Immunology Guides Skeletal Muscle Regeneration
Abstract
:1. Clinical Background
2. Muscle Regeneration
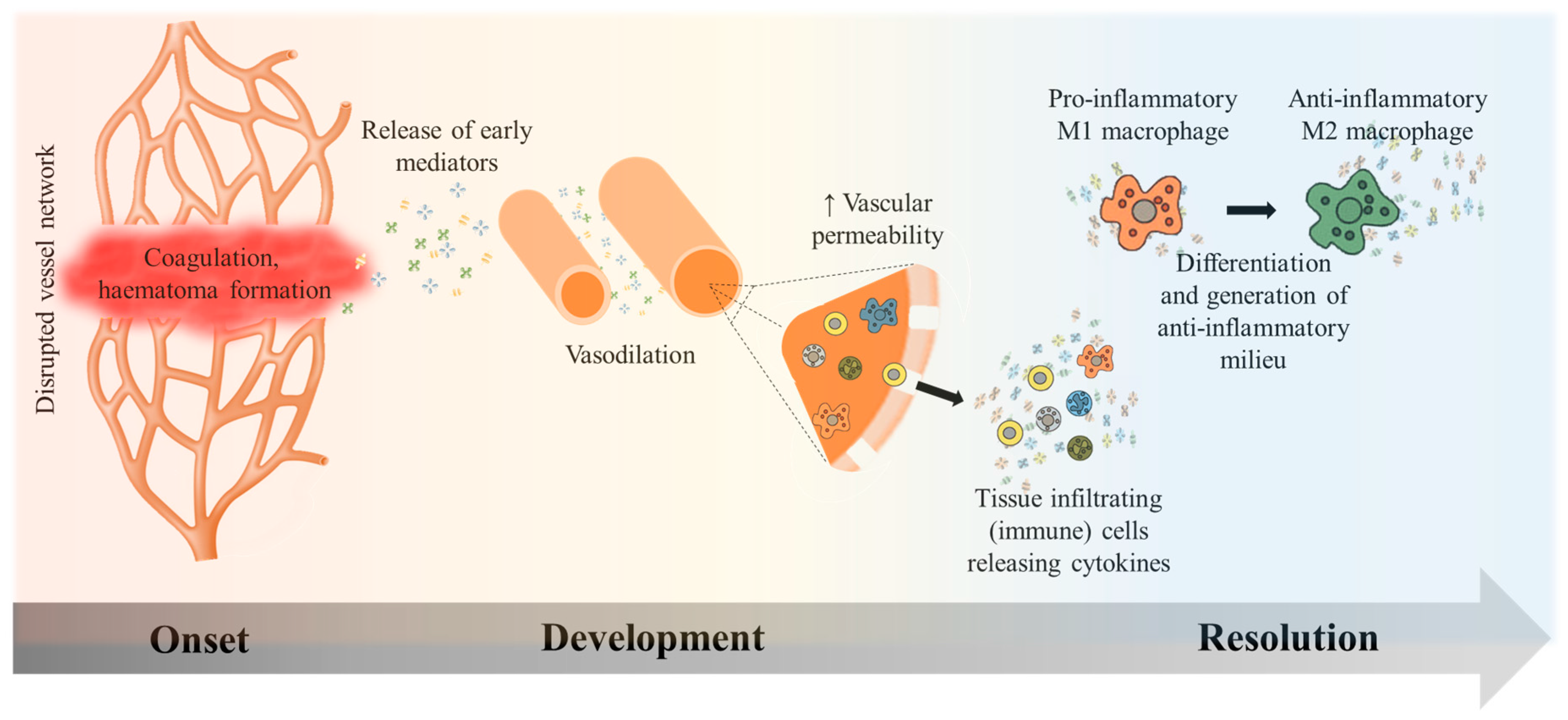
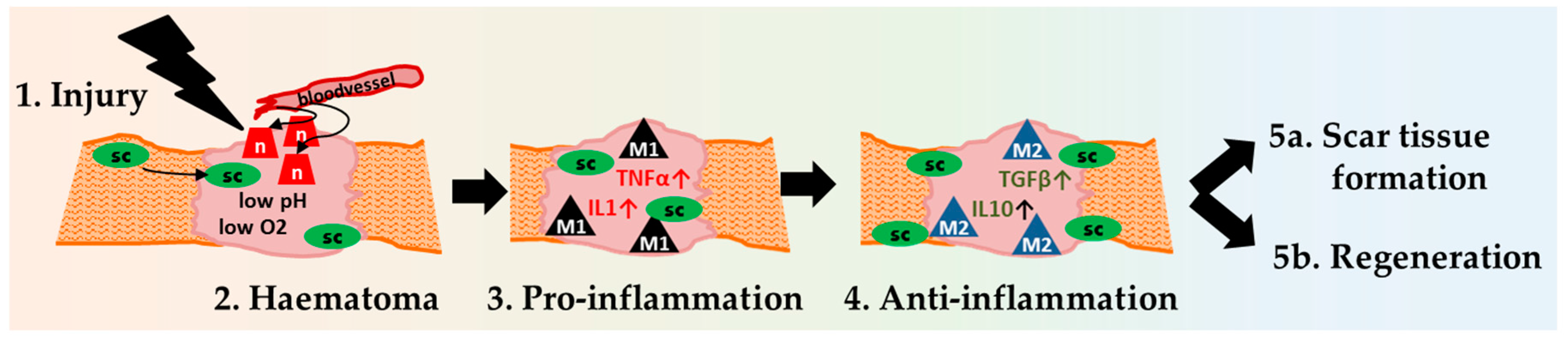
2.1. Inflammation Onset, Development, and Resolution
2.2. Satellite Cells—Multipotent, Muscle Specific Stem Cells
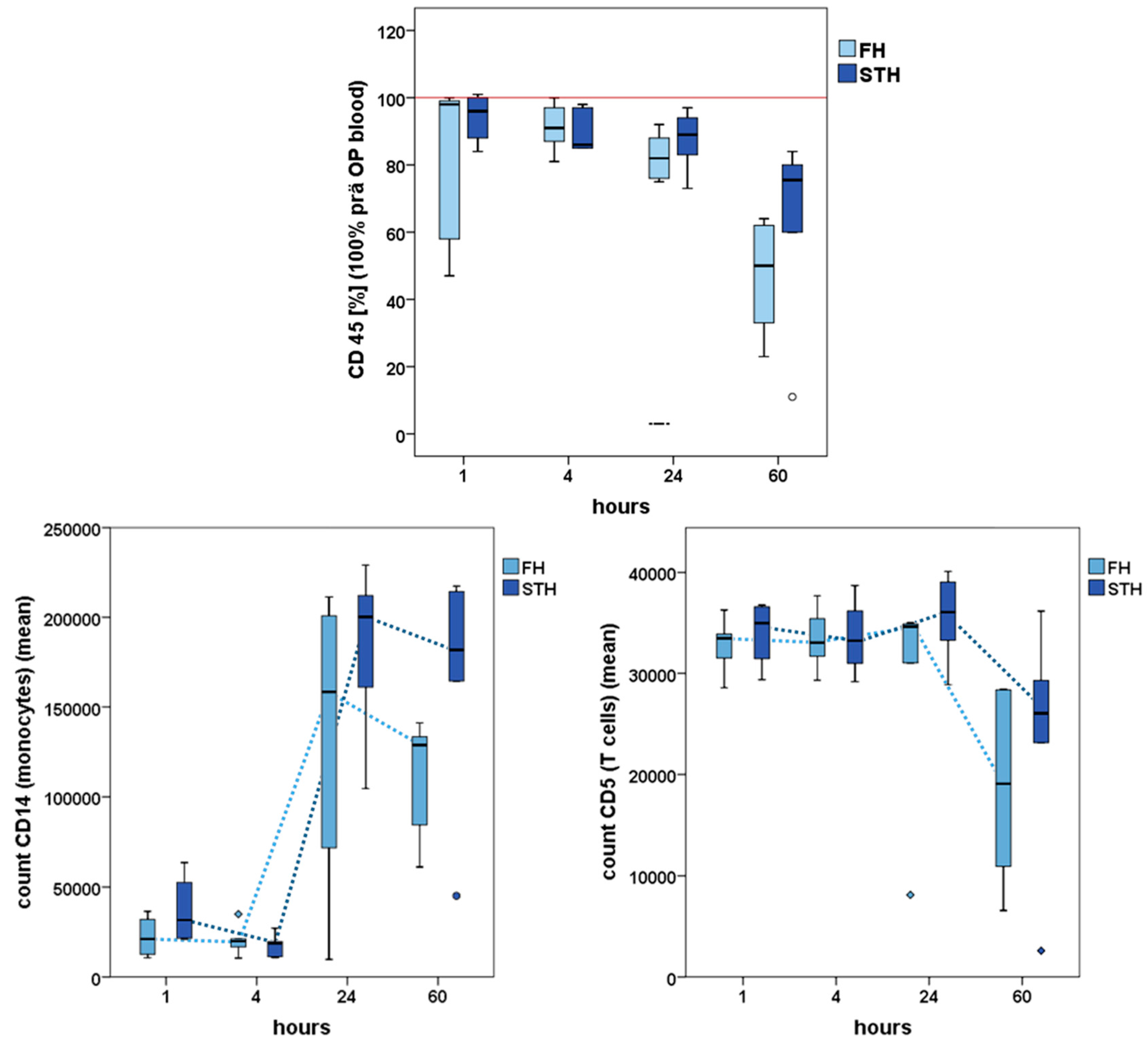
2.3. Interstitial Cells Partake in the Healing Process
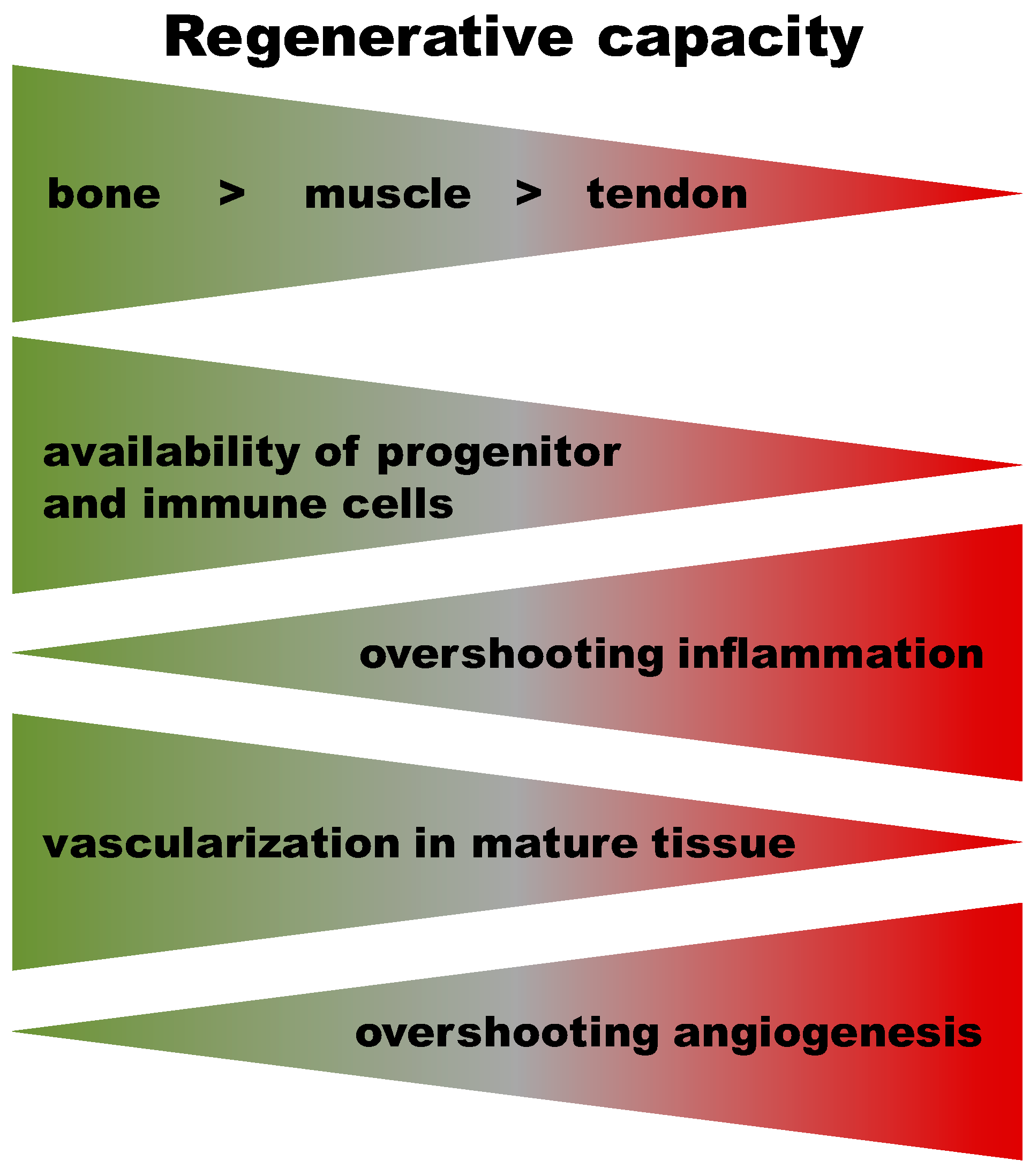
3. Commonalities and Differences in Healing Cascades of Different Tissues
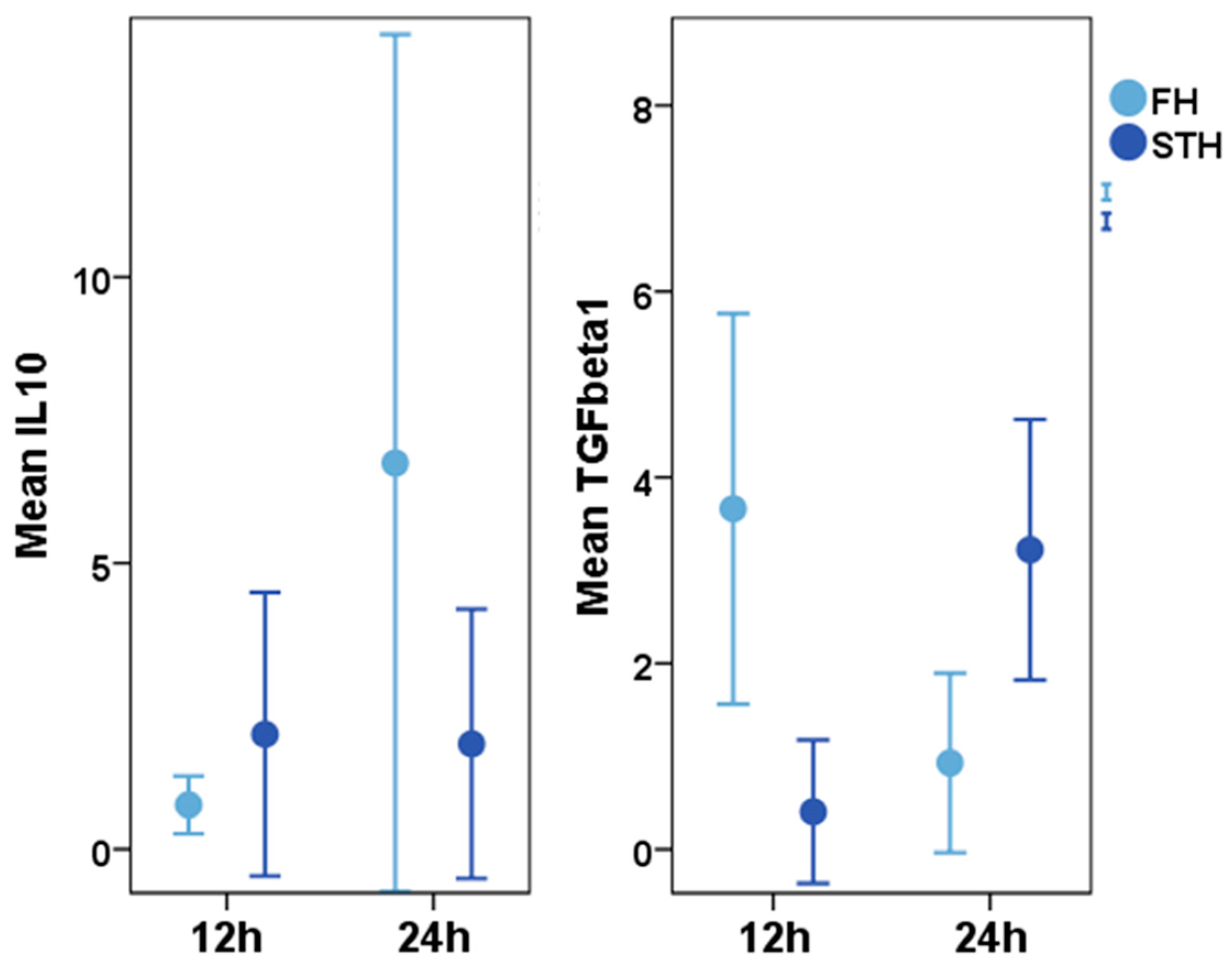
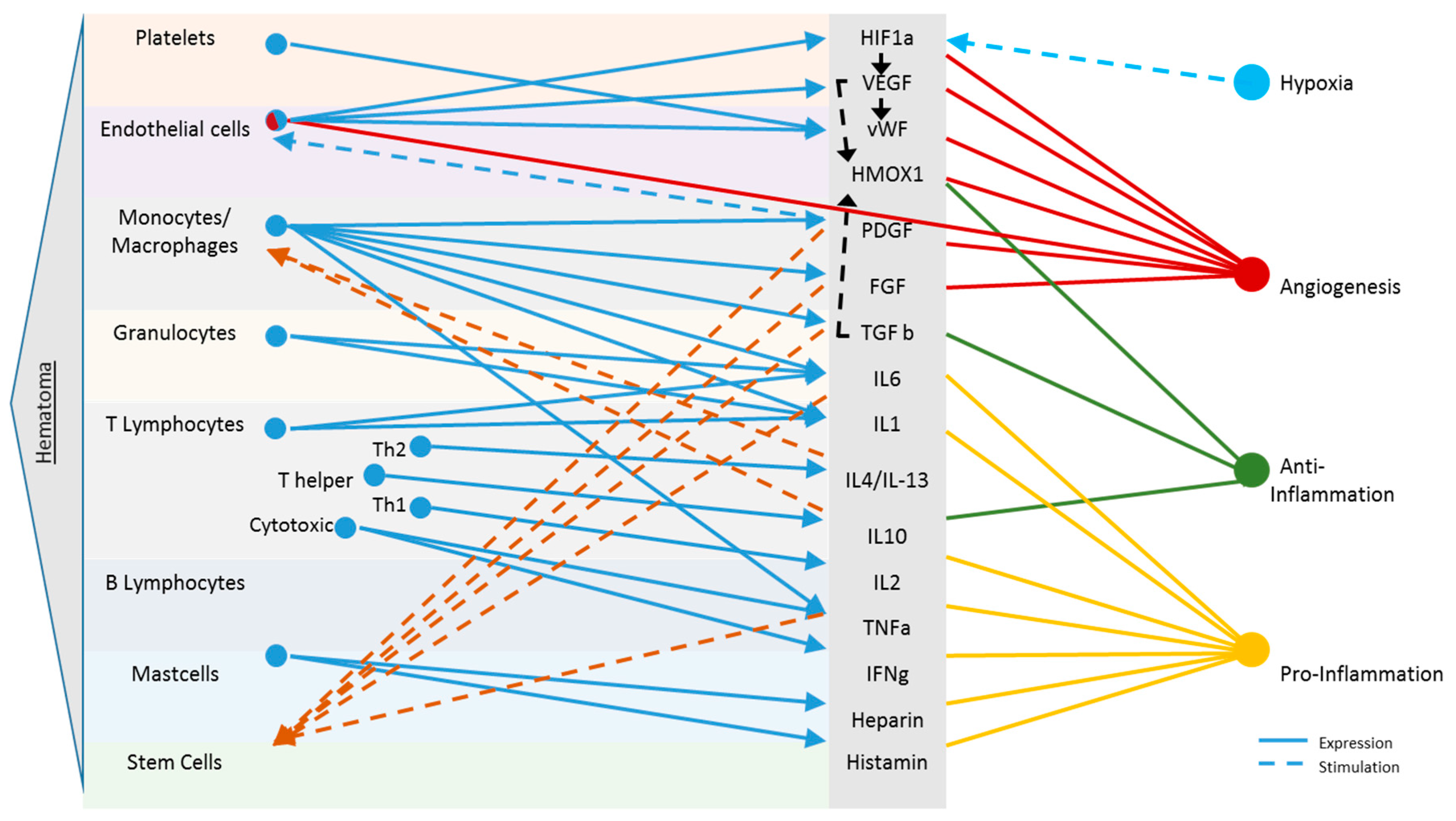
Early Haematoma, Molecular Interplay, and Differential Regulation in Regeneration
4. Myoimmunology-Based (Cell) Therapeutic Options and Future Directions
5. Concluding Remarks
Acknowledgments
Conflicts of Interest
References
- Janssen, I.; Heymsfield, S.B.; Wang, Z.M.; Ross, R. Skeletal muscle mass and distribution in 468 men and women aged 18-88 yr. J. Appl. Physiol. 2000, 89, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Garrett, W.E., Jr.; Swiontkowski, M.F.; Weinstein, J.N.; Callaghan, J.; Rosier, R.N.; Berry, D.J.; Harrast, J.; Derosa, G.P. American Board of Orthopaedic Surgery Practice of the Orthopaedic Surgeon: Part-II, certification examination case mix. J. Bone Jt. Surg. Am. 2006, 88, 660–667. [Google Scholar] [CrossRef]
- Pumberger, M.; Chiu, Y.L.; Ma, Y.; Girardi, F.P.; Mazumdar, M.; Memtsoudis, S.G. National in-hospital morbidity and mortality trends after lumbar fusion surgery between 1998 and 2008. J. Bone Jt. Surg. Br. 2012, 94, 359–364. [Google Scholar] [CrossRef] [PubMed]
- Memtsoudis, S.G.; Pumberger, M.; Ma, Y.; Chiu, Y.L.; Fritsch, G.; Gerner, P.; Poultsides, L.; Valle, A.G. Epidemiology and risk factors for perioperative mortality after total hip and knee arthroplasty. J. Orthop. Res. 2012, 30, 1811–1821. [Google Scholar] [CrossRef] [PubMed]
- De Anta-Diaz, B.; Serralta-Gomis, J.; Lizaur-Utrilla, A.; Benavidez, E.; Lopez-Prats, F.A. No differences between direct anterior and lateral approach for primary total hip arthroplasty related to muscle damage or functional outcome. Int. Orthop. 2016, 40, 2025–2030. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; Chen, H.; Chen, C.; Liang, X.; Huang, W. The strength and function of hip abductors following anterolateral minimally invasive total hip arthroplasty. Chin. J. Traumatol. 2014, 17, 73–78. [Google Scholar] [PubMed]
- Von Roth, P.; Abdel, M.P.; Wauer, F.; Winkler, T.; Wassilew, G.; Diederichs, G.; Perka, C. Significant muscle damage after multiple revision total hip replacements through the direct lateral approach. Bone Jt. J. 2014, 96-B, 1618–1622. [Google Scholar] [CrossRef] [PubMed]
- Perka, C.; Heller, M.; Wilke, K.; Taylor, W.R.; Haas, N.P.; Zippel, H.; Duda, G.N. Surgical approach influences periprosthetic femoral bone density. Clin. Orthop. Relat. Res. 2005, 432, 153–159. [Google Scholar] [CrossRef]
- Muller, M.; Tohtz, S.; Dewey, M.; Springer, I.; Perka, C. Evidence of reduced muscle trauma through a minimally invasive anterolateral approach by means of MRI. Clin. Orthop. Relat. Res. 2010, 468, 3192–3200. [Google Scholar] [CrossRef] [PubMed]
- Odak, S.; Ivory, J. Management of abductor mechanism deficiency following total hip replacement. Bone Jt. J. 2013, 95-B, 343–347. [Google Scholar] [CrossRef] [PubMed]
- Muller, M.; Tohtz, S.; Dewey, M.; Springer, I.; Perka, C. Age-related appearance of muscle trauma in primary total hip arthroplasty and the benefit of a minimally invasive approach for patients older than 70 years. Int. Orthop. 2011, 35, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Hartwig, T.; Streitparth, F.; Gross, C.; Muller, M.; Perka, C.; Putzier, M.; Strube, P. Digital 3-dimensional analysis of the paravertebral lumbar muscles after circumferential single-level fusion. J. Spinal Disord. Tech. 2011, 24, 451–454. [Google Scholar] [CrossRef] [PubMed]
- Vasilakis, I.; Solomou, E.; Vitsas, V.; Fennema, P.; Korovessis, P.; Siamblis, D.K. Correlative analysis of MRI-evident abductor hip muscle degeneration and power after minimally invasive versus conventional unilateral cementless THA. Orthopedics 2012, 35, e1684–e1691. [Google Scholar] [CrossRef] [PubMed]
- Weber, C.O. Ueber die Neubildung quergestreifter Muskelfasern, insbesondere die regenerative Neubildung derselben nach Verletzungen. Virchows Arch. 1867, 39, 216–253. [Google Scholar] [CrossRef]
- Heiderscheit, B.C.; Sherry, M.A.; Silder, A.; Chumanov, E.S.; Thelen, D.G. Hamstring strain injuries: Recommendations for diagnosis, rehabilitation, and injury prevention. J. Orthop. Sports Phys. Ther. 2010, 40, 67–81. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Bleek, K.; Schell, H.; Lienau, J.; Schulz, N.; Hoff, P.; Pfaff, M.; Schmidt, G.; Martin, C.; Perka, C.; Buttgereit, F.; et al. Initial immune reaction and angiogenesis in bone healing. J. Tissue Eng. Regen. Med. 2014, 8, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Schell, H.; Duda, G.N.; Peters, A.; Tsitsilonis, S.; Johnson, K.A.; Schmidt-Bleek, K. The haematoma and its role in bone healing. J. Exp. Orthop. 2017, 4, 5. [Google Scholar] [CrossRef] [PubMed]
- Kolar, P.; Schmidt-Bleek, K.; Schell, H.; Gaber, T.; Toben, D.; Schmidmaier, G.; Perka, C.; Buttgereit, F.; Duda, G.N. The early fracture hematoma and its potential role in fracture healing. Tissue Eng. Part B Rev. 2010, 16, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Opal, S.M. Phylogenetic and functional relationships between coagulation and the innate immune response. Crit. Care Med. 2000, 28 (Suppl. S9), S77–S80. [Google Scholar] [CrossRef] [PubMed]
- Gaudenzio, N.; Sibilano, R.; Marichal, T.; Starkl, P.; Reber, L.L.; Cenac, N.; McNeil, B.D.; Dong, X.; Hernandez, J.D.; Sagi-Eisenberg, R.; et al. Different activation signals induce distinct mast cell degranulation strategies. J. Clin. Investig. 2016, 126, 3981–3998. [Google Scholar] [CrossRef] [PubMed]
- Duchesne, E.; Bouchard, P.; Roussel, M.P.; Cote, C.H. Mast cells can regulate skeletal muscle cell proliferation by multiple mechanisms. Muscle Nerve 2013, 48, 403–414. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.Y.; Nunez, G. Sterile inflammation: Sensing and reacting to damage. Nat. Rev. Immunol. 2010, 10, 826–837. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Bleek, K.; Kwee, B.J.; Mooney, D.J.; Duda, G.N. Boon and Bane of Inflammation in Bone Tissue Regeneration and Its Link with Angiogenesis. Tissue Eng. Part B Rev. 2015. [Google Scholar] [CrossRef] [PubMed]
- Hunt, T.K.; Twomey, P.; Zederfeldt, B.; Dunphy, J.E. Respiratory gas tensions and pH in healing wounds. Am. J. Surg. 1967, 114, 302–307. [Google Scholar] [CrossRef]
- Woo, Y.C.; Park, S.S.; Subieta, A.R.; Brennan, T.J. Changes in tissue pH and temperature after incision indicate acidosis may contribute to postoperative pain. Anesthesiology 2004, 101, 468–475. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Lee, D.; Theusch, B.E.; Arpey, C.J.; Brennan, T.J. Wound hypoxia in deep tissue after incision in rats. Wound Repair Regen. 2013, 21, 730–739. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Zhao, Q.; Yang, T.; Ding, W.; Zhao, Y. Cellular metabolism and macrophage functional polarization. Int. Rev. Immunol. 2015, 34, 82–100. [Google Scholar] [CrossRef] [PubMed]
- Marshall, J.S. Mast-cell responses to pathogens. Nat. Rev. Immunol. 2004, 4, 787–799. [Google Scholar] [CrossRef] [PubMed]
- Luster, A.D.; Alon, R.; von Andrian, U.H. Immune cell migration in inflammation: Present and future therapeutic targets. Nat. Immunol. 2005, 6, 1182–1190. [Google Scholar] [CrossRef] [PubMed]
- Fielding, R.A.; Manfredi, T.J.; Ding, W.; Fiatarone, M.A.; Evans, W.J.; Cannon, J.G. Acute phase response in exercise. III. Neutrophil and IL-1 beta accumulation in skeletal muscle. Am. J. Physiol. 1993, 265, R166–R172. [Google Scholar] [CrossRef] [PubMed]
- Tidball, J.G.; Villalta, S.A. Regulatory interactions between muscle and the immune system during muscle regeneration. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 298, R1173–R1187. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Li, Y.; Wu, Y.; Wang, L.; Wang, X.; Du, J. Interleukin-6/signal transducer and activator of transcription 3 (STAT3) pathway is essential for macrophage infiltration and myoblast proliferation during muscle regeneration. J. Biol. Chem. 2013, 288, 1489–1499. [Google Scholar] [CrossRef] [PubMed]
- Arnold, L.; Henry, A.; Poron, F.; Baba-Amer, Y.; van Rooijen, N.; Plonquet, A.; Gherardi, R.K.; Chazaud, B. Inflammatory monocytes recruited after skeletal muscle injury switch into antiinflammatory macrophages to support myogenesis. J. Exp. Med. 2007, 204, 1057–1069. [Google Scholar] [CrossRef] [PubMed]
- Chazaud, B.; Brigitte, M.; Yacoub-Youssef, H.; Arnold, L.; Gherardi, R.; Sonnet, C.; Lafuste, P.; Chretien, F. Dual and beneficial roles of macrophages during skeletal muscle regeneration. Exerc. Sport Sci. Rev. 2009, 37, 18–22. [Google Scholar] [CrossRef] [PubMed]
- Sunderkotter, C.; Steinbrink, K.; Goebeler, M.; Bhardwaj, R.; Sorg, C. Macrophages and angiogenesis. J. Leukoc. Biol. 1994, 55, 410–422. [Google Scholar] [CrossRef] [PubMed]
- Shimokado, K.; Raines, E.W.; Madtes, D.K.; Barrett, T.B.; Benditt, E.P.; Ross, R. A significant part of macrophage-derived growth factor consists of at least two forms of PDGF. Cell 1985, 43, 277–286. [Google Scholar] [CrossRef]
- Grounds, M.D. Phagocytosis of necrotic muscle in muscle isografts is influenced by the strain, age, and sex of host mice. J. Pathol. 1987, 153, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Duchesne, E.; Tremblay, M.H.; Cote, C.H. Mast cell tryptase stimulates myoblast proliferation; a mechanism relying on protease-activated receptor-2 and cyclooxygenase-2. BMC Musculoskelet. Disord. 2011, 12, 235. [Google Scholar] [CrossRef] [PubMed]
- Dumont, N.; Bouchard, P.; Frenette, J. Neutrophil-induced skeletal muscle damage: A calculated and controlled response following hindlimb unloading and reloading. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 295, R1831–R1838. [Google Scholar] [CrossRef] [PubMed]
- Rigamonti, E.; Zordan, P.; Sciorati, C.; Rovere-Querini, P.; Brunelli, S. Macrophage plasticity in skeletal muscle repair. Biomed. Res. Int. 2014, 2014, 560629. [Google Scholar] [CrossRef] [PubMed]
- Sciorati, C.; Rigamonti, E.; Manfredi, A.A.; Rovere-Querini, P. Cell death, clearance and immunity in the skeletal muscle. Cell Death Differ. 2016, 23, 927–937. [Google Scholar] [CrossRef] [PubMed]
- Cantini, M.; Giurisato, E.; Radu, C.; Tiozzo, S.; Pampinella, F.; Senigaglia, D.; Zaniolo, G.; Mazzoleni, F.; Vitiello, L. Macrophage-secreted myogenic factors: A promising tool for greatly enhancing the proliferative capacity of myoblasts in vitro and in vivo. Neurol. Sci. 2002, 23, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Rocheteau, P.; Gayraud-Morel, B.; Siegl-Cachedenier, I.; Blasco, M.A.; Tajbakhsh, S. A subpopulation of adult skeletal muscle stem cells retains all template DNA strands after cell division. Cell 2012, 148, 112–125. [Google Scholar] [CrossRef] [PubMed]
- Mauro, A. Satellite cell of skeletal muscle fibers. J. Biophys. Biochem. Cytol. 1961, 9, 493–495. [Google Scholar] [CrossRef] [PubMed]
- Dumont, N.A.; Bentzinger, C.F.; Sincennes, M.C.; Rudnicki, M.A. Satellite Cells and Skeletal Muscle Regeneration. Compr. Physiol. 2015, 5, 1027–1059. [Google Scholar] [CrossRef] [PubMed]
- Relaix, F.; Zammit, P.S. Satellite cells are essential for skeletal muscle regeneration: The cell on the edge returns centre stage. Development 2012, 139, 2845–2856. [Google Scholar] [CrossRef] [PubMed]
- Lepper, C.; Partridge, T.A.; Fan, C.M. An absolute requirement for Pax7-positive satellite cells in acute injury-induced skeletal muscle regeneration. Development 2011, 138, 3639–3646. [Google Scholar] [CrossRef] [PubMed]
- Chazaud, B.; Sonnet, C.; Lafuste, P.; Bassez, G.; Rimaniol, A.C.; Poron, F.; Authier, F.J.; Dreyfus, P.A.; Gherardi, R.K. Satellite cells attract monocytes and use macrophages as a support to escape apoptosis and enhance muscle growth. J. Cell. Biol. 2003, 163, 1133–1143. [Google Scholar] [CrossRef] [PubMed]
- Robertson, T.A.; Maley, M.A.; Grounds, M.D.; Papadimitriou, J.M. The role of macrophages in skeletal muscle regeneration with particular reference to chemotaxis. Exp. Cell Res. 1993, 207, 321–331. [Google Scholar] [CrossRef] [PubMed]
- Hawke, T.J.; Garry, D.J. Myogenic satellite cells: Physiology to molecular biology. J. Appl. Physiol. 2001, 91, 534–551. [Google Scholar] [CrossRef] [PubMed]
- Ceafalan, L.C.; Fertig, T.E.; Popescu, A.C.; Popescu, B.O.; Hinescu, M.E.; Gherghiceanu, M. Skeletal muscle regeneration involves macrophage-myoblast bonding. Cell Adh. Migr. 2017, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.M.; Lawson, J.A.; Mathew, S.J.; Hutcheson, D.A.; Kardon, G. Satellite cells, connective tissue fibroblasts and their interactions are crucial for muscle regeneration. Development 2011, 138, 3625–3637. [Google Scholar] [CrossRef] [PubMed]
- Hinz, B.; Mastrangelo, D.; Iselin, C.E.; Chaponnier, C.; Gabbiani, G. Mechanical tension controls granulation tissue contractile activity and myofibroblast differentiation. Am. J. Pathol. 2001, 159, 1009–1020. [Google Scholar] [CrossRef]
- Tomasek, J.J.; Gabbiani, G.; Hinz, B.; Chaponnier, C.; Brown, R.A. Myofibroblasts and mechano-regulation of connective tissue remodelling. Nat. Rev. Mol. Cell Biol. 2002, 3, 349–363. [Google Scholar] [CrossRef] [PubMed]
- Bagalad, B.S.; Mohan Kumar, K.P.; Puneeth, H.K. Myofibroblasts: Master of disguise. J. Oral Maxillofac. Pathol. 2017, 21, 462–463. [Google Scholar] [CrossRef] [PubMed]
- Munoz-Canoves, P.; Serrano, A.L. Macrophages decide between regeneration and fibrosis in muscle. Trends Endocrinol. Metab. 2015, 26, 449–450. [Google Scholar] [CrossRef] [PubMed]
- Barnes, A.E. The specificity of pH and ionic strength effects on the kinetics of the Rh (D)-anti-Rh (D) system. J. Immunol. 1966, 96, 854–864. [Google Scholar] [PubMed]
- Judson, R.N.; Low, M.; Eisner, C.; Rossi, F.M. Isolation, Culture, and Differentiation of Fibro/Adipogenic Progenitors (FAPs) from Skeletal Muscle. Methods Mol. Biol. 2017, 1668, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Gilland, O. [Principles for amantadine therapy in parkinsonism]. Lakartidningen 1972, 69, 5343–5345. [Google Scholar] [PubMed]
- Schlundt, C.; El Khassawna, T.; Serra, A.; Dienelt, A.; Wendler, S.; Schell, H.; van Rooijen, N.; Radbruch, A.; Lucius, R.; Hartmann, S.; et al. Macrophages in bone fracture healing: Their essential role in endochondral ossification. Bone 2015. [Google Scholar] [CrossRef] [PubMed]
- Popescu, L.M.; Manole, E.; Serboiu, C.S.; Manole, C.G.; Suciu, L.C.; Gherghiceanu, M.; Popescu, B.O. Identification of telocytes in skeletal muscle interstitium: Implication for muscle regeneration. J. Cell Mol. Med. 2011, 15, 1379–1392. [Google Scholar] [CrossRef] [PubMed]
- Meeson, A.P.; Hawke, T.J.; Graham, S.; Jiang, N.; Elterman, J.; Hutcheson, K.; Dimaio, J.M.; Gallardo, T.D.; Garry, D.J. Cellular and molecular regulation of skeletal muscle side population cells. Stem Cells 2004, 22, 1305–1320. [Google Scholar] [CrossRef] [PubMed]
- Claes, L.; Recknagel, S.; Ignatius, A. Fracture healing under healthy and inflammatory conditions. Nat. Rev. Rheumatol. 2012, 8, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Hu, P. Hierarchical signaling transduction of the immune and muscle cell crosstalk in muscle regeneration. Cell. Immunol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Reinke, S.; Geissler, S.; Taylor, W.R.; Schmidt-Bleek, K.; Juelke, K.; Schwachmeyer, V.; Dahne, M.; Hartwig, T.; Akyuz, L.; Meisel, C.; et al. Terminally Differentiated CD8+ T Cells Negatively Affect Bone Regeneration in Humans. Sci. Transl. Med. 2013, 5, 177ra36. [Google Scholar] [CrossRef] [PubMed]
- Han, A.A.; Currie, H.N.; Loos, M.S.; Scardoni, G.; Miller, J.V.; Prince, N.; Mouch, J.A.; Boyd, J.W. The impact of cytokine responses in the intra- and extracellular signaling network of a traumatic injury. Cytokine 2017. [Google Scholar] [CrossRef] [PubMed]
- Behm, B.; Babilas, P.; Landthaler, M.; Schreml, S. Cytokines, chemokines and growth factors in wound healing. J. Eur. Acad. Dermatol. Venereol. 2012, 26, 812–820. [Google Scholar] [CrossRef] [PubMed]
- Vandercappellen, J.; Van Damme, J.; Struyf, S. The role of the CXC chemokines platelet factor-4 (CXCL4/PF-4) and its variant (CXCL4L1/PF-4var) in inflammation, angiogenesis and cancer. Cytokine Growth Factor Rev. 2011, 22, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Aloui, C.; Prigent, A.; Sut, C.; Tariket, S.; Hamzeh-Cognasse, H.; Pozzetto, B.; Richard, Y.; Cognasse, F.; Laradi, S.; Garraud, O. The signaling role of CD40 ligand in platelet biology and in platelet component transfusion. Int. J. Mol. Sci. 2014, 15, 22342–22364. [Google Scholar] [CrossRef] [PubMed]
- Karin, N. The multiple faces of CXCL12 (SDF-1alpha) in the regulation of immunity during health and disease. J. Leukoc. Biol. 2010, 88, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Fujio, K.; Komai, T.; Inoue, M.; Morita, K.; Okamura, T.; Yamamoto, K. Revisiting the regulatory roles of the TGF-beta family of cytokines. Autoimmun. Rev. 2016, 15, 917–922. [Google Scholar] [CrossRef] [PubMed]
- Rutz, S.; Ouyang, W. Regulation of interleukin-10 and interleukin-22 expression in T helper cells. Curr. Opin. Immunol. 2011, 23, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Chillemi, C.; Petrozza, V.; Garro, L.; Sardella, B.; Diotallevi, R.; Ferrara, A.; Gigante, A.; Di Cristofano, C.; Castagna, A.; Della Rocca, C. Rotator cuff re-tear or non-healing: Histopathological aspects and predictive factors. Knee Surg. Sports Traumatol. Arthrosc. 2011, 19, 1588–1596. [Google Scholar] [CrossRef] [PubMed]
- Klatte-Schulz, F.; Gerhardt, C.; Scheibel, M.; Wildemann, B.; Pauly, S. Relationship between muscle fatty infiltration and the biological characteristics and stimulation potential of tenocytes from rotator cuff tears. J. Orthop. Res. 2014, 32, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Sayegh, E.T.; Sandy, J.D.; Virk, M.S.; Romeo, A.A.; Wysocki, R.W.; Galante, J.O.; Trella, K.J.; Plaas, A.; Wang, VM. Recent Scientific Advances Towards the Development of Tendon Healing Strategies. Curr. Tissue Eng. 2015, 4, 128–143. [Google Scholar] [CrossRef] [PubMed]
- Thomopoulos, S.; Parks, W.C.; Rifkin, D.B.; Derwin, K.A. Mechanisms of tendon injury and repair. J. Orthop. Res. 2015, 33, 832–839. [Google Scholar] [CrossRef] [PubMed]
- Paredes, J.J.; Andarawis-Puri, N. Therapeutics for tendon regeneration: A multidisciplinary review of tendon research for improved healing. Ann. N. Y. Acad. Sci. 2016, 1383, 125–138. [Google Scholar] [CrossRef] [PubMed]
- Moyer, A.L.; Wagner, K.R. Regeneration versus fibrosis in skeletal muscle. Curr. Opin. Rheumatol. 2011, 23, 568–573. [Google Scholar] [CrossRef] [PubMed]
- Gharaibeh, B.; Chun-Lansinger, Y.; Hagen, T.; Ingham, S.J.; Wright, V.; Fu, F.; Huard, J. Biological approaches to improve skeletal muscle healing after injury and disease. Birth Defects Res. Part C Embryo Today Rev. 2012, 96, 82–94. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Bleek, K.; Schell, H.; Kolar, P.; Pfaff, M.; Perka, C.; Buttgereit, F.; Duda, G.; Lienau, J. Cellular composition of the initial fracture hematoma compared to a muscle hematoma: A study in sheep. J. Orthop. Res. 2009, 27, 1147–1151. [Google Scholar] [CrossRef] [PubMed]
- Hurtgen, B.J.; Henderson, B.E.P.; Ward, C.L.; Goldman, S.M.; Garg, K.; McKinley, T.O.; Greising, S.M.; Wenke, J.C.; Corona, B.T. Impairment of early fracture healing by skeletal muscle trauma is restored by FK506. BMC Musculoskelet. Disord. 2017, 18, 253. [Google Scholar] [CrossRef] [PubMed]
- Hurtgen, B.J.; Ward, C.L.; Garg, K.; Pollot, B.E.; Goldman, S.M.; McKinley, T.O.; Wenke, J.C.; Corona, B.T. Severe muscle trauma triggers heightened and prolonged local musculoskeletal inflammation and impairs adjacent tibia fracture healing. J. Musculoskelet. Neuronal. Interact. 2016, 16, 122–134. [Google Scholar] [PubMed]
- Granger, D.; Neil, S.E. Inflammation and Microcirculation; Morgan & Claypole Life Sciences: Williston, VT, USA, 2010. [Google Scholar]
- DiPietro, L.A. Angiogenesis and scar formation in healing wounds. Curr.Opin. Rheumatol. 2013, 25, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Muller, S.A.; Todorov, A.; Heisterbach, P.E.; Martin, I.; Majewski, M. Tendon healing: An overview of physiology, biology, and pathology of tendon healing and systematic review of state of the art in tendon bioengineering. Knee Surg. Sports Traumatol. Arthrosc. 2015, 23, 2097–2105. [Google Scholar] [CrossRef] [PubMed]
- Matthews, T.J.; Hand, G.C.; Rees, J.L.; Athanasou, N.A.; Carr, A.J. Pathology of the torn rotator cuff tendon. Reduction in potential for repair as tear size increases. J. Bone Jt. Surg. Br. 2006, 88, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Salazar-Onfray, F.; Lopez, M.N.; Mendoza-Naranjo, A. Paradoxical effects of cytokines in tumor immune surveillance and tumor immune escape. Cytokine Growth Factor Rev. 2007, 18, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Roberts, A.B.; Lamb, L.C.; Newton, D.L.; Sporn, M.B.; De Larco, J.E.; Todaro, G.J. Transforming growth factors: Isolation of polypeptides from virally and chemically transformed cells by acid/ethanol extraction. Proc. Natl. Acad. Sci. USA 1980, 77, 3494–3498. [Google Scholar] [CrossRef] [PubMed]
- Wynn, T.A. Cellular and molecular mechanisms of fibrosis. J. Pathol. 2008, 214, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Zanotti, S.; Saredi, S.; Ruggieri, A.; Fabbri, M.; Blasevich, F.; Romaggi, S.; Morandi, L.; Mora, M. Altered extracellular matrix transcript expression and protein modulation in primary Duchenne muscular dystrophy myotubes. Matrix Biol. 2007, 26, 615–624. [Google Scholar] [CrossRef] [PubMed]
- Foster, W.; Li, Y.; Usas, A.; Somogyi, G.; Huard, J. Gamma interferon as an antifibrosis agent in skeletal muscle. J. Orthop. Res. 2003, 21, 798–804. [Google Scholar] [CrossRef]
- Sassoli, C.; Chellini, F.; Pini, A.; Tani, A.; Nistri, S.; Nosi, D.; Zecchi-Orlandini, S.; Bani, D.; Formigli, L. Relaxin prevents cardiac fibroblast-myofibroblast transition via notch-1-mediated inhibition of TGF-beta/Smad3 signaling. PLoS ONE 2013, 8, e63896. [Google Scholar] [CrossRef] [PubMed]
- Taniguti, A.P.; Pertille, A.; Matsumura, C.Y.; Santo Neto, H.; Marques, M.J. Prevention of muscle fibrosis and myonecrosis in mdx mice by suramin, a TGF-beta1 blocker. Muscle Nerve 2011, 43, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Kelc, R.; Trapecar, M.; Gradisnik, L.; Rupnik, M.S.; Vogrin, M. Platelet-rich plasma, especially when combined with a TGF-beta inhibitor promotes proliferation, viability and myogenic differentiation of myoblasts in vitro. PLoS ONE 2015, 10, e0117302. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Lakhia, R.; Thomas, S.S.; Dong, Y.; Wang, X.H.; Silva, K.A.; Zhang, L. Interactions between p-Akt and Smad3 in injured muscles initiate myogenesis or fibrogenesis. Am. J. Physiol. Endocrinol. Metab. 2013, 305, E367–E375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Delaney, K.; Kasprzycka, P.; Ciemerych, M.A.; Zimowska, M. The role of TGF-beta1 during skeletal muscle regeneration. Cell Biol. Int. 2017, 41, 706–715. [Google Scholar] [CrossRef] [PubMed]
- Zimowska, M.; Kasprzycka, P.; Bocian, K.; Delaney, K.; Jung, P.; Kuchcinska, K.; Kaczmarska, K.; Gladysz, D.; Streminska, W.; Ciemerych, M.A. Inflammatory response during slow- and fast-twitch muscle regeneration. Muscle Nerve 2017, 55, 400–409. [Google Scholar] [CrossRef] [PubMed]
- Veldhoen, M.; Hocking, R.J.; Atkins, C.J.; Locksley, R.M.; Stockinger, B. TGFbeta in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17-producing T cells. Immunity 2006, 24, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Jarnicki, A.G.; Lysaght, J.; Todryk, S.; Mills, K.H. Suppression of antitumor immunity by IL-10 and TGF-beta-producing T cells infiltrating the growing tumor: Influence of tumor environment on the induction of CD4+ and CD8+ regulatory T cells. J. Immunol. 2006, 177, 896–904. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, M.W. Scar wars. Br. Dent. J. 2002, 192, 475. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, M.W.; O’Kane, S. Scar-free healing: From embryonic mechanisms to adult therapeutic intervention. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2004, 359, 839–850. [Google Scholar] [CrossRef] [PubMed]
- De Lima Rodrigues, D.; Alves, A.N.; Guimaraes, B.R.; de Alcantara Araujo Amorim, W.W.; Bussadori, S.K.; Fernandes, K.P.S.; Mesquita-Ferrari, R.A. Effect of prior application with and without post-injury treatment with low-level laser on the modulation of key proteins in the muscle repair process. Lasers Med. Sci. 2018. [Google Scholar] [CrossRef] [PubMed]
- Demonbreun, A.R.; McNally, E.M. Muscle cell communication in development and repair. Curr. Opin. Pharmacol. 2017, 34, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.B.; Kollias, H.D.; Wagner, K.R. Myostatin directly regulates skeletal muscle fibrosis. J. Biol. Chem. 2008, 283, 19371–19378. [Google Scholar] [CrossRef] [PubMed]
- Tsao, J.; Kovanecz, I.; Awadalla, N.; Gelfand, R.; Sinha-Hikim, I.; White, R.A.; Gonzalez-Cadavid, N.F. Muscle Derived Stem Cells Stimulate Muscle Myofiber Repair and Counteract Fat Infiltration in a Diabetic Mouse Model of Critical Limb Ischemia. J. Stem Cell Res. Ther. 2016, 6. [Google Scholar] [CrossRef]
- Qin, Y.; Peng, Y.; Zhao, W.; Pan, J.; Ksiezak-Reding, H.; Cardozo, C.; Wu, Y.; Divieti Pajevic, P.; Bonewald, L.F.; Bauman, W.A.; et al. Myostatin inhibits osteoblastic differentiation by suppressing osteocyte-derived exosomal microRNA-218: A novel mechanism in muscle-bone communication. J. Biol. Chem. 2017, 292, 11021–11033. [Google Scholar] [CrossRef] [PubMed]
- Wallner, C.; Jaurich, H.; Wagner, J.M.; Becerikli, M.; Harati, K.; Dadras, M.; Lehnhardt, M.; Behr, B. Inhibition of GDF8 (Myostatin) accelerates bone regeneration in diabetes mellitus type 2. Sci. Rep. 2017, 7, 9878. [Google Scholar] [CrossRef] [PubMed]
- Braun, A.C.; Gutmann, M.; Ebert, R.; Jakob, F.; Gieseler, H.; Luhmann, T.; Meinel, L. Matrix Metalloproteinase Responsive Delivery of Myostatin Inhibitors. Pharm. Res. 2017, 34, 58–72. [Google Scholar] [CrossRef] [PubMed]
- Konnecke, I.; Serra, A.; El Khassawna, T.; Schlundt, C.; Schell, H.; Hauser, A.; Ellinghaus, A.; Volk, H.D.; Radbruch, A.; Duda, G.N.; et al. T and B cells participate in bone repair by infiltrating the fracture callus in a two-wave fashion. Bone 2014, 64, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Bleek, K.; Schell, H.; Schulz, N.; Hoff, P.; Perka, C.; Buttgereit, F.; Volk, H.D.; Lienau, J.; Duda, G.N. Inflammatory phase of bone healing initiates the regenerative healing cascade. Cell Tissue Res. 2012, 347, 567–573. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Bleek, K.; Petersen, A.; Dienelt, A.; Schwarz, C.; Duda, G.N. Initiation and early control of tissue regeneration—Bone healing as a model system for tissue regeneration. Expert Opin. Biol. Ther. 2014, 14, 247–259. [Google Scholar] [CrossRef] [PubMed]
- Mountziaris, P.M.; Spicer, P.P.; Kasper, F.K.; Mikos, A.G. Harnessing and Modulating Inflammation in Strategies for Bone Regeneration. Tissue Eng. Part B Rev. 2011. [Google Scholar] [CrossRef] [PubMed]
- Mountziaris, P.M.; Mikos, A.G. Modulation of the Inflammatory Response for Enhanced Bone Tissue Regeneration. Tissue Eng. Part B Rev. 2008, 14, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Gerstenfeld, L.C.; Cullinane, D.M.; Barnes, G.L.; Graves, D.T.; Einhorn, T.A. Fracture healing as a post-natal developmental process: Molecular, spatial, and temporal aspects of its regulation. J. Cell. Biochem. 2003, 88, 873–884. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wan, C.; Deng, L.; Liu, X.; Cao, X.; Gilbert, S.R.; Bouxsein, M.L.; Faugere, M.C.; Guldberg, R.E.; Gerstenfeld, L.C.; et al. The hypoxia-inducible factor alpha pathway couples angiogenesis to osteogenesis during skeletal development. J. Clin. Investig. 2007, 117, 1616–1626. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.R.; Dragoo, J.L. The effect of nonsteroidal anti-inflammatory drugs on tissue healing. Knee Surg. Sports Traumatol. Arthrosc. 2013, 21, 540–549. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, L.; Kikuiri, T.; Akiyama, K.; Chen, C.; Xu, X.; Yang, R.; Chen, W.; Wang, S.; Shi, S. Mesenchymal stem cell-based tissue regeneration is governed by recipient T lymphocytes via IFN-gamma and TNF-alpha. Nat. Med. 2011, 17, 1594–1601. [Google Scholar] [CrossRef] [PubMed]
- Sass, F.A.; Schmidt-Bleek, K.; Ellinghaus, A.; Filter, S.; Rose, A.; Preininger, B.; Reinke, S.; Geissler, S.; Volk, H.D.; Duda, G.N.; et al. CD31+ Cells From Peripheral Blood Facilitate Bone Regeneration in Biologically Impaired Conditions Through Combined Effects on Immunomodulation and Angiogenesis. J. Bone Miner. Res. 2016. [Google Scholar] [CrossRef] [PubMed]
- Toben, D.; Schroeder, I.; El Khassawna, T.; Mehta, M.; Hoffmann, J.E.; Frisch, J.T.; Schell, H.; Lienau, J.; Serra, A.; Radbruch, A.; et al. Fracture healing is accelerated in the absence of the adaptive immune system. J. Bone Miner. Res. 2011, 26, 113–124. [Google Scholar] [CrossRef] [PubMed]
- El Khassawna, T.; Serra, A.; Bucher, C.H.; Petersen, A.; Schlundt, C.; Konnecke, I.; Malhan, D.; Wendler, S.; Schell, H.; Volk, H.D.; et al. T Lymphocytes Influence the Mineralization Process of Bone. Front. Immunol. 2017, 8, 562. [Google Scholar] [CrossRef] [PubMed]
- Burzyn, D.; Kuswanto, W.; Kolodin, D.; Shadrach, J.L.; Cerletti, M.; Jang, Y.; Sefik, E.; Tan, T.G.; Wagers, A.J.; Benoist, C. A special population of regulatory T cells potentiates muscle repair. Cell 2013, 155, 1282–1295. [Google Scholar] [CrossRef] [PubMed]
- Deng, B.; Wehling-Henricks, M.; Villalta, S.A.; Wang, Y.; Tidball, J.G. IL-10 triggers changes in macrophage phenotype that promote muscle growth and regeneration. J. Immunol. 2012, 189, 3669–3680. [Google Scholar] [CrossRef] [PubMed]
- Biswas, S.K.; Mantovani, A. Macrophage plasticity and interaction with lymphocyte subsets: Cancer as a paradigm. Nat. Immunol. 2010, 11, 889–896. [Google Scholar] [CrossRef] [PubMed]
- Novak, M.L.; Weinheimer-Haus, E.M.; Koh, T.J. Macrophage activation and skeletal muscle healing following traumatic injury. J. Pathol. 2014, 232, 344–355. [Google Scholar] [CrossRef] [PubMed]
- Thomas, J.A.; Pope, C.; Wojtacha, D.; Robson, A.J.; Gordon-Walker, T.T.; Hartland, S.; Ramachandran, P.; Van Deemter, M.; Hume, D.A.; Iredale, J.P.; et al. Macrophage therapy for murine liver fibrosis recruits host effector cells improving fibrosis, regeneration, and function. Hepatology 2011, 53, 2003–2015. [Google Scholar] [CrossRef] [PubMed]
- Dezawa, M.; Ishikawa, H.; Itokazu, Y.; Yoshihara, T.; Hoshino, M.; Takeda, S.; Ide, C.; Nabeshima, Y. Bone marrow stromal cells generate muscle cells and repair muscle degeneration. Science 2005, 309, 314–317. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Menocal, L.; Shareef, S.; Salgado, M.; Shabbir, A.; Van Badiavas, E. Role of whole bone marrow, whole bone marrow cultured cells, and mesenchymal stem cells in chronic wound healing. Stem Cell Res. Ther. 2015, 6, 24. [Google Scholar] [CrossRef] [PubMed]
- Kasper, G.; Dankert, N.; Tuischer, J.; Hoeft, M.; Gaber, T.; Glaeser, J.D.; Zander, D.; Tschirschmann, M.; Thompson, M.; Matziolis, G.; et al. Mesenchymal stem cells regulate angiogenesis according to their mechanical environment. Stem Cells 2007, 25, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Qu-Petersen, Z.; Deasy, B.; Jankowski, R.; Ikezawa, M.; Cummins, J.; Pruchnic, R.; Mytinger, J.; Cao, B.; Gates, C.; Wernig, A.; et al. Identification of a novel population of muscle stem cells in mice: Potential for muscle regeneration. J. Cell Biol. 2002, 157, 851–864. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Huard, J. Differentiation of muscle-derived cells into myofibroblasts in injured skeletal muscle. Am. J. Pathol. 2002, 161, 895–907. [Google Scholar] [CrossRef]
- Caplan, A.I. Adult Mesenchymal Stem Cells: When, Where, and How. Stem Cells Int. 2015, 2015, 628767. [Google Scholar] [CrossRef] [PubMed]
- Crisan, M.; Yap, S.; Casteilla, L.; Chen, C.W.; Corselli, M.; Park, T.S.; Andriolo, G.; Sun, B.; Zheng, B.; Zhang, L.; et al. A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell 2008, 3, 301–313. [Google Scholar] [CrossRef] [PubMed]
- Caplan, A.I. All MSCs are pericytes? Cell Stem Cell 2008, 3, 229–230. [Google Scholar] [CrossRef] [PubMed]
- Murray, I.R.; Baily, J.E.; Chen, W.C.W.; Dar, A.; Gonzalez, Z.N.; Jensen, A.R.; Petrigliano, F.A.; Deb, A.; Henderson, N.C. Skeletal and cardiac muscle pericytes: Functions and therapeutic potential. Pharmacol. Ther. 2017, 171, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Winkler, T.; von Roth, P.; Matziolis, G.; Schumann, M.R.; Hahn, S.; Strube, P.; Stoltenburg-Didinger, G.; Perka, C.; Duda, G.N.; Tohtz, S.V. Time course of skeletal muscle regeneration after severe trauma. Acta Orthop. 2011, 82, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Von Roth, P.; Duda, G.N.; Radojewski, P.; Preininger, B.; Perka, C.; Winkler, T. Mesenchymal stem cell therapy following muscle trauma leads to improved muscular regeneration in both male and female rats. Gend. Med. 2012, 9, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Matziolis, G.; Winkler, T.; Schaser, K.; Wiemann, M.; Krocker, D.; Tuischer, J.; Perka, C.; Duda, G.N. Autologous bone marrow-derived cells enhance muscle strength following skeletal muscle crush injury in rats. Tissue Eng. 2006, 12, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Winkler, T.; von Roth, P.; Schuman, M.R.; Sieland, K.; Stoltenburg-Didinger, G.; Taupitz, M.; Perka, C.; Duda, G.N.; Matziolis, G. In vivo visualization of locally transplanted mesenchymal stem cells in the severely injured muscle in rats. Tissue Eng. Part A 2008, 14, 1149–1160. [Google Scholar] [CrossRef] [PubMed]
- Winkler, T.; von Roth, P.; Matziolis, G.; Mehta, M.; Perka, C.; Duda, G.N. Dose-response relationship of mesenchymal stem cell transplantation and functional regeneration after severe skeletal muscle injury in rats. Tissue Eng. Part A 2009, 15, 487–492. [Google Scholar] [CrossRef] [PubMed]
- Winkler, T.; von Roth, P.; Radojewski, P.; Urbanski, A.; Hahn, S.; Preininger, B.; Duda, G.N.; Perka, C. Immediate and delayed transplantation of mesenchymal stem cells improve muscle force after skeletal muscle injury in rats. J. Tissue Eng. Regen. Med. 2012, 6 (Suppl. S3), S60–S67. [Google Scholar] [CrossRef] [PubMed]
- Reuters. Pluristem Stem Cell Trial to Treat Muscle Injury Meets Main Goal. 2014. Available online: http://www.reuters.com/article/us-pluristem-cells-trial-idUSBREA0K09S20140121 (accessed on 12 February 2018).
- Pumberger, M.; Qazi, T.H.; Ehrentraut, M.C.; Textor, M.; Kueper, J.; Stoltenburg-Didinger, G.; Winkler, T.; von Roth, P.; Reinke, S.; Borselli, C.; et al. Synthetic niche to modulate regenerative potential of MSCs and enhance skeletal muscle regeneration. Biomaterials 2016, 99, 95–108. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sass, F.A.; Fuchs, M.; Pumberger, M.; Geissler, S.; Duda, G.N.; Perka, C.; Schmidt-Bleek, K. Immunology Guides Skeletal Muscle Regeneration. Int. J. Mol. Sci. 2018, 19, 835. https://doi.org/10.3390/ijms19030835
Sass FA, Fuchs M, Pumberger M, Geissler S, Duda GN, Perka C, Schmidt-Bleek K. Immunology Guides Skeletal Muscle Regeneration. International Journal of Molecular Sciences. 2018; 19(3):835. https://doi.org/10.3390/ijms19030835
Chicago/Turabian StyleSass, F. Andrea, Michael Fuchs, Matthias Pumberger, Sven Geissler, Georg N. Duda, Carsten Perka, and Katharina Schmidt-Bleek. 2018. "Immunology Guides Skeletal Muscle Regeneration" International Journal of Molecular Sciences 19, no. 3: 835. https://doi.org/10.3390/ijms19030835
APA StyleSass, F. A., Fuchs, M., Pumberger, M., Geissler, S., Duda, G. N., Perka, C., & Schmidt-Bleek, K. (2018). Immunology Guides Skeletal Muscle Regeneration. International Journal of Molecular Sciences, 19(3), 835. https://doi.org/10.3390/ijms19030835





