Detection of the Mitochondrial Membrane Potential by the Cationic Dye JC-1 in L1210 Cells with Massive Overexpression of the Plasma Membrane ABCB1 Drug Transporter
Abstract
:1. Introduction
2. Results
2.1. Characterization of S, R and T Variants of L1210 Cells
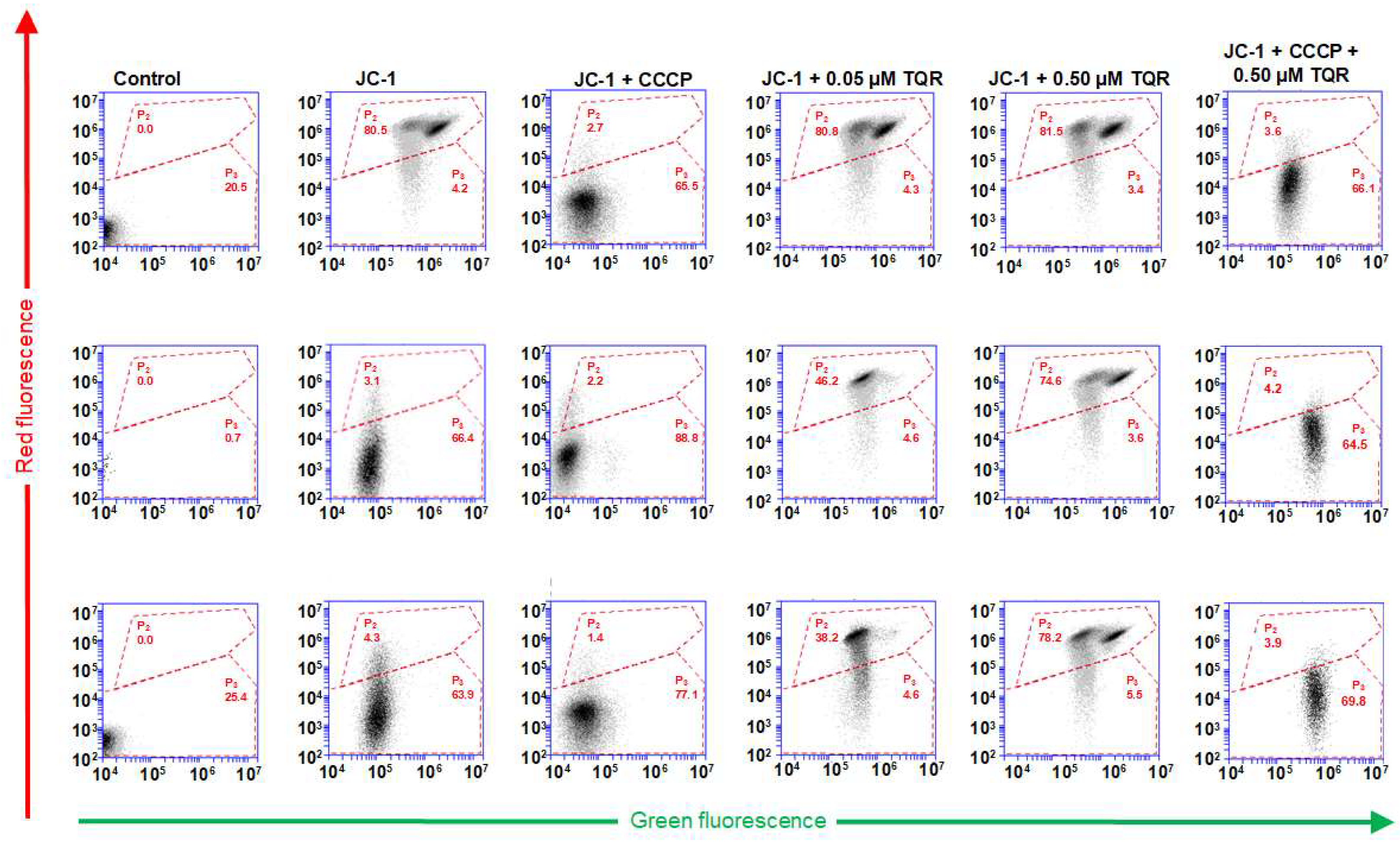
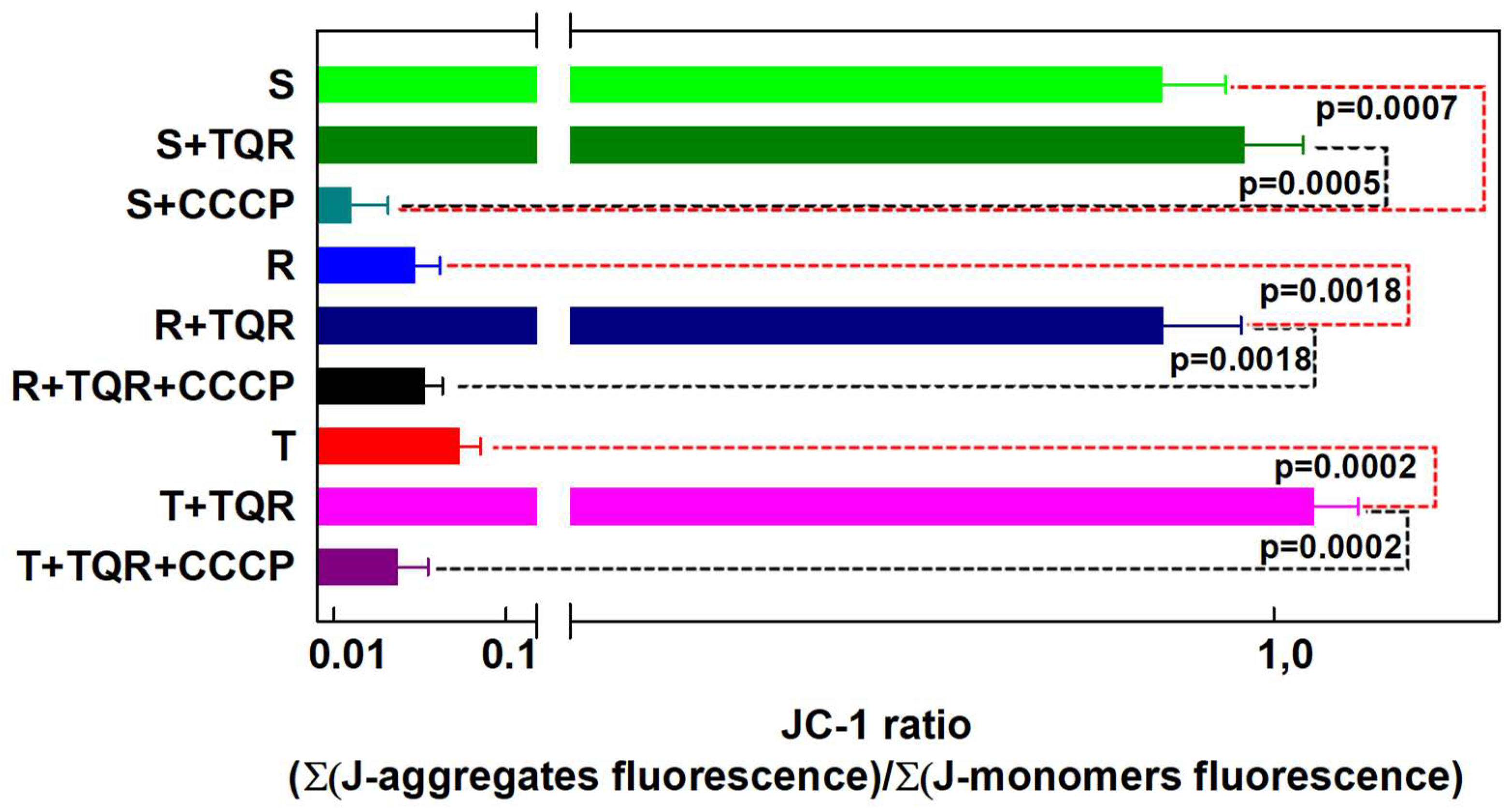
2.2. Measurement of JC-1 Fluorescence in S, R and T Cells by Fluorescence Cytometry
2.3. TQR as a Reversal Agent of P-gp-Mediated Drug Resistance in R and T Cells
2.4. Confocal Microscopy Detection JC-1 Labeling of S, R and T Cells
3. Discussion
4. Materials and Methods
4.1. Cell Cultivation Conditions and the Determination of P-gp Expression
4.2. Flow Cytometry Detection of JC-1 Fluorescence in S, R and T Cells
4.3. Confocal Microscopy of JC-1 Stained S, R and T Cells
4.4. Examination of the TQR Inhibitory Effect on P-gp Efflux Activity in S, R and T Cells using the Calcein/AM Assay
4.5. Measurement of the P-gp-Mediated Vincristine Resistance Reversal Induced by TQR
4.6. Statistical Analysis and Data Processing
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BCRP | Breast cancer resistance protein, ABCG2 protein |
| CCCP | Carbonyl cyanide 3-chlorophenylhydrazone |
| CSA | Cyclosporine A |
| DAPI | 4′,6-Diamidino-2-phenylindole |
| ΔΨ | Mitochondrial membrane potential |
| JC-1 | 5,6-Dichloro-2-[(E)-3-(5,6-dichloro-1,3-diethylbenzimidazol-3-ium-2-yl)prop-2-enylidene]-1,3-diethylbenzimidazole iodide |
| MDR | Multidrug resistance |
| MRP(1-3) | Multidrug resistance-associated proteins (1-3), ABCC (1-3) proteins |
| MTT | ([3-(4,5-Dimethyldiazol-2-yl)-2,5 diphenyltetrazolium bromide]) |
| P-gp | P-glycoprotein, ABCB1 protein |
| R | P-gp-positive variant of L1210 cells obtained by selection with vincristine |
| S | P-gp-negative L1210 |
| T | P-gp-positive variants of L1210 cells obtained by transfection gene encoding P-gp |
| TQR | Tariquidar |
| VCR | Vincristine |
| VER | Verapamil |
References
- Seppet, E.; Gruno, M.; Peetsalu, A.; Gizatullina, Z.; Nguyen, H.P.; Vielhaber, S.; Wussling, M.H.; Trumbeckaite, S.; Arandarcikaite, O.; Jerzembeck, D.; et al. Mitochondria and energetic depression in cell pathophysiology. Int. J. Mol. Sci. 2009, 10, 2252–2303. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Lazareno, P.C.; Bravo-Cuellar, A.; Lerma-Diaz, J.M.; Jave-Suarez, L.F.; Aguilar-Lemarroy, A.; Dominguez-Rodriguez, J.R.; Gonzalez-Ramella, O.; de Celis, R.; Gomez-Lomeli, P.; Hernandez-Flores, G. Sensitization of u937 leukemia cells to doxorubicin by the mg132 proteasome inhibitor induces an increase in apoptosis by suppressing NF-κB and mitochondrial membrane potential loss. Cancer Cell Int. 2014, 14, 13. [Google Scholar] [CrossRef] [PubMed]
- Salvioli, S.; Ardizzoni, A.; Franceschi, C.; Cossarizza, A. JC-1, but not DiOC6(3) or rhodamine 123, is a reliable fluorescent probe to assess delta psi changes in intact cells: Implications for studies on mitochondrial functionality during apoptosis. FEBS Lett. 1997, 411, 77–82. [Google Scholar] [CrossRef]
- Chazotte, B. Labeling mitochondria with JC-1. Cold Spring Harb Protoc 2011, 2011. [Google Scholar] [CrossRef] [PubMed]
- Cottet-Rousselle, C.; Ronot, X.; Leverve, X.; Mayol, J.F. Cytometric assessment of mitochondria using fluorescent probes. Cytometry A 2011, 79, 405–425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Du, Y.; Chen, B. Detection approaches for multidrug resistance genes of leukemia. Drug Des. Devel. Ther. 2017, 11, 1255–1261. [Google Scholar] [CrossRef] [PubMed]
- Breier, A.; Barancik, M.; Sulova, Z.; Uhrik, B. P-glycoprotein—Implications of metabolism of neoplastic cells and cancer therapy. Curr. Cancer Drug Targets 2005, 5, 457–468. [Google Scholar] [CrossRef] [PubMed]
- Breier, A.; Gibalova, L.; Seres, M.; Barancik, M.; Sulova, Z. New insight into P-glycoprotein as a drug target. Anticancer Agents Med. Chem. 2013, 13, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Gillet, J.P.; Gottesman, M.M. Advances in the molecular detection of ABC transporters involved in multidrug resistance in cancer. Curr. Pharm. Biotechnol. 2011, 12, 686–692. [Google Scholar] [CrossRef] [PubMed]
- Sharom, F.J. Abc multidrug transporters: Structure, function and role in chemoresistance. Pharmacogenomics 2008, 9, 105–127. [Google Scholar] [CrossRef] [PubMed]
- Nerada, Z.; Hegyi, Z.; Szepesi, A.; Toth, S.; Hegedus, C.; Varady, G.; Matula, Z.; Homolya, L.; Sarkadi, B.; Telbisz, A. Application of fluorescent dye substrates for functional characterization of ABC multidrug transporters at a single cell level. Cytometry A 2016, 89, 826–834. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuhnel, J.M.; Perrot, J.Y.; Faussat, A.M.; Marie, J.P.; Schwaller, M.A. Functional assay of multidrug resistant cells using JC-1, a carbocyanine fluorescent probe. Leukemia 1997, 11, 1147–1155. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Legrand, O.; Perrot, J.Y.; Simonin, G.; Baudard, M.; Marie, J.P. JC-1: A very sensitive fluorescent probe to test P-gp activity in adult acute myeloid leukemia. Blood 2001, 97, 502–508. [Google Scholar] [CrossRef] [PubMed]
- Chaoui, D.; Faussat, A.M.; Majdak, P.; Tang, R.; Perrot, J.Y.; Pasco, S.; Klein, C.; Marie, J.P.; Legrand, O. JC-1, a sensitive probe for a simultaneous detection of P-glycoprotein activity and apoptosis in leukemic cells. Cytom. B Clin. Cytom. 2006, 70, 189–196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Polekova, L.; Barancik, M.; Mrazova, T.; Pirker, R.; Wallner, J.; Sulova, Z.; Breier, A. Adaptation of mouse leukemia cells l1210 to vincristine. Evidence for expression of P-glycoprotein. Neoplasma 1992, 39, 73–77. [Google Scholar] [PubMed]
- Sulova, Z.; Ditte, P.; Kurucova, T.; Polakova, E.; Rogozanova, K.; Gibalova, L.; Seres, M.; Skvarkova, L.; Sedlak, J.; Pastorek, J.; et al. The presence of P-glycoprotein in l1210 cells directly induces down-regulation of cell surface saccharide targets of concanavalin a. Anticancer Res. 2010, 30, 3661–3668. [Google Scholar] [PubMed]
- Anonymous. Application note: JC-1 mitochondrial membrane potential. Accuri Cytometers Inc. 2009, 10, 1–6. [Google Scholar]
- Schuetz, J.D.; Schuetz, E.G. Extracellular matrix regulation of multidrug resistance in primary monolayer cultures of adult rat hepatocytes. Cell Growth Differ. 1993, 4, 31–40. [Google Scholar] [PubMed]
- Pasquier, J.; Rioult, D.; Abu-Kaoud, N.; Marie, S.; Rafii, A.; Guerrouahen, B.S.; Le Foll, F. P-glycoprotein-activity measurements in multidrug resistant cell lines: Single-cell versus single-well population fluorescence methods. Biomed. Res. Int. 2013, 2013, 676845. [Google Scholar] [CrossRef] [PubMed]
- Silva, R.; Vilas-Boas, V.; Carmo, H.; Dinis-Oliveira, R.J.; Carvalho, F.; de Lourdes Bastos, M.; Remiao, F. Modulation of P-glycoprotein efflux pump: Induction and activation as a therapeutic strategy. Pharmacol. Ther. 2015, 149, 1–123. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.W.; Pan, Q.C.; Lin, G.Y. Compared study on Fura-2/AM assay and mtt assay for screening multidrug resistant modulators. Yao Xue Xue Bao 1997, 32, 401–405. [Google Scholar] [PubMed]
- Nelson, E.J.; Zinkin, N.T.; Hinkle, P.M. Fluorescence methods to assess multidrug resistance in individual cells. Cancer Chemother. Pharmacol. 1998, 42, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Orlicky, J.; Sulova, Z.; Dovinova, I.; Fiala, R.; Zahradnikova, A., Jr.; Breier, A. Functional Fluo-3/AM assay on P-glycoprotein transport activity in l1210/vcr cells by confocal microscopy. Gen. Physiol. Biophys. 2004, 23, 357–366. [Google Scholar] [PubMed]
- Marques-Santos, L.F.; Oliveira, J.G.; Maia, R.C.; Rumjanek, V.M. Mitotracker green is a P-glycoprotein substrate. Biosci. Rep. 2003, 23, 199–212. [Google Scholar] [CrossRef] [PubMed]
- Sulova, Z.; Orlicky, J.; Fiala, R.; Dovinova, I.; Uhrik, B.; Seres, M.; Gibalova, L.; Breier, A. Expression of P-glycoprotein in l1210 cells is linked with rise in sensitivity to Ca2+. Biochem. Biophys. Res. Commun. 2005, 335, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Sulova, Z.; Seres, M.; Barancik, M.; Gibalova, L.; Uhrik, B.; Polekova, L.; Breier, A. Does any relationship exist between P-glycoprotein-mediated multidrug resistance and intracellular calcium homeostasis. Gen. Physiol. Biophys. 2009, 28, F89–F95. [Google Scholar] [PubMed]
- Michaelis, M.; Rothweiler, F.; Wurglics, M.; Aniceto, N.; Dittrich, M.; Zettl, H.; Wiese, M.; Wass, M.; Ghafourian, T.; Schubert-Zsilavecz, M.; et al. Substrate-specific effects of pirinixic acid derivatives on abcb1-mediated drug transport. Oncotarget 2016, 7, 11664–11676. [Google Scholar] [CrossRef] [PubMed]
- Didziapetris, R.; Japertas, P.; Avdeef, A.; Petrauskas, A. Classification analysis of P-glycoprotein substrate specificity. J. Drug Target. 2003, 11, 391–406. [Google Scholar] [CrossRef] [PubMed]
- Kim, R.B. Drugs as P-glycoprotein substrates, inhibitors, and inducers. Drug Metab. Rev. 2002, 34, 47–54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Litman, T.; Zeuthen, T.; Skovsgaard, T.; Stein, W.D. Competitive, non-competitive and cooperative interactions between substrates of P-glycoprotein as measured by its atpase activity. Biochim. Biophys. Acta. 1997, 1361, 169–176. [Google Scholar] [CrossRef]
- Swerts, K.; de Moerloose, B.; Dhooge, C.; Noens, L.; Laureys, G.; Benoit, Y.; Philippe, J. Comparison of two functional flow cytometric assays to assess P-gp activity in acute leukemia. Leuk. Lymphoma. 2004, 45, 2221–2228. [Google Scholar] [CrossRef] [PubMed]
- Wang, E.J.; Casciano, C.N.; Clement, R.P.; Johnson, W.W. Active transport of fluorescent P-glycoprotein substrates: Evaluation as markers and interaction with inhibitors. Biochem. Biophys. Res. Commun. 2001, 289, 580–585. [Google Scholar] [CrossRef] [PubMed]
- Kosztyu, P.; Bukvova, R.; Dolezel, P.; Mlejnek, P. Resistance to daunorubicin, imatinib, or nilotinib depends on expression levels of ABCB1 and ABCG2 in human leukemia cells. Chem. Biol. Interact. 2014, 219, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Kosztyu, P.; Dolezel, P.; Mlejnek, P. Can P-glycoprotein mediate resistance to nilotinib in human leukaemia cells? Pharmacol. Res. 2013, 67, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Pavlikova, L.; Seres, M.; Imrichova, D.; Hano, M.; Rusnak, A.; Zamorova, M.; Katrlik, J.; Breier, A.; Sulova, Z. The expression of P-gp in leukemia cells is associated with cross-resistance to protein n-glycosylation inhibitor tunicamycin. Gen. Physiol. Biophys. 2016, 35, 497–510. [Google Scholar] [CrossRef] [PubMed]
- Mlejnek, P.; Kosztyu, P.; Dolezel, P.; Bates, S.E.; Ruzickova, E. Reversal of ABCB1 mediated efflux by imatinib and nilotinib in cells expressing various transporter levels. Chem. Biol. Interact. 2017, 273, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Fox, E.; Bates, S.E. Tariquidar (XR9576): A P-glycoprotein drug efflux pump inhibitor. Expert Rev. Anticancer Ther. 2007, 7, 447–459. [Google Scholar] [CrossRef] [PubMed]
- Martin, C.; Berridge, G.; Mistry, P.; Higgins, C.; Charlton, P.; Callaghan, R. The molecular interaction of the high affinity reversal agent xr9576 with P-glycoprotein. Br. J. Pharmacol. 1999, 128, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Weidner, L.D.; Fung, K.L.; Kannan, P.; Moen, J.K.; Kumar, J.S.; Mulder, J.; Innis, R.B.; Gottesman, M.M.; Hall, M.D. Tariquidar is an inhibitor and not a substrate of human and mouse P-glycoprotein. Drug Metab. Dispos. 2016, 44, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Mistry, P.; Stewart, A.J.; Dangerfield, W.; Okiji, S.; Liddle, C.; Bootle, D.; Plumb, J.A.; Templeton, D.; Charlton, P. In vitro and in vivo reversal of P-glycoprotein-mediated multidrug resistance by a novel potent modulator, xr9576. Cancer Res. 2001, 61, 749–758. [Google Scholar] [PubMed]
- Tenopoulou, M.; Kurz, T.; Doulias, P.T.; Galaris, D.; Brunk, U.T. Does the calcein-am method assay the total cellular ‘labile iron pool’ or only a fraction of it? Biochem. J. 2007, 403, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Eneroth, A.; Astrom, E.; Hoogstraate, J.; Schrenk, D.; Conrad, S.; Kauffmann, H.M.; Gjellan, K. Evaluation of a vincristine resistant caco-2 cell line for use in a calcein am extrusion screening assay for P-glycoprotein interaction. Eur. J. Pharm. Sci. 2001, 12, 205–214. [Google Scholar] [CrossRef]
- Seres, M.; Cholujova, D.; Bubencikova, T.; Breier, A.; Sulova, Z. Tunicamycin depresses P-glycoprotein glycosylation without an effect on its membrane localization and drug efflux activity in l1210 cells. Int. J. Mol. Sci. 2011, 12, 7772–7784. [Google Scholar] [CrossRef] [PubMed]
- Glavinas, H.; von Richter, O.; Vojnits, K.; Mehn, D.; Wilhelm, I.; Nagy, T.; Janossy, J.; Krizbai, I.; Couraud, P.; Krajcsi, P. Calcein assay: A high-throughput method to assess P-gp inhibition. Xenobiotica 2011, 41, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Strouse, J.J.; Ivnitski-Steele, I.; Waller, A.; Young, S.M.; Perez, D.; Evangelisti, A.M.; Ursu, O.; Bologa, C.G.; Carter, M.B.; Salas, V.M.; et al. Fluorescent substrates for flow cytometric evaluation of efflux inhibition in abcb1, abcc1, and abcg2 transporters. Anal. Biochem. 2013, 437, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Bohacova, V.; Sulova, Z.; Dovinova, I.; Polakova, E.; Barancik, M.; Uhrik, B.; Orlicky, J.; Breier, A. L1210 cells cultivated under the selection pressure of doxorubicin or vincristine express common mechanisms of multidrug resistance based on the overexpression of P-glycoprotein. Toxicol. In Vitro 2006, 20, 1560–1568. [Google Scholar] [CrossRef] [PubMed]
- Kannan, P.; Telu, S.; Shukla, S.; Ambudkar, S.V.; Pike, V.W.; Halldin, C.; Gottesman, M.M.; Innis, R.B.; Hall, M.D. The "specific" P-glycoprotein inhibitor tariquidar is also a substrate and an inhibitor for breast cancer resistance protein (BCRP/ABCG2). ACS Chem. Neurosci. 2011, 2, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Gibalova, L.; Seres, M.; Rusnak, A.; Ditte, P.; Labudova, M.; Uhrik, B.; Pastorek, J.; Sedlak, J.; Breier, A.; Sulova, Z. P-glycoprotein depresses cisplatin sensitivity in l1210 cells by inhibiting cisplatin-induced caspase-3 activation. Toxicol. In Vitro 2012, 26, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Keil, V.C.; Funke, F.; Zeug, A.; Schild, D.; Muller, M. Ratiometric high-resolution imaging of JC-1 fluorescence reveals the subcellular heterogeneity of astrocytic mitochondria. Pflugers Arch 2011, 462, 693–708. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pastan, I.; Gottesman, M.M.; Ueda, K.; Lovelace, E.; Rutherford, A.V.; Willingham, M.C. A retrovirus carrying an mdr1 cdna confers multidrug resistance and polarized expression of P-glycoprotein in mdck cells. Proc. Natl. Acad. Sci. USA 1988, 85, 4486–4490. [Google Scholar] [CrossRef] [PubMed]
- Breier, A.; Stetka, J.; Bohacova, V.; Macejova, D.; Brtko, J.; Sulova, Z. Effect of 9-cis retinoic acid and all-trans retinoic acid in combination with verapamil on P-glycoprotein expression in l1210 cells. Neoplasma 2014, 61, 553–565. [Google Scholar] [CrossRef] [PubMed]
- Turakova, K.; Pavlikova, L.; Messingerova, L.; Lakatos, B.; Breier, A.; Sulova, Z. Reduced UDP-glucose levels are associated with P-glycoprotein over-expression in l1210 cells and limit glucosylceramide synthase activity. Anticancer Res. 2015, 35, 2627–2634. [Google Scholar] [PubMed]



| Control | TQR (0.05 μM) | TQR (0.50 μM) | |||||
|---|---|---|---|---|---|---|---|
| VCR (0.25 μM) | VCR (0.25 μM) | VCR (0.25 μM) | |||||
| S cells | means ± SD | 100.0 ± 14.7 | 10.5 ± 3.4 | 95.2 ± 12.8 | 11.5 ± 6.5 | 95.2 ± 10.3 | 11.8 ± 5.1 |
| Student‘s t-test | t-value | 11.9 | 11.7 | 13.2 | |||
| p-value | 2.1 × 10−5 | 2.4 × 10−5 | 1.2 × 10−5 | ||||
| R cells | means ± SD | 100.0 ± 17.9 | 95.6 ± 9.2 | 103.4 ± 9.3 | 67.8 ± 9.5 | 95.8 ± 18.0 | 11.3 ± 5.6 |
| Student‘s t-test | t-value | 0.43 | 5.4 | 9.0 | |||
| p-value | 0.68 | 1.7 × 10−3 | 1.1 × 10−4 | ||||
| T-cells | means±SD | 100.0 ± 17.2 | 97.2 ± 15.1 | 104.4 ± 16.6 | 36.5 ± 12.0 | 104.2 ± 15.2 | 11.5 ± 4.3 |
| Student‘s t-test | t-value | 0.24 | 6.6 | 11.7 | |||
| p-value | 0.82 | 5.6 × 10−4 | 2.3 × 10−5 | ||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elefantova, K.; Lakatos, B.; Kubickova, J.; Sulova, Z.; Breier, A. Detection of the Mitochondrial Membrane Potential by the Cationic Dye JC-1 in L1210 Cells with Massive Overexpression of the Plasma Membrane ABCB1 Drug Transporter. Int. J. Mol. Sci. 2018, 19, 1985. https://doi.org/10.3390/ijms19071985
Elefantova K, Lakatos B, Kubickova J, Sulova Z, Breier A. Detection of the Mitochondrial Membrane Potential by the Cationic Dye JC-1 in L1210 Cells with Massive Overexpression of the Plasma Membrane ABCB1 Drug Transporter. International Journal of Molecular Sciences. 2018; 19(7):1985. https://doi.org/10.3390/ijms19071985
Chicago/Turabian StyleElefantova, Katarina, Boris Lakatos, Jana Kubickova, Zdena Sulova, and Albert Breier. 2018. "Detection of the Mitochondrial Membrane Potential by the Cationic Dye JC-1 in L1210 Cells with Massive Overexpression of the Plasma Membrane ABCB1 Drug Transporter" International Journal of Molecular Sciences 19, no. 7: 1985. https://doi.org/10.3390/ijms19071985






