Phytochemical Analysis by HPLC–HRESI-MS and Anti-Inflammatory Activity of Tabernaemontana catharinensis
Abstract
:1. Introduction
2. Results
2.1. Thin-Layer Chromatography (TLC) Profile of the Hydroethanolic Extract and Organic Fractions of T. catharinensis Leaves
2.2. High-Performance Liquid Chromatography–High-Resolution Electrospray Ionization-Mass Spectrometry (HPLC–HRESI-MS) Analysis of the Hydroethanolic Extract of T. catharinensis Leaves
2.3. Cytotoxicity Assay
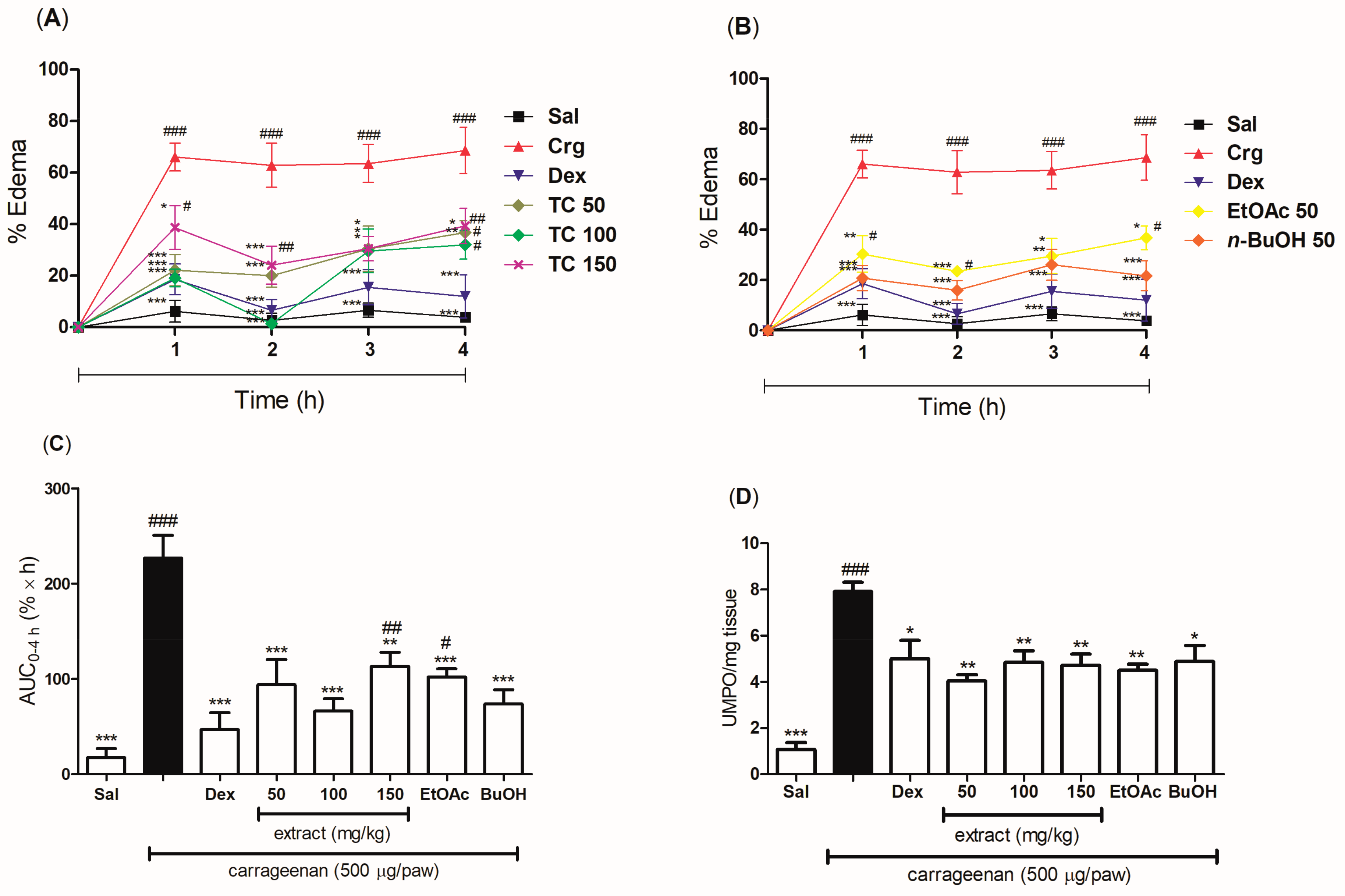
2.4. Anti-Inflammatory Effect of the Pretreatment with the Hydroethanolic Extract and the Fractions of T. catharinensis Leaves in the Carrageenan-Induced Paw Edema Model
2.5. Anti-Inflammatory Effect of the Post-Treatment with the Hydroethanolic Extract and the Fractions of T. catharinensis Leaves in the Carrageenan-Induced Paw Edema Model
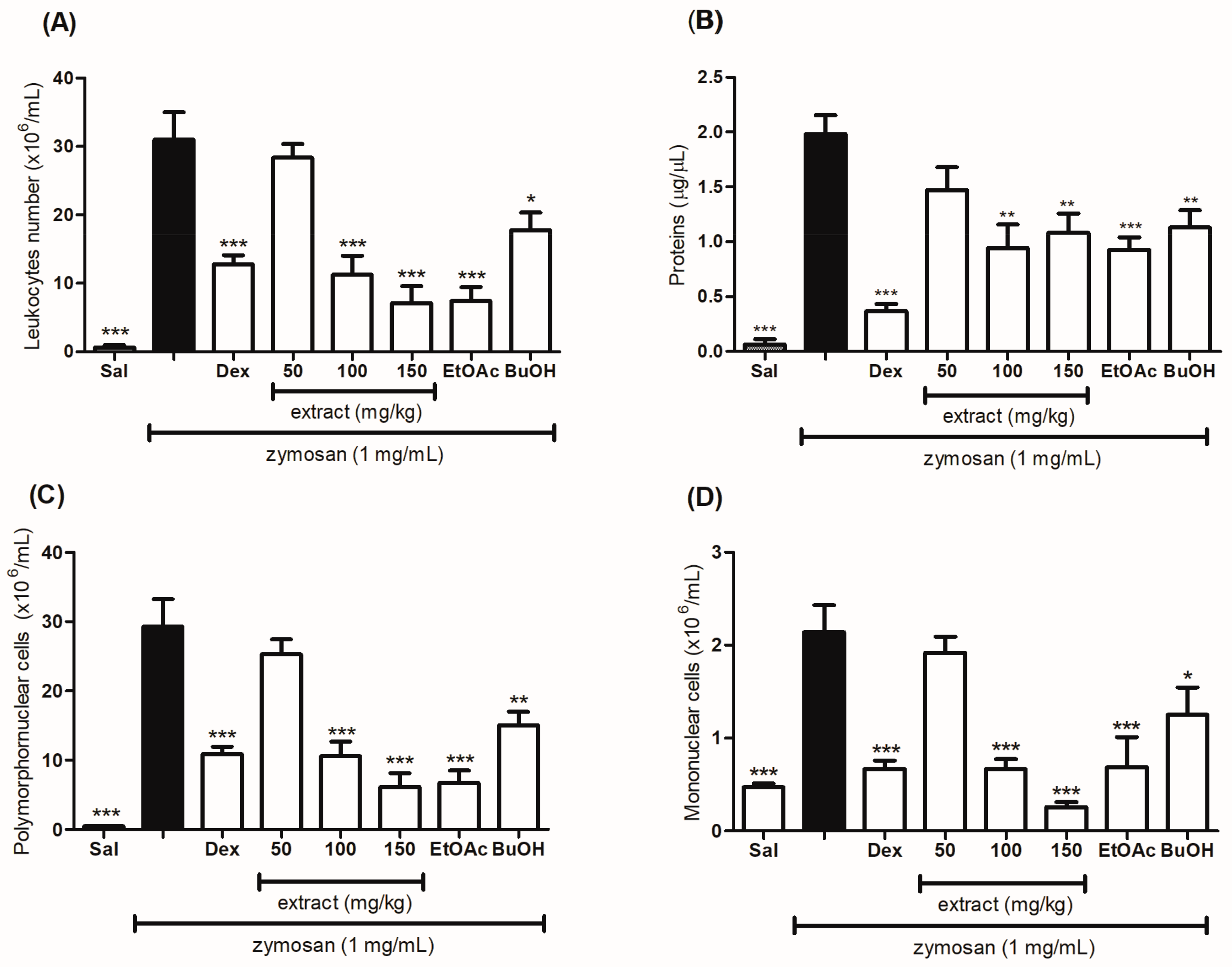
2.6. Evaluation of the Anti-Inflammatory Effect of the Hydroethanolic Extract and the Fractions of T. catharinensis Leaves in the Zymosan-Induced Air-Pouch Model
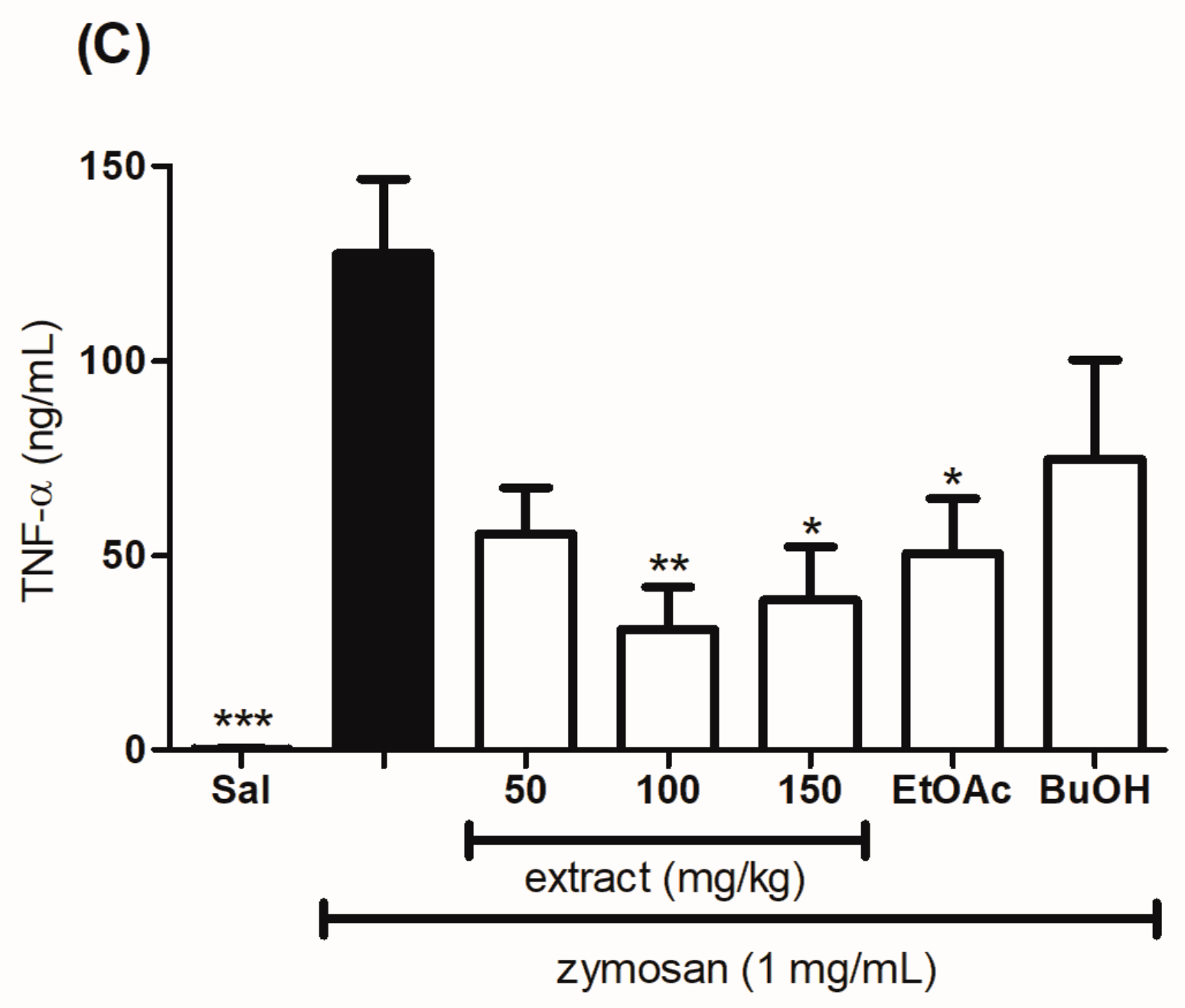
2.7. Evaluation of the Effect of the Hydroethanolic Extract and the Fractions of T. catharinensis Leaves on the Levels of Myeloperoxidase and Cytokines
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Extract Preparation
4.3. Thin-Layer Chromatography (TLC) Profile of the Leaves Extract
4.4. High-Performance Liquid Chromatography–High-Resolution Electrospray Ionization-Mass Spectrometry (HPLC–HRESI-MS) Profile of the Leaves Extract
4.5. MTT Assay
4.6. Animals
4.7. Carrageenan-Induced Paw Edema Model
4.8. Zymosan-Induced Air-Pouch Model
4.9. Differential Cell Count in the Exudate
4.10. Determination of Total Proteins
4.11. Quantitative Determination of Myeloperoxidase Levels
4.12. Determination of Cytokines Concentration
4.13. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflict of Interest
References
- Medzhitov, R. Inflammation 2010: New Adventures of an Old Flame. Cell 2010, 140, 771–776. [Google Scholar] [CrossRef] [PubMed]
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Gilligan, J.P.; Lovato, S.J.; Erion, M.D.; Jeng, A.Y. Modulation of carrageenan-induced hind paw edema by substance P. Inflammation 1994, 18, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Sun, J.; Jiang, L.; Duan, L.; Huo, M.; Chen, N.; Zhong, W.; Wassy, L.; Yang, Z.; Feng, H. Imperatorin Attenuates LPS-Induced Inflammation by Suppressing NF-κB and MAPKs Activation in RAW 264.7 Macrophages. Inflammation 2012, 35, 1764–1772. [Google Scholar] [CrossRef] [PubMed]
- Naik, A.D.; Woofter, A.L.; Skinner, J.M.; Abraham, N.S. Pharmaceutical company influence on nonsteroidal anti-inflammatory drug prescribing behaviors. Am. J. Manag. Care 2009, 15, e9–15. [Google Scholar] [CrossRef] [PubMed]
- Ong, C.K.S.; Lirk, P.; Tan, C.H.; Seymour, R.A. An evidence-based update on nonsteroidal anti-inflammatory drugs. Clin. Med. Res. 2007, 5, 19–34. [Google Scholar] [CrossRef] [PubMed]
- Horl, W.H. Nonsteroidal Anti-Inflammatory Drugs and the Kidney. Pharmaceuticals 2010, 3, 2291–2321. [Google Scholar] [CrossRef] [PubMed]
- Atanasov, A.G.; Waltenberger, B.; Pferschy-Wenzig, E.M.; Linder, T.; Wawrosch, C.; Uhrin, P.; Temml, V.; Wang, L.; Schwaiger, S.; Heiss, E.H.; et al. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol. Adv. 2015, 33, 1582–1614. [Google Scholar] [CrossRef] [PubMed]
- Rathee, P.; Chaudhary, H.; Rathee, S.; Rathee, D.; Kumar, V.; Kohli, K. Mechanism of Action of Flavonoids as Anti-inflammatory Agents: A Review. Inflamm. Allergy Drug Targets 2009, 8, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Bellik, Y.; Boukraâ, L.; Alzahrani, H.A.; Bakhotmah, B.A.; Abdellah, F.; Hammoudi, S.M.; Iguer-Ouada, M. Molecular mechanism underlying anti-inflammatory and anti-allergic activities of phytochemicals: An update. Molecules 2012, 18, 322–353. [Google Scholar] [CrossRef] [PubMed]
- Koch, I.; Rapini, A.; Simões, A.O.; Kinoshita, L.S.; Spina, A.P.; Castello, A.C. Apocynaceae in Lista de Espécies da Flora do Brasil. Jadim Botânico do Rio de Janeiro. Available online: http://floradobrasil.jbrj.gov.br/jabot/floradobrasil/FB4861 (accessed on 5 February 2016).
- Rates, S.M.K.; Schopoval, E.E.S.; Sousa, I.A.; Henrigue, A.T. Chemical constituentis and phartmacologial activities of Peschiera australis. Int. J. Pharmacogn. 1993, 31, 284–288. [Google Scholar] [CrossRef]
- Gomes, R.C.; Neto, A.C.; Melo, V.L.; Fernandes, V.C.; Dagrava, G.; Santos, W.S.; Pereira, P.S.; Couto, L.B.; Beleboni, R.O. Antinociceptive and anti-inflammatory activities of Tabernaemontana catharinensis. Pharm. Biol. 2009, 47, 372–376. [Google Scholar] [CrossRef]
- Pereira, C.G.; Marques, M.O.M.; Barreto, A.S.; Siani, A.C.; Fernandes, E.C.; Meireles, M.A.A. Extraction of indole alkaloids from Tabernaemontana catharinensis using supercritical CO2+ ethanol: An evaluation of the process variables and the raw material origin. J. Supercrit. Fluids 2004, 30, 51–61. [Google Scholar] [CrossRef]
- Gonçalves, M.S.; Vieira, I.J.C.; Oliveira, R.R.; Braz-Filho, R. Application of preparative High-Speed Counter-Current Chromatography for the separation of two alkaloids from the roots of Tabernaemontana catharinensis (Apocynaceae). Molecules 2011, 16, 7480–7487. [Google Scholar] [CrossRef] [PubMed]
- Delorenzi, J.C.; Attias, M.; Gattass, C.R.; Andrade, M.; Rezende, C.; Da Cunha Pinto, A.; Henriques, A.T.; Bou-Habib, D.C.; Saraiva, E.M.B. Antileishmanial activity of an indole alkaloid from Peschiera australis. Antimicrob. Agents Chemother. 2001, 45, 1349–1354. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.G.; Rosa, P.T.V.; Meireles, M.A.A. Extraction and isolation of indole alkaloids from Tabernaemontana catharinensis A.DC: Technical and economical analysis. J. Supercrit. Fluids 2007, 40, 232–238. [Google Scholar] [CrossRef]
- Singh, B.; Sharma, R.A.; Vyas, K.G. Antimicrobial, Antineoplastic and Cytotoxic Activities of Indole Alkaloids from Tabernaemontana divaricata (L.) R.Br. Curr. Pharm. Anal. 2011, 7, 125–132. [Google Scholar] [CrossRef]
- Pereira, P.S.; França, S.C.; Oliveira, P.V.A.; Brves, C.M.S.; Pereira, S.I.V. Chemical constituents from Tabernaemontana catharinensis root bark: A brief NMR review of indole alkaloids and in vitro cytotoxicity. Quim. Nova 2008, 31, 20–24. [Google Scholar] [CrossRef]
- Mabry, T.J.; Markham, K.R.; Thomas, M.B. The Systematic Identification of Flavonoids; Springer: New York, NY, USA, 1970; p. 354. [Google Scholar]
- Wagner, H.; Bladt, S. Plant Drug Analysis: A Thin Layer Chromatography Atlas, 2nd ed.; Springer: Berlin, Germany, 2001; p. 384. [Google Scholar]
- Serafini, M.; Peluso, I.; Raguzzini, A. Flavonoids as anti-inflammatory agents. Proc. Nutr. Soc. 2010, 69, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Suleyman, H.; Demircan, B.; Karagöz, Y. Anti-inflammatory and side effects of cyclooxygenase inhibitors. Pharmacol. Rep. 2007, 59, 247–258. [Google Scholar] [PubMed]
- Zarghi, A.; Arfaei, S. Selective COX-2 inhibitors: A review of their structure-activity relationships. Iran. J. Pharm. Res. 2011, 10, 655–683. [Google Scholar] [PubMed]
- Hawkey, C.J. COX-1 and COX-2 inhibitors. Best Pract. Res. Clin. Gastroenterol. 2001, 15, 801–820. [Google Scholar] [CrossRef] [PubMed]
- Schlansky, B.; Hwang, J.H. Prevention of nonsteroidal anti-inflammatory drug-induced gastropathy. J. Gastroenterol. 2009, 44, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.; Duggan, A. Gastrointestinal adverse effects of non-steroidal anti-inflammatory drugs. Expert Opin. Drug Saf. 2005, 4, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef] [PubMed]
- Boligon, A.A.; Schwanz, T.G.; Piana, M.; Bandeira, R.V.; Frohlich, J.K.; Brum, T.F.D.; Zadra, M.; Athayde, M.L. Chemical composition and antioxidant activity of the essential oil of Tabernaemontana catharinensis A. DC. leaves. Nat. Prod. Res. 2013, 27, 68–71. [Google Scholar] [CrossRef] [PubMed]
- Boligon, A.A.; Athayde, M.L. Phytochemical Invertigation and Cytotoxic Properties of Tabernemontana catharinensis A. DC. Cultivated in Brasil. Res. J. Phytochem. 2012, 6, 127–131. [Google Scholar] [CrossRef]
- Belke, B.V.; Piana, M.; Boligon, A.A.; Zadra, M.; Fröhlich, J.K.; de Brum, T.F.; Athayde, M.L. Doseamento de flavonóides nos ramos de Tabernaemontana catharinensis A. DC. Rev. Context. Saúde 2011, 11, 911–914. [Google Scholar]
- Morris, C.J. Carrageenan-induced paw edema in the rat and mouse. Methods Mol. Biol. 2003, 225, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Posadas, I.; Bucci, M.; Roviezzo, F.; Rossi, A.; Parente, L.; Sautebin, L.; Cirino, G. Carrageenan-induced mouse paw oedema is biphasic, age-weight dependent and displays differential nitric oxide cyclooxygenase-2 expression. Br. J. Pharmacol. 2004, 142, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Pereira, L. Population studies and carrageenan properties in eight gigartinales (Rhodophyta) from western coast of Portugal. Sci. World J. 2013, 2013, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Konno, S.; Tsurufuji, S. Induction of zymosan-air-pouch inflammation in rats and its characterization with reference to the effects of anticomplementary and anti-inflammatory agents. Br. J. Pharmacol. 1983, 80, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Pulli, B.; Ali, M.; Forghani, R.; Schob, S.; Hsieh, K.L.C.; Wojtkiewicz, G.; Linnoila, J.J.; Chen, J.W. Measuring Myeloperoxidase Activity in Biological Samples. PLoS ONE 2013, 8, e67976. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Francischi, J.N.; Queiroz-Junior, C.M.; Pacheco, C.M.D.F.; Fonseca, A.H.; Klein, A.; Caliari, M.V. Myeloperoxidase content is a marker of systemic inflammation in a chronic condition: The example given by the periodontal disease in rats. Mediators Inflamm. 2009, 2009, 1–7. [Google Scholar] [CrossRef]
- Stendahl, O.; Coble, B.I.; Dahlgren, C.; Hed, J.; Molin, L. Myeloperoxidase modulates the phagocytic activity of polymorphonuclear neutrophil leukocytes. Studies with cells from a myeloperoxidase-deficient patient. J. Clin. Investig. 1984, 73, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Hamalainen, M.; Nieminen, R.; Asmawi, M.Z.; Vuorela, P.; Vapaatalo, H.; Moilanen, E. Effects of flavonoids on prostaglandin E2 production and on COX-2 and mPGES-1 expressions in activated macrophages. Planta Med. 2011, 77, 1504–1511. [Google Scholar] [CrossRef] [PubMed]
- Kong, L.; Luo, C.; Li, X.; Zhou, Y.; He, H. The anti-inflammatory effect of kaempferol on early atherosclerosis in high cholesterol fed rabbits. Lipids Health Dis. 2013, 12, 115. [Google Scholar] [CrossRef] [PubMed]
- Leyva-López, N.; Gutierrez-Grijalva, E.P.; Ambriz-Perez, D.L.; Basilio Heredia, J. Flavonoids as cytokine modulators: A possible therapy for inflammation-related diseases. Int. J. Mol. Sci. 2016, 17, 921. [Google Scholar] [CrossRef] [PubMed]
- Borthwick, L.A. The IL-1 cytokine family and its role in inflammation and fibrosis in the lung. Semin. Immunopathol. 2016, 38, 517–534. [Google Scholar] [CrossRef] [PubMed]
- Antunes-Ricardo, M.; Moreno-García, B.E.; Gutiérrez-Uribe, J.A.; Aráiz-Hernández, D.; Alvarez, M.M.; Serna-Saldivar, S.O. Induction of Apoptosis in Colon Cancer Cells Treated with Isorhamnetin Glycosides from Opuntia Ficus-indica Pads. Plant Foods Hum. Nutr. 2014, 69, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.L.; Zhu, G.L.; Wang, J.A.; Zhang, G.D.; Liu, H.F.; Chen, J.R.; Wang, Y.; He, X.L. Protective effects of isorhamnetin on apoptosis and inflammation in TNF-α-induced HUVECs injury. Int. J. Clin. Exp. Pathol. 2015, 8, 2311–2320. [Google Scholar] [PubMed]
- Devi, K.P.; Malar, D.S.; Nabavi, S.F.; Sureda, A.; Xiao, J.; Nabavi, S.M.; Daglia, M. Kaempferol and inflammation: From chemistry to medicine. Pharmacol. Res. 2015, 99, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Boesch-Saadatmandi, C.; Loboda, A.; Wagner, A.E.; Stachurska, A.; Jozkowicz, A.; Dulak, J.; Döring, F.; Wolffram, S.; Rimbach, G. Effect of quercetin and its metabolites isorhamnetin and quercetin-3-glucuronide on inflammatory gene expression: Role of miR-155. J. Nutr. Biochem. 2011, 22, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Poetker, D.M.; Reh, D.D. A comprehensive review of the adverse effects of systemic corticosteroids. Otolaryngol. Clin. N. Am. 2010, 43, 753–768. [Google Scholar] [CrossRef] [PubMed]
- Sarnes, E.; Crofford, L.; Watson, M.; Dennis, G.; Kan, H.; Bass, D. Incidence and US Costs of Corticosteroid-Associated Adverse Events: A Systematic Literature Review. Clin. Ther. 2011, 33, 1413–1432. [Google Scholar] [CrossRef] [PubMed]
- Winter, C.A.; Risley, E.A.; Nuss, G.W. Carrageenin-induced edema in hind paw of the rat as an assay for anti-inflammatory drugs. Exp. Biol. Med. 1962, 111, 544–547. [Google Scholar] [CrossRef]
- Torres-Rêgo, M.; Furtado, A.A.; Bitencourt, M.A.; Lima, M.C.; Andrade, R.C.; Azevedo, E.P.; Soares Tda, C.; Tomaz, J.C.; Lopes, N.P.; da Silva-Júnior, A.A.; et al. Anti-inflammatory activity of aqueous extract and bioactive compounds identified from the fruits of Hancornia speciosa Gomes (Apocynaceae). BMC Complement. Altern. Med. 2016, 16, 275. [Google Scholar] [CrossRef] [PubMed]
- Fialho, E.M.S.; Maciel, M.C.G.; Silva, A.C.B.; Reis, A.S.; Assunção, A.K.M.; Fortes, T.S.; Silva, L.A.; Guerra, R.N.M.; Kwasniewski, F.H.; Nascimento, F.R.F. Immune cells recruitment and activation by Tityus serrulatus scorpion venom. Toxicon 2011, 58, 480–485. [Google Scholar] [CrossRef] [PubMed]
- Furtado, A.A.; Torres-Rêgo, M.; Lima, M.C.J.S.; Bitencourt, M.A.O.; Estrela, A.B.; Souza da Silva, N.; da Siqueira, E.M.S.; Tomaz, J.C.; Lopes, N.P.; Silva-Júnior, A.A.; et al. Aqueous extract from Ipomoea asarifolia (Convolvulaceae) leaves and its phenolic compounds have anti-inflammatory activity in murine models of edema, peritonitis and air-pouch inflammation. J. Ethnopharmacol. 2016, 192, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein using the principle of protein dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Bradley, P.P.; Priebat, D.A.; Christensen, R.D.; Rothstein, G. Measurement of cutaneous inflammation: Estimation of neutrophil content with an enzyme marker. J. Investig. Dermatol. 1982, 78, 206–209. [Google Scholar] [CrossRef] [PubMed]
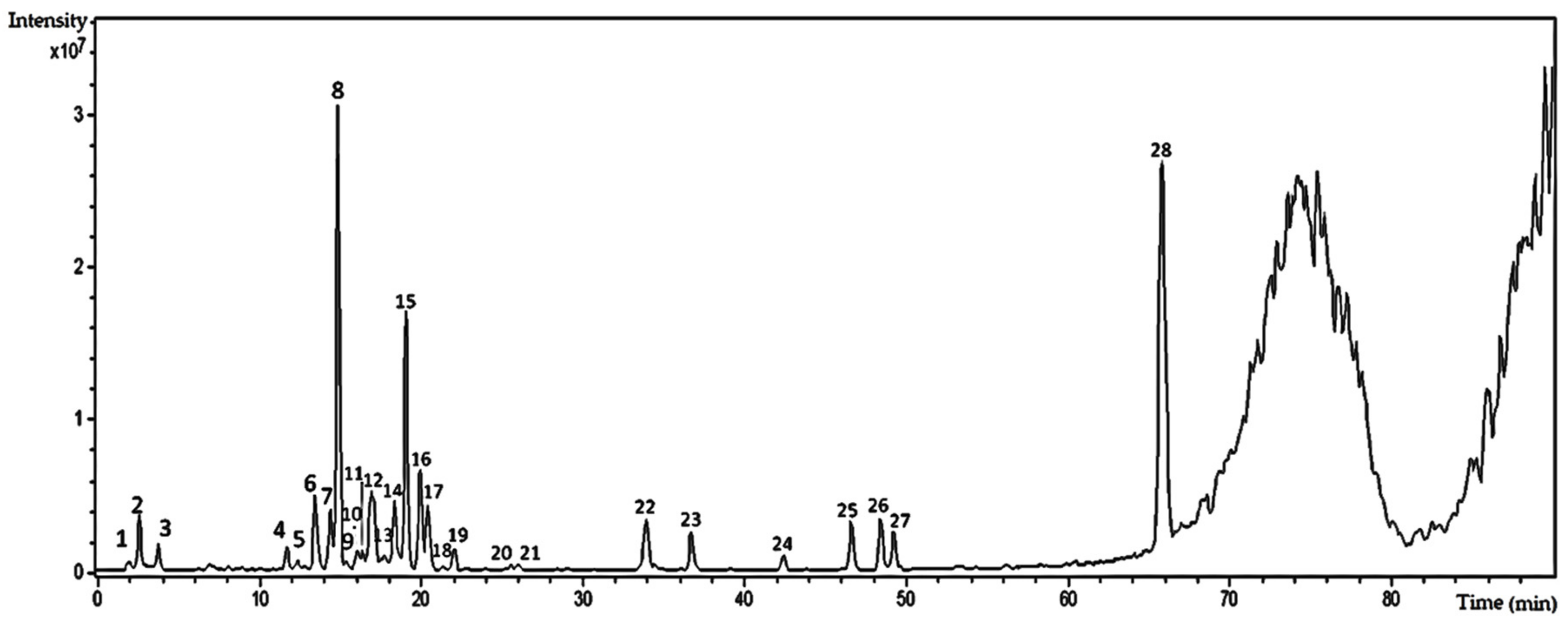



| Peak | (TR) Retention Time (min) | (MS) Mass Spectrometry (M/Z) | MS2 | UV Max (nm) | Compounds |
|---|---|---|---|---|---|
| 5 | 13.5 | [M − H]—739.17 | 739.17–575.09; 500.15; 473.04; 393.02; 338.92; 284.92; 256.94 | kaempferol-3-robinobioside-7-rhamnoside | |
| 6 | 13.8 | [M − H]—609.07 | 609.07–564.41; 488.97; 462.96; 408.87; 344.06; 300.95; 270.97; 216.99 | 211, 264 | quercetin-O-rutinoside |
| 7 | 14.5 | [M – H]—769.12 | 769.12–623.13; 477.09; 357.00; 314.98 | 211, 351 | 7-methylquercetin-3-galactoside-6″-rhamnoside-3′′′-rhamnoside |
| 8 | 15.0 | [M – H]—769.12 | 769.12–754.11; 701.12; 638.06; 623.12; 605.10; 579.02; 503.00; 400.05; 356.95; 314.94 | 211, 351 | isorhamnetin-3-galactoside-6″-rhamnoside-3′′′-rhamnoside |
| 12 | 16.8 | [M – H]—593.06 | 593.06–326.98; 298.96; 284.91 | 211, 253 | kaempferol-O-rutinoside |
| 13 | 16.8 | [M – H]—623.05 | 623.05–595.09; 477.02; 411.93; 356.95; 327.96; 314.95; 299.89; 270.87 | isorhamnetin-3-hexoside-6″-rhamnoside | |
| 14 | 19.4 | [M – H]—477.00 | 477.00–357; 326.95; 284.95; 254.93 | 249 | isorhamnetin-3-O-glicoside |
| 15 | 20.1 | [M − H]—477.00 | 477.00–449.05; 356.95; 315.00 | 210, 253 | isorhamnetin-3-O-glicoside |
| 16 | 20.5 | [M − H]—477.00 | 477.00–356.96; 314.92 | 211, 251 | isorhamnetin-3-O-glicoside |
| 12 | 22.3 | [M − H]—563.18 | 563.18–360.71 | isoschaftoside |
| Groups | Dose (mg/kg) | Cell Migration (×106/mL) | Inhibition (%) |
|---|---|---|---|
| Saline | - | 31.00 ± 4.000 | - |
| Dexamethasone | 2 | 12.80 ± 1.300 *** | 59 |
| Hydroethanolic extract | 50 | 28.40 ± 1.965 | 9 |
| Hydroethanolic extract | 100 | 11.25 ± 2.780 *** | 64 |
| Hydroethanolic extract | 150 | 7.125 ± 2.461 *** | 77 |
| EtOAc fraction | 50 | 7.400 ± 2.040 *** | 76 |
| BuOH fraction | 50 | 17.75 ± 2.602 ** | 43 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marques, J.I.; Alves, J.S.F.; Torres-Rêgo, M.; Furtado, A.A.; Siqueira, E.M.d.S.; Galinari, E.; Araújo, D.F.d.S.; Guerra, G.C.B.; Azevedo, E.P.d.; Fernandes-Pedrosa, M.D.F.; et al. Phytochemical Analysis by HPLC–HRESI-MS and Anti-Inflammatory Activity of Tabernaemontana catharinensis. Int. J. Mol. Sci. 2018, 19, 636. https://doi.org/10.3390/ijms19020636
Marques JI, Alves JSF, Torres-Rêgo M, Furtado AA, Siqueira EMdS, Galinari E, Araújo DFdS, Guerra GCB, Azevedo EPd, Fernandes-Pedrosa MDF, et al. Phytochemical Analysis by HPLC–HRESI-MS and Anti-Inflammatory Activity of Tabernaemontana catharinensis. International Journal of Molecular Sciences. 2018; 19(2):636. https://doi.org/10.3390/ijms19020636
Chicago/Turabian StyleMarques, José Ivan, Jovelina Samara Ferreira Alves, Manoela Torres-Rêgo, Allanny Alves Furtado, Emerson Michell da Silva Siqueira, Eder Galinari, Daline Fernandes de Souza Araújo, Gerlane Coelho Bernardo Guerra, Eduardo Pereira de Azevedo, Matheus De Freitas Fernandes-Pedrosa, and et al. 2018. "Phytochemical Analysis by HPLC–HRESI-MS and Anti-Inflammatory Activity of Tabernaemontana catharinensis" International Journal of Molecular Sciences 19, no. 2: 636. https://doi.org/10.3390/ijms19020636







