Emerging Roles of G Protein-Coupled Receptors in Hepatocellular Carcinoma
Abstract
:1. Introduction
2. Structure and Signal Transduction of G Protein-Coupled Receptors (GPCRs)
2.1. Structure and Classification of GPCRs
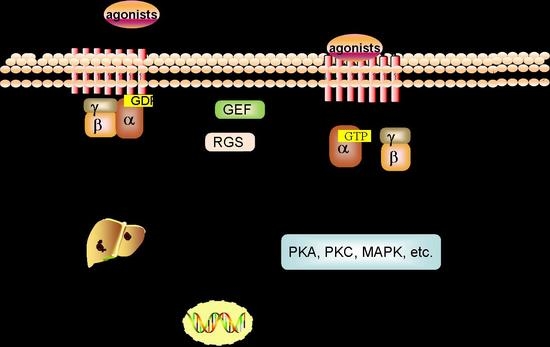
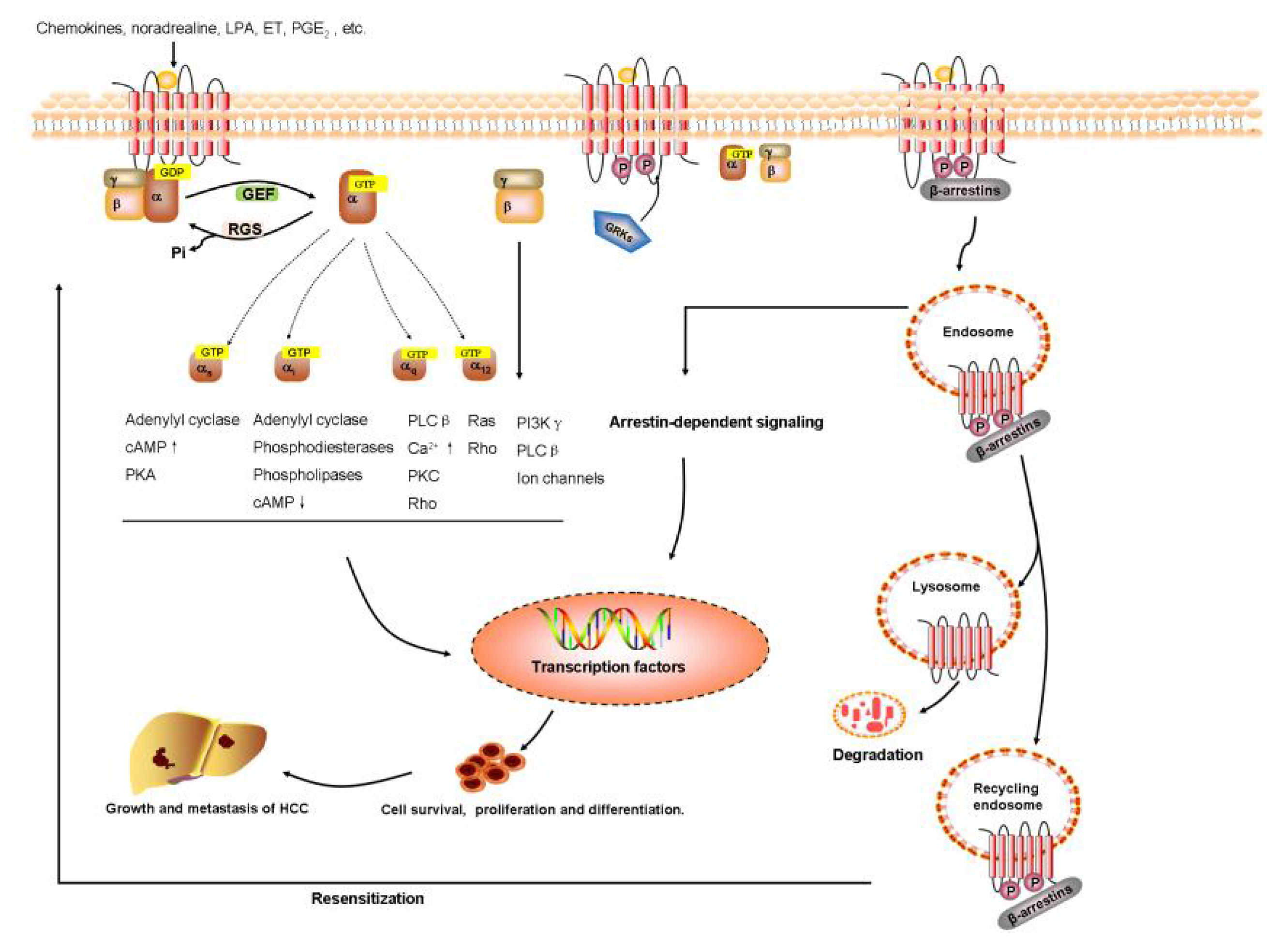
2.2. Signal Transduction of GPCRs
3. Role of GPCRs in the Occurrence and Development of Hepatocellular Carcinoma (HCC)
3.1. Chemokine Receptors
3.1.1. CCRn
3.1.2. CXCRn
3.1.3. CX3CRn
3.1.4. CXRn
3.2. E-Prostanoid Receptors
3.3. Lysophosphatidic Acid Receptors
3.4. Adrenergic Receptors
3.5. Angiotensin II Receptors
3.6. Smoothened (Smo) Receptors
3.7. Orphan GPCRs
4. GPCR-Based Treatments in HCC
5. Conclusions
Acknowledgments
Conflicts of Interest
Abbreviations
| GPCRs | G protein-coupled receptors |
| HCC | hepatocellular carcinoma |
| GRKs | G protein receptor kinases |
| AC | adenylyl cyclases |
| GABAB | γ-aminobutyric acid type B |
| GDP | guanosine diphosphate |
| GTP | guanosine triphosphate |
| DCs | dendritic cells |
| IL-12 | interleukin-12 |
| IFN-γ | interferon-γ |
| 3‘-UTR | 3'-untranslated region |
| p-JNK | phosphorylase c-jun N-terminal kinase |
| PLC | phospholipase C |
| TGF-β | transforming growth factor-β |
| MAPK | mitogen-activated protein kinase |
| DEN | N-nitrosodiethylamine |
| HBs | hepatitis B virus surface |
| MMPs | matrix metalloproteinases |
| RNAi | RNA interference |
| EMT | epithelial-mesenchymal transition |
| EGFR | epidermal growth factor receptor |
| PDGFR | platelet-derived growth factor receptor |
| IGF1R | insulin-like growth factor 1 receptor |
| siRNAs | small interfering RNAs |
| RTKs | receptor tyrosine kinases |
| VEGF | vascular endothelial growth factor |
| PGE2 | prostaglandin E2 |
| PKA | protein kinase A |
| YB-1 | Y-box-binding protein 1 |
| LPA | lysophosphatidic acid |
| Edg | endothelial differentiation gene |
| PTFs | peritumoral tissue fibroblasts |
| PI3K | phosphoinositide 3-kinase |
| PKCδ | protein kinase Cδ |
| HIF-1 | hypoxia-inducible factor-1 |
| ETAR | endothelin type A receptor |
| ARs | adrenergic receptors |
| SNS | sympathetic nervous system |
| cAMP | cyclic 3′,5′-adenosine monophsphate |
| CREB | cAMP response element binding protein |
| Ang II | angiotensin II |
| AT1 | angiotensin II type 1 |
| Ang-2 | angiopoietin-2 |
| Hh | hedgehog |
| Ptc | patched |
| oGPCRs | orphan GPCRs |
| TAMs | tumor associated macrophages |
| Met-RANTES | methionylated RANTES |
| Shh | sonic hedgehog |
References
- Chen, W.; Zheng, R.; Baade, P.D.; Zhang, S.; Zeng, H.; Bray, F.; Jemal, A.; Yu, X.Q.; He, J. Cancer statistics in China. CA Cancer J. Clin. 2015, 66, 115–132. [Google Scholar] [CrossRef] [PubMed]
- El-Serag, H.B. Hepatocellular carcinoma. N. Eng. J. Med. 2011, 365, 1118–1127. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.M.; Booe, J.M.; Pioszak, A.A. Structural insights into ligand recognition and selectivity for classes A, B, and C GPCRs. Eur. J. Pharmacol. 2015, 763, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Mogha, A.; D’Rozario, M.; Monk, K.R. G Protein-Coupled Receptors in Myelinating Glia. Trends Pharmacol. Sci. 2016, 37, 977–987. [Google Scholar] [CrossRef] [PubMed]
- Gough, N.R. Focus Issue: New insights in GPCR to G protein signaling. Sci. Signal 2016, 9, G6. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.J.; Sun, W.Y.; Zhang, S.; Wu, J.J.; Wei, W. The emerging roles of β-arrestins in fibrotic diseases. Acta Pharmacol. Sin. 2015, 36, 1277–1287. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Sun, L.; Jiao, Y. The Role of G Protein-coupled Receptor Kinases in Cancer. Int. J. Biol. Sci. 2018, 14, 189–203. [Google Scholar] [CrossRef] [PubMed]
- Cattaneo, F.; Guerra, G.; Parisi, M.; Marinis, M.D.; Tafuri, D.; Cinelli, M.; Ammendola, R. Cell-surface receptors transactivation mediated by G protein-coupled receptors. Int. J. Mol. Sci. 2014, 15, 19700–19728. [Google Scholar] [CrossRef] [PubMed]
- Bareja, A.; Patel, S.; Hodgkinson, C.P.; Payne, A.; Dzau, V.J. Understanding the mechanism of bias signaling of the insulin-like growth factor 1 receptor: Effects of LL37 and HASF. Cell Signal. 2018, 46, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Xia, S.; He, C.; Zhu, Y.; Wang, S.; Li, H.; Zhang, Z.; Jiang, X.; Liu, J. GABAR-induced EGFR transactivation promotes migration of human prostate cancer cells. Mol. Pharmacol. 2017, 92, 265–277. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.L.; Chen, W.C.; Lee, I.T.; Chi, P.L.; Cheng, S.E.; Yang, C.M. c-Src-dependent transactivation of PDGFR contributes to TNF-α-induced MMP-9 expression and functional impairment in osteoblasts. Bone 2014, 60, 186–197. [Google Scholar] [CrossRef] [PubMed]
- Cattaneo, F.; Parisi, M.; Ammendola, R. WKYMVm-induced cross-talk between FPR2 and HGF receptor in human prostate epithelial cell line PNT1A. FEBS Lett. 2013, 587, 1536–1542. [Google Scholar] [CrossRef] [PubMed]
- Castan, L.; Magnan, A. Chemokine receptors in allergic diseases. Allergy 2017, 72, 682–690. [Google Scholar] [CrossRef] [PubMed]
- Melik, P.S.; Rivat, C.; Rostene, W.; Reaux-Le, G.A. Opioid and chemokine receptor crosstalk: A promising target for pain therapy? Nat. Rev. Neurosci. 2015, 16, 69–78. [Google Scholar] [CrossRef]
- Wu, X.; Fan, J.; Wang, X.; Zhou, J.; Qiu, S.; Yu, Y.; Liu, Y.; Tang, Z. Downregulation of CCR1 inhibits human hepatocellular carcinoma cell invasion. Biochem. Biophys. Res. Commun. 2007, 355, 866–871. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.L.; Zhou, Z.J.; Hu, Z.Q.; Li, X.; Huang, X.W.; Wang, Z.; Fan, J.; Dai, Z.; Zhou, J. CXCR2/CXCL5 axis contributes to epithelial-mesenchymal transition of HCC cells through activating PI3K/Akt/GSK-3β/Snail signaling. Cancer Lett. 2015, 358, 124–135. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.L.; Dai, Z.; Zhou, Z.J.; Wang, X.Y.; Yang, G.H.; Wang, Z.; Huang, X.W.; Fan, J.; Zhou, J. Overexpression of CXCL5 mediates neutrophil infiltration and indicates poor prognosis for hepatocellular carcinoma. Hepatology 2012, 56, 2242–2254. [Google Scholar] [CrossRef] [PubMed]
- Yanru, W.; Zhenyu, B.; Zhengchuan, N.; Qi, Q.; Chunmin, L. Transcriptomic analyses of chemokines reveal that down-regulation of XCR1 is associated with advanced hepatocellular carcinoma. Biochem. Biophys. Res. Commun. 2018, 496, 1314–1321. [Google Scholar] [CrossRef] [PubMed]
- Ryschich, E.; Lizdenis, P.; Ittrich, C.; Benner, A.; Stahl, S.; Hamann, A.; Schmidt, J.; Knolle, P.; Arnold, B.; Hammerling, G.J.; et al. Molecular fingerprinting and autocrine growth regulation of endothelial cells in a murine model of hepatocellular carcinoma. Cancer Res. 2006, 66, 198–211. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wu, J.; Zhang, W.; Zhang, N.; Guo, H. Identification of serum CCL15 in hepatocellular carcinoma. Br. J. Cancer 2013, 108, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Rodero, M.P.; Auvynet, C.; Poupel, L.; Combadiere, B.; Combadiere, C. Control of both myeloid cell infiltration and angiogenesis by CCR1 promotes liver cancer metastasis development in mice. Neoplasia 2013, 15, 641–648. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Lim, S.Y.; Gordon-Weeks, A.N.; Tapmeier, T.T.; Im, J.H.; Cao, Y.; Beech, J.; Allen, D.; Smart, S.; Muschel, R.J. Recruitment of a myeloid cell subset (CD11b/Gr1 mid) via CCL2/CCR2 promotes the development of colorectal cancer liver metastasis. Hepatology 2013, 57, 829–839. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Lu, P.; Ishida, Y.; Kuziel, W.A.; Fujii, C.; Mukaida, N. Attenuated liver tumor formation in the absence of CCR2 with a concomitant reduction in the accumulation of hepatic stellate cells, macrophages and neovascularization. Int. J. Cancer 2006, 118, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Barashi, N.; Weiss, I.D.; Wald, O.; Wald, H.; Beider, K.; Abraham, M.; Klein, S.; Goldenberg, D.; Axelrod, J.; Pikarsky, E.; et al. Inflammation-induced hepatocellular carcinoma is dependent on CCR5 in mice. Hepatology 2013, 58, 1021–1030. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.P.; Yan, J.; Xu, J.; Pang, X.H.; Chen, M.S.; Li, L.; Wu, C.; Li, S.P.; Zhen, L. Increased intratumoral IL-17-producing cells correlate with poor survival in hepatocellular carcinoma patients. J. Hepatol. 2009, 50, 980–989. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.J.; Lin, S.Z.; Zhou, L.; Xie, H.Y.; Zhou, W.H.; Taki-Eldin, A.; Zheng, S.S. Selective recruitment of regulatory T cell through CCR6-CCL20 in hepatocellular carcinoma fosters tumor progression and predicts poor prognosis. PLoS ONE 2011, 6, e24671. [Google Scholar] [CrossRef]
- Schimanski, C.C.; Bahre, R.; Gockel, I.; Junginger, T.; Simiantonaki, N.; Biesterfeld, S.; Achenbach, T.; Wehler, T.; Galle, P.R.; Moehler, M. Chemokine receptor CCR7 enhances intrahepatic and lymphatic dissemination of human hepatocellular cancer. Oncol. Rep. 2006, 16, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhou, S.; Qin, J.; Hu, H.; Ma, H.; Liu, B.; Wang, X.; Ma, J.; Ye, S.; Zhong, C.; et al. Combination of SLC administration and Tregs depletion is an attractive strategy for targeting hepatocellular carcinoma. Mol. Cancer 2013, 12, 153. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Qin, C.; Wu, Y.; Su, Z.; Xian, G.; Hu, B. CCR9 as a prognostic marker and therapeutic target in hepatocellular carcinoma. Oncol. Rep. 2014, 31, 1629–1636. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Yang, L.; Xu, J.; Zhang, X.; Wang, B. Enhanced expression and clinical significance of chemokine receptor CXCR2 in hepatocellular carcinoma. J. Surg. Res. 2011, 166, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Kuang, D.M.; Zhao, Q.; Wu, Y.; Peng, C.; Wang, J.; Xu, Z.; Yin, X.Y.; Zheng, L. Peritumoral neutrophils link inflammatory response to disease progression by fostering angiogenesis in hepatocellular carcinoma. J. Hepatol. 2011, 54, 948–955. [Google Scholar] [CrossRef] [PubMed]
- Ding, Q.; Xia, Y.; Ding, S.; Lu, P.; Sun, L.; Liu, M. An alternatively spliced variant of CXCR3 mediates the metastasis of CD133+ liver cancer cells induced by CXCL9. Oncotarget 2016, 7, 14405–14414. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Pan, Z.; Li, A.; Fu, S.; Lei, Y.; Sun, H.; Wu, M.; Zhou, W. Roles of chemokine receptor 4 (CXCR4) and chemokine ligand 12 (CXCL12) in metastasis of hepatocellular carcinoma cells. Cell. Mol. Immunol. 2008, 5, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Guo, W.; Shi, J.; Xue, J.; Hu, H.; Xie, D.; Wu, M.; Cheng, S.M. Expression of the chemokine receptor CXCR4 in human hepatocellular carcinoma and its role in portal vein tumor thrombus. J. Exp. Clin. Cancer Res. 2010, 29, 156. [Google Scholar] [CrossRef] [PubMed]
- Schimanski, C.C.; Bahre, R.; Gockel, I.; Muller, A.; Frerichs, K.; Horner, V.; Teufel, A.; Simiantonaki, N.; Biesterfeld, S.; Wehler, T.; et al. Dissemination of hepatocellular carcinoma is mediated via chemokine receptor CXCR4. Br. J. Cancer 2006, 95, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Pan, X.; Huang, Z.; Weber, G.F.; Zhang, G. Osteopontin enhances the expression and activity of MMP-2 via the SDF-1/CXCR4 axis in hepatocellular carcinoma cell lines. PLoS ONE 2011, 6, e23831. [Google Scholar] [CrossRef] [PubMed]
- Bertran, E.; Caja, L.; Navarro, E.; Sancho, P.; Mainez, J.; Murillo, M.M.; Vinyals, A.; Fabra, A.; Fabregat, I. Overactivation of the TGF-beta pathway confers a mesenchymal-like phenotype and CXCR4-dependent migratory properties to liver tumor cells. Hepatology 2013, 58, 2032–2044. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, P.; Chang, Y.; Xu, Q.; Wu, Z.; Ma, Q.; Wang, Z. The SDF-1/CXCR4 axis induces epithelial-mesenchymal transition in hepatocellular carcinoma. Mol. Cell. Biochem. 2014, 392, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Xue, T.C.; Chen, R.X.; Han, D.; Chen, J.; Xue, Q.; Gao, D.M.; Sun, R.X.; Tang, Z.Y.; Ye, S.L. Down-regulation of CXCR7 inhibits the growth and lung metastasis of human hepatocellular carcinoma cells with highly metastatic potential. Exp. Ther. Med. 2012, 3, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Han, M.M.; Wang, F.; Xu, L.L.; Yu, H.X.; Yang, P.Y. CXCR7 stimulates MAPK signaling to regulate hepatocellular carcinoma progression. Cell Death Dis. 2014, 5, e1488. [Google Scholar] [CrossRef] [PubMed]
- Zheng, K.; Li, H.Y.; Su, X.L.; Wang, X.Y.; Tian, T.; Li, F.; Ren, G.S. Chemokine receptor CXCR7 regulates the invasion, angiogenesis and tumor growth of human hepatocellular carcinoma cells. J. Exp. Clin. Cancer Res. 2010, 29, 31. [Google Scholar] [CrossRef] [PubMed]
- Monnier, J.; Boissan, M.; L’Helgoualc’h, A.; Lacombe, M.L.; Turlin, B.; Zucman-Rossi, J.; Theret, N.; Piquet-Pellorce, C.; Samson, M. CXCR7 is up-regulated in human and murine hepatocellular carcinoma and is specifically expressed by endothelial cells. Eur. J. Cancer 2012, 48, 138–148. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Zhao, Y.J.; Wang, X.Y.; Qiu, S.J.; Shi, Y.H.; Sun, J.; Yi, Y.; Shi, J.Y.; Shi, G.M.; Ding, Z.B.; et al. CXCR6 upregulation contributes to a proinflammatory tumor microenvironment that drives metastasis and poor patient outcomes in hepatocellular carcinoma. Cancer Res. 2012, 72, 3546–3556. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, T.; Ono, T.; Yamanoi, A.; Tachibana, M.; Nagasue, N. Fractalkine-CX3CR1 axis regulates tumor cell cycle and deteriorates prognosis after radical resection for hepatocellular carcinoma. J. Surg. Oncol. 2007, 95, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.; Hu, H.D.; Hu, P.; Lan, Y.H.; Peng, M.L.; Chen, M.; Ren, H. Gene therapy with CX3CL1/Fractalkine induces antitumor immunity to regress effectively mouse hepatocellular carcinoma. Gene Ther. 2007, 14, 1226–1234. [Google Scholar] [CrossRef] [PubMed]
- Xia, S.; Ma, J.; Bai, X.; Zhang, H.; Cheng, S.; Zhang, M.; Zhang, L.; Du, M.; Wang, Y.; Li, H.; et al. Prostaglandin E2 promotes the cell growth and invasive ability of hepatocellular carcinoma cells by upregulating c-Myc expression via EP4 receptor and the PKA signaling pathway. Oncol. Rep. 2014, 32, 1521–1530. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Cheng, S.; Zhang, M.; Ma, X.; Zhang, L.; Wang, Y.; Rong, R.; Ma, J.; Xia, S.; Du, M.; et al. Prostaglandin E2 promotes hepatocellular carcinoma cell invasion through upregulation of YB-1 protein expression. Int. J. Oncol. 2014, 44, 769–780. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Wang, J.; Guo, Y.; Pan, J.; Yang, Q.; Zhang, M.; Li, H.; Zhang, L.; Ma, J.; Shi, F.; et al. Prostaglandin E2 stimulates beta1-integrin expression in hepatocellular carcinoma through the EP1 receptor/PKC/NF-kappaB pathway. Sci. Rep. 2014, 4, 6538. [Google Scholar] [CrossRef] [PubMed]
- Breinig, M.; Rieker, R.; Eiteneuer, E.; Wertenbruch, T.; Haugg, A.M.; Helmke, B.M.; Schirmacher, P.; Kern, M.A. Differential expression of E-prostanoid receptors in human hepatocellular carcinoma. Int. J. Cancer 2008, 122, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.Y.; Zhang, H.; Zhang, M.; Xia, S.K.; Bai, X.M.; Zhang, L.; Ma, J.; Rong, R.; Wang, Y.P.; Du, M.Z.; et al. Prostaglandin E2 receptor EP2 mediates Snail expression in hepatocellular carcinoma cells. Oncol. Rep. 2014, 31, 2099–2106. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Sun, W.; Wei, W.; Wang, D.; Jin, J.; Wu, J.; Chen, J.; Wu, H.; Wang, Q. Involvement of the prostaglandin E receptor EP2 in paeoniflorin-induced human hepatoma cell apoptosis. Anticancer Drugs 2013, 24, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Yanagida, K.; Ishii, S. Non-Edg family LPA receptors: The cutting edge of LPA research. J. Biochem. 2011, 150, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Sokolov, E.; Eheim, A.L.; Ahrens, W.A.; Walling, T.L.; Swet, J.H.; McMillan, M.T.; Simo, K.A.; Thompson, K.J.; Sindram, D.; McKillop, I.H. Lysophosphatidic acid receptor expression and function in human hepatocellular carcinoma. J. Surg. Res. 2013, 180, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Mazzocca, A.; Dituri, F.; Lupo, L.; Quaranta, M.; Antonaci, S.; Giannelli, G. Tumor-secreted lysophostatidic acid accelerates hepatocellular carcinoma progression by promoting differentiation of peritumoral fibroblasts in myofibroblasts. Hepatology 2011, 54, 920–930. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Jeong, K.J.; Panupinthu, N.; Yu, S.; Lee, J.; Han, J.W.; Kim, J.M.; Lee, J.S.; Kang, J.; Park, C.G.; et al. Lysophosphatidic acid augments human hepatocellular carcinoma cell invasion through LPA1 receptor and MMP-9 expression. Oncogene 2011, 30, 1351–1359. [Google Scholar] [CrossRef] [PubMed]
- Zuckerman, V.; Sokolov, E.; Swet, J.H.; Ahrens, W.A.; Showlater, V.; Iannitti, D.A.; Mckillop, I.H. Expression and function of lysophosphatidic acid receptors (LPARs) 1 and 3 in human hepatic cancer progenitor cells. Oncotarget 2016, 7, 2951–2967. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Park, S.Y.; Lee, E.K.; Park, C.G.; Chung, H.C.; Rha, S.Y.; Kim, Y.K.; Bae, G.U.; Kim, B.K.; Han, J.W. Activation of hypoxia-inducible factor-1alpha is necessary for lysophosphatidic acid-induced vascular endothelial growth factor expression. Clin. Cancer Res. 2006, 12, 6351–6358. [Google Scholar] [CrossRef] [PubMed]
- Scanzano, A.; Cosentino, M. Adrenergic regulation of innate immunity: A review. Front. Pharmacol. 2015, 6, 171. [Google Scholar] [CrossRef] [PubMed]
- Kassahun, W.T.; Gunl, B.; Jonas, S.; Ungemach, F.R.; Abraham, G. Altered liver α1-adrenoceptor density and phospholipase C activity in the human hepatocellular carcinoma. Eur. J. Pharmacol. 2011, 670, 92–95. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yang, X.M.; Wang, Y.H.; Feng, M.X.; Liu, X.J.; Zhang, Y.L.; Huang, S.; Wu, Z.; Xue, F.; Qin, W.X. Monoamine oxidase A suppresses hepatocellular carcinoma metastasis by inhibiting the adrenergic system and its transactivation of EGFR signaling. J. Hepatol. 2014, 60, 1225–1234. [Google Scholar] [CrossRef] [PubMed]
- Yuan, A.; Li, Z.; Li, X.; Yi, S.; Wang, S.; Cai, Y.; Cao, H. The mitogenic effectors of isoproterenol in human hepatocellular carcinoma cells. Oncol. Rep. 2010, 23, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wu, W.K.; Yu, L.; Li, Z.J.; Sung, J.J.; Zhang, S.T.; Cho, C.H. Epidermal growth factor-induced esophageal cancer cell proliferation requires transactivation of beta-adrenoceptors. J. Pharmacol. Exp. Ther. 2008, 326, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.Q.; Fang, T.; Yu, L.X.; Lv, G.S.; Lv, H.W.; Liang, D.; Li, T.; Wang, C.Z.; Tan, Y.X.; Ding, J.; et al. ADRB2 signaling promotes HCC progression and sorafenib resistance by inhibiting autophagic degradation of HIF1alpha. J. Hepatol. 2016, 65, 314–324. [Google Scholar] [CrossRef] [PubMed]
- Neo, J.H.; Ager, E.I.; Angus, P.W.; Zhu, J.; Herath, C.B.; Christophi, C. Changes in the renin angiotensin system during the development of colorectal cancer liver metastases. BMC Cancer 2010, 10, 134. [Google Scholar] [CrossRef] [PubMed]
- Otake, A.H.; Mattar, A.L.; Freitas, H.C.; Machado, C.M.; Nonogaki, S.; Fujihara, C.K.; Zatz, R.; Chammas, R. Inhibition of angiotensin II receptor 1 limits tumor-associated angiogenesis and attenuates growth of murine melanoma. Cancer Chemother. Pharmacol. 2010, 66, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.; Wang, Z.; Li, Z.; Li, K.; Le, X.; Zhang, T. Angiotensin II induces angiogenic factors production partly via AT1/JAK2/STAT3/SOCS3 signaling pathway in MHCC97H cells. Cell Physiol. Biochem. 2012, 29, 863–874. [Google Scholar] [CrossRef] [PubMed]
- Deshayes, F.; Nahmias, C. Angiotensin receptors: A new role in cancer? Trends Endocrinol. Metab. 2005, 16, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Rizkalla, B.; Forbes, J.M.; Cooper, M.E.; Cao, Z. Increased renal vascular endothelial growth factor and angiopoietins by angiotensin II infusion is mediated by both AT1 and AT2 receptors. J. Am. Soc. Nephrol. 2003, 14, 3061–3071. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Lassila, M.; Cooper, M.E.; Cao, Z. Retinal expression of vascular endothelial growth factor is mediated by angiotensin type 1 and type 2 receptors. Hypertension 2004, 43, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Silvestre, J.S.; Tamarat, R.; Senbonmatsu, T.; Icchiki, T.; Ebrahimian, T.; Iglarz, M.; Besnard, S.; Duriez, M.; Inagami, T.; Levy, B.I. Antiangiogenic effect of angiotensin II type 2 receptor in ischemia-induced angiogenesis in mice hindlimb. Circ. Res. 2002, 90, 1072–1089. [Google Scholar] [CrossRef] [PubMed]
- Du, H.; Liang, Z.; Zhang, Y.; Jie, F.; Li, J.; Fei, Y.; Huang, Z.; Pei, N.; Wang, S.; Li, A.; et al. Effects of angiotensin II type 2 receptor overexpression on the growth of hepatocellular carcinoma cells in vitro and in vivo. PLoS ONE 2013, 8, e83754. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Wu, H.; Katritch, V.; Han, G.W.; Huang, X.P.; Liu, W.; Siu, F.Y.; Roth, B.L.; Cherezov, V.; Stevens, R.C. Structure of the human smoothened receptor bound to an antitumour agent. Nature 2013, 497, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Che, L.; Yuan, Y.H.; Jia, J.; Ren, J. Activation of sonic hedgehog signaling pathway is an independent potential prognosis predictor in human hepatocellular carcinoma patients. Chin. J. Cancer Res. 2012, 24, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Jeng, K.S.; Sheen, I.S.; Jeng, W.J.; Lin, C.C.; Lin, C.K.; Su, J.C.; Yu, M.C.; Fang, H.Y. High expression of patched homolog-1 messenger RNA and glioma-associated oncogene-1 messenger RNA of sonic hedgehog signaling pathway indicates a risk of postresection recurrence of hepatocellular carcinoma. Ann. Surg. Oncol. 2013, 20, 464–473. [Google Scholar] [CrossRef] [PubMed]
- Philips, G.M.; Chan, I.S.; Swiderska, M.; Schroder, V.T.; Guy, C.; Karaca, G.F.; Moylan, C.; Venkatraman, T.; Feuerlein, S.; Syn, W.K.; et al. Hedgehog signaling antagonist promotes regression of both liver fibrosis and hepatocellular carcinoma in a murine model of primary liver cancer. PLoS ONE 2011, 6, e23943. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Chenna, V.; Hu, C.; Sun, H.X.; Khan, M.; Bai, H.; Yang, X.R.; Zhu, Q.F.; Sun, Y.F.; Maitra, A.; et al. Polymeric nanoparticle-encapsulated hedgehog pathway inhibitor HPI-1 (NanoHHI) inhibits systemic metastases in an orthotopic model of human hepatocellular carcinoma. Clin. Cancer Res. 2012, 18, 1291–1302. [Google Scholar] [CrossRef] [PubMed]
- Sicklick, J.K.; Li, Y.X.; Jayaraman, A.; Kannangai, R.; Qi, Y.; Vivekanandan, P.; Ludlow, J.W.; Owzar, K.; Chen, W.; Torbenson, M.S.; et al. Dysregulation of the Hedgehog pathway in human hepatocarcinogenesis. Carcinogenesis 2006, 27, 748–757. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Song, W.; Li, H.; Wang, C.; Shi, B.; Guo, W.; Zhong, L. Association between donor and recipient smoothened gene polymorphisms and the risk of hepatocellular carcinoma recurrence following orthotopic liver transplantation in a Han Chinese population. Tumour. Biol. 2015, 36, 7807–7815. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.H.; Chen, J.S.; Wang, Q.; Chen, X.L.; Wen, L.; Chen, L.Z.; Bi, J.; Zhang, L.J.; Su, Q.; Zeng, W.T. miR-338-3p suppresses invasion of liver cancer cell by targeting smoothened. J. Pathol. 2011, 225, 463–472. [Google Scholar] [CrossRef] [PubMed]
- Ngo, T.; Kufareva, I.; James, L.J.; Graham, R.M.; Abagyan, R.; Smith, N.J. Identifying ligands at orphan GPCRs: Current status using structure-based approaches. Br. J. Pharmacol. 2016, 173, 2934–2951. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, Y.; Sakamoto, M.; Fujii, G.; Tsuiji, H.; Kenetaka, K.; Asaka, M.; Hirohashi, S. Overexpression of orphan G-protein-coupled receptor, Gpr49, in human hepatocellular carcinomas with beta-catenin mutations. Hepatology 2003, 37, 528–533. [Google Scholar] [CrossRef] [PubMed]
- Shao, X.; Liu, Y.; Huang, H.; Zhuang, L.; Luo, T.; Huang, H.; Ge, X. Down-regulation of G protein-coupled receptor 137 by RNA interference inhibits cell growth of two hepatoma cell lines. Cell Biol. Int. 2015, 39, 418–426. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Zhu, C.; Huang, X.; Cai, J.; Wang, H.; Wang, X.; He, S.; Liu, C.; Yang, X.; Zhang, Y.; et al. A low level of GPR37 is associated with human hepatocellular carcinoma progression and poor patient survival. Pathol. Res. Pract. 2014, 210, 885–892. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yao, W.; Yuan, Y.; Chen, P.; Li, B.; Li, J.; Chu, R.; Song, H.; Xie, D.; Jiang, X. Targeting of tumour-infiltrating macrophages via CCL2/CCR2 signalling as a therapeutic strategy against hepatocellular carcinoma. Gut 2017, 66, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Ochoa-Callejero, L.; Perez-Martinez, L.; Rubio-Mediavilla, S.; Oteo, J.A.; Martinez, A.; Blanco, J.R. Maraviroc, a CCR5 antagonist, prevents development of hepatocellular carcinoma in a mouse model. PLoS ONE 2013, 8, e53992. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.Y.; Chiang, T.; Liu, C.H.; Chern, G.G.; Lin, T.; Gao, D.Y.; Chen, Y. Delivery of siRNA using CXCR4-targeted nanoparticles modulates tumor microenvironment and achieves a potent antitumor response in liver cancer. Mol. Ther. 2015, 23, 1772–1782. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.Y.; Lin, T.; Sung, Y.C.; Liu, Y.C.; Chiang, W.H.; Chang, C.C.; Liu, J.Y.; Chen, Y. CXCR4-targeted lipid-coated PLGA nanoparticles deliver sorafenib and overcome acquired drug resistance in liver cancer. Biomaterials 2015, 67, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Chang, Y.; Wei, W.; He, Y.F.; Hu, S.S.; Wang, D.; Wu, Y.J. Prostanoid EP1 receptor as the target of (−)-epigallocatechin-3-gallate in suppressing hepatocellular carcinoma cells in vitro. Acta Pharmacol. Sin. 2012, 33, 701–709. [Google Scholar] [CrossRef] [PubMed]
- Han, C.; Michalopoulos, G.K.; Wu, T. Prostaglandin E2 receptor EP1 transactivates EGFR/MET receptor tyrosine kinases and enhances invasiveness in human hepatocellular carcinoma cells. J. Cell Physiol. 2006, 207, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Cusimano, A.; Fodera, D.; Lampiasi, N.; Azzolina, A.; Notarbartolo, M.; Giannitrapani, L.; D’Alessandro, N.; Montalto, G.; Cervello, M. Prostaglandin E2 receptors and COX enzymes in human hepatocellular carcinoma: Role in the regulation of cell growth. Ann. N. Y. Acad. Sci. 2009, 1155, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zhao, W.; Xu, J.; Li, J.; Hong, Z.; Yin, Z.; Wang, X. Activated hepatic stellate cells promote liver cancer by induction of myeloid-derived suppressor cells through cyclooxygenase-2. Oncotarget 2016, 7, 8866–8878. [Google Scholar] [CrossRef] [PubMed]
- Cong, N.; Li, Z.; Shao, W.; Li, J.; Yu, S. Activation of ETA receptor by endothelin-1 induces hepatocellular carcinoma cell migration and invasion via ERK1/2 and AKT signaling pathways. J. Membr. Biol. 2016, 249, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Jeng, K.S.; Jeng, C.J.; Jeng, W.J.; Sheen, I.S.; Chang, C.F.; Hsiau, H.I.; Hung, Z.H.; Yu, M.C.; Chang, F.Y. Sonic hedgehog pathway inhibitor mitigates mouse hepatocellular carcinoma. Am. J. Surg. 2015, 210, 554–560. [Google Scholar] [CrossRef] [PubMed]
| GPCRs | Protein Expression | Cognate Ligands | Role in HCC | Biological Model |
|---|---|---|---|---|
| CCR1 | Up | CCL2, CCL3, CCL5 | Increased migration [21], increased angiogenesis [19,20], increased invasion [15] | HCC patients, DEN-induced mice model, myelo-ablated syngeneic HBs antigen transgenic mice, CCR1-deficient mice, human HCC cell line (HCCLM3) |
| CCR2 | Up | CCL2 | Increased metastasis [22], increased angiogenesis [23] | HCC patients, CD11b/Gr1mid cells, transgenic CD11b-diphtheria toxin receptor mouse model, CCR2- deficient mice |
| CCR5 | Up | CCL3 | Alter tumor microenvironment: increased IL-6 and TNF-α [24] | HCC patients, Mdr2-knockout mice |
| CCR6 | Up | CCL20 | Increased migration [25], increased invasion, migration and angiogenesis [26] | HCC patients, Tregs cell, human HCC cell line (HepG2, MCF-7) |
| CCR7 | Up | CCL21, CCL19 | Increased angiogenesis [27], alter tumor microenvironment: increased IL-10 and TGF-β1, decreased level of IL-12 and IFN-γ [28] | HCC patients, DEN-induced mice model |
| CCR9 | Up | CCL25 | Increased proliferation [29] | HCC patients, human HCC cell lines (HepG2, Huh7, Hepan3B) |
| CXCR2 | Up | CXCL5 | Increased EMT [16], increased migration and invasion [16,17,30], increased angiogenesis [31] | HCC patients, xenograft mouse models, orthotropic nude mice, human HCC cell lines (MHCC97-L, MHCC97-H, HCCLM3, HepG2, Huh7) |
| CXCR3 | Up | CXCL9, CXCL10 | Increased invasion and migration [32] | HCC patients, metastasis mice model, human HCC cell lines (Hep3B, PLC/PRF/5, Huh7) |
| CXCR4 | Up | CXCL12 | Increased invasion and migration [33,35,36,38], increased EMT [34,36,37,38] | HCC patients, human HCC cell lines (Huh7, Hep3B, MHCC97, SMMC7721), hepatic stellate cell (LX-2) |
| CXCR6 | Up | CXCL16 | Increased invasion, alter tumor microenvironment: increased proinflammatory cytokine production such as IL-17, IL-6, and IL-8 [43] | HCC patients, xenograft mouse models, human HCC cell lines (Hep3B, HepG2, Huh7, PLC/PRF/5, SK-HEP-1, MHCC97L, MHCC97H, HCCLM3), human liver cell line (L-02) |
| CXCR7 | Up | CXCL12 | Increased proliferation, migration and invasion [39,40,41], increased angiogenesis and EMT [42] | HCC patients, DEN-induced mice model, human micro-vascular endothelial cell line (HMEC-1) |
| CX3CR1 | Down | CX3CL1 | Immunoprevention [44,45] | HCC patients |
| XCR1 | Down | XCL1 | Suppresses liver cancer growth and tumorigenesis, increased metastasis [18] | human HCC cell lines (Huh-7, HepG2, Hep3B, SMMC7721, HCLM3, HCCLM6, MHCC-97L, MHCC-97H) |
| EP1 | Up | PGE2 | Increased invasion [47], increased migration [48], increased apoptosis [49] | HCC patients, human HCC cell lines (Huh-7, Hep3B, HEK293 cells) |
| EP2 | Up | PGE2 | Decrease apoptosis [50,51] | Human HCC cell lines (SMMC-7721, HepG2) |
| EP4 | Up | PGE2 | Increased proliferation and invasion [46] | Human HCC cell line (Huh-7) |
| LPAR1 | Up | LPA | Increased invasion and migration [55], increased proliferation [56] | HCC patients, human HCC cell lines (SK-Hep1, HepG2 and HuH-7) |
| LPAR3 | Up | LPA | Increased invasion and migration [56] | HCC patients, human HCC cell lines (SK-Hep1, HepG2 and HuH-7) |
| LPAR6 | Up | LPA | Increased proliferation, invasion and migration [53,56] | HCC patients, subcutaneous implantation HCC female CD-1 nude athymic mice, human HCC cell lines (HepG2, HuH-7 PLC/PRF/5, and HLE) |
| α1-AR | Down | Catecholamines | Increased ALT, alter tumor microenvironment: decrease energy expenditure, low levels of thyroid hormones, induce hepatocyte injury [59] | HCC patients |
| β1-AR | Up | Catecholamines | Increased proliferation [60] | HCC patients |
| β2-AR | Up | Catecholamines | Increased proliferation, invasion and migration [61,62,63] | HCC patients, DEN-induced HCC mice model, xenograft male nude mice model, human HCC cell lines (SMMC-7721, HepG2, MHCC-97L) human normal liver cell line (L-02) |
| AT1R | Up | Ang II | Increased angiogenesis [67] | human HCC cell lines (MHCC97-L, Bel-7402) |
| AT2R | Up or down | Ang II | Increased proliferation [68], increased angiogenesis [69,70,71] | Intrahepatic tumor mice model, wild-type and AT2-deleted mice, human HCC cell lines (SMMC-7721, Bel7402, HepG2), human fetal liver cell line (L-02) |
| Smo | Up | Sonic hedgehog | Increased invasion and migration [77,78,79,80] | HCC patients, normal human hepatic cell (L-02), human liver cancer cell lines (PLC/PRF/5, Hep3B, SK-HEP-1, Huh7, Bel-7402, and SMMC-7721) |
| GPR49 | Up | Unknown | Increased proliferation [81] | HCC patients, human HCC cell lines (PLC/PRF/5 and HepG2) |
| GPR137 | Up | Unknown | Increased proliferation, decreased apoptosis [82] | Human HCC cell lines (HepG2, Bel7402, Bel7404, SK-HEP-1, Hep3B, SMMC-7721), human embryonic kidney cell line (HEK293T) |
| GPR37 | Up | Unknown | Increased proliferation, decreased apoptosis [83] | HCC patients, human HCC cell line (Huh-7) |
| Antagonists | Targeted-GPCR | Mechanism | Biological Model | Reference |
|---|---|---|---|---|
| RDC018 | CCR2 | Inhibits the recruitment of inflammatory monocytes, infiltration, and M2-polarisation of tumor-associated macrophages resulting in reversal of the immunosuppression status of the tumor microenvironment and activation of an antitumorous CD8+ T cell response. | HCC patients, orthotopic HCC model, murine HCC cell line (Hepa1-6, LPC-H12, and H22), human HCC cell lines (Hep3B, HepG2, BEL-7404, SMMC-7721, Huh-7, PVTT-1, MHCC-97L, MHCC-97H, and MHCC-LM3), human liver cell lines (HL-7702 and L-02) | [84] |
| Maraviroc | CCR5 | Inhibits hepatic stellate cells activation markers such as phosphorylation of p38 and ERK, and increases hepatocyte survival. | Choline-containing diet male C57BL/6 mice | [24,85] |
| Met-RANTEs | CCR1, CCR5 | Reduced inflammation by reducing periductal accumulation of CD24+ oval cells and abrogation of fibrosis. | Wild-type C57Bl/6J, CCR5-deficient mice, double mutant Mdr2:CCR5 and Mdr2:CCR1 DKO mice | [24] |
| AMD3100 | CXCR4 | In combination with sorafenib treatment block CXCR4/SDF1α, prevents the infiltration of tumor-associated macrophages. enhanced antiangiogenic effect and suppress local and distant tumor growth in HCC. | Orthotopically implanted HCC Male C3H/HeNCrNarl mice, murine HCC cell line(HCA-1), human HCC cell line(JHH-7) | [86,87] |
| CCX771 | CXCR7 | Inhibited CXCR7-induced phosphorylation of ERK1/2 signaling and secretion of the proangiogenic factors VEGF-A. | HCC patients, hepatic cell lines (L-02 and QSG7701), human HCC cell lines (QGY7703, HepG2, Hep3B, MHCC97L, MHCC97H, HCCLM3, HCCLM6) | [40] |
| ONO-8711 | EP1 | Decreased EGFR phosphorylation and tumor cell invasion. | HCC patients, human HCC cell lines (Hep3B, Huh7, and HepG2) | [88,89] |
| AH6809 | EP1 | Combination of EP1 receptor antagonist and COX inhibitors produced a significantly greater cell growth inhibition than the single agent alone. | HCC patients with hepatitis virus-associated chronic liver disease, human HCC cell lines (HuH-6 and Huh7) | [90] |
| AH23848 | EP4 | Inhibition of HSC-derived PGE2 could inhibit HSC-induced MDSC accumulation and HCC growth. | Adult male BALB/c mice injected intra-hepatically with cell suspension, mouse hepatoma cell line (H22) | [91] |
| Propranolol | β2-AR | Enhanced autophagy, HIF-1α destabilization, tumor growth suppression, and improved anti-tumor activity of sorafenib. | Human patients, DEN-induced HCC mouse model, human HCC cell line (SMMC-7721) | [63] |
| BQ123 | ETAR | Blockade of ETAR inhibits HCC cell migration and invasion via blocking ERK1/2 and Akt signaling pathways and lowering MMP-3 expression. | Human liver cell line (L-02), human HCC cell lines (MHCC97-L, Bel-7402, SMMC-7721, and HepG2) | [92] |
| GDC-0449 (vismodegib) | Smo | Blockade of Smo and Shh signaling pathway result in a successful mitigation of ML-1 growth. | Implantation of mice hepatoma ML-1 cells C57BL/6 mice model | [93] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peng, W.-T.; Sun, W.-Y.; Li, X.-R.; Sun, J.-C.; Du, J.-J.; Wei, W. Emerging Roles of G Protein-Coupled Receptors in Hepatocellular Carcinoma. Int. J. Mol. Sci. 2018, 19, 1366. https://doi.org/10.3390/ijms19051366
Peng W-T, Sun W-Y, Li X-R, Sun J-C, Du J-J, Wei W. Emerging Roles of G Protein-Coupled Receptors in Hepatocellular Carcinoma. International Journal of Molecular Sciences. 2018; 19(5):1366. https://doi.org/10.3390/ijms19051366
Chicago/Turabian StylePeng, Wen-Ting, Wu-Yi Sun, Xin-Ran Li, Jia-Chang Sun, Jia-Jia Du, and Wei Wei. 2018. "Emerging Roles of G Protein-Coupled Receptors in Hepatocellular Carcinoma" International Journal of Molecular Sciences 19, no. 5: 1366. https://doi.org/10.3390/ijms19051366
APA StylePeng, W.-T., Sun, W.-Y., Li, X.-R., Sun, J.-C., Du, J.-J., & Wei, W. (2018). Emerging Roles of G Protein-Coupled Receptors in Hepatocellular Carcinoma. International Journal of Molecular Sciences, 19(5), 1366. https://doi.org/10.3390/ijms19051366