Aggregation of Human Trophoblast Cells into Three-Dimensional Culture System Enhances Anti-Inflammatory Characteristics through Cytoskeleton Regulation
Abstract
:1. Introduction
2. Results
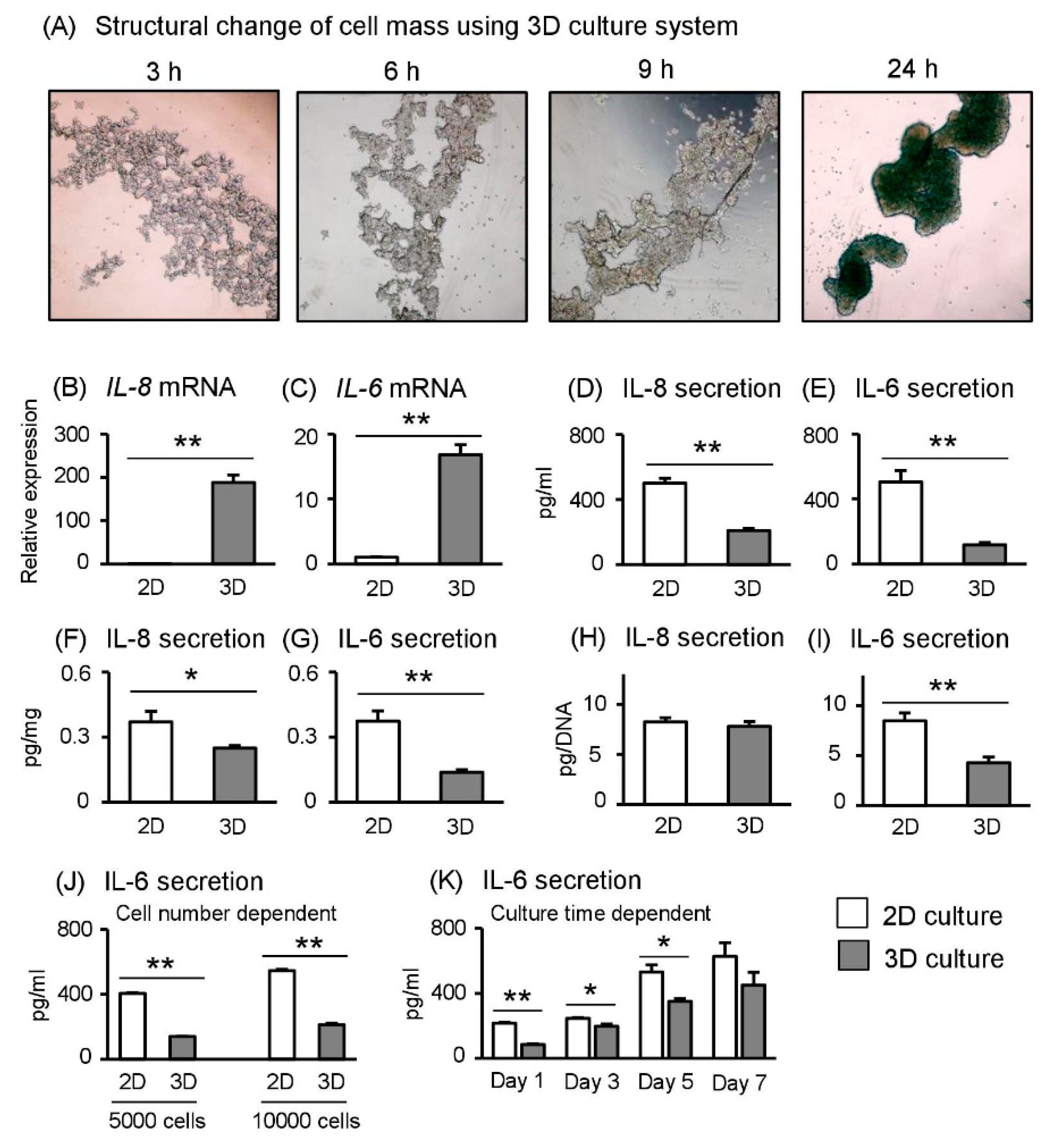
2.1. Aggregation of Human Trophoblast Cells in 3D Cultures
2.2. 3D–Cultured Sw.71 Cells Possess Distinct Gene Expression Patterns Compared with 2D–Cultured Cells
2.3. Spheroid Sw.71 Cells Grown under 3D Culture Conditions Reduce Inflammatory Cytokine Secretion
2.4. NF-κB Levels are Higher in Spheroid Sw.71 Cells
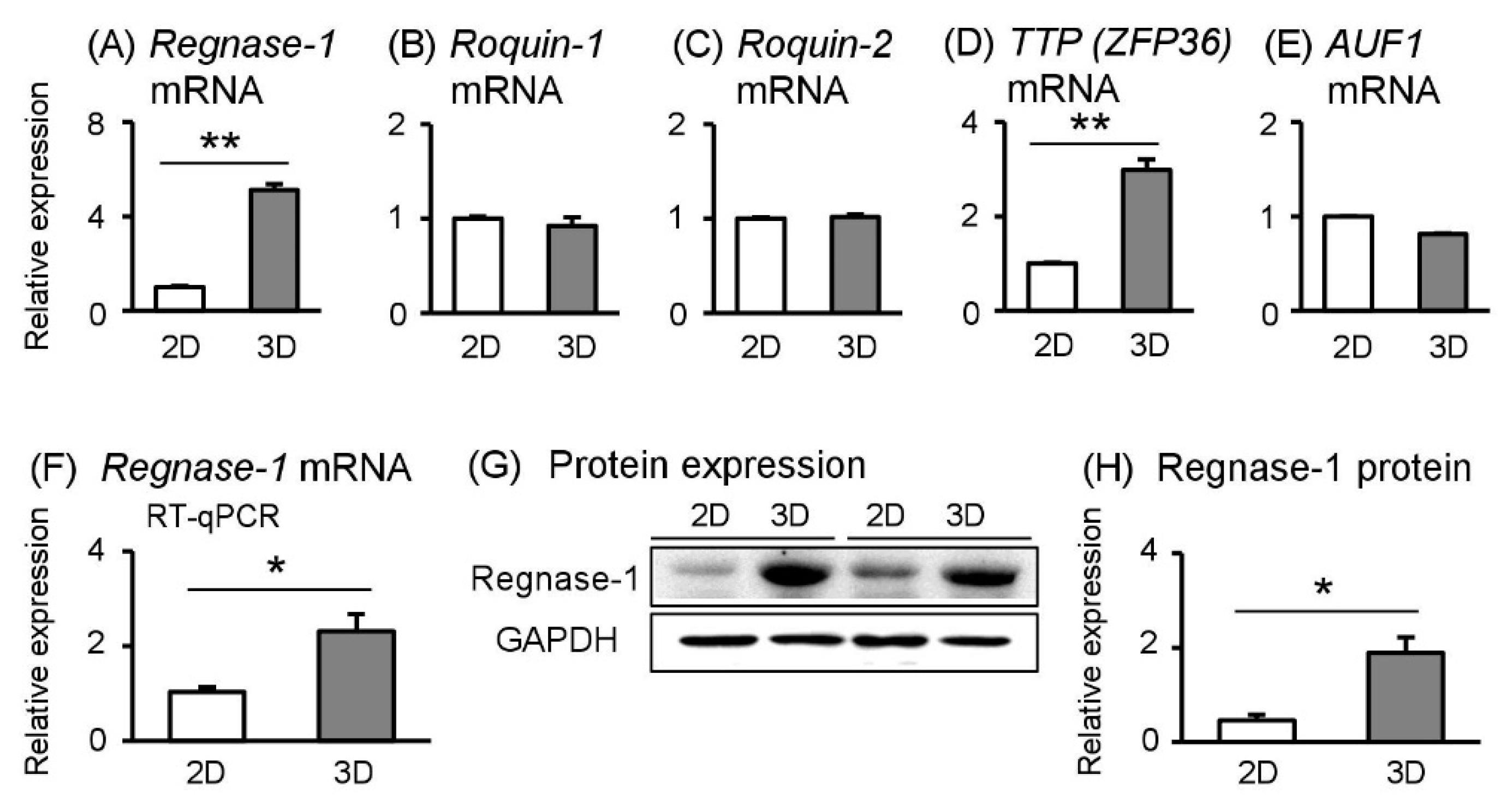
2.5. Post–Transcriptional Factor Regnase-1 More Abundant in Spheroid Sw.71 Cells
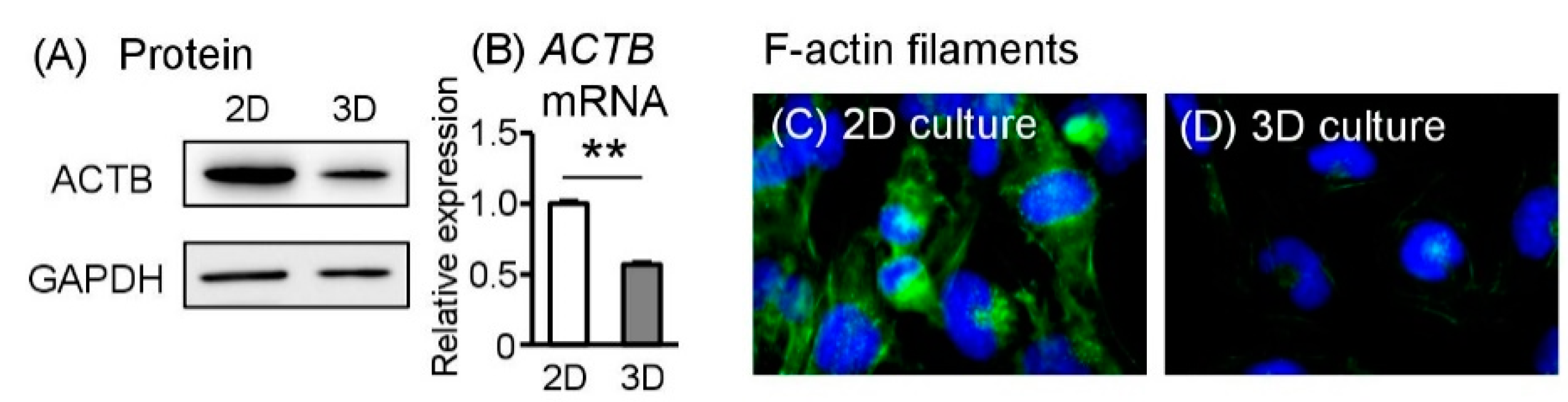
2.6. Spheroid Sw.71 Cells Change Cytoskeletal–Related Molecules
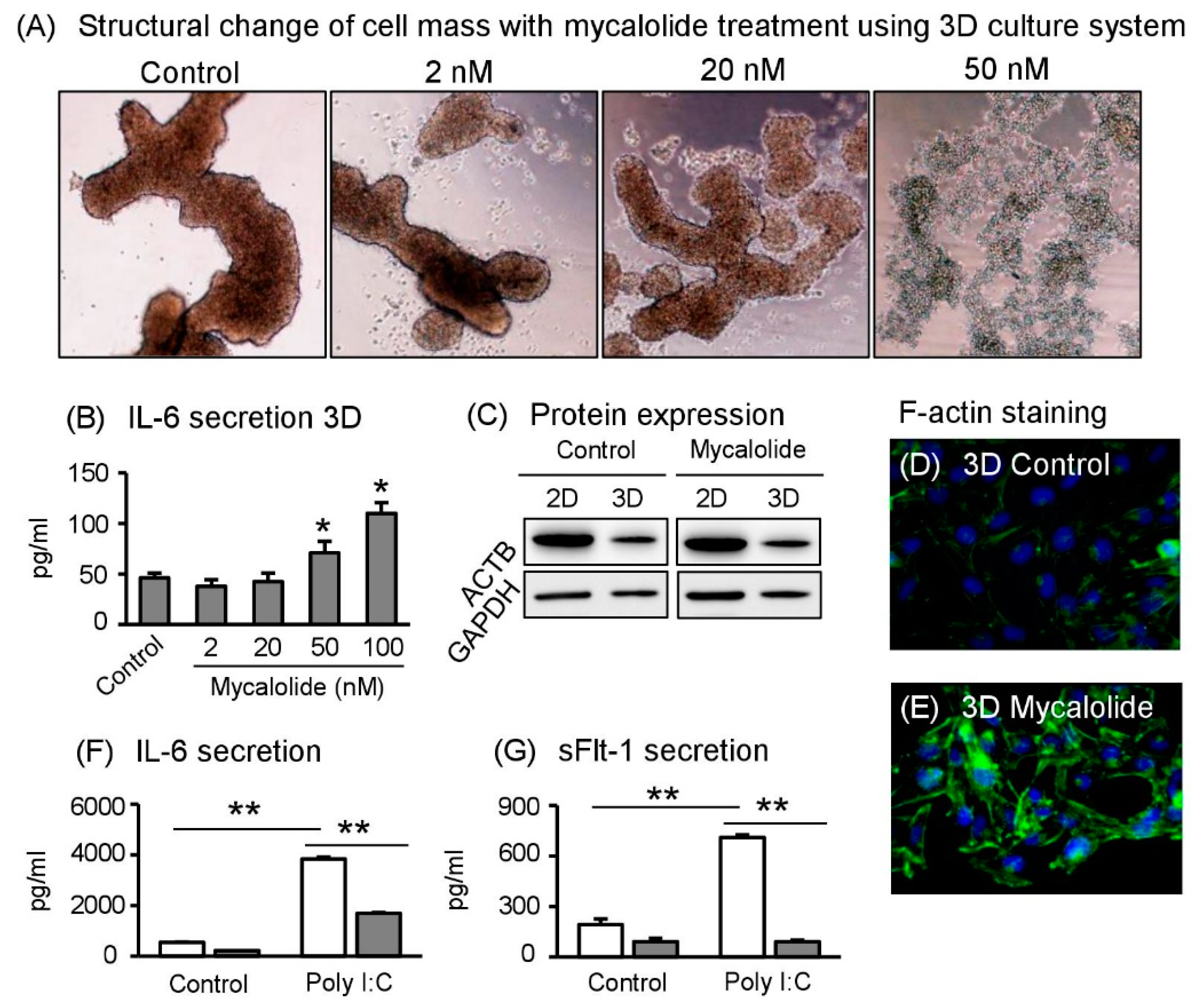
2.7. Actin Cytoskeleton Regulates 3D Cell Formation and IL–6 Secretion
2.8. Spheroid Sw.71 Cells Modulate Inflammatory Responses
3. Discussion
4. Materials and Methods
4.1. Human Trophoblast Cell Culture
4.2. Transcriptome Analysis of Human Trophoblast Cells
4.3. Real–Time RT-PCR
4.4. Determination of Cytokines
4.5. NF-κB p65 Activation Assay
4.6. Western Blot Analysis
4.7. Immunocytochemistry
4.8. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lee, K.Y.; DeMayo, F.J. Animal models of implantation. Reproduction 2004, 128, 679–695. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, H.; Pilla, F.; Anderson, S.; Martinez-Escribano, S.; Herrer, I.; Moreno-Moya, J.M.; Musti, S.; Bocca, S.; Oehninger, S.; Horcajadas, J.A. A novel model of human implantation: 3D endometrium-like culture system to study attachment of human trophoblast (Jar) cell spheroids. Mol. Hum. Reprod. 2012, 18, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Hutson, J.R.; Garcia-Bournissen, F.; Davis, A.; Koren, G. The human placental perfusion model: A systematic review and development of a model to predict in vivo transfer of therapeutic drugs. Clin. Pharmacol. Ther. 2011, 90, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Grafmueller, S.; Manser, P.; Diener, L.; Diener, P.A.; Maeder-Althaus, X.; Maurizi, L.; Jochum, W.; Krug, H.F.; Buerki-Thurnherr, T.; von Mandach, U.; et al. Bidirectional Transfer Study of Polystyrene Nanoparticles across the Placental Barrier in an ex Vivo Human Placental Perfusion Model. Environ. Health Perspect. 2015, 123, 1280–1286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muoth, C.; Wichser, A.; Monopoli, M.; Correia, M.; Ehrlich, N.; Loeschner, K.; Gallud, A.; Kucki, M.; Diener, L.; Manser, P.; et al. A 3D co-culture microtissue model of the human placenta for nanotoxicity assessment. Nanoscale 2016, 8, 17322–17332. [Google Scholar] [CrossRef] [PubMed]
- McConkey, C.A.; Delorme-Axford, E.; Nickerson, C.A.; Kim, K.S.; Sadovsky, Y.; Boyle, J.P.; Coyne, C.B. A three-dimensional culture system recapitulates placental syncytiotrophoblast development and microbial resistance. Sci. Adv. 2016, 2, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.M.; Cukierman, E. Modeling tissue morphogenesis and cancer in 3D. Cell 2007, 130, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Straszewski-Chavez, S.L.; Abrahams, V.M.; Alvero, A.B.; Aldo, P.B.; Ma, Y.; Guller, S.; Romero, R.; Mor, G. The isolation and characterization of a novel telomerase immortalized first trimester trophoblast cell line, Swan 71. Placenta 2009, 30, 939–948. [Google Scholar] [CrossRef] [PubMed]
- Shirasuna, K.; Seno, K.; Ohtsu, A.; Shiratsuki, S.; Ohkuchi, A.; Suzuki, H.; Matsubara, S.; Nagayama, S.; Iwata, H.; Kuwayama, T. AGEs and HMGB1 Increase Inflammatory Cytokine Production from Human Placental Cells, Resulting in an Enhancement of Monocyte Migration. Am. J. Reprod. Immunol. 2016, 75, 557–568. [Google Scholar] [CrossRef] [PubMed]
- Brasier, A.R. The nuclear factor-kappaB-interleukin-6 signalling pathway mediating vascular inflammation. Cardiovasc. Res. 2010, 86, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Uehata, T.; Akira, S. mRNA degradation by the endoribonuclease Regnase-1/ZC3H12a/MCPIP-1. Biochim. Biophys. Acta 2013, 1829, 708–713. [Google Scholar] [CrossRef] [PubMed]
- Matsushita, K.; Takeuchi, O.; Standley, D.M.; Kumagai, Y.; Kawagoe, T.; Miyake, T.; Satoh, T.; Kato, H.; Tsujimura, T.; Nakamura, H.; et al. Zc3h12a is an RNase essential for controlling immune responses by regulating mRNA decay. Nature 2009, 458, 1185–1190. [Google Scholar] [CrossRef] [PubMed]
- Saito, S.; Watabe, S.; Ozaki, H.; Fusetani, N.; Karaki, H.; Mycalolide, B. A novel actin depolymerizing agent. J. Biol. Chem. 1994, 269, 29710–29714. [Google Scholar] [PubMed]
- Wang, B.; Koga, K.; Osuga, Y.; Cardenas, I.; Izumi, G.; Takamura, M.; Hirata, T.; Yoshino, O.; Hirota, Y.; Harada, M.; et al. Toll-like receptor-3 ligation-induced indoleamine 2,3-dioxygenase expression in human trophoblasts. Endocrinology 2011, 152, 4984–4992. [Google Scholar] [CrossRef] [PubMed]
- Maynard, S.E.; Min, J.Y.; Merchan, J.; Lim, K.H.; Li, J.; Mondal, S.; Libermann, T.A.; Morgan, J.P.; Sellke, F.W.; Stillman, I.E.; et al. Excess placental soluble fms-like tyrosine kinase 1 (sFlt1) may contribute to endothelial dysfunction, hypertension, and proteinuria in preeclampsia. J. Clin. Investig. 2003, 111, 649–658. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Venkatesha, S.; Toporsian, M.; Lam, C.; Hanai, J.; Mammoto, T.; Kim, Y.M.; Bdolah, Y.; Lim, K.H.; Yuan, H.T.; Libermann, T.A.; et al. Soluble endoglin contributes to the pathogenesis of preeclampsia. Nat. Med. 2006, 12, 642–649. [Google Scholar] [CrossRef] [PubMed]
- Muthuswamy, S.K. 3D culture reveals a signaling network. Breast Cancer Res. 2011, 13, 103–105. [Google Scholar] [CrossRef] [PubMed]
- Multhaup, A.; Huppertz, B.; Gohner, C.; Bohringer, M.; Mai, M.; Markert, U.; Schleussner, E.; Groten, T. N-cadherin knockdown leads to disruption of trophoblastic and endothelial cell interaction in a 3D cell culture model–New insights in trophoblast invasion failure. Cell Adh. Migr. 2017, 6, 1–12. [Google Scholar] [CrossRef] [PubMed]
- DelNero, P.; Lane, M.; Verbridge, S.S.; Kwee, B.; Kermani, P.; Hempstead, B.; Stroock, A.; Fischbach, C. 3D culture broadly regulates tumor cell hypoxia response and angiogenesis via pro-inflammatory pathways. Biomaterials 2015, 55, 110–118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lipert, B.; Wilamowski, M.; Gorecki, A.; Jura, J. MCPIP1, alias Regnase-1 binds and cleaves mRNA of C./EBPbeta. PLoS ONE 2017, 12, 17–19. [Google Scholar] [CrossRef] [PubMed]
- Mino, T.; Murakawa, Y.; Fukao, A.; Vandenbon, A.; Wessels, H.H.; Ori, D.; Uehata, T.; Tartey, S.; Akira, S.; Suzuki, Y.; et al. Regnase-1 and Roquin Regulate a Common Element in Inflammatory mRNAs by Spatiotemporally Distinct Mechanisms. Cell 2015, 161, 1058–1073. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iwasaki, H.; Takeuchi, O.; Teraguchi, S.; Matsushita, K.; Uehata, T.; Kuniyoshi, K.; Satoh, T.; Saitoh, T.; Matsushita, M.; Standley, D.M.; et al. The IkappaB kinase complex regulates the stability of cytokine-encoding mRNA induced by TLR-IL-1R by controlling degradation of regnase-1. Nat. Immunol. 2011, 12, 1167–1175. [Google Scholar] [CrossRef] [PubMed]
- Bartosh, T.J.; Ylostalo, J.H.; Mohammadipoor, A.; Bazhanov, N.; Coble, K.; Claypool, K.; Lee, R.H.; Choi, H.; Prockop, D.J. Aggregation of human mesenchymal stromal cells (MSCs) into 3D spheroids enhances their antiinflammatory properties. Proc. Natl. Acad. Sci. USA 2010, 107, 13724–13729. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Redondo-Castro, E.; Cunningham, C.J.; Miller, J.; Brown, H.; Allan, S.M.; Pinteaux, E. Changes in the secretome of tri-dimensional spheroid-cultured human mesenchymal stem cells in vitro by interleukin-1 priming. Stem Cell Res. Ther. 2018, 9, 11–13. [Google Scholar] [CrossRef] [PubMed]
- Widera, D.; Mikenberg, I.; Kaus, A.; Kaltschmidt, C.; Kaltschmidt, B. Nuclear Factor-kappaB controls the reaggregation of 3D neurosphere cultures in vitro. Eur. Cell Mater. 2006, 11, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Chen, H.; Li, H.; Wu, Y. 3D culture increases pluripotent gene expression in mesenchymal stem cells through relaxation of cytoskeleton tension. J. Cell Mol. Med. 2017, 21, 1073–1084. [Google Scholar] [CrossRef] [PubMed]
- McBeath, R.; Pirone, D.M.; Nelson, C.M.; Bhadriraju, K.; Chen, C.S. Cell shape, cytoskeletal tension, and RhoA regulate stem cell lineage commitment. Dev. Cell 2004, 6, 483–495. [Google Scholar] [CrossRef]
- Song, Q.; Jiang, Z.; Li, N.; Liu, P.; Liu, L.; Tang, M.; Cheng, G. Anti-inflammatory effects of three-dimensional graphene foams cultured with microglial cells. Biomaterials 2014, 35, 6930–6940. [Google Scholar] [CrossRef] [PubMed]
- Chia, S.L.; Tay, C.Y.; Setyawati, M.I.; Leong, D.T. Biomimicry 3D gastrointestinal spheroid platform for the assessment of toxicity and inflammatory effects of zinc oxide nanoparticles. Small 2015, 11, 702–712. [Google Scholar] [CrossRef] [PubMed]
- Htwe, S.S.; Harrington, H.; Knox, A.; Rose, F.; Aylott, J.; Haycock, J.W.; Ghaemmaghami, A.M. Investigating NF-kappaB signaling in lung fibroblasts in 2D and 3D culture systems. Respir. Res. 2015, 16, 144–156. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Jackson, S.; Haycock, J.W.; MacNeil, S. Culture of skin cells in 3D rather than 2D improves their ability to survive exposure to cytotoxic agents. J. Biotechnol. 2006, 122, 372–381. [Google Scholar] [CrossRef] [PubMed]
- Grasso, E.; Paparini, D.; Hauk, V.; Salamone, G.; Leiros, C.P.; Ramhorst, R. Differential migration and activation profile of monocytes after trophoblast interaction. PLoS ONE 2014, 9, e97147. [Google Scholar] [CrossRef] [PubMed]
- Tanikawa, N.; Seno, K.; Kawahara-Miki, R.; Kimura, K.; Matsuyama, S.; Iwata, H.; Kuwayama, T.; Shirasuna, K. Interferon Tau Regulates Cytokine Production and Cellular Function in Human Trophoblast Cell Line. J. Interf. Cytokine Res. 2017, 37, 456–466. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta, C.(T.)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Seno, K.; Tanikawa, N.; Takahashi, H.; Ohkuchi, A.; Suzuki, H.; Matsubara, S.; Iwata, H.; Kuwayama, T.; Shirasuna, K. Oxygen concentration modulates cellular senescence and autophagy in human trophoblast cells. Am. J. Reprod. Immunol. 2018, 79, e128–e129. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seno, K.; Munakata, Y.; Sano, M.; Kawahara-Miki, R.; Takahashi, H.; Ohkuchi, A.; Iwata, H.; Kuwayama, T.; Shirasuna, K. Aggregation of Human Trophoblast Cells into Three-Dimensional Culture System Enhances Anti-Inflammatory Characteristics through Cytoskeleton Regulation. Int. J. Mol. Sci. 2018, 19, 2322. https://doi.org/10.3390/ijms19082322
Seno K, Munakata Y, Sano M, Kawahara-Miki R, Takahashi H, Ohkuchi A, Iwata H, Kuwayama T, Shirasuna K. Aggregation of Human Trophoblast Cells into Three-Dimensional Culture System Enhances Anti-Inflammatory Characteristics through Cytoskeleton Regulation. International Journal of Molecular Sciences. 2018; 19(8):2322. https://doi.org/10.3390/ijms19082322
Chicago/Turabian StyleSeno, Kotomi, Yasuhisa Munakata, Michiya Sano, Ryouka Kawahara-Miki, Hironori Takahashi, Akihide Ohkuchi, Hisataka Iwata, Takehito Kuwayama, and Koumei Shirasuna. 2018. "Aggregation of Human Trophoblast Cells into Three-Dimensional Culture System Enhances Anti-Inflammatory Characteristics through Cytoskeleton Regulation" International Journal of Molecular Sciences 19, no. 8: 2322. https://doi.org/10.3390/ijms19082322







