VEGF Triggers the Activation of Cofilin and the Arp2/3 Complex within the Growth Cone
Abstract
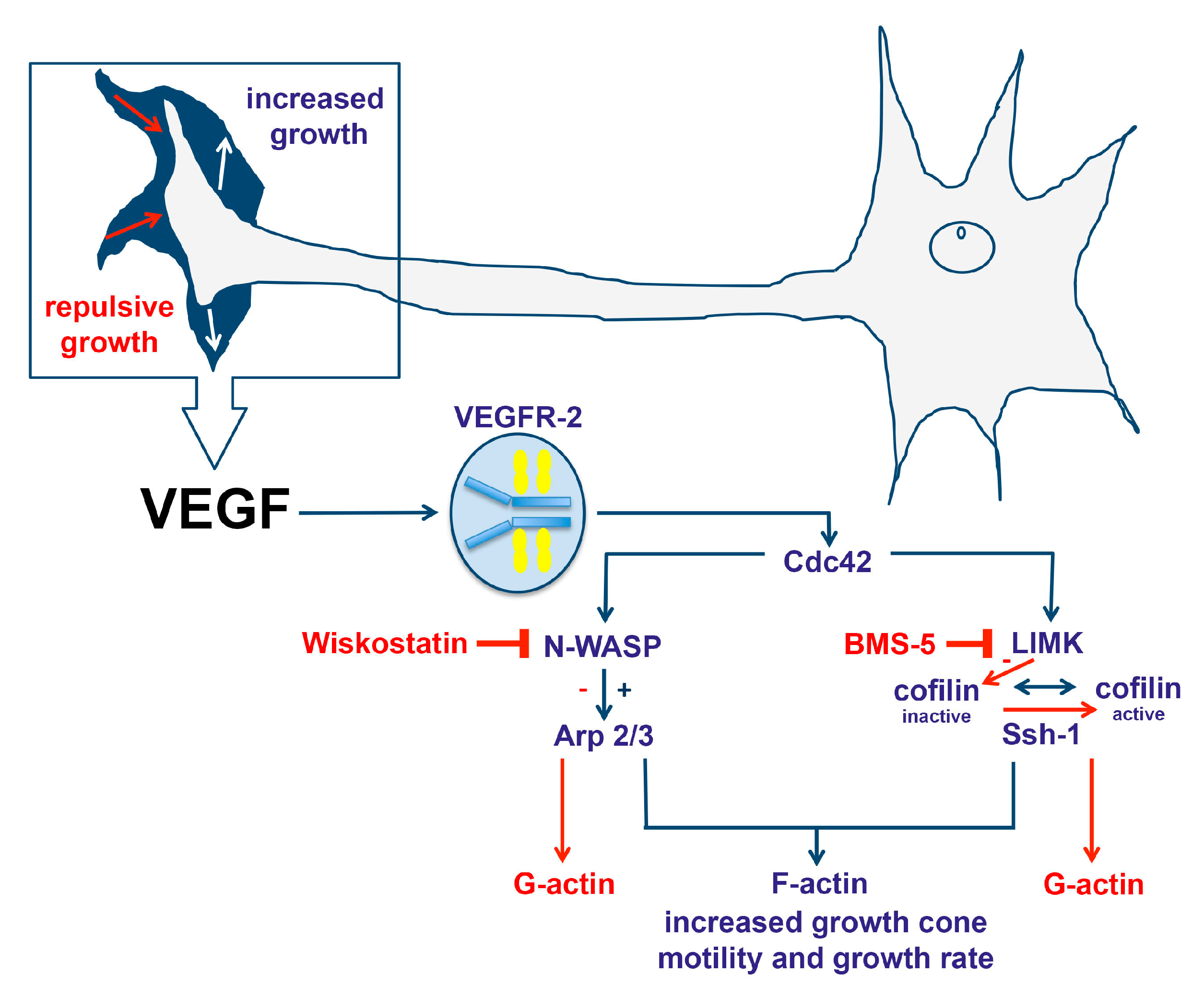
:1. Introduction
2. Results
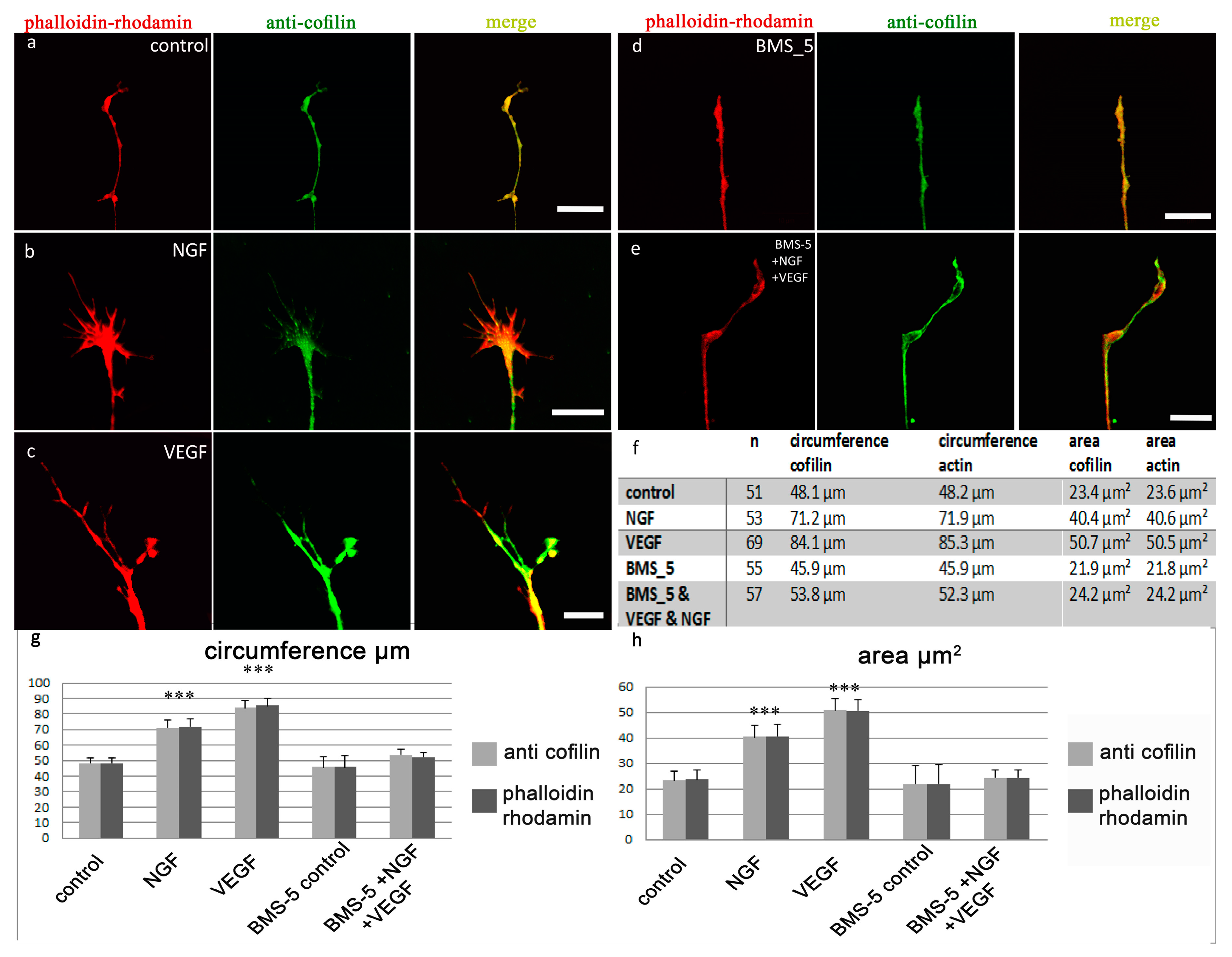
2.1. Analysis of the Distribution of Cofilin and Actin in the Growth Cone
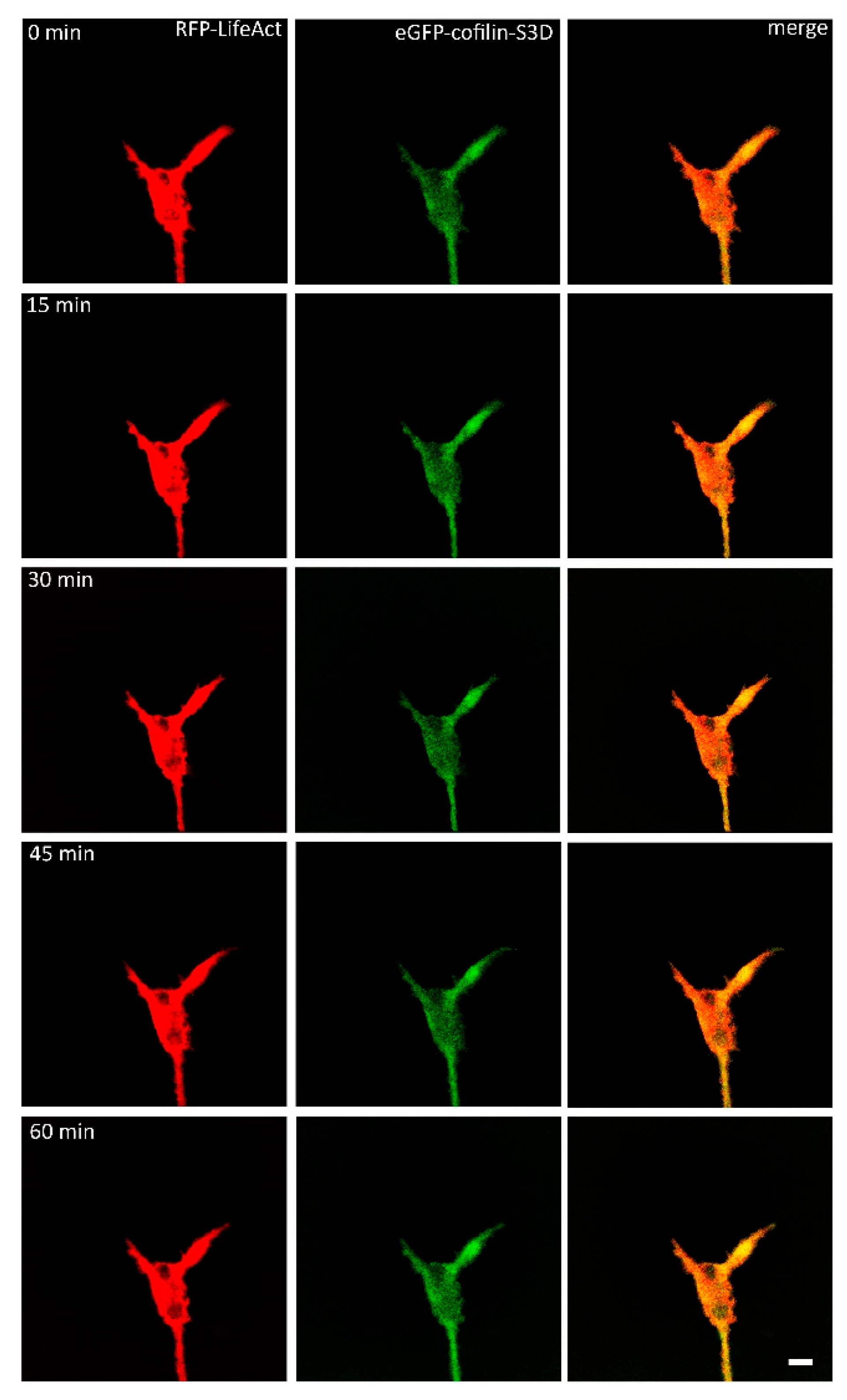
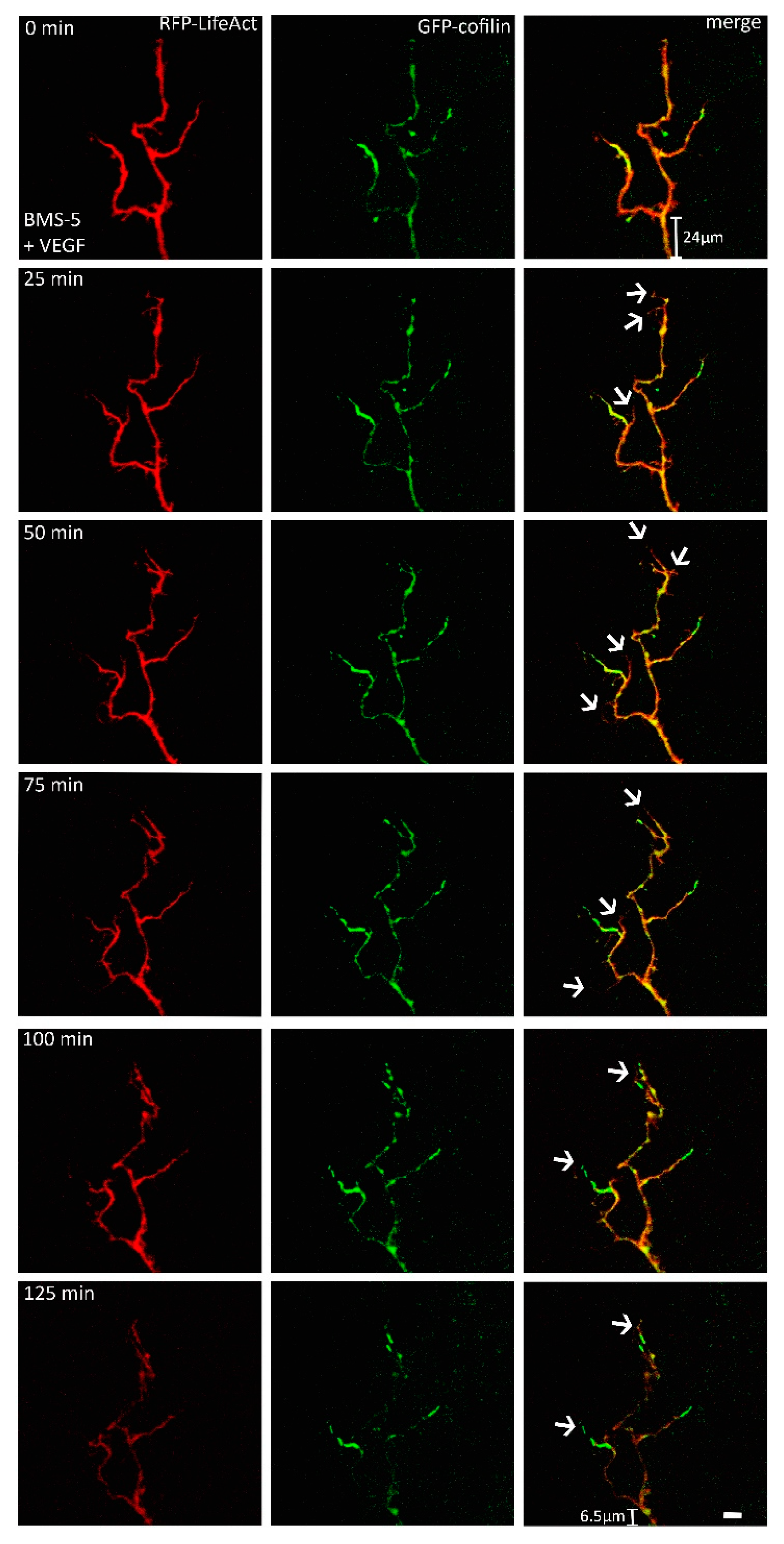
2.2. VEGF Leads to Rapid Changes in Cofilin Distribution and Repels Growth Cones When LIMK is Inhibited
2.3. Analysis of the Distribution of the Arp 2/3-Complex within the Axonal Growth Cone
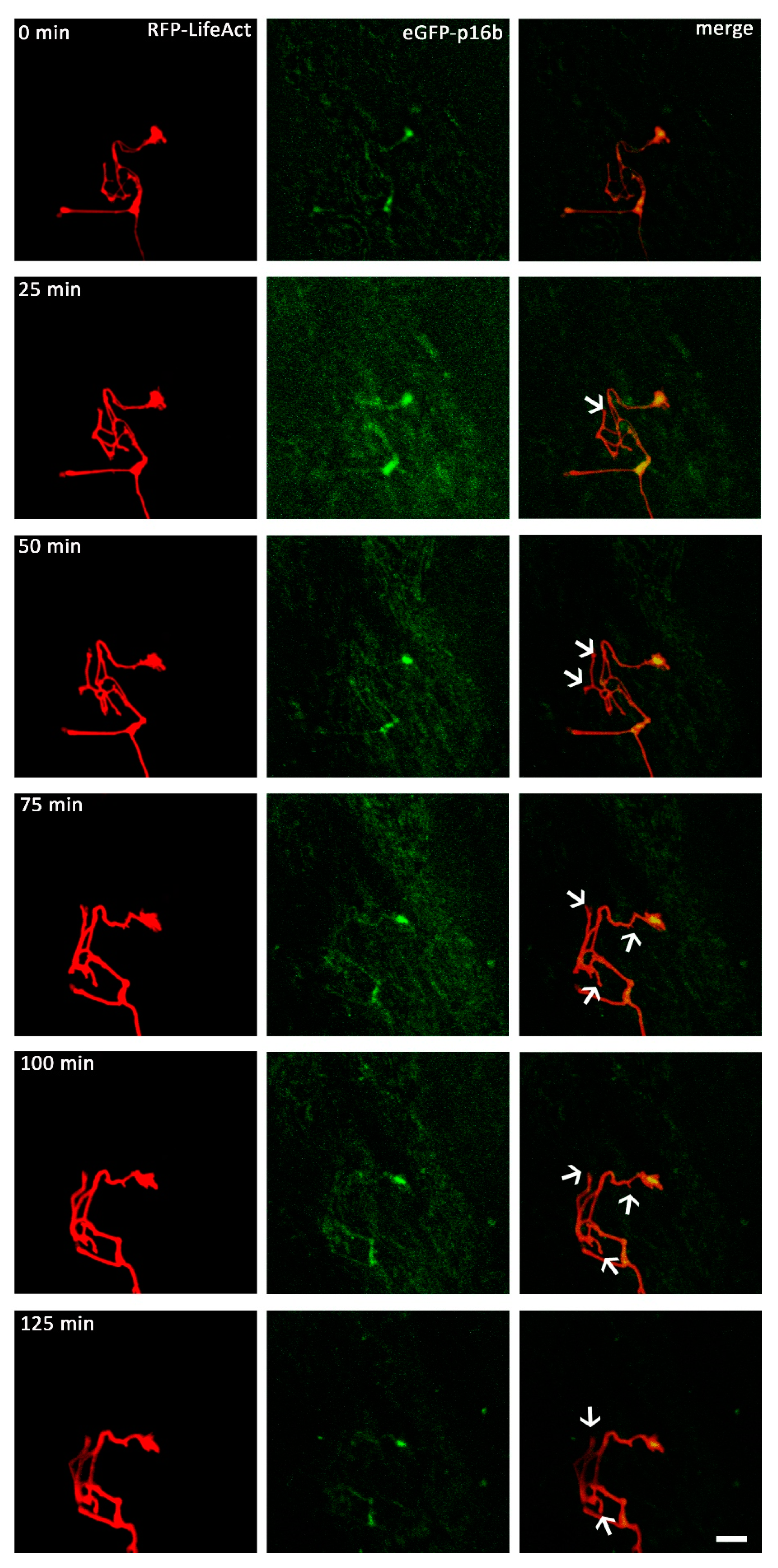
2.4. VEGF Triggers the Redistribution of Arp2/3 and Repels Growth Cones When N-WASP is Inhibited
3. Discussion
3.1. VEGF Triggers a Change in the Distribution of Cofilin in a LIMK-Dependent Pathway
3.2. VEGF Triggers the Distribution of the Arp2/3 Complex within the Growth Cone in an N-WASP Dependent Pathway
4. Materials and Methods
4.1. Immunohistochemistry and Evaluation of Morphology
4.1.1. DRG Explants
4.1.2. Immunohistochemistry
4.1.3. Analysis of Growth Cone Morphology
4.2. Live Cell Imaging
4.2.1. Dissociated DRG Cell Cultures
4.2.2. Microinjection
4.2.3. Live Cell Imaging with the Confocal Laser Scanning Microscope
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dent, E.W.; Gertler, F.B. Cytoskeletal dynamics and transport in growth cone motility and axon guidance. Neuron 2003, 40, 209–227. [Google Scholar] [CrossRef]
- Lowery, L.A.; van Vactor, D. The trip of the tip: Understanding the growth cone machinery. Nat. Rev. Mol. Cell Biol. 2009, 10, 332–343. [Google Scholar] [CrossRef] [PubMed]
- Wegner, A. Treadmilling of actin at physiological salt concentrations. An analysis of the critical concentrations of actin filaments. J. Mol. Biol. 1982, 161, 607–615. [Google Scholar] [CrossRef]
- Dominguez, R. Actin filament nucleation and elongation factors—Structure-function relationships. Crit. Rev. Biochem. Mol. Biol. 2009, 44, 351–366. [Google Scholar] [CrossRef] [PubMed]
- Machesky, L.M.; Atkinson, S.J.; Ampe, C.; Vandekerckhove, J.; Pollard, T.D. Purification of a cortical complex containing two unconventional actins from Acanthamoeba by affinity chromatography on profilin-agarose. J. Cell Biol. 1994, 127, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Machesky, L.M.; Insall, R.H. Scar1 and the related Wiskott-Aldrich syndrome protein, WASP, regulate the actin cytoskeleton through the Arp2/3 complex. Curr. Biol. 1998, 8, 1347–1356. [Google Scholar] [CrossRef]
- Wear, M.A.; Schafer, D.A.; Cooper, J.A. Actin dynamics: Assembly and disassembly of actin networks. Curr. Biol. 2000, 10, R891–R895. [Google Scholar] [CrossRef]
- Machesky, L.M.; Mullins, R.D.; Higgs, H.N.; Kaiser, D.A.; Blanchoin, L.; May, R.C.; Hall, M.E.; Pollard, T.D. Scar, a WASp-related protein, activates nucleation of actin filaments by the Arp2/3 complex. Proc. Natl. Acad. Sci. USA 1999, 96, 3739–3744. [Google Scholar] [CrossRef] [PubMed]
- Rohatgi, R.; Ma, L.; Miki, H.; Lopez, M.; Kirchhausen, T.; Takenawa, T.; Kirschner, M.W. The interaction between N-WASP and the Arp2/3 complex links Cdc42-dependent signals to actin assembly. Cell 1999, 97, 221–231. [Google Scholar] [CrossRef]
- Brown, J.A.; Bridgman, P.C. Disruption of the cytoskeleton during Semaphorin 3A induced growth cone collapse correlates with differences in actin organization and associated binding proteins. Dev. Neurobiol. 2009, 69, 633–646. [Google Scholar] [CrossRef] [PubMed]
- Andrianantoandro, E.; Pollard, T.D. Mechanism of Actin filament turnover by severing and nucleation at different concentrations of ADF/cofilin. Mol. Cell. 2006, 24, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.Q.; Kwiatkowska, K.; Yin, H.L. Actin monomer binding proteins. Curr. Opin. Cell Biol. 1995, 7, 102–110. [Google Scholar] [CrossRef]
- Cooper, J.A.; Schafer, D.A. Control of actin assembly and disassembly at filament ends. Curr. Opin. Cell Biol. 2000, 12, 97–103. [Google Scholar] [CrossRef]
- Svitkina, T.M.; Borisy, G.G. Arp2/3 complex and actin depolymerizing factor/cofilin in dendritic organization and treadmilling of actin filament array in lamellipodia. J. Cell Biol. 1999, 145, 1009–1026. [Google Scholar] [CrossRef] [PubMed]
- Al Haj, A.; Mazur, A.J.; Buchmeier, S.; App, C.; Theiss, C.; Jokusch, B.M.; Hannappel, E.; Weeds, A.G.; Mannherz, H.G. Thymosin β4 inhibits ADF/cofilin stimulated F-actin dycling and cell migration: Reversal by Arp2/3 complex. Cytoskeleton 2014, 71, 95–107. [Google Scholar] [CrossRef] [PubMed]
- Sarmiere, P.D.; Bamburg, J.R. Regulation of the neuronal actin cytoskeleton by ADF/cofilin. J. Neurobiol. 2004, 58, 103–117. [Google Scholar] [CrossRef] [PubMed]
- Endo, M.; Ohashi, K.; Sasaki, Y.; Goshima, Y.; Niwa, R.; Uemura, T.; Mizuno, K. Control of growth cone motility and morphology by LIM kinase and Slingshot via phosphorylation and dephosphorylation of cofilin. J. Neurosci. 2003, 23, 2527–2537. [Google Scholar] [PubMed]
- Marsick, B.M.; Flynn, K.C.; Santiago-Medina, M.; Bamburg, J.R.; Letourneau, P.C. Activation of ADF/cofilin mediates attractive growth cone turning toward nerve growth factor and netrin-1. Dev. Neurobiol. 2010, 70, 565–588. [Google Scholar] [CrossRef] [PubMed]
- Chai, X.; Zhao, S.; Fan, L.; Zhang, W.; Lu, X.; Shao, H.; Wang, S.; Song, L.; Failla, A.V.; Zobiak, B.; et al. Reelin and cofilin cooperate during the migration of cortical neurons: A quantitative morphological analysis. Development 2016, 143, 1029–1040. [Google Scholar] [CrossRef] [PubMed]
- Senger, D.R.; Galli, S.J.; Dvorak, A.M.; Perruzzi, C.A.; Harvey, V.S.; Dvorak, H.F. Tumor cells secrete a vascular permeability factor that promotes accumulation of ascites fluid. Science 1983, 219, 983–985. [Google Scholar] [CrossRef] [PubMed]
- Dumpich, M.; Mannherz, H.G.; Theiss, C. VEGF Signaling Regulates Cofilin and the Arp2/3-complex within the Axonal Growth Cone. Curr. Neurovasc. Res. 2015, 12, 293–307. [Google Scholar] [CrossRef] [PubMed]
- Sondell, M.; Lundborg, G.; Kanje, M. Vascular endothelial growth factor has neurotrophic activity and stimulates axonal outgrowth, enhancing cell survival and Schwann cell proliferation in the peripheral nervous system. J. Neurosci. 1999, 19, 5731–5740. [Google Scholar] [PubMed]
- Sondell, M.; Sundler, F.; Kanje, M. Vascular endothelial growth factor is a neurotrophic factor which stimulates axonal outgrowth through the flk-1 receptor. Eur. J. Neurosci. 2000, 12, 4243–4254. [Google Scholar] [CrossRef] [PubMed]
- Sondell, M.; Lundborg, G.; Kanje, M. Vascular endothelial growth factor stimulates Schwann cell invasion and neovascularization of acellular nerve grafts. Brain Res. 1999, 846, 219–228. [Google Scholar] [CrossRef]
- Hayakawa, K.; Pham, L.D.; Som, A.T.; Lee, B.J.; Guo, S.; Lo, E.H.; Arai, K. Vascular endothelial growth factor regulates the migration of oligodendrocyte precursor cells. J. Neurosci. 2011, 31, 10666–10670. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Jiao, X.; Zuzga, D.S.; Liu, Y.; Fong, D.M.; Young, D.; During, M.J. VEGF links hippocampal activity with neurogenesis, learning and memory. Nat. Genet. 2004, 36, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Erskine, L.; Reijntjes, S.; Pratt, T.; Denti, L.; Schwarz, Q.; Vieira, J.M.; Alakakone, B.; Shewan, D.; Ruhrberg, C. VEGF signaling through neuropilin 1 guides commissural axon crossing at the optic chiasm. Neuron 2011, 70, 951–965. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Almodovar, C.R.; Fabre, P.J.; Knevels, E.; Coulon, C.; Segura, I.; Haddick, P.C.; Aerts, L.; Delattin, N.; Strasser, G.; Oh, W.J.; et al. VEGF mediates commissural axon chemoattraction through its receptor Flk1. Neuron 2011, 70, 966–978. [Google Scholar]
- Olbrich, L.; Foehring, D.; Happel, P.; Brand-Saberi, B.; Theiss, C. Fast rearrangement of the neuronal growth cone’s actin cytoskeleton following VEGF stimulation. Histochem. Cell Biol. 2013, 139, 431–445. [Google Scholar] [CrossRef] [PubMed]
- Foehring, D.; Brand-Saberi, B.; Theiss, C. VEGF-induced growth cone enhancement is diminished by inhibiting tyrosine-residue 1214 of VEGFR-2. Cells Tissues Organs 2012, 196, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Lamalice, L.; Houle, F.; Jourdan, G.; Huot, J. Phosphorylation of tyrosine 1214 on VEGFR2 is required for VEGF-induced activation of Cdc42 upstream of SAPK2/p38. Oncogene 2004, 23, 434–445. [Google Scholar] [CrossRef] [PubMed]
- Nobes, C.D.; Hall, A. Rho, Rac, and cdc42 GTPases regulate the assembly of multimolecular focal complexes associated with actin stress fibers, lamellipodia, and filopodia. Cell 1995, 81, 53–62. [Google Scholar] [CrossRef]
- Masyuk, M.; Morosan-Puopolo, G.; Brand-Saberi, B.; Theiss, C. Combination of in ovo electroporation and time-lapse imaging to study migrational events in chicken embryos. Dev. Dyn. 2014, 243, 690–698. [Google Scholar] [CrossRef] [PubMed]
- Theiss, C.; Napirei, M.; Meller, K. Impairment of anterograde and retrograde neurofilament transport after anti-kinesin and anti-dynein antibody microinjection in chicken dorsal root ganglia. Eur. J. Cell Biol. 2005, 84, 29–43. [Google Scholar] [CrossRef] [PubMed]
- Storkebaum, E.; Carmeliet, P. VEGF: A critical player in neurodegeneration. J. Clin. Investig. 2004, 113, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Oosthuyse, B.; Moons, L.; Storkebaum, E.; Beck, H.; Nuyens, D.; Brusselmans, K.; van Dorpe, J.; Hellings, P.; Gorselink, M.; Heymans, S.; et al. Deletion of the hypoxia-response element in the vascular endothelial growth factor promoter causes motor neuron degeneration. Nat. Genet. 2001, 28, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, B.W.; Painter, W.B.; Chen, H.; Minamide, L.S.; Abe, H.; Bamburg, J.R. Intracellular pH modulation of ADF/cofilin proteins. Cell Motil. Cytoskelet. 2000, 47, 319–336. [Google Scholar] [CrossRef]
- Ressad, F.; Didry, D.; Egile, C.; Pantaloni, D.; Carlier, M.F. Control of actin filament length and turnover by actin depolymerizing factor (ADF/cofilin) in the presence of capping proteins and ARP2/3 complex. J. Biol. Chem. 1999, 274, 20970–20976. [Google Scholar] [CrossRef] [PubMed]
- Gohla, A.; Bokoch, G.M. 14-3-3 regulates actin dynamics by stabilizing phosphorylated cofilin. Curr. Biol. 2002, 12, 1704–1710. [Google Scholar] [CrossRef]
- Yonezawa, N.; Homma, Y.; Yahara, I.; Sakai, H.; Nishida, E. A short sequence responsible for both phosphoinositide binding and actin binding activities of cofilin. J. Biol. Chem. 1991, 266, 17218–17221. [Google Scholar] [PubMed]
- Zhang, H.H.; Wang, W.; Feng, L.; Yang, Y.; Zheng, J.; Huang, L.; Chen, D.B. S-nitrosylation of Cofilin-1 Serves as a Novel Pathway for VEGF-Stimulated Endothelial Cell Migration. J. Cell. Physiol. 2014, 230, 406–417. [Google Scholar] [CrossRef] [PubMed]
- Morgan, T.E.; Lockerbie, R.O.; Minamide, L.S.; Browning, M.D.; Bamburg, J.R. Isolation and characterization of a regulated form of actin depolymerizing factor. J. Cell Biol. 1993, 122, 623–633. [Google Scholar] [CrossRef] [PubMed]
- Bamburg, J.R.; Bernstein, B.W. ADF/cofilin. Curr. Biol. 2008, 18, R273–R275. [Google Scholar] [CrossRef] [PubMed]
- Niwa, R.; Nagata-Ohashi, K.; Takeichi, M.; Mizuno, K.; Uemura, T. Control of actin reorganization by Slingshot, a family of phosphatases that dephosphorylate ADF/cofilin. Cell 2002, 108, 233–246. [Google Scholar] [CrossRef]
- Ono, S.; Minami, N.; Abe, H.; Obinata, T. Characterization of a novel cofilin isoform that is predominantly expressed in mammalian skeletal muscle. J. Biol. Chem. 1994, 269, 15280–15286. [Google Scholar] [PubMed]
- Abe, H.; Endo, T.; Yamamoto, K.; Obinata, T. Sequence of cDNAs encoding actin depolymerizing factor and cofilin of embryonic chicken skeletal muscle: Two functionally distinct actin regulatory proteins exhibit high structural homology. Biochemistry 1990, 29, 720–725. [Google Scholar] [CrossRef]
- Li, L.; Zhang, W.; Chai, B.; Zhang, Q.; Xie, J.; Chen, S.; Zhao, S. Neuronal maturation and laminar formation in the chicken optic tectum are accompanied by the transition of phosphorylated cofilin from cytoplasm to nucleus. Gene Expr. Patterns 2014, 16, 75–78. [Google Scholar] [CrossRef] [PubMed]
- Ross-Macdonald, P.; de Silva, H.; Guo, Q.; Xiao, H.; Hung, C.Y.; Penhallow, B.; Markwalder, J.; He, L.; Attar, R.M.; Lin, T.A.; et al. Identification of a nonkinase target mediating cytotoxicity of novel kinase inhibitors. Mol. Cancer Ther. 2008, 7, 3490–3498. [Google Scholar] [CrossRef] [PubMed]
- Mannherz, H.G.; Gonsior, S.M.; Gremm, D.; Wu, X.; Pope, B.J.; Weeds, A.G. Activated cofilin colocalises with Arp2/3 complex in apoptotic blebs during programmed cell death. Eur. J. Cell Biol. 2005, 84, 503–515. [Google Scholar] [CrossRef] [PubMed]
- Pröschel, C.; Blouin, M.J.; Gutowski, N.J.; Ludwig, R.; Noble, M. Limk1 is predominantly expressed in neural tissues and phosphorylates serine, threonine and tyrosine residues in vitro. Oncogene 1995, 11, 1271–1281. [Google Scholar] [PubMed]
- Aizawa, H.; Wakatsuki, S.; Ishii, A.; Sasaki, K.Y.; Ohashi, K.; Sekine-Aizawa, Y.; Sehara-Fujisawa, A.; Mizuno, K.; Goshima, Y.; Yahara, I. Phosphorylation of cofilin by LIM-kinase is necessary for semaphorin 3A-induced growth cone collapse. Nat. Neurosci. 2001, 4, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Lai, F.P.L.; Szczodrak, M.; Block, J.; Faix, J.; Breitsprecher, D.; Mannherz, H.G.; Stradal, E.E.B.; Dunn, G.A.; Small, J.V.; Rottner, K. Arp2/3 complex interactions and actin netword turnover in lamellipodia. EMBO J. 2008, 27, 982–992. [Google Scholar] [CrossRef] [PubMed]
- Wen, Z.; Han, L.; Bamburg, J.R.; Shim, S.; Ming, G.L.; Zheng, J.Q. BMP gradients steer nerve growth cones by a balancing act of LIM kinase and Slingshot phosphatase on ADF/cofilin. J. Cell Biol. 2007, 178, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Bernstein, B.W.; Bamburg, J.R. Regulating actin-filament dynamics in vivo. Trends Biochem. Sci. 2000, 25, 19–23. [Google Scholar] [CrossRef]
- Argiro, V.; Bunge, M.B.; Johnson, M.I. Correlation between growth form and movement and their dependence on neuronal age. J. Neurosci. 1984, 4, 3051–3062. [Google Scholar] [PubMed]
- Mongiu, A.K.; Weitzke, E.L.; Chaga, O.Y.; Borisy, G.G. Kinetic structural analysis of neuronal growth cone veil motility. J. Cell Sci. 2007, 120, 1113–1125. [Google Scholar] [CrossRef] [PubMed]
- DeMali, K.A.; Barlow, C.A.; Burridge, K. Recruitment of the Arp2/3 complex to vinculin: Coupling membrane protrusion to matrix adhesion. J. Cell Biol. 2002, 159, 881–891. [Google Scholar] [CrossRef] [PubMed]
- LeClaire, L.L.; Rana, M.; Baumgartner, M.; Barber, D.L. The Nck interacting kinase NIK increases Arp2/3 complex activity by phosphorylating the Arp2 subunit. J. Cell Biol. 2015, 208, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Mashiach-Farkash, E.; Rak, R.; Elad-Sfadia, G.; Haklai, R.; Carmeli, S.; Kloog, Y.; Wolfson, H.J. Computer-based identification of a novel LIMK1/2 inhibitor that synergizes with salirasib to destabilize the actin cytoskeleton. Oncotarget 2012, 3, 629–639. [Google Scholar] [PubMed]
- Pfannes, E.K.; Theves, M.; Wegner, C.; Beta, C. Impact of the carbazole derivative wiskostatin on mechanical stability and dynamics of motile cells. J. Muscle Res. Cell Motil. 2012, 33, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Rottner, K.; Kaverina, I.N.; Stradal, T.E.B. Cytoskeleton proteins. In Cell Biology, A Laboratory Handbook; Celis, J.E., Ed.; Elsevier Academic Press: London, UK, 2006; Volume 3, pp. 111–119. [Google Scholar]



© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schlau, M.; Terheyden-Keighley, D.; Theis, V.; Mannherz, H.G.; Theiss, C. VEGF Triggers the Activation of Cofilin and the Arp2/3 Complex within the Growth Cone. Int. J. Mol. Sci. 2018, 19, 384. https://doi.org/10.3390/ijms19020384
Schlau M, Terheyden-Keighley D, Theis V, Mannherz HG, Theiss C. VEGF Triggers the Activation of Cofilin and the Arp2/3 Complex within the Growth Cone. International Journal of Molecular Sciences. 2018; 19(2):384. https://doi.org/10.3390/ijms19020384
Chicago/Turabian StyleSchlau, Matthias, Daniel Terheyden-Keighley, Verena Theis, Hans Georg Mannherz, and Carsten Theiss. 2018. "VEGF Triggers the Activation of Cofilin and the Arp2/3 Complex within the Growth Cone" International Journal of Molecular Sciences 19, no. 2: 384. https://doi.org/10.3390/ijms19020384






