The Study of Anti-/Pro-Oxidant, Lipophilic, Microbial and Spectroscopic Properties of New Alkali Metal Salts of 5-O-Caffeoylquinic Acid
Abstract
:1. Introduction
2. Results
2.1. Spectroscopic Study
2.2. Antimicrobial Activity
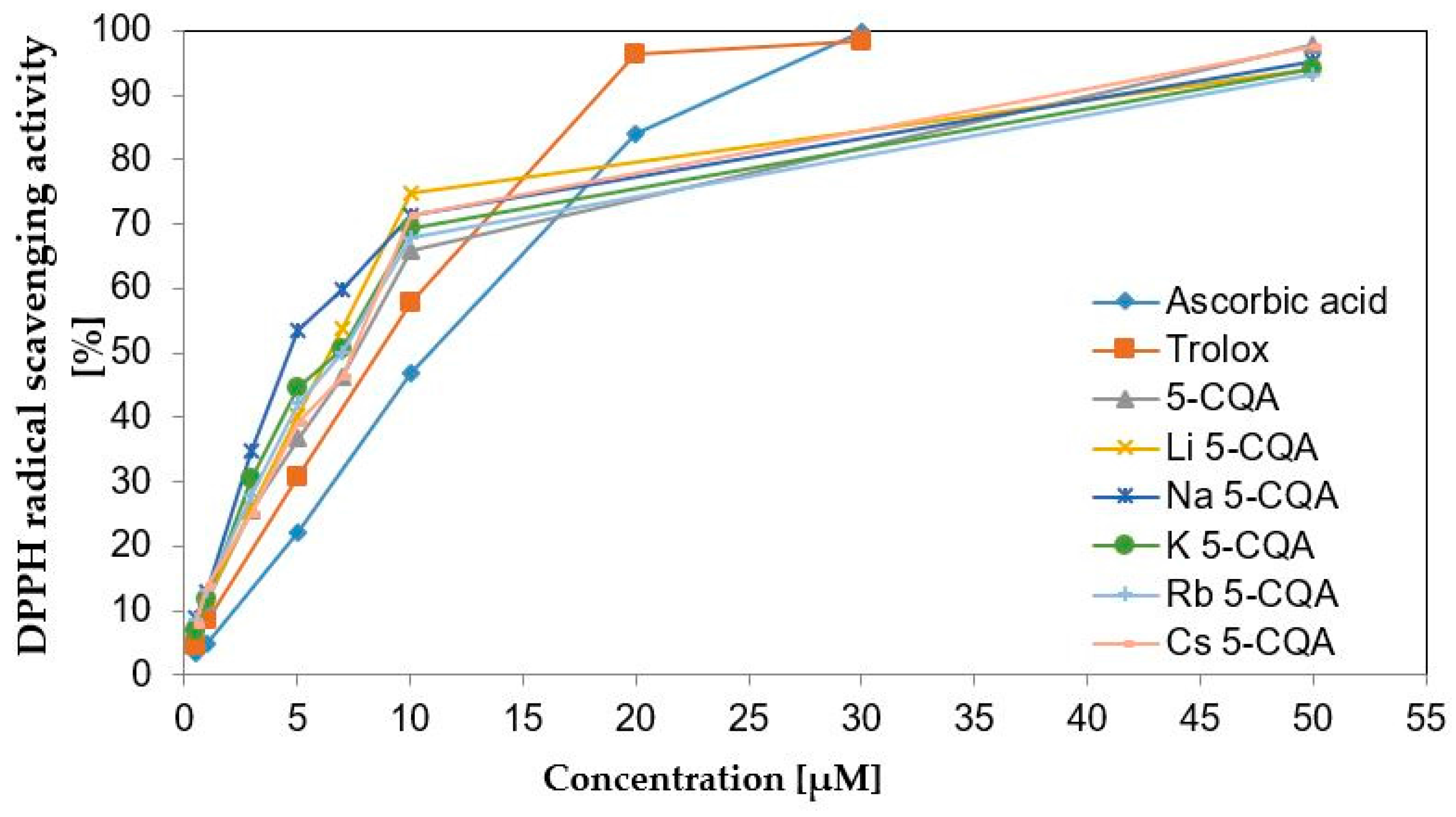
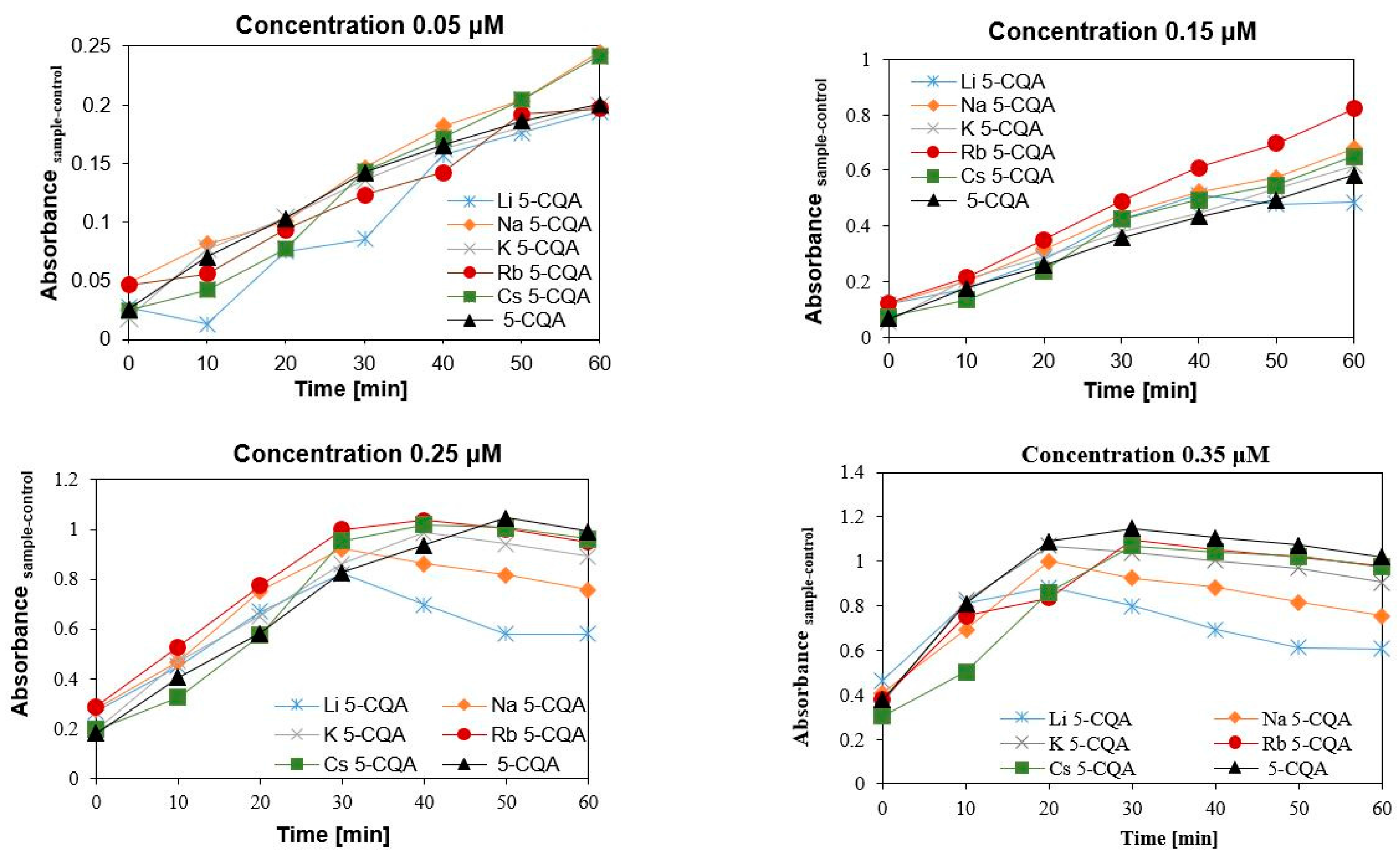
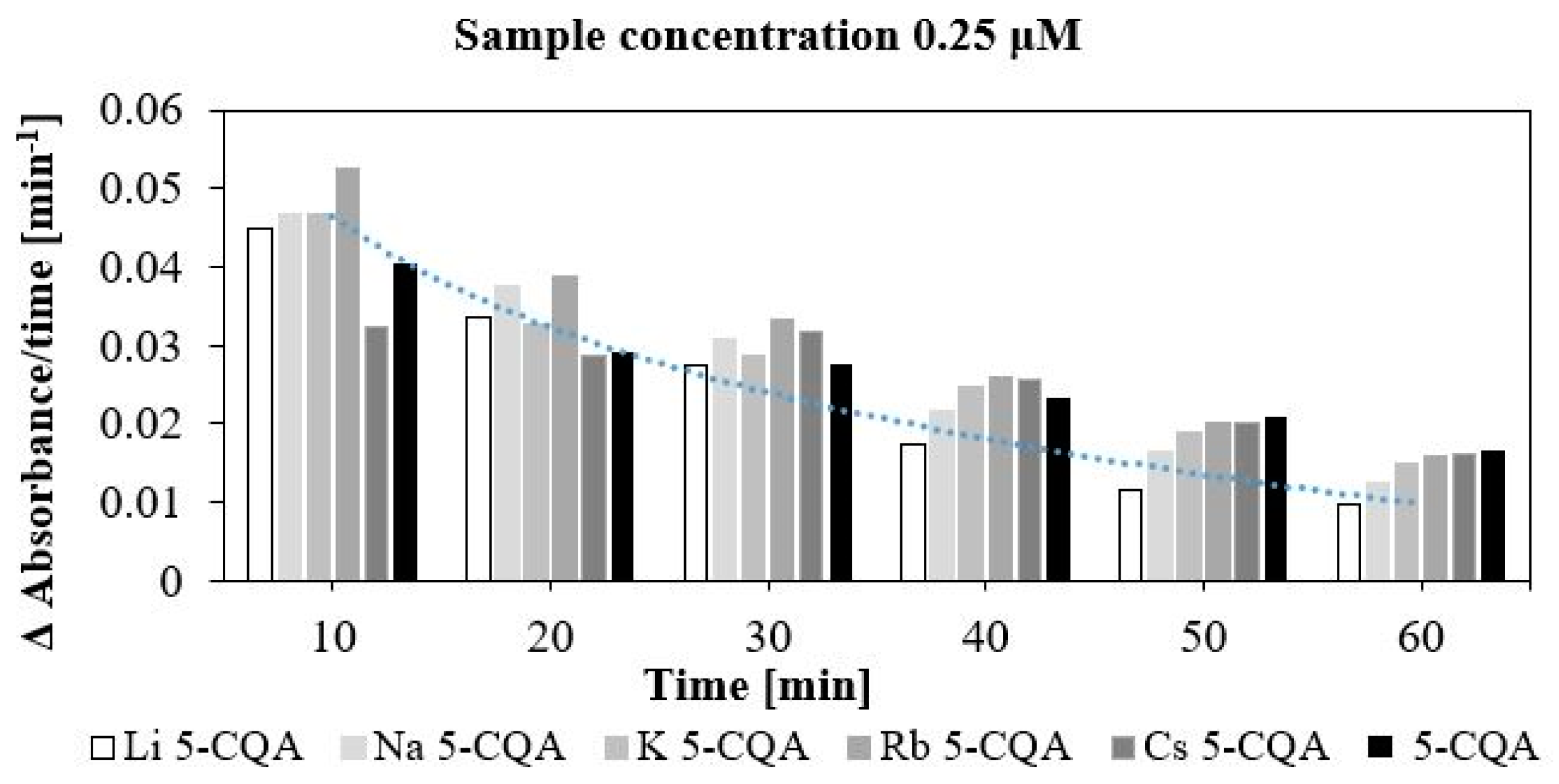
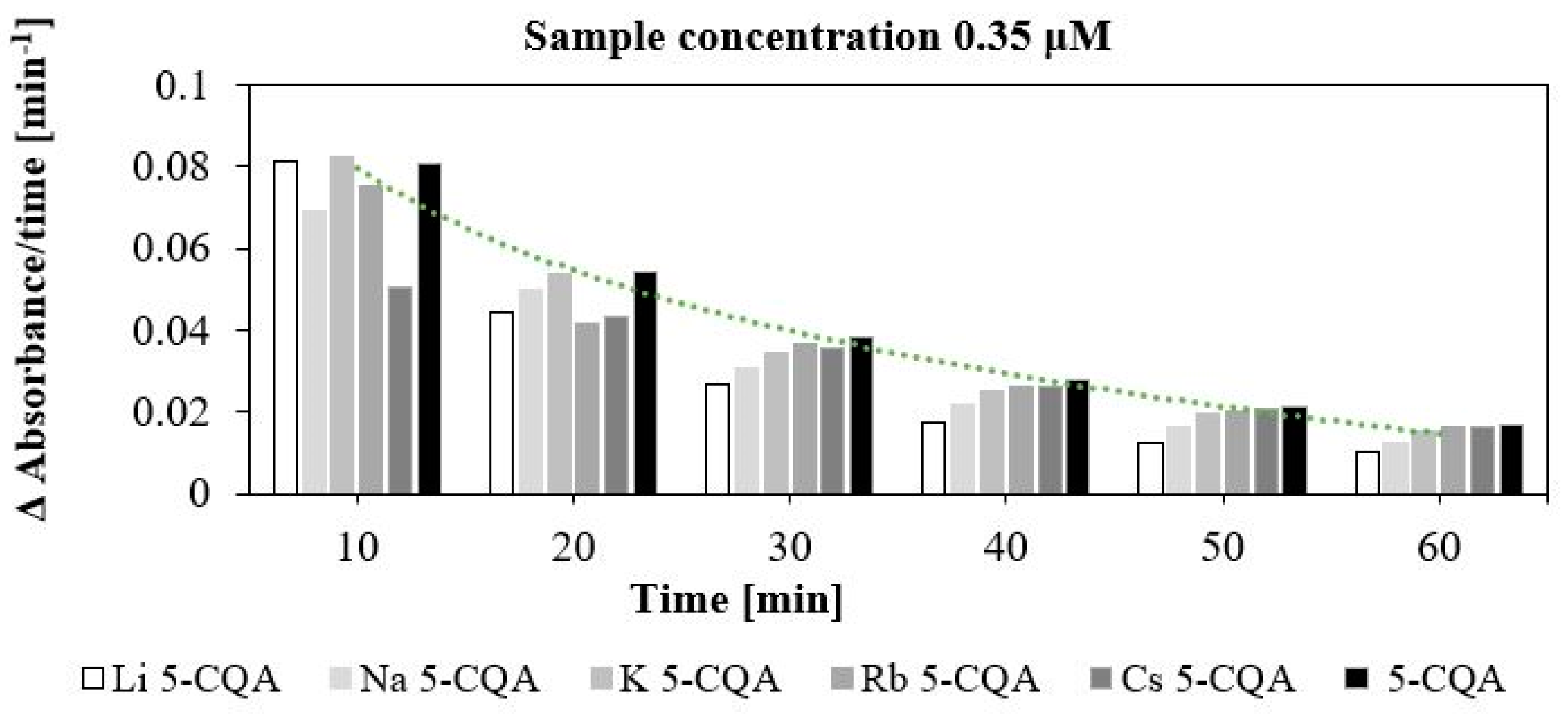
2.3. Antioxidant (DPPH and FRAP Assays) and Pro-Oxidant (Trolox Assay) Properties
2.4. Lipophilicity Studies
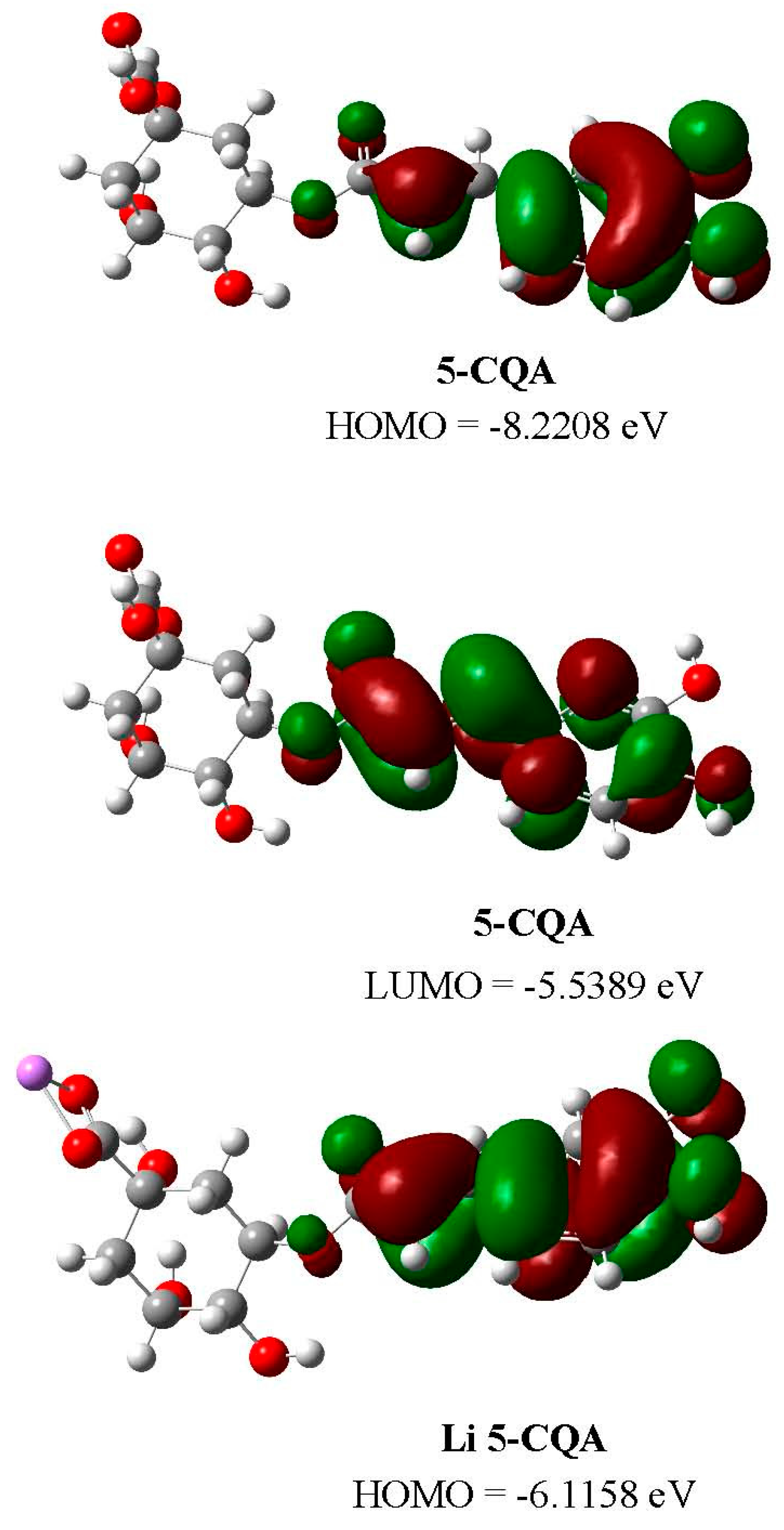
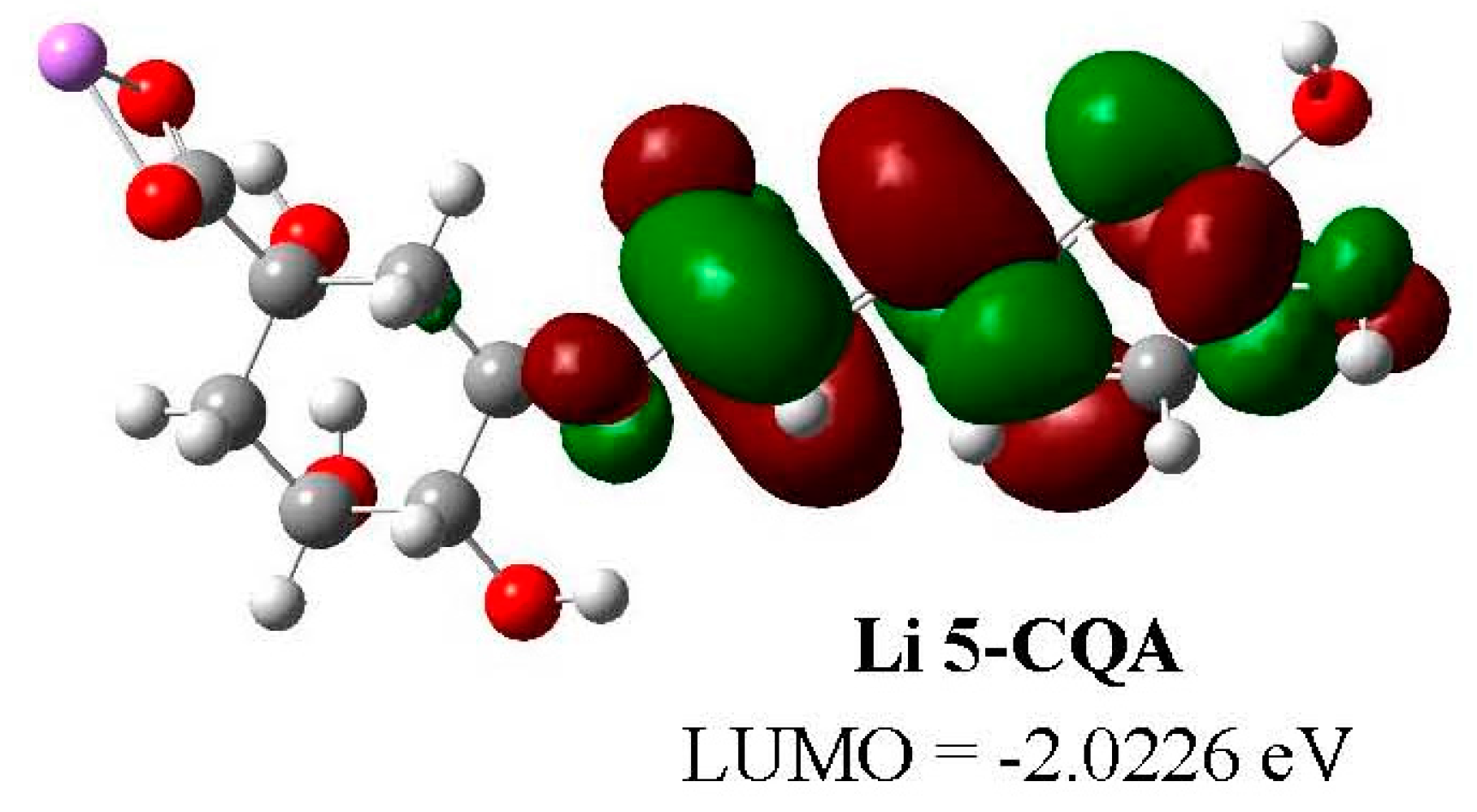
2.5. DFT Studies
3. Discussion
4. Materials and Methods
4.1. Materials Synthesis and Characterization
4.2. Evaluation of Biological Activity
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jeszka-Skowron, M.; Sentkowska, A.; Pyrzyńska, A.; Paz De Pena, M. Chlorogenic acid, caffeine content and antioxidant properties of green coffee extracts: Influence of green coffee bean preparation. Eur. Food Res. Technol. 2016, 242, 1403–1409. [Google Scholar] [CrossRef]
- Bajko, E.; Kalinowska, M.; Borowski, P.; Siergiejczyk, L.; Lewandowski, W. 5-O-Caffeoylquinic acid: A spectroscopic study and biological screening for antimicrobial activity. LWT Food Sci. Technol. 2016, 65, 471–479. [Google Scholar] [CrossRef]
- Erdem, S.A.; Senol, F.S.; Budakoglu, E.; Orhan, I.E.; Sener, B. Exploring in vitro neurobiological effects and high-presure liquid chromatography-assisted quantitation of chlorogenic acid in 18 Turkish coffee brands. J. Food Drug Anal. 2016, 24, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Wang, H.; Yang, B. Identification of cyclodextrin inclusion complex of chlorogenic acid and its antimicrobial activity. Food Chem. 2010, 120, 1138–1142. [Google Scholar] [CrossRef]
- Da Silva, F.M.; Iorio, N.L.P.; Lobo, L.A. Antibacterial effect of aqueous extracts and bioactive chemical compounds of coffea canephora against microorganisms involved in dental caries and periodontal disease. Adv. Microbiol. 2014, 4, 978–985. [Google Scholar] [CrossRef]
- Mikami, Y.; Yamazawa, T. Chlorogenic acid, a polyphenol in coffee, protects neuron against glutamate neurotoxicity. Life Sci. 2015, 139, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Kang, Q.; Ren, J.; Li, Z.; Xu, X. Antitumor molecular mechanism of chlorogenic acid on inducting genes gsk-3β and apc and inhibiting gene β-catenin. J. Anal. Methods Chem. 2013, 2013, 1–7. [Google Scholar]
- Hou, N.; Liu, N.; Han, J.; Yan, Y.; Li, J. Chlorogenic acid induces reactive oxygen species generation and inhibits the viability of human colon cancer cells. Anticancer Drugs 2017, 28, 59–65. [Google Scholar] [CrossRef] [PubMed]
- de Magalhaes, P.M.; Dupont, I.; Hendrickx, A.; Joly, A.; Dessy, S.; Sergent, T.; Schneider, Y.J. Anti-inflammatory effect and modulation of cytochrome P450 activties by Artemisia annua tea infusions in human intestinal Caco-2 cells. Food Chem. 2012, 134, 864–871. [Google Scholar] [CrossRef] [PubMed]
- Clarke, R.J.; Macrae, R. Coffee: Vol. 1: Chemistry; Elsevier: London, UK, 1985. [Google Scholar]
- Van Buren, J.; De Vos, L.; Pilnik, W. Chlorogenic acid and flavonol glycosides in juice from enzyme-treated golden delicious apple pulp. J. Food Sci. 1973, 38, 656–674. [Google Scholar]
- De Maria, C.A.B.; Trugo, L.C.; De Mariz e Miranda, L.S. The content of individual caffeoylquinic acids in edible vegetables. J. Food Comp. Anal. 1999, 12, 289–292. [Google Scholar] [CrossRef]
- Marques, V.; Farah, A. Chlorogenic acids and related compounds in medicinal plants and infusions. Food Chem. 2009, 113, 1370–1376. [Google Scholar] [CrossRef]
- Kalinowska, M.; Bielawska, A.; Lewandowska-Siwkiewicz, H.; Priebe, W.; Lewandowski, W. Apple: Phenolic compounds, extraction and health benefits—A review. Plant Phys. Biochem. 2014, 84, 169–188. [Google Scholar] [CrossRef] [PubMed]
- Liang, N.; Kitts, D.D. Antioxidant property of coffee components: assessment of methods that define mechanisms of action. Molecules 2014, 19, 19180–19208. [Google Scholar] [CrossRef] [PubMed]
- Kono, Y.; Kobayashi, K.; Tagawa, S.; Adachi, K.; Ueda, A. Antioxidant activity of polyphenolics in diets. Rate constants of reactions of chlorogenic acid and caffeic acid with reactive species of oxygen and nitrogen. Biochim. Biophys. Acta 1997, 1335, 335–342. [Google Scholar] [CrossRef]
- Bajko, E.; Kalinowska, M.; Świderski, G.; Lewandowski, W. Analytical Methods to Study Oxidative Damage, Antioxidants and Drugs; Medical University of Bialystok: Bialystok, Poland, 2015. [Google Scholar]
- Liang, N.; Kitts, D.D. Role of chlorogenic acids in controlling oxidative and inflammatory stress conditions. Nutrients 2016, 8, 16. [Google Scholar] [CrossRef] [PubMed]
- Jiang, R.; Hodgson, J.M.; Mas, E.; Croft, K.D.; Ward, N.C. Chlorogenic acid improves ex vivo vessel function and protects endothelial cells against HOCl-induced oxidative damage, via increased production of nitric oxide and induction of Hmox-1. J. Nutr. Biochem. 2016, 27, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Cha, J.W.; Piao, M.J.; Kim, K.C.; Yao, C.W.; Zheng, J.; Kim, S.M.; Hyun, C.L.; Ahn, Y.S.; Hyun, J.W. The polyphenol chlorogenic acid attenuates UVB-mediated oxidative stress in human HaCaT keratinocytes. Biomol. Ther. 2014, 22, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Pavlica, S.; Gebhardt, R. Protective effects of ellagic and chlorogenic acids against oxidative stress in PC12 cells. Free Radic. Res. 2005, 39, 1377–1390. [Google Scholar] [CrossRef] [PubMed]
- Sakihama, Y.; Cohen, M.F.; Grace, S.C.; Yamasaki, H. Plant phenolic antioxidant and prooxidant activities: phenolics-induced oxidative damage mediated by metals in plants. Toxicology 2002, 177, 67–80. [Google Scholar] [CrossRef]
- Li, X.; Hu, Q.; Jiang, S.; Li, F.; Lin, J.; Han, J.; Hong, Y.; Lu, W.; Gao, Y.; Chen, D. Flos Chrysanthemi indiciprotects against hydroxyl-induced damages to DNA and MSCs via antioxidant mechanism: A chemistry study. J. Saudi Chem. Soc. 2015, 19, 454–460. [Google Scholar] [CrossRef]
- Pirker, K.F.; Goodman, B.A. Caffeoylquinic acid derived free radicals identified during antioxidant reactions of bitter tea (Ilex latifolia and Ilex kudincha). Food Funct. 2010, 1, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Hockertz, J.; Steenken, S.; Wieghardt, K.; Hildebrandt, P. (Photo)ionization of tris(phenolata)iron(III) complexes: Generation of phenoxyl radical as ligand. J. Am. Chem. Soc. 1993, 115, 11222–11230. [Google Scholar] [CrossRef]
- Shimazaki, Y.; Yamauchi, O. Recent advances in metal-phenoxyl radical chemistry. Indian J. Chem. A 2011, 50, 383–394. [Google Scholar]
- Watson, J. Oxidants, antioxidants and the current incurability of metastatic cancers. Open Biol. 2013, 3, 120144. [Google Scholar] [CrossRef] [PubMed]
- Anantharaju, P.G.; Prathima, C.G.; Vimalambike, M.G.; Madhunapantula, S.V. An overview on the role of dietary phenolics for the treatment of cancers. Nutr. J. 2016, 15, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Kohanski, M.A.; Dwyer, D.J.; Hayeta, B.; Lawerence, C.A.; Collins, J.J. A common mechanism of cellular death induced by bactericidal antibiotics. Cell 2007, 130, 797–810. [Google Scholar] [CrossRef] [PubMed]
- Foti, J.; Devadoss, B.; Winkler, J.A.; Collins, J.J.; Walker, G.C. Oxidation of the guanine nucleotide pool underlies cell death by bactericidal antibiotics. Science 2012, 336, 315–319. [Google Scholar] [CrossRef] [PubMed]
- Vatansever, F.; de Mel, W.C.M.A.; Avci, P.; Vecchio, D.; Sadasivam, M.; Gupta, A.; Chandran, R.; Karimi, M.; Parizotto, N.A.; Yin, R.; et al. Antimicrobial strategies centered around reactive oxygen species—Bactericidal antibiotics, photodynamic therapy, and beyond. FEMS Microbiol. Rev. 2013, 37, 955–989. [Google Scholar] [CrossRef] [PubMed]
- Santana-Gálvez, J.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. Chlorogenic Acid: Recent Advances on Its Dual Role as a Food Additive and a Nutraceutical against Metabolic Syndrome. Molecules 2017, 22, 358. [Google Scholar] [CrossRef] [PubMed]
- Muthuswamy, S.; Vasantha-Rupasinghe, H.P. Fruit phenolics as natural antimicrobial agents: Selective antimicrobial activity of catechin, chlorogenic acid and phloridzin. J. Food Agric. Environ. 2007, 5, 81–85. [Google Scholar]
- Karunanidhi, A.; Thomas, R.; van Belkum, A.; Neela, V. In vitro antibacterial and antibiofilm activities of chlorogenic acid against clinical isolates of Stenotrophomonas maltophilia including the trimethoprim/sulfamethoxazole resistant strain. Biomed. Res. Int. 2013, 2013, 392058. [Google Scholar] [CrossRef] [PubMed]
- Fiamegos, Y.C.; Kastritis, P.L.; Exarchou, V.; Han, H.; Bonvin, A.M.J.J.; Vervoort, J.; Lewis, K.; Hamblin, M.R.; Tegos, G.P. Antimicrobial and efflux pump inhibitory activity of caffeoylquinic acids from Artemisia absinthium against gram-positive pathogenic bacteria. PLoS ONE 2011, 6, e18127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lou, Z.; Wang, H.; Zhu, S.; Ma, C.; Wang, Z. Antibacterial activity and mechanism of action of chlorogenic acid. J. Food Sci. 2011, 76, M398–M403. [Google Scholar] [CrossRef] [PubMed]
- ur Rahman, A. Frontiers in Clinical Drug Research; Bentham Science Publishers: Sharjah, UAE, 2016. [Google Scholar]
- Wang, G.F.; Shi, L.P.; Ren, Y.D.; Liu, Q.F.; Liu, H.F.; Zhang, R.J.; Li, Z.; Zhu, F.H.; He, P.L.; Tang, W.; et al. Anti-hepatitis B virus activity of chlorogenic acid, quinic acid and caffeic acid in vivo and in vitro. Antivir. Res. 2009, 83, 186–190. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Liu, Y.; Hou, X.; Peng, H.; Zhang, L.; Jiang, Q.; Shi, M.; Ji, Y.; Wang, Y.; Shi, W. Chlorogenic acid inhibits the replication and viability of enterovirus 71 in vitro. PLoS ONE 2013, 8, e76007. [Google Scholar] [CrossRef] [PubMed]
- Sung, W.S.; Lee, D.G. Antifungal action of chlorogenic acid against pathogenic fungi, mediated by membrane disruption. Pure Appl. Chem. 2010, 82, 219–226. [Google Scholar] [CrossRef]
- Mujtaba, A.; Masud, T.; Ahmad, A.; Ahmed, W.; Jabbar, S.; Levin, R.E. Antibacterial activity by chlorogenic acid isolated through resin from apricot (Prunus Armeniaca L.). Pak. J. Agric. Res. 2017, 30, 144–148. [Google Scholar] [CrossRef]
- Silva, I.F.; Oliveira, R.G.; Soaresm, I.M.; Alvim, T.C.; Ascencio, S.D.; Martins, D.T.O. Evaluation of acute toxicity, antibacterial activity, and mode of action of the hydroethanolic extract of Piper umbellatum. J. Ethnopharmacol. 2014, 151, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Bi, C.; Ca, H.; Liu, B.; Zhong, X.; Deng, X.; Wang, T.; Xiang, H.; Niu, X.; Wang, D. The therapeutic effect of chlorogenic acid against Staphylococcus aureus infection through sortase a inhibition. Front. Microbiol. 2015, 16, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Ameziane, J.; Aplincourt, M.; Dupont, L.; Heirman, F.; Pierrard, J.-C. Thermodynamic stability of copper (II), manganese (II), zinc (II) and iron. (III) complexes with chlorogenic acid. Bull. Soc. Fr. 1996, 133, 243–249. [Google Scholar]
- Mahal, H.S.; Kappor, S.; Satpati, S.K.; Mukherjee, T. Radical scavenging and catalytic activity of metal-phenolic complexes. J. Phys. Chem. B 2005, 109, 24197–24202. [Google Scholar] [CrossRef] [PubMed]
- Kiss, T.; Nagy, G.; Pesci, M.; Kozłowski, H.; Micera, G.; Erre, L.S. Complexes of 3,4-dihydroxylphenyl derivatives—X. Copper(II) complexes of chlorogenic acid and related compounds. Polyhedron 1989, 8, 2345–2349. [Google Scholar] [CrossRef]
- Milić, S.Z.; Potkonjak, N.I.; Gorjanović, S.Ž.; Veljović-Jovanović, S.D.; Pastor, F.T.; Sužnjević, D.Ž. A polarographic study of chlorogenic acid and its interaction with some heavy metal ions. Electroanalysis 2011, 23, 2935–2940. [Google Scholar] [CrossRef]
- Naso, L.G.; Valcarcel, M.; Roura-Ferre, M.; Kortazar, D.; Salado, C.; Lezama, L.; Rojo, T.; Gonzalez-Baro, A.C.; Williams, P.A.M.; Ferrer, E.G. Promising antioxidant and anticancer (human breast cancer) oxidovanadium(IV) complex of chlorogenic acid. Synthesis, characterization and spectroscopic examination on the transport mechanism with bovine serum albumin. J. Inorg. Biochem. 2014, 135, 86–99. [Google Scholar] [CrossRef] [PubMed]
- Belay, A. Determination of self—Associated 5-caffeoylquinic acid and its complexation with sodium hydroxide using UV-Vis spectroscopy. Int. J. Phys. Sci. 2010, 5, 459–464. [Google Scholar]
- Martin, R.; Lilley, T.H.; Falshaw, C.P.; Haslam, E.; Begley, M.J.; Magnolato, D. The caffeine-potassium chlorogenate molecular complex. Phytochem 1986, 26, 273–279. [Google Scholar] [CrossRef]
- Gaussian, Inc. Gaussian 09, Revision A.02; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Zhan, C.; Nichols, J.A.; Dixon, D.A. Ionization potential, ionization potential, electron affinity, electronegativity, hardness, and electron excitation energy: Molecular properties from density functional theory orbital energies. J. Phys. Chem. A 2033, 107, 4184–4195. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Diplock, A.T.; Symons, M.C.R. Techniques in Free Radical Research; Elsevier: Amsterdam, The Netherlands; London, UK; New York, NY, USA; Tokyo, Japan, 1991. [Google Scholar]
- Zeraik, M.L.; Petrono, M.S.; Coelho, D.; Regasini, L.O.; Silva, D.H.S.; da Fonseca, L.M.; Machado, S.A.S.; Bolzani, V.S.; Ximenes, V.F. Improvement of pro-oxidant capacity of protocatechuic acid by esterification. PLoS ONE 2014, 9, e110277. [Google Scholar] [CrossRef] [PubMed]
- Soczewiński, E.; Wachtmeister, C.A. The relation between the composition of certainternary twophase solvent systems and R. values. J. Chromatogr. 1962, 7, 311–320. [Google Scholar]
- Sato, Y.; Itagaki, S.; Kurokawa, T.; Ogura, J.; Kobayashi, M.; Hirano, T. In vitro and in vivo antioxidant properties of chlorogenic acid and caffeic acid. Int. J. Pharm. 2011, 403, 136–138. [Google Scholar] [CrossRef] [PubMed]
- Kabir, F.; Katayama, S.; Tanji, N.; Nakamur, S. Antimicrobial effects of chlorogenic acid and related compounds. J. Korean Soc. Appl. Biol. Chem. 2014, 57, 359–365. [Google Scholar] [CrossRef]
- Li, G.; Wang, X.; Xu, Y.; Zhang, B.; Xia, X. Antimicrobial effect and mode of action of chlorogenic acid on Staphylococcus aureus. Eur. Food Res. Technol. 2014, 238, 589–596. [Google Scholar] [CrossRef]
- Ohnishi, M.; Sugawara, R.; Kusano, T. Structure-Activity Relationship between the Hydrophobicity of Alkali Metal Salts of Warfarin [3-(α-Acetonyl-benzyl)-4-hydroxycoumarin] and the Effectiveness of the Taste Response to These Salts in Mice. Biosci. Biotechnol. Biochem. 1995, 56, 995–1001. [Google Scholar] [CrossRef]
- Mähler, J.; Persson, I. A study of the hydration of the alkali metal ions in aqueous solution. Inorg. Chem. 2012, 2, 425–438. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, K.; Cheynier, V.; Quideau, S. Recent Advances in Polyphenol Research; Wiley-Blackwell: Hoboken, NJ, USA, 2017. [Google Scholar]
- Litwinienko, G.; Ingold, K.U. Solvent effects on the rates and mechanisms of reaction of phenols with free radicals. Acc. Chem. Res. 2007, 40, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Prior, R.L. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef] [PubMed]
- Rutkowska, E.; Pająk, K.; Jóźwiak, K. Lipophilicity-methods of determination and its role in medicinal chemistry. Acta Pol. Pharm. Drug Res. 2013, 70, 3–18. [Google Scholar]
- Tsopelas, F.; Giaginis, C.; Tsantili-Kakoulidou, A. Lipophilicity and biomimetic properties to support drug discovery. Expert Opin. Drug Discov. 2017, 12, 885–896. [Google Scholar] [CrossRef] [PubMed]
- Reiner, R.N.; Labuckas, D.O.; Garcia, D.A. Lipophilicity of some GABAergic phenols and related compounds determined by HPLC and partition coefficients in different systems. J. Pharm. Biomed. Anal. 2009, 49, 686–691. [Google Scholar] [CrossRef] [PubMed]
- Jóźwiak, K.; Szumiło, H.; Senczyna, B. Lipophilicity parameter from high-performance liquid chromatography on an immobilized artificial membrane column and its relationships to bioactivity of the group of 2,4-dihydroxythiobenzanilides. Acta Pol. Pharm. 2002, 59, 341–346. [Google Scholar] [PubMed]



| Li 5-CQA | Na 5-CQA | K 5-CQA | Rb 5-CQA | Cs 5-CQA | 5-CQA | Assignments | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IR Calc. | Int. Calc. | IR Exp. | Int. Exp. | IR Calc. | Int. Calc. | Ir Exp. | Int. Exp. | IR Calc. | Int. Calc. | IR Exp. | Int. Exp. | IR Exp. | Int. Exp. | IR Exp. | Int. Exp. | IR Exp. | Int. Exp. | |
| 1725 | s | ν(C=O)COOH | ||||||||||||||||
| 1761 | 209.4 | 1687 | S | 1761 | 209.8 | 1694 | s | 1761 | 207.1 | 1692 | s | 1690 | s | 1685 | s | 1687 | vs | ν(C=O)ester group |
| 1679 | 204.9 | 1637 | Sh | 1680 | 197.7 | 1636 | s | 1681 | 194.9 | 1634 | sh | 1628 | sh | 1626 | sh | 1640 | s | ν(C=C) + ν(CC)ar |
| 1641 | 354.6 | 1642 | 327.5 | 1642 | 310.9 | 1602 | m | ν(CC)ar + ν(C=C) | ||||||||||
| 1633 | 55.7 | 1633 | 55.6 | 1633 | 55.8 | 1613 | m | ν(CC)ar | ||||||||||
| 1581 | 333.4 | 1603 | vs | 1602 | 360.2 | 1598 | vs | 1613 | 326.4 | 1599 | vs | 1596 | vs | 1598 | vs | - | νas(COO−) | |
| 1559 | 134.3 | 1518 | m | 1558 | 134.3 | 1528 | m | 1558 | 135.2 | 1522 | w | 1522 | m | 1522 | w | 1530 | m | ν(CC)ar |
| 1462 | 81.9 | 1446 | m | 1460 | 84.8 | 1450 | w | 1465 | 79.8 | 1447 | w | 1447 | m | 1447 | w | 1443 | m | δ(C3-H3) + δ(O2-H8) |
| 1443 | 237.1 | 1385 | sh | 1423 | 132.3 | 1390 | sh | 1421 | 116.4 | 1395 | m | 1395 | m | 1395 | m | - | νs(COO−) | |
| 1415 | 4.7 | 1378 | m | 1414 | 8.0 | 1384 | m | 1412 | 28.9 | 1375 | m | 1375 | m | 1375 | m | 1382 | w | δ(C-H)quin + δ(O1-H1)quin |
| 1304 | 142.8 | 1273 | vs | 1303 | 296.4 | 1281 | vs | 1303 | 326.9 | 1268 | vs | 1266 | vs | 1268 | vs | 1289 | vs | ν(C4’-O2’) + δ(C-H)ar |
| 1208 | 80.5 | 1184 | s | 1207 | 93.5 | 1178 | s | 1207 | 105.5 | 1181 | m | 1181 | m | 1118 | m | 1190 | m | ν(C1’-C7’) + δ(C-H)ar + ν(C3’-O1’) + δ(O2’-H4’) |
| 1144 | 667.7 | 1119 | m | 1143 | 618.7 | 1119 | m | 1145 | 600.1 | 1119 | m | 1119 | m | 1120 | m | 1114 | s | (C8’-H8’) + δ(O3-H9) + δ(C-H)quin + δ(O1-H1)quin |
| 1101 | 44.3 | 1083 | w | 1099 | 48.2 | 1081 | m | 1101 | 51.5 | 1083 | w | 1083 | w | 1083 | w | 1086 | m | ν(C5-C6) + ν(C4-O3) |
| 1055 | 16.5 | 1059 | vw | 1054 | 12.0 | 1059 | w | 1055 | 12.6 | 1055 | vw | 1057 | sh | ν(C3-C3) + ν(C5-O4’) + δ(C-H)quin | ||||
| 1035 | 79.2 | 1041 | w | 1030 | 131.9 | 1037 | w | 1029 | 133.5 | 1037 | w | 1038 | w | 1037 | w | 1038 | sh | ν(C5-O4’) + δ(C-H)quin |
| 1019 | 28.5 | 1018 | 29.4 | 997 | vw | 1020 | 29.6 | 1000 | w | oop(C7’-H7’) + oop(C8’-H8’) | ||||||||
| 973 | 48.3 | 976 | w | 972 | 49 | 969 | w | 973 | 50.6 | 974 | w | 976 | w | 976 | w | 977 | m | ν(C1-O1) + δ(C-H)quin + δ(C-H)ar |
| 893 | 5.6 | 853 | w | 890 | 4.9 | 854 | w | 894 | 6.5 | 851 | vw | 851 | vw | 853 | w | 854 | w | oop(C8’-H8’) + oop(C7’-H7’) + oop(C2’-H2’) |
| 819 | 18.4 | 812 | w | 812 | 14.9 | 808 | w | 827 | 1.5 | 810 | w | 812 | w | 813 | w | 819 | s | δ(C-H)quin + δ(COO−) |
| 790 | 24.3 | 769 | w | 790 | 23.6 | 772 | vw | 790 | 22.4 | 783 | w | 769 | vw | 768 | vw | δ(C4’-C3’) | ||
| 764 | 13.8 | 613 | w | 766 | 14.2 | 615 | w | 766 | 15.4 | oop(COO−) | ||||||||
| 672 | 199.4 | 668 | vw | 670 | 4.1 | 668 | wv | 672 | 4.8 | 668 | vw | 668 | vw | 668 | vw | 670 | vw | δ(C-C)quin |
| 631 | 174.8 | 623 | 63.6 | 603 | vw | 623 | 63.9 | 603 | vw | 605 | vw | 606 | vw | 603 | m | δ(C-C)quin + oop(O5-H7) | ||
| 580 | 18.0 | 571 | w | 581 | 14.8 | 565 | vw | 581 | 13.0 | 569 | vw | 571 | vw | 569 | vw | 564 | w | δ(C-C)ar |
| Proton No. | 5-CQA [2] | 5-CQAs | Carbon No. | 5-CQA | 5-CQAs | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Li | Na | K | Rb | Cs | Li | Na | K | Rb | Cs | ||||
| H2eq | 1.80 | 1.68 | 1.73 | 1.72 | 1.61 | 1.60 | C1 | 73.6 | 75.8 | 75.2 | 75.2 | 75.2 | 75.2 |
| H2ax | 2.03 | 1.99 | 1.99 | 1.98 | 1.98 | 1.97 | C2 | 37.3 | 39.5 | 39.5 | 39.5 | 39.5 | 39.5 |
| H3 | 3.94 | 3.92 | 3.91 | 3.90 | 3.88 | 3.88 | C3 | 68.2 | 71.4 | 71.4 | 71.5 | 71.6 | 71.6 |
| H4 | 3.58 | 3.48 | 3.49 | 3.48 | 3.47 | 3.48 | C4 | 70.5 | 71.5 | 71.6 | 71.6 | 71.7 | 71.6 |
| H5 | 5.08 | 5.17 | 5.17 | 5.17 | 5.16 | 5.16 | C5 | 71.0 | 73.2 | 73.3 | 73.4 | 73.5 | 73.5 |
| H6eq | 1.96 | 1.81 | 1.81 | 1.81 | 1.90 | 1.81 | C6 | 36.4 | 38.0 | 38.1 | 38.2 | 38.2 | 38.2 |
| H6ax | 2.01 | 1.85 | 1.87 | 1.87 | 1.94 | 1.94 | C7 | 175.0 | 176.5 | 176.7 | 176.5 | 176.2 | 176.0 |
| H7 | 12.41 | C1′ | 125.7 | 125.4 | 125.5 | 125.4 | 124.4 | 124.5 | |||||
| H2′ | 7.04 | 7.06 | 7.07 | 7.08 | 7.01 | 7.02 | C2′ | 114.9 | 114.5 | 114.7 | 114.7 | 114.0 | 114.1 |
| H3′ | 9.13 | C3′ | 145.6 | 145.8 | 145.8 | 145.9 | 146.3 | 146.2 | |||||
| H4′ | 9.56 | C4′ | 148.4 | 148.8 | 148.5 | 148.8 | 150.2 | 150.0 | |||||
| H5′ | 6.77 | 6.75 | 6.75 | 6.72 | 6.69 | 6.70 | C5′ | 115.9 | 115.9 | 115.8 | 115.9 | 115.6 | 115.6 |
| H6′ | 6.98 | 6.97 | 6.97 | 6.96 | 6.93 | 6.93 | C6′ | 121.5 | 121.2 | 121.2 | 121.2 | 121.5 | 121.4 |
| H7′ | 7.43 | 7.46 | 7.44 | 7.44 | 7.44 | 7.44 | C7′ | 145.0 | 144.7 | 144.6 | 144.7 | 144.9 | 144.9 |
| H8′ | 6.16 | 6.22 | 6.22 | 6.22 | 6.21 | 6.22 | C8′ | 114.4 | 114.5 | 114.6 | 114.5 | 113.7 | 114.1 |
| Compounds | E. coli (Gram−) | Bacillus sp. (Gram+) | Staphylococcus Epidermidis (Gram+) | Streptococcus Pyogenes (Gram+) | Candida sp. (Gram+) |
|---|---|---|---|---|---|
| 5-CQA | 7.06 (2.5 mg/mL) | ˃7.06 (2.5 mg/mL) | ˃7.06 (2.5 mg/mL) | ˃7.06 (2.5 mg/mL) | ˃14.11 (5 mg/mL) |
| Li 5-CQA | 6.46 | ˃6.46 | ˃6.46 | ˃6.46 | ˃12.91 |
| Na 5-CQA | 6.20 | ˃6.20 | ˃6.20 | ˃6.20 | ˃12.40 |
| K 5-CQA | 5.96 | 5.96 | 2.98 | ˃5.96 | 5.96 |
| Rb 5-CQA | 2.63 | 5.27 | 5.27 | 2.63 | 5.27 |
| Cs 5-CQA | 4.79 | ˃4.79 | ˃4.79 | ˃4.79 | ˃9.58 |
| Kanamycin (positive control) | 0.21 | 0.21 | 0.21 | 0.21 | 0.41 |
| Ampicillin (positive control) | 0.43 | 0.57 | 0.57 | 0.57 | 0.57 |
| Compound | DPPH | FRAP Values | |
|---|---|---|---|
| EC50 [µM] | Ccompound [µM] | C Fe2+ [µM] | |
| 5-CQA | 7.39 ± 0.71 | 5 2.5 | 114.22 ± 8.09 72.53 ± 2.42 |
| Li 5-CQA | 5.28 ± 0.375 | 5 2.5 | 106.92 ± 1.28 78.93 ± 4.23 |
| Na 5-CQA | 4.50 ± 0.36 | 5 2.5 | 141.13 ± 5.78 103.08 ± 2.79 |
| K 5-CQA | 5.32 ± 0.49 | 5 2.5 | 132.00 ± 6.07 99.89 ± 7.05 |
| Rb 5-CQA | 5.40 ± 0.68 | 5 2.5 | 123.16 ± 9.56 86.70 ± 2.86 |
| Cs 5-CQA | 5.89 ± 1.57 | 5 2.5 | 117. 00 ± 6.22 80.47 ± 5.68 |
| Trolox | 8.25 ± 0.75 | 5 | 70.85 ± 3.57 |
| l-ascorbic acid | 10.87 ± 0.53 | 5 | 81.70 ± 4.23 |
| BHT | 52.80 ± 2.83 | 5 | 69.66 ± 5.54 |
| Compound | logkw | logP | |||||
|---|---|---|---|---|---|---|---|
| C18 | C8 | CN | IAM | PHE | |||
| 5-CQA | 3.88 | 1.11 | 2.09 | 0.74 | 1.45 | Exp. | 0.6 [1] |
| Calc. | −0.45 [2] 0.07 [3] 0.69 [4] | ||||||
| Li 5-CQA | 1.84 | 0.21 | 0.84 | 1.24 | 1.23 | ||
| Na 5-CQA | 2.24 | 1.11 | 1.35 | 1.84 | 1.43 | ||
| K 5-CQA | 2.36 | 1.24 | 1.84 | 1.91 | 1.84 | ||
| Rb 5-CQA | 3.21 | 1.84 | 2.23 | 2.84 | 2.24 | ||
| Cs 5-CQA | 3.74 | 2.13 | 2.64 | 3.84 | 2.64 | ||
| Parameters | 5-CQA | Li 5-CQA | Na 5-CQA | K 5-CQA | ||||
|---|---|---|---|---|---|---|---|---|
| Gas | MeOH | Gas | MeOH | Gas | MeOH | Gas | MeOH | |
| LUMO (hartree) | −0.2036 | −0.0812 | −0.0743 | −0.0799 | −0.0719 | −0.0797 | −0.0687 | −0.0796 |
| HOMO (hartree) | −0.3021 | −0.2269 | −0.2248 | −0.2264 | −0.2222 | −0.2263 | −0.2206 | −0.2262 |
| LUMO (eV) | −5.5389 | −2.2093 | −2.0226 | −2.1747 | −1.9554 | −2.1685 | −1.8697 | −2.1647 |
| HOMO (eV) | −8.2208 | −6.1743 | −6.1158 | −6.1607 | −6.0450 | −6.1579 | −6.0037 | −6.1555 |
| Energy gap (eV) | 2.6820 | 3.9650 | 4.0931 | 3.9859 | 4.0896 | 3.9895 | 4.1340 | 3.9908 |
| Ionisation potential (eV) | 8.2208 | 6.1743 | 6.1158 | 6.1607 | 6.0450 | 6.1579 | 6.0037 | 6.1555 |
| Electron affinity (eV) | 5.5389 | 2.2093 | 2.0226 | 2.1747 | 1.9554 | 2.1685 | 1.8697 | 2.1647 |
| Electronegativity (eV) | 6.8799 | 4.1918 | 4.0692 | 4.1677 | 4.0002 | 4.1632 | 3.9367 | 4.1601 |
| Electronic chemical potential (eV) | −6.8799 | −4.1918 | −4.0692 | −4.1677 | −4.0002 | −4.1632 | −3.9367 | −4.1601 |
| Chemical hardness (eV) | 1.3410 | 1.9825 | 2.0466 | 1.9930 | 2.0448 | 1.9947 | 2.0670 | 1.9954 |
| Chemical softness (eV) | 0.3729 | 0.2522 | 0.2443 | 0.2509 | 0.2445 | 0.2507 | 0.2419 | 0.2506 |
| Electrophilicity index (eV) | 17.6485 | 4.4316 | 4.0454 | 4.3578 | 3.9128 | 4.3445 | 3.7488 | 4.3365 |
| Total energy (a.u.) | −1297.950 | −1297.985 | −1304.95 | −1305.029 | −1459.7200 | −1459.795 | −1897.370 | −1897.433 |
| Dipole moment (Debay) | 6.1992 | 8.6622 | 9.0114 | 13.4233 | 11.7279 | 15.4030 | 14.0710 | 17.1000 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kalinowska, M.; Bajko, E.; Matejczyk, M.; Kaczyński, P.; Łozowicka, B.; Lewandowski, W. The Study of Anti-/Pro-Oxidant, Lipophilic, Microbial and Spectroscopic Properties of New Alkali Metal Salts of 5-O-Caffeoylquinic Acid. Int. J. Mol. Sci. 2018, 19, 463. https://doi.org/10.3390/ijms19020463
Kalinowska M, Bajko E, Matejczyk M, Kaczyński P, Łozowicka B, Lewandowski W. The Study of Anti-/Pro-Oxidant, Lipophilic, Microbial and Spectroscopic Properties of New Alkali Metal Salts of 5-O-Caffeoylquinic Acid. International Journal of Molecular Sciences. 2018; 19(2):463. https://doi.org/10.3390/ijms19020463
Chicago/Turabian StyleKalinowska, Monika, Ewelina Bajko, Marzena Matejczyk, Piotr Kaczyński, Bożena Łozowicka, and Włodzimierz Lewandowski. 2018. "The Study of Anti-/Pro-Oxidant, Lipophilic, Microbial and Spectroscopic Properties of New Alkali Metal Salts of 5-O-Caffeoylquinic Acid" International Journal of Molecular Sciences 19, no. 2: 463. https://doi.org/10.3390/ijms19020463







