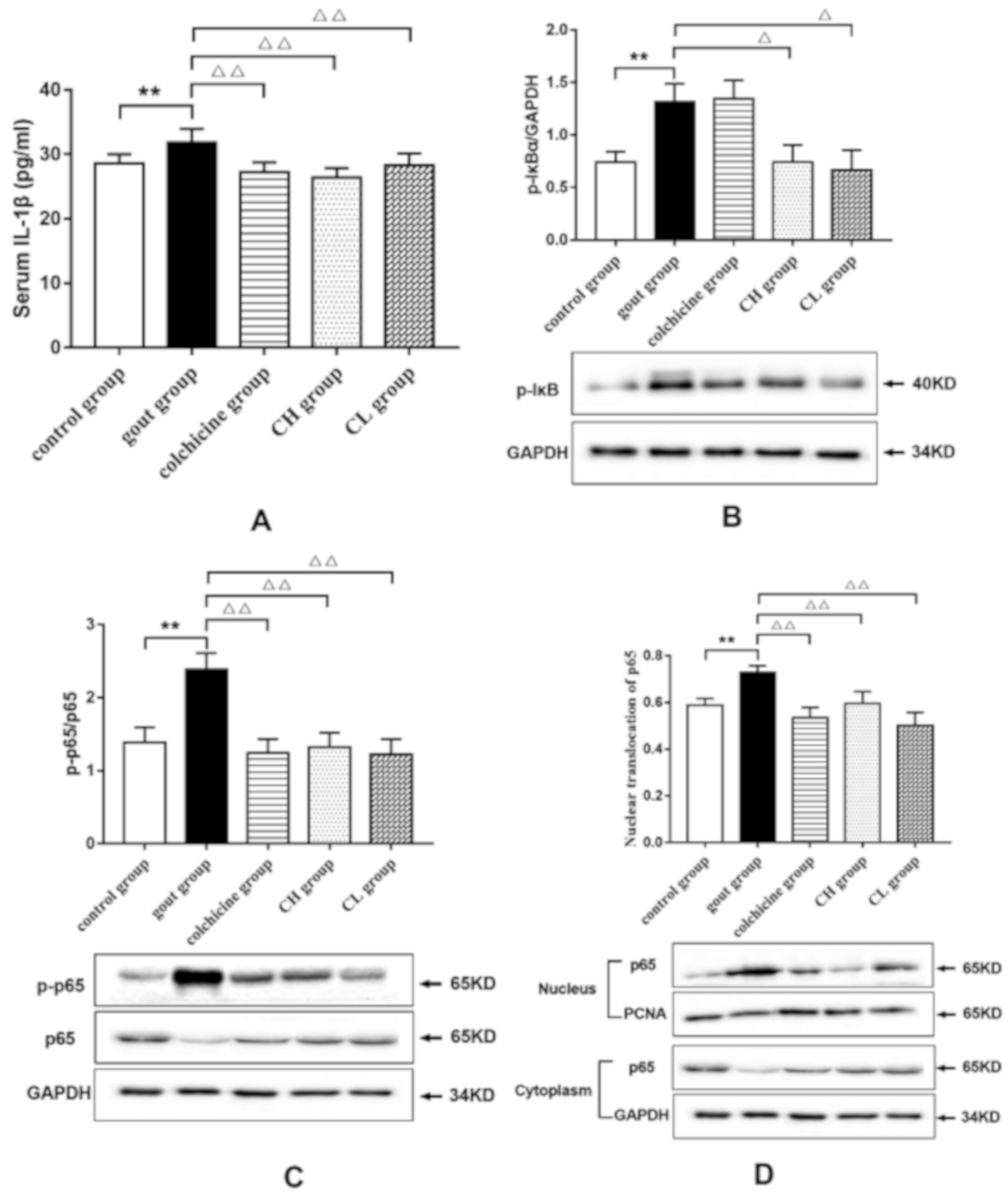
2.1.3. Effect of Chicory Extract on IL-1β in Gout Rats
2.1.4. Effect of Chicory Extract on the NF-κB Signaling Pathway in Gout Rats
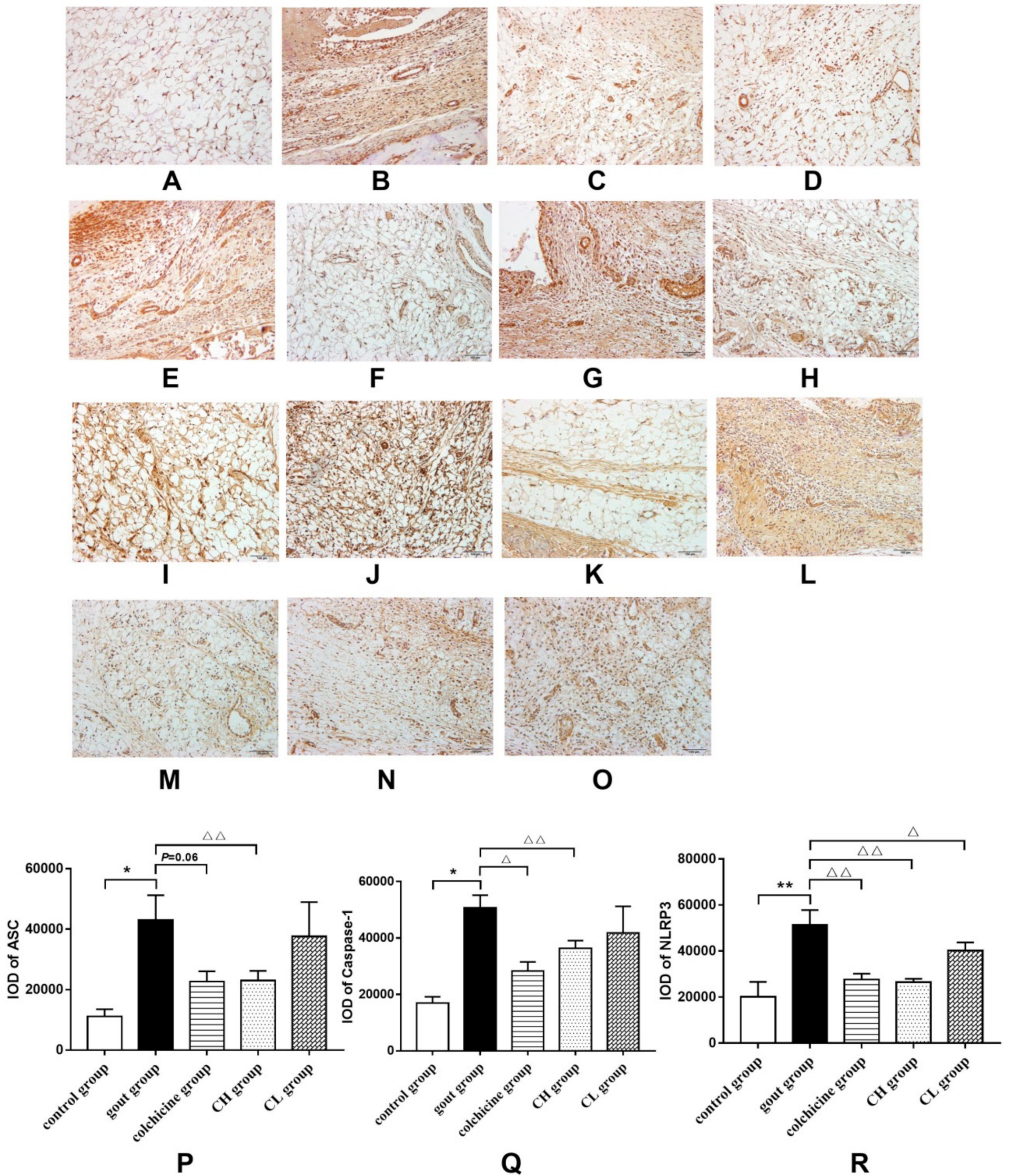
2.1.5. Effect of Chicory on the NLRP3 Signaling Pathway in Gout Rats
2.2. Cell Experiments
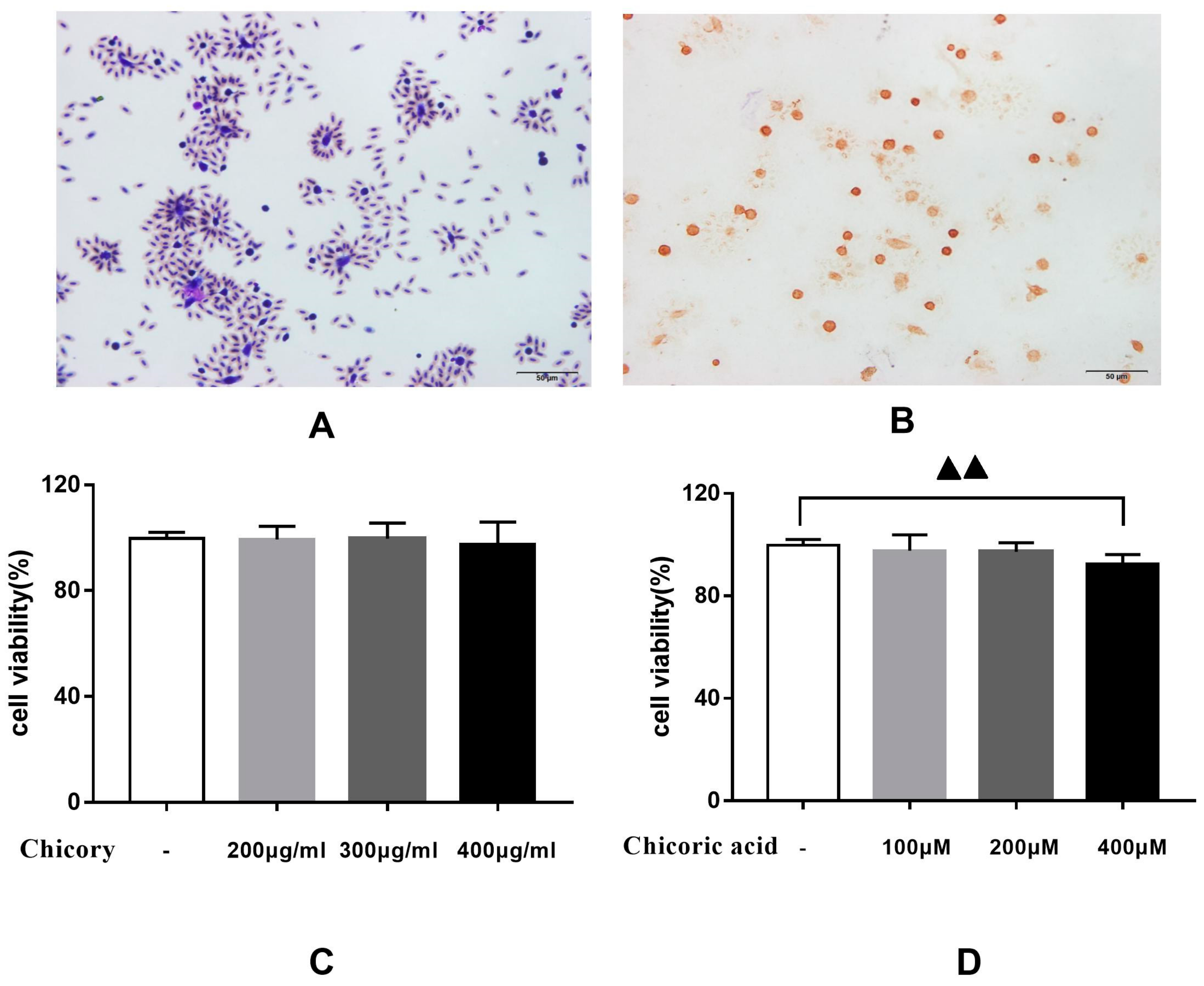
2.2.1. Effects of Chicory Extract and Chicoric Acid on the Proliferation of Macrophages
2.2.2. Effects of Chicory Extract and Chicoric Acid on the NF-κB and the NLRP3 Signaling Pathways in Macrophages
3. Discussion
4. Materials and Methods
4.1. Animal Experiments
4.1.1. Chicory Extract Preparation
4.1.2. Induction of Gouty Arthritis with MSU Crystals and Drug Treatment
4.1.3. Assessment of Ankle Edema
4.1.4. Assessment of Inflammatory Activity
4.1.5. Histopathological Analysis
4.1.6. Measurement of IL-1β in Gout Rats
4.1.7. Analysis of the NF-κB Signaling Pathway in Synovial Tissues of Gout Rats by Western Blotting
4.1.8. Analysis of the NLRP3 Signaling Pathway in Synovial Tissues of Gout Rats by Immunohistochemistry
4.2. Cell Experiments
4.2.1. Isolation, Characterization, and Cultivation of Macrophages
4.2.2. Cell Viability Assay by CCK8
4.2.3. Measurement of IL-1β in Isolated Macrophages
4.2.4. Analysis of the NF-κB and NLRP3 Signaling Pathways in Isolated Macrophages by Western Blotting
4.3. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| MSU | monosodium urate |
| NF-κB | nuclear factor kappa-B |
| NLRP3 | nucleotide-binding oligomerization domain leucine-rich repeat and pyrin domain-containing protein 3 |
| IL-1β | interleukin-1β |
| ASD | ankle swelling degree |
| IAI | inflammatory activity index |
| GAPDH | glyceraldehyde 3-phosphate dehydrogenase |
References
- Perez-Ruiz, F.; Dalbeth, N.; Bardin, T. A review of uric acid, crystal deposition disease, and gout. Adv. Ther. 2015, 32, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Burke, B.T.; Köttgen, A.; Law, A.; Windham, B.G.; Segev, D.; Baer, A.N.; Coresh, J.; McAdams-DeMarco, M.A. Physical Function, Hyperuricemia, and Gout in Older Adults. Arthritis Care Res. 2015, 67, 1730–1738. [Google Scholar] [CrossRef] [PubMed]
- Rai, S.K.; Burns, L.C.; De Vera, M.A.; Haji, A.; Giustini, D.; Choi, H.K. The economic burden of gout: A systematic review. Semin. Arthritis Rheum. 2015, 45, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Roughley, M.J.; Belcher, J.; Mallen, C.D.; Roddy, E. Gout and risk of chronic kidney disease and nephrolithiasis: meta-analysis of observational studies. Arthritis Res. Ther. 2015, 17, 90. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.F.; See, L.C.; Luo, S.F.; Ko, Y.S.; Lin, Y.S.; Wang, J.S.; Lin, C.M.; Chen, H.W.; Yu, K.H. Gout: an independent risk factor for all-cause and cardiovascular mortality. Rheumatology. 2010, 49, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.C.; Rai, S.K.; Lu, N.; Zhang, Y.; Choi, H.K. The unclosing premature mortality gap in gout: a general population-based study. Ann. Rheum. Dis. 2017, 76, 1289–1294. [Google Scholar] [CrossRef] [PubMed]
- Doherty, M. New insights into the epidemiology of gout. Rheumatology 2009, 48, ii2–ii8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rai, S.K.; Aviña-Zubieta, J.A.; McCormick, N.; De Vera, M.A.; Shojania, K.; Sayre, E.C.; Choi, H.K. The rising prevalence and incidence of gout in British Columbia, Canada: Population-based trends from 2000 to 2012. Semin Arthritis Rheum. 2017, 46, 451–456. [Google Scholar] [CrossRef]
- Kuo, C.F.; Grainge, M.J.; Mallen, C.; Zhang, W.; Doherty, M. Rising burden of gout in the UK but continuing suboptimal management: a nationwide population study. Ann. Rheum. Dis. 2015, 74, 661–667. [Google Scholar] [CrossRef]
- So, A.; De Smedt, T.; Revaz, S.; Tschopp, J. A pilot study of IL-1 inhibition by anakinra in acute gout. Arthritis Res. Ther. 2007, 9, R28. [Google Scholar] [CrossRef]
- Jaramillo, M.; Godbout, M.; Naccache, P.; Olivier, M. Signaling events involved in macrophage chemokine expression in response to monosodium urate crystals. J. Biol. Chem. 2004, 279, 52797–52805. [Google Scholar] [CrossRef] [PubMed]
- Skeldon, A.M.; Faraj, M.; Saleh, M. Caspases and inflammasomes in metabolic inflammation. Immunol. Cell Biol. 2014, 92, 304–313. [Google Scholar] [CrossRef] [PubMed]
- Perez-Ruiz, F.; Mielgo, F.J.; Beites, A.M. Optimisation of the Treatment of Acute Gout. Biodrugs. 2000, 13, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Aran, S.; Malekzadeh, S.; Seifirad, S. A double-blind randomized controlled trial appraising the symptom-modifying effects of colchicine on osteoarthritis of the knee. Clin. Exp. Rheumatol. 2011, 29, 513–518. [Google Scholar] [PubMed]
- Douros, A.; Bronder, E.; Klimpel, A.; Erley, C.; Garbe, E.; Kreutz, R. Drug-induced kidney injury: A large case series from the Berlin Case-Control Surveillance Study. Clin Nephrol. 2018, 89, 18–26. [Google Scholar] [CrossRef]
- Zhong, H.; Zhong, Z.; Li, H.; Zhou, T.; Xie, W. A rare case report of heavy dose colchicine induced acute kidney injury. BMC Pharmacol Toxicol. 2018, 19, 69. [Google Scholar] [CrossRef] [PubMed]
- Subhash, C.; Mukesh, K.; Pradeep, D.; Ku, A. Studies on Industrial Importance and Medicinal Value of Chicory Plant (Cichorium intybus L.). Int. J. Adv. Res. 2016, 4, 1060–1071. [Google Scholar]
- Liu, Q.; Chen, Y.; Shen, C.; Xiao, Y.; Wang, Y.; Liu, Z.; Liu, X. Chicoric acid supplementation prevents systemic inflammation-induced memory impairment and amyloidogenesis via inhibition of NF-κB. Faseb J. 2017, 31, 1494–1507. [Google Scholar] [CrossRef]
- El-Sayed, Y.S.; Lebda, M.A.; Mohammed, H. Neoman, S.A. Chicory (Cichorium intybus L.) Root Extract Regulates the Oxidative Status and Antioxidant Gene Transcripts in CCl4-Induced Hepatotoxicity. PLOS ONE 2015, 10, e0121549. [Google Scholar] [CrossRef]
- Rammal, H.; Younos, C.; Bouayed, J.; Chakou, A.; Bedouhene, S.; Soulimani, R. Aperçu ethnobotanique et phytopharmacologique sur Cichorium intybus L. Phytotherapie. 2008, 6, 184–186. [Google Scholar] [CrossRef]
- Schmidt, B.M.; Ilic, N.; Poulev, A.; Raskin, I. Toxicological evaluation of a chicory root extract. Food Chem. Toxicol. 2007, 45, 1131–1139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, C.S.; Zhang, B.; Lin, Z.J.; Wang, X.J.; Zhou, Y.; Sun, X.X.; Xiao, M.L. Relationship between High-Performance Liquid Chromatography Fingerprints and Uric Acid-Lowering Activities of Cichorium intybus L. Molecules 2015, 20, 9455–9467. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.J.; Lin, Z.J.; Zhang, B.; Zhu, C.S.; Niu, H.J.; Zhou, Y.; Nie, A.Z.; Wang, Y. Molecular docking analysis of xanthine oxidase inhibition by constituents of cichory. China J. Chin. Mater. Med. 2015, 40, 3818–3825. [Google Scholar]
- Jin, Y.N.; Lin, Z.J.; Zhang, B.; Bai, Y.F. Effects of Chicory on Serum Uric Acid, Renal Function, and GLUT9 Expression in Hyperuricaemic Rats with Renal Injury and In Vitro Verification with Cells. Evid. Based Complement. Alternat. Med. 2018, 2018, 1764212. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lin, Z.; Zhang, B.; Nie, A.Z.; Bian, M. Cichorium intybus L. promotes intestinal uric acid excretion by modulating ABCG2 in experimental hyperuricemia. Nutr. Metab. 2017, 14, 38. [Google Scholar] [CrossRef] [PubMed]
- Saad, E.M.; Madbouly, A.; Ayoub, N.; El Nashar, R.M. Preparation and application of molecularly imprinted polymer for isolation of chicoric acid from Chicorium intybus L. medical plant. Anal. Chim. Acta. 2015, 877, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, W.; Fayazuddin, M.; Shariq, S.; Singh, O.; Moin, S.; Akhtar, K.; Kumar, A. Anti-inflammatory activity of roots of Cichorium intybus due to its inhibitory effect on various cytokines and antioxidant activity. Anc. Sci. Life. 2014, 34, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Tsai, K.L.; Kao, C.L.; Hung, C.H.; Cheng, Y.H.; Lin, H.C.; Chu, P.M. Chicoric acid is a potent anti-atherosclerotic ingredient by anti-oxidant action and anti-inflammation capacity. Oncotarget 2017, 8, 29600–29612. [Google Scholar] [CrossRef] [Green Version]
- Abhishek, A.; Roddy, E.; Doherty, M. Gout - a guide for the general and acute physicians. Clin Med 2017, 17, 54–59. [Google Scholar] [CrossRef]
- Chuang, S.Y.; Lee, S.C.; Hsieh, Y.T.; Pan, W.H. Trends in Hyperuricemia and Gout Prevalence: Nutrition and Health Survey in Taiwan from 1993-1996 to 2005-2008. Asia Pac. J Clin. Nutr. 2011, 20, 301–308. [Google Scholar]
- Kiadaliri, A.A.; Uhlig, T.; Englund, M. Burden of gout in the Nordic region, 1990–2015: findings from the Global Burden of Disease Study 2015. Scand. J Rheumatol. 2018, 47, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Elfishawi, M.M.; Zleik, N.; Kvrgic, Z.; Michet, C.J.J.; Crowson, C.S.; Matteson, E.L.; Bongartz, T. The Rising Incidence of Gout and the Increasing Burden of Comorbidities: A Population-based Study over 20 Years. J Rheumatol. 2018, 45, 574–579. [Google Scholar] [CrossRef] [PubMed]
- Gaffo, A.L.; Saag, K.G. Febuxostat: the evidence for its use in the treatment of hyperuricaemia and gout. Core Evid. 2009, 4, 25–36. [Google Scholar]
- Martinon, F. Mechanisms of uric acid crystal-mediated autoinflammation. Immunol. Rev. 2010, 233, 218–232. [Google Scholar] [CrossRef] [PubMed]
- Popa-Nita, O.; Naccache, P.H. Crystal-induced neutrophil activation. Immunolo Cell Biol. 2010, 88, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, H.R.; Sundy, J.; Terkeltaub, R.; Knapp, H.R.; Mellis, S.; Soo, Y.; King-Davis, S.; Weinstein, S.; Radin, A. Placebo-controlled study of rilonacept for gout flare prophylaxis during intiation of urate-lowering therapy. Ann. Rheum. Dis. 2009, 60, S410. [Google Scholar]
- Lo, Y.H.; Huang, Y.W.; Wu, Y.H.; Tsai, C.S.; Lin, Y.C.; Mo, S.T.; Kuo, W.C.; Chuang, Y.T.; Jiang, S.T.; Shih, H.M.; et al. Selective inhibition of the NLRP3 inflammasome by targeting to promyelocytic leukemia protein in mouse and human. Blood. 2013, 121, 3185–3194. [Google Scholar] [CrossRef] [Green Version]
- Van de Veerdonk, F.L.; Netea, M.G.; Dinarello, C.A.; Joosten, L.A. Inflammasome activation and IL-1beta and IL-18 processing during infection. Trends Immunol. 2011, 32, 110–116. [Google Scholar] [CrossRef]
- Busso, N.; So, A. Mechanisms of inflammation in gout. Rheumatology. 2005, 44, 1090–1096. [Google Scholar] [CrossRef]
- Di Giovine, F.S.; Malawista, S.E.; Nuki, G.; Duff, G.W. Interleukin 1 (IL 1) as a mediator of crystal arthritis. Stimulation of T cell and synovial fi broblast mitogenesis by urate crystal-induced IL 1. J Immunol. 1987, 138, 3213–3218. [Google Scholar]
- Coderre, T.J.; Wall, P.D. Ankle joint urate arthritis (AJUA) in rats: an alternative animal model of arthritis to that produced by Freund’s adjuvant. Pain. 1987, 28, 379–393. [Google Scholar] [CrossRef]
- Zhang, X.; Goncalves, R.; Mosser, D.M. Current Protocols in Immunology, 1st ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2008; pp. 14.1.1–14.1.14. [Google Scholar]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Lin, Z.; Zhang, B.; Jiang, Z.; Guo, F.; Yang, T. Cichorium intybus L. Extract Suppresses Experimental Gout by Inhibiting the NF-κB and NLRP3 Signaling Pathways. Int. J. Mol. Sci. 2019, 20, 4921. https://doi.org/10.3390/ijms20194921
Wang Y, Lin Z, Zhang B, Jiang Z, Guo F, Yang T. Cichorium intybus L. Extract Suppresses Experimental Gout by Inhibiting the NF-κB and NLRP3 Signaling Pathways. International Journal of Molecular Sciences. 2019; 20(19):4921. https://doi.org/10.3390/ijms20194921
Chicago/Turabian StyleWang, Yu, Zhijian Lin, Bing Zhang, Zhuoxi Jiang, Fanfan Guo, and Ting Yang. 2019. "Cichorium intybus L. Extract Suppresses Experimental Gout by Inhibiting the NF-κB and NLRP3 Signaling Pathways" International Journal of Molecular Sciences 20, no. 19: 4921. https://doi.org/10.3390/ijms20194921




