Administration of Oxygen Ultra-Fine Bubbles Improves Nerve Dysfunction in a Rat Sciatic Nerve Crush Injury Model
Abstract
:1. Introduction
2. Results
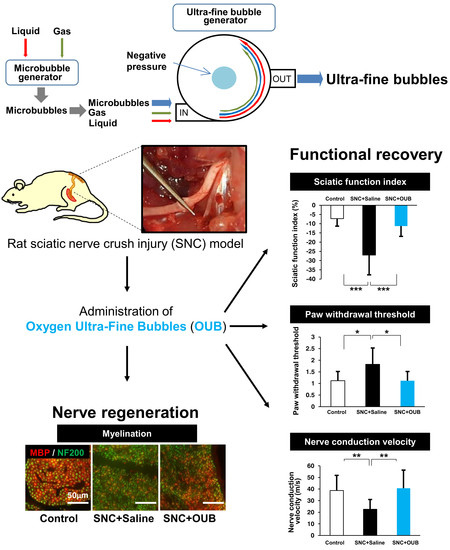
2.1. OUBs Improve Dysfunction after Sciatic Nerve Crush Injury in Rats
2.2. OUBs Promote Neurite Outgrowth in DRG Neurons
2.3. OUBs Have No Effects on the Differentiation of SCs
2.4. OUBs Stimulate the Proliferation of SCs
2.5. OUBs Induce the Expression of Regeneration-Related Factors in SCs
2.6. OUBs Accelerate Proliferation and Inhibit Apoptosis of SCs under Hypoxic Conditions In Vitro
3. Discussion
4. Materials and Methods
4.1. Preparation of UFBs Diluted in Medium and Saline
4.2. Animals
4.3. Surgical Procedures
4.4. SFI Analysis
4.5. von Frey Filament Test
4.6. Electrophysiological Analysis
4.7. Immunostaining of Sciatic Nerves
4.8. Primary Culture of DRG Neurons and SCs
4.9. Immunocytochemistry
4.10. Neurite Outgrowth Assay
4.11. WB
4.12. Gene Expression Analysis
4.13. SC Differentiation Assay
4.14. Cell Proliferation Assay
4.15. BrdU Uptake Assay
4.16. Apoptosis Assay
4.17. Statistics
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| ANOVA | analysis of variance |
| BDNF | brain-derived neurotrophic factor |
| BrdU | 5-bromo-2-deoxyuridine |
| BSA | bovine serum albumin |
| CMAP | compound muscle action potential |
| db-cAMP | dibutyryl cyclic AMP |
| DMEM | Dulbecco’s Modified Eagle’s Medium |
| DRG | dorsal root ganglion |
| ERK | extracellular signal-regulated kinase |
| GDNF | glial cell-derived neurotrophic factor |
| HIF1α | hypoxia-inducible factor 1α |
| HPRT1 | hypoxanthine phosphoribosyltransferase 1 |
| IGF-1 | insulin-like growth factor-1 |
| ITS | intermediary toe spread |
| JNK | c-Jun-N-terminal kinase |
| MAG | myelin-associated glycoprotein |
| MBP | myelin basic protein |
| NCV | nerve conduction velocity |
| NF200 | neurofilament 200 |
| NGF | nerve growth factor |
| ns | not significant |
| NUBs | nitrogen ultra-fine bubbles |
| Oct-6 | octamer transcription factor-6 |
| OUBs | oxygen ultra-fine bubbles |
| P0 | protein zero |
| PBS | phosphate-buffered saline |
| PCR | polymerase chain reaction |
| PDGF-BB | platelet-derived growth factor-beta |
| PL | print length |
| Pmp22 | peripheral myelin protein 22 |
| PNI | peripheral nerve injury |
| PNS | peripheral nervous system |
| SCs | Schwann cells |
| SD | standard deviation |
| SFI | sciatic functional index |
| SNC | sciatic nerve crush injury |
| TL | terminal latency |
| TS | toe spread |
| Tuj1 | neuronal class III β-tubulin |
| UFBs | ultra-fine bubbles |
| VEGF | vascular endothelial growth factor |
| WB | western blotting |
References
- Agarwal, A.; Ng, W.J.; Liu, Y. Principle and applications of microbubble and nanobubble technology for water treatment. Chemosphere 2011, 84, 1175–1180. [Google Scholar] [CrossRef] [PubMed]
- Ebina, K.; Shi, K.; Hirao, M.; Hashimoto, J.; Kawato, Y.; Kaneshiro, S.; Morimoto, T.; Koizumi, K.; Yoshikawa, H. Oxygen and air nanobubble water solution promote the growth of plants, fishes, and mice. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, M.; Chiba, K.; Li, P. Free-radical generation from collapsing microbubbles in the absence of a dynamic stimulus. J. Phys. Chem. B 2007, 111, 1343–1347. [Google Scholar] [CrossRef] [PubMed]
- Matsuki, N.; Ichiba, S.; Ishikawa, T.; Nagano, O.; Takeda, M.; Ujike, Y.; Yamaguchi, T. Blood oxygenation using microbubble suspensions. Eur. Biophys. J. 2012, 41, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Bitterman, H. Bench-to-bedside review: Oxygen as a drug. Crit. Care 2009, 13, 205. [Google Scholar] [CrossRef] [PubMed]
- Abdelsalam, M.; Cheifetz, I.M. Goal-directed therapy for severely hypoxic patients with acute respiratory distress syndrome: Permissive hypoxemia. Respir. Care 2010, 55, 1483–1490. [Google Scholar] [PubMed]
- Guo, S.; Dipietro, L.A. Factors affecting wound healing. J. Dent. Res. 2010, 89, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, X.; Zhou, Y.; Huang, P.; Xu, Y. Preparation of nanobubbles for ultrasound imaging and intracelluar drug delivery. Int. J. Pharm. 2010, 384, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, T.; Ebina, K.; Hirao, M.; Morimoto, T.; Koizumi, K.; Kitaguchi, K.; Matsuoka, H.; Iwahashi, T.; Yoshikawa, H. Oxygen ultra-fine bubbles water administration prevents bone loss of glucocorticoid-induced osteoporosis in mice by suppressing osteoclast differentiation. Osteoporos. Int. 2017, 28, 1063–1075. [Google Scholar] [CrossRef] [PubMed]
- Hirose, Y.; Yasui, T.; Taguchi, K.; Fujii, Y.; Niimi, K.; Hamamoto, S.; Okada, A.; Kubota, Y.; Kawai, N.; Itoh, Y.; et al. Oxygen nano-bubble water reduces calcium oxalate deposits and tubular cell injury in ethylene glycol-treated rat kidney. Urolithiasis 2013, 41, 279–294. [Google Scholar] [CrossRef] [PubMed]
- Eser, F.; Aktekin, L.A.; Bodur, H.; Atan, C. Etiological factors of traumatic peripheral nerve injuries. Neurol. India 2009, 57, 434–437. [Google Scholar] [CrossRef] [PubMed]
- Martyn, C.N.; Hughes, R.A. Epidemiology of peripheral neuropathy. J. Neurol. Neurosurg. Psychiatry 1997, 62, 310–318. [Google Scholar] [CrossRef] [PubMed]
- Painter, M.W.; Brosius Lutz, A.; Cheng, Y.C.; Latremoliere, A.; Duong, K.; Miller, C.M.; Posada, S.; Cobos, E.J.; Zhang, A.X.; Wagers, A.J.; et al. Diminished schwann cell repair responses underlie age-associated impaired axonal regeneration. Neuron 2014, 83, 331–343. [Google Scholar] [CrossRef] [PubMed]
- Heinen, A.; Beyer, F.; Tzekova, N.; Hartung, H.P.; Kury, P. Fingolimod induces the transition to a nerve regeneration promoting schwann cell phenotype. Exp. Neurol. 2015, 271, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Hoyng, S.A.; de Winter, F.; Tannemaat, M.R.; Blits, B.; Malessy, M.J.; Verhaagen, J. Gene therapy and peripheral nerve repair: A perspective. Front. Mol. Neurosci. 2015, 8, 32. [Google Scholar] [CrossRef] [PubMed]
- Eguiluz-Ordonez, R.; Sanchez, C.E.; Venegas, A.; Figueroa-Granados, V.; Hernandez-Pando, R. Effects of hyperbaric oxygen on peripheral nerves. Plast. Reconstr. Surg. 2006, 118, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Haapaniemi, T.; Nylander, G.; Kanje, M.; Dahlin, L. Hyperbaric oxygen treatment enhances regeneration of the rat sciatic nerve. Exp. Neurol. 1998, 149, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Bajrovic, F.F.; Sketelj, J.; Jug, M.; Gril, I.; Mekjavic, I.B. The effect of hyperbaric oxygen treatment on early regeneration of sensory axons after nerve crush in the rat. J. Peripher. Nerv. Syst. 2002, 7, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.W. Therapeutic effect of hyperbaric oxygen on recovery of surgically repaired peripheral nerve injury. Zhonghua Wai Ke Za Zhi 1991, 29, 118–120. [Google Scholar] [PubMed]
- Viera, C.; Galvez, C.; Carrasco, B.; Santos, C.; Castellanos, R. A study of peripheral neural conduction, motor and sensory, in diabetic patients treated with hyperbaric oxygenation. Rev. Neurol. 1999, 28, 868–872. [Google Scholar] [PubMed]
- Wang, Y.; Qi, F.; Zhu, S.; Ye, Z.; Ma, T.; Hu, X.; Huang, J.; Luo, Z. A synthetic oxygen carrier in fibrin matrices promotes sciatic nerve regeneration in rats. Acta Biomater. 2013, 9, 7248–7263. [Google Scholar] [CrossRef] [PubMed]
- Gaudet, A.D.; Popovich, P.G.; Ramer, M.S. Wallerian degeneration: Gaining perspective on inflammatory events after peripheral nerve injury. J. Neuroinflamm. 2011, 8, 110. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Tanaka, H.; Ebara, M.; Uto, K.; Matsuoka, H.; Nishimoto, S.; Okada, K.; Murase, T.; Yoshikawa, H. Electrospun nanofiber sheets incorporating methylcobalamin promote nerve regeneration and functional recovery in a rat sciatic nerve crush injury model. Acta Biomater. 2017, 53, 250–259. [Google Scholar] [CrossRef] [PubMed]
- Fontana, X.; Hristova, M.; Da Costa, C.; Patodia, S.; Thei, L.; Makwana, M.; Spencer-Dene, B.; Latouche, M.; Mirsky, R.; Jessen, K.R.; et al. C-jun in schwann cells promotes axonal regeneration and motoneuron survival via paracrine signaling. J. Cell Biol. 2012, 198, 127–141. [Google Scholar] [CrossRef] [PubMed]
- Lim, T.K.; Shi, X.Q.; Johnson, J.M.; Rone, M.B.; Antel, J.P.; David, S.; Zhang, J. Peripheral nerve injury induces persistent vascular dysfunction and endoneurial hypoxia, contributing to the genesis of neuropathic pain. J. Neurosci. 2015, 35, 3346–3359. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Lin, W.; Liu, H.; Deng, J.; Chen, Y.; Liu, H.; Fu, X.; Yang, Y. Ultrasound-responsive nanobubbles contained with peptide-camptothecin conjugates for targeted drug delivery. Drug. Deliv. 2016, 23, 2756–2764. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.Y.; Liu, H.L.; Hsu, P.H.; Chiang, C.S.; Tsai, C.H.; Chi, H.S.; Chen, S.Y.; Chen, Y.Y. A multitheragnostic nanobubble system to induce blood-brain barrier disruption with magnetically guided focused ultrasound. Adv. Mater. 2015, 27, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Geis, N.A.; Katus, H.A.; Bekeredjian, R. Microbubbles as a vehicle for gene and drug delivery: Current clinical implications and future perspectives. Curr. Pharm. Des. 2012, 18, 2166–2183. [Google Scholar] [CrossRef] [PubMed]
- Dayton, P.A.; Chomas, J.E.; Lum, A.F.; Allen, J.S.; Lindner, J.R.; Simon, S.I.; Ferrara, K.W. Optical and acoustical dynamics of microbubble contrast agents inside neutrophils. Biophys. J. 2001, 80, 1547–1556. [Google Scholar] [CrossRef]
- Lindner, J.R.; Coggins, M.P.; Kaul, S.; Klibanov, A.L.; Brandenburger, G.H.; Ley, K. Microbubble persistence in the microcirculation during ischemia/reperfusion and inflammation is caused by integrin- and complement-mediated adherence to activated leukocytes. Circulation 2000, 101, 668–675. [Google Scholar] [CrossRef] [PubMed]
- Markus, A.; Patel, T.D.; Snider, W.D. Neurotrophic factors and axonal growth. Curr. Opin. Neurobiol. 2002, 12, 523–531. [Google Scholar] [CrossRef]
- Zhou, F.Q.; Snider, W.D. Intracellular control of developmental and regenerative axon growth. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2006, 361, 1575–1592. [Google Scholar] [CrossRef] [PubMed]
- Auer, M.; Schweigreiter, R.; Hausott, B.; Thongrong, S.; Holtje, M.; Just, I.; Bandtlow, C.; Klimaschewski, L. Rho-independent stimulation of axon outgrowth and activation of the erk and akt signaling pathways by c3 transferase in sensory neurons. Front. Cell. Neurosci. 2012, 6, 43. [Google Scholar] [CrossRef] [PubMed]
- Campbell, M.; Allen, W.E.; Sawyer, C.; Vanhaesebroeck, B.; Trimble, E.R. Glucose-potentiated chemotaxis in human vascular smooth muscle is dependent on cross-talk between the pi3k and mapk signaling pathways. Circ. Res. 2004, 95, 380–388. [Google Scholar] [CrossRef] [PubMed]
- Merighi, S.; Benini, A.; Mirandola, P.; Gessi, S.; Varani, K.; Leung, E.; Maclennan, S.; Borea, P.A. A3 adenosine receptor activation inhibits cell proliferation via phosphatidylinositol 3-kinase/akt-dependent inhibition of the extracellular signal-regulated kinase 1/2 phosphorylation in a375 human melanoma cells. J. Biol. Chem. 2005, 280, 19516–19526. [Google Scholar] [CrossRef] [PubMed]
- Rommel, C.; Clarke, B.A.; Zimmermann, S.; Nunez, L.; Rossman, R.; Reid, K.; Moelling, K.; Yancopoulos, G.D.; Glass, D.J. Differentiation stage-specific inhibition of the raf-mek-erk pathway by akt. Science 1999, 286, 1738–1741. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, S.; Moelling, K. Phosphorylation and regulation of raf by akt (protein kinase b). Science 1999, 286, 1741–1744. [Google Scholar] [CrossRef] [PubMed]
- Boerboom, A.; Dion, V.; Chariot, A.; Franzen, R. Molecular mechanisms involved in schwann cell plasticity. Front. Mol. Neurosci. 2017, 10, 38. [Google Scholar] [CrossRef] [PubMed]
- Ogata, T.; Iijima, S.; Hoshikawa, S.; Miura, T.; Yamamoto, S.; Oda, H.; Nakamura, K.; Tanaka, S. Opposing extracellular signal-regulated kinase and akt pathways control schwann cell myelination. J. Neurosci. 2004, 24, 6724–6732. [Google Scholar] [CrossRef] [PubMed]
- Harrisingh, M.C.; Perez-Nadales, E.; Parkinson, D.B.; Malcolm, D.S.; Mudge, A.W.; Lloyd, A.C. The ras/raf/erk signalling pathway drives schwann cell dedifferentiation. EMBO J. 2004, 23, 3061–3071. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Shin, Y.K.; Park, H.T. Mitogen activated protein kinase family proteins and c-jun signaling in injury-induced schwann cell plasticity. Exp. Neurobiol. 2014, 23, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Parkinson, D.B.; Bhaskaran, A.; Droggiti, A.; Dickinson, S.; D’Antonio, M.; Mirsky, R.; Jessen, K.R. Krox-20 inhibits jun-nh2-terminal kinase/c-jun to control schwann cell proliferation and death. J. Cell Biol. 2004, 164, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, J.; Chan, J.R.; Shooter, E.M. Neurotrophin 3 activation of trkc induces schwann cell migration through the c-jun n-terminal kinase pathway. Proc. Natl. Acad. Sci. USA 2003, 100, 14421–14426. [Google Scholar] [CrossRef] [PubMed]
- Fei, W.; Aixi, Y.; Danmou, X.; Wusheng, K.; Zhengren, P.; Ting, R. The mood stabilizer valproic acid induces proliferation and myelination of rat schwann cells. Neurosci. Res. 2011, 70, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Leitman, E.M.; Tewari, A.; Horn, M.; Urbanski, M.; Damanakis, E.; Einheber, S.; Salzer, J.L.; de Lanerolle, P.; Melendez-Vasquez, C.V. Mlck regulates schwann cell cytoskeletal organization, differentiation and myelination. J. Cell Sci. 2011, 124, 3784–3796. [Google Scholar] [CrossRef] [PubMed]
- Henderson, C.E.; Phillips, H.S.; Pollock, R.A.; Davies, A.M.; Lemeulle, C.; Armanini, M.; Simmons, L.; Moffet, B.; Vandlen, R.A.; Simmons, L.; et al. Gdnf: A potent survival factor for motoneurons present in peripheral nerve and muscle. Science 1994, 266, 1062–1064. [Google Scholar] [CrossRef] [PubMed]
- Iihara, K.; Hashimoto, N.; Tsukahara, T.; Sakata, M.; Yanamoto, H.; Taniguchi, T. Platelet-derived growth factor-bb, but not -aa, prevents delayed neuronal death after forebrain ischemia in rats. J. Cereb. Blood Flow Metab. 1997, 17, 1097–1106. [Google Scholar] [CrossRef] [PubMed]
- Apel, P.J.; Ma, J.; Callahan, M.; Northam, C.N.; Alton, T.B.; Sonntag, W.E.; Li, Z. Effect of locally delivered igf-1 on nerve regeneration during aging: An experimental study in rats. Muscle Nerve 2010, 41, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Yi, S.; Yuan, Y.; Chen, Q.; Wang, X.; Gong, L.; Liu, J.; Gu, X.; Li, S. Regulation of schwann cell proliferation and migration by mir-1 targeting brain-derived neurotrophic factor after peripheral nerve injury. Sci. Rep. 2016, 6, 29121. [Google Scholar] [CrossRef] [PubMed]
- Webber, C.; Zochodne, D. The nerve regenerative microenvironment: Early behavior and partnership of axons and schwann cells. Exp. Neurol. 2010, 223, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, H.C.; Hoke, A. Use of engineered schwann cells in peripheral neuropathy: Hopes and hazards. Brain Res. 2016, 1638, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Storkebaum, E.; Lambrechts, D.; Carmeliet, P. Vegf: Once regarded as a specific angiogenic factor, now implicated in neuroprotection. Bioessays 2004, 26, 943–954. [Google Scholar] [CrossRef] [PubMed]
- Arthur-Farraj, P.J.; Latouche, M.; Wilton, D.K.; Quintes, S.; Chabrol, E.; Banerjee, A.; Woodhoo, A.; Jenkins, B.; Rahman, M.; Turmaine, M.; et al. C-jun reprograms schwann cells of injured nerves to generate a repair cell essential for regeneration. Neuron 2012, 75, 633–647. [Google Scholar] [CrossRef] [PubMed]
- Waxman, S.G. Determinants of conduction velocity in myelinated nerve fibers. Muscle Nerve 1980, 3, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Navarro, X. Functional evaluation of peripheral nerve regeneration and target reinnervation in animal models: A critical overview. Eur. J. Neurosci. 2016, 43, 271–286. [Google Scholar] [CrossRef] [PubMed]
- Varejao, A.S.; Meek, M.F.; Ferreira, A.J.; Patricio, J.A.; Cabrita, A.M. Functional evaluation of peripheral nerve regeneration in the rat: Walking track analysis. J. Neurosci. Methods 2001, 108, 1–9. [Google Scholar] [CrossRef]
- Pereira Lopes, F.R.; Martin, P.K.; Frattini, F.; Biancalana, A.; Almeida, F.M.; Tomaz, M.A.; Melo, P.A.; Borojevic, R.; Han, S.W.; Martinez, A.M. Double gene therapy with granulocyte colony-stimulating factor and vascular endothelial growth factor acts synergistically to improve nerve regeneration and functional outcome after sciatic nerve injury in mice. Neuroscience 2013, 230, 184–197. [Google Scholar] [CrossRef] [PubMed]
- Goulart, C.O.; Jurgensen, S.; Souto, A.; Oliveira, J.T.; de Lima, S.; Tonda-Turo, C.; Marques, S.A.; de Almeida, F.M.; Martinez, A.M. A combination of schwann-cell grafts and aerobic exercise enhances sciatic nerve regeneration. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Pitcher, G.M.; Ritchie, J.; Henry, J.L. Paw withdrawal threshold in the von frey hair test is influenced by the surface on which the rat stands. J. Neurosci. Methods 1999, 87, 185–193. [Google Scholar] [CrossRef]
- Noda, K.; Akita, H.; Ogata, M.; Saji, M. Paclitaxel-induced hyperalgesia modulates negative affective component of pain and nr1 receptor expression in the frontal cortex in rats. Neurosci. Res. 2014, 80, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Okada, K.; Tanaka, H.; Temporin, K.; Okamoto, M.; Kuroda, Y.; Moritomo, H.; Murase, T.; Yoshikawa, H. Methylcobalamin increases erk1/2 and akt activities through the methylation cycle and promotes nerve regeneration in a rat sciatic nerve injury model. Exp. Neurol. 2010, 222, 191–203. [Google Scholar] [CrossRef] [PubMed]
- Nishimoto, S.; Tanaka, H.; Okamoto, M.; Okada, K.; Murase, T.; Yoshikawa, H. Methylcobalamin promotes the differentiation of schwann cells and remyelination in lysophosphatidylcholine-induced demyelination of the rat sciatic nerve. Front. Cell. Neurosci. 2015, 9, 298. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, J.; Miyamoto, Y.; Hamasaki, H.; Sanbe, A.; Kusakawa, S.; Nakamura, A.; Tsumura, H.; Maeda, M.; Nemoto, N.; Kawahara, K.; et al. The atypical guanine-nucleotide exchange factor, dock7, negatively regulates schwann cell differentiation and myelination. J. Neurosci. 2011, 31, 12579–12592. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Wang, Y.; Qi, F.; Zhu, S.; Huang, L.; Liu, Z.; Huang, J.; Luo, Z. The effect of synthetic oxygen carrier-enriched fibrin hydrogel on schwann cells under hypoxia condition In Vitro. Biomaterials 2013, 34, 10016–10027. [Google Scholar] [CrossRef] [PubMed]








© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matsuoka, H.; Ebina, K.; Tanaka, H.; Hirao, M.; Iwahashi, T.; Noguchi, T.; Suzuki, K.; Nishimoto, S.; Murase, T.; Yoshikawa, H. Administration of Oxygen Ultra-Fine Bubbles Improves Nerve Dysfunction in a Rat Sciatic Nerve Crush Injury Model. Int. J. Mol. Sci. 2018, 19, 1395. https://doi.org/10.3390/ijms19051395
Matsuoka H, Ebina K, Tanaka H, Hirao M, Iwahashi T, Noguchi T, Suzuki K, Nishimoto S, Murase T, Yoshikawa H. Administration of Oxygen Ultra-Fine Bubbles Improves Nerve Dysfunction in a Rat Sciatic Nerve Crush Injury Model. International Journal of Molecular Sciences. 2018; 19(5):1395. https://doi.org/10.3390/ijms19051395
Chicago/Turabian StyleMatsuoka, Hozo, Kosuke Ebina, Hiroyuki Tanaka, Makoto Hirao, Toru Iwahashi, Takaaki Noguchi, Koji Suzuki, Shunsuke Nishimoto, Tsuyoshi Murase, and Hideki Yoshikawa. 2018. "Administration of Oxygen Ultra-Fine Bubbles Improves Nerve Dysfunction in a Rat Sciatic Nerve Crush Injury Model" International Journal of Molecular Sciences 19, no. 5: 1395. https://doi.org/10.3390/ijms19051395