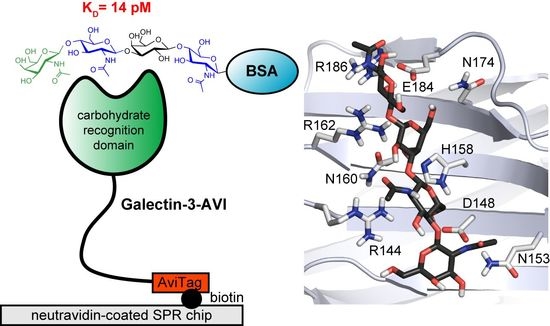
Poly-N-Acetyllactosamine Neo-Glycoproteins as Nanomolar Ligands of Human Galectin-3: Binding Kinetics and Modeling
Abstract
:1. Introduction
2. Results
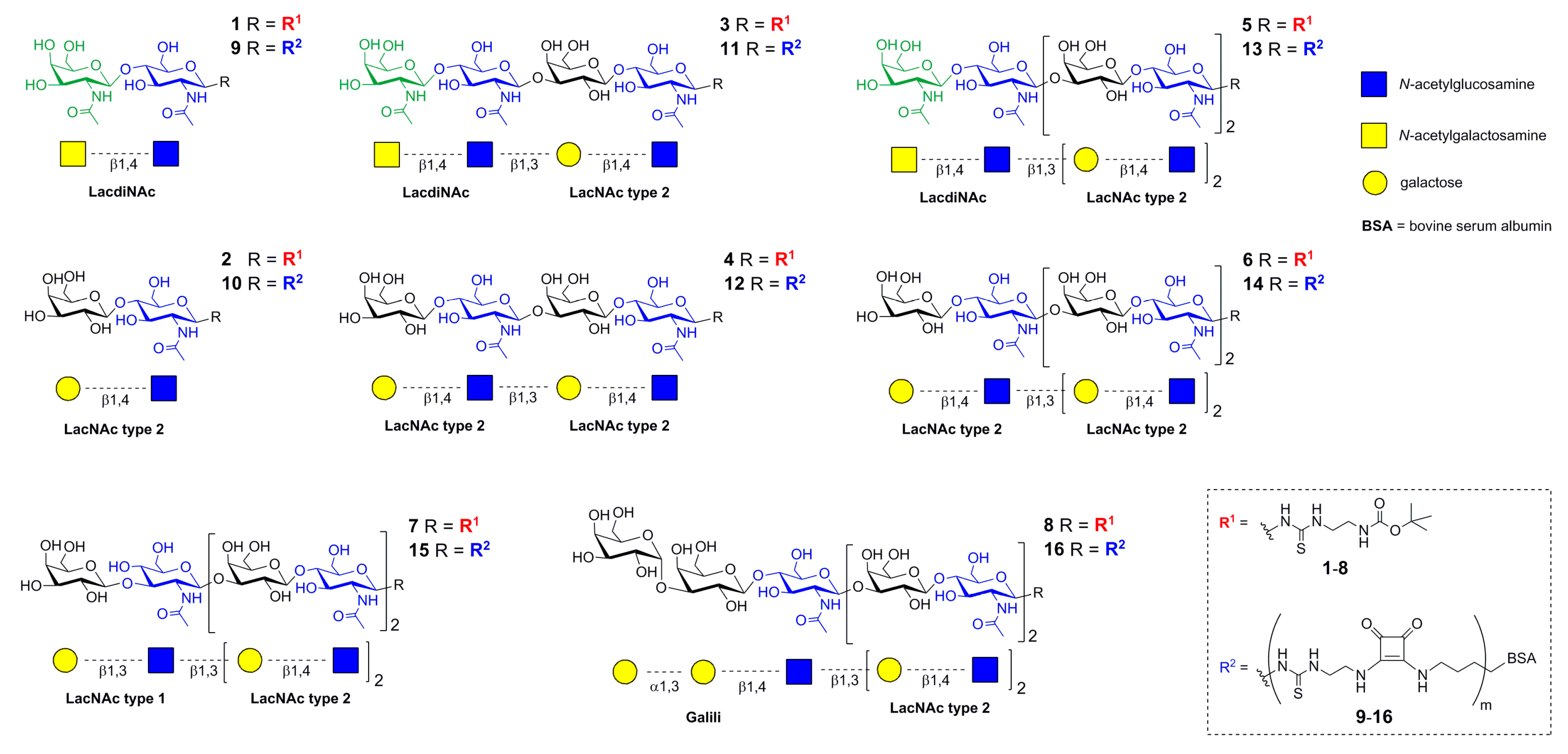
2.1. Preparation of functionalized Poly-LacNAc glycans 1–8 and Neo-Glycoproteins 9–16
2.2. Binding Properties of Glycans 1–8 and Neo-Glycoproteins 9–16 to Gal-3 in ELISA Assay
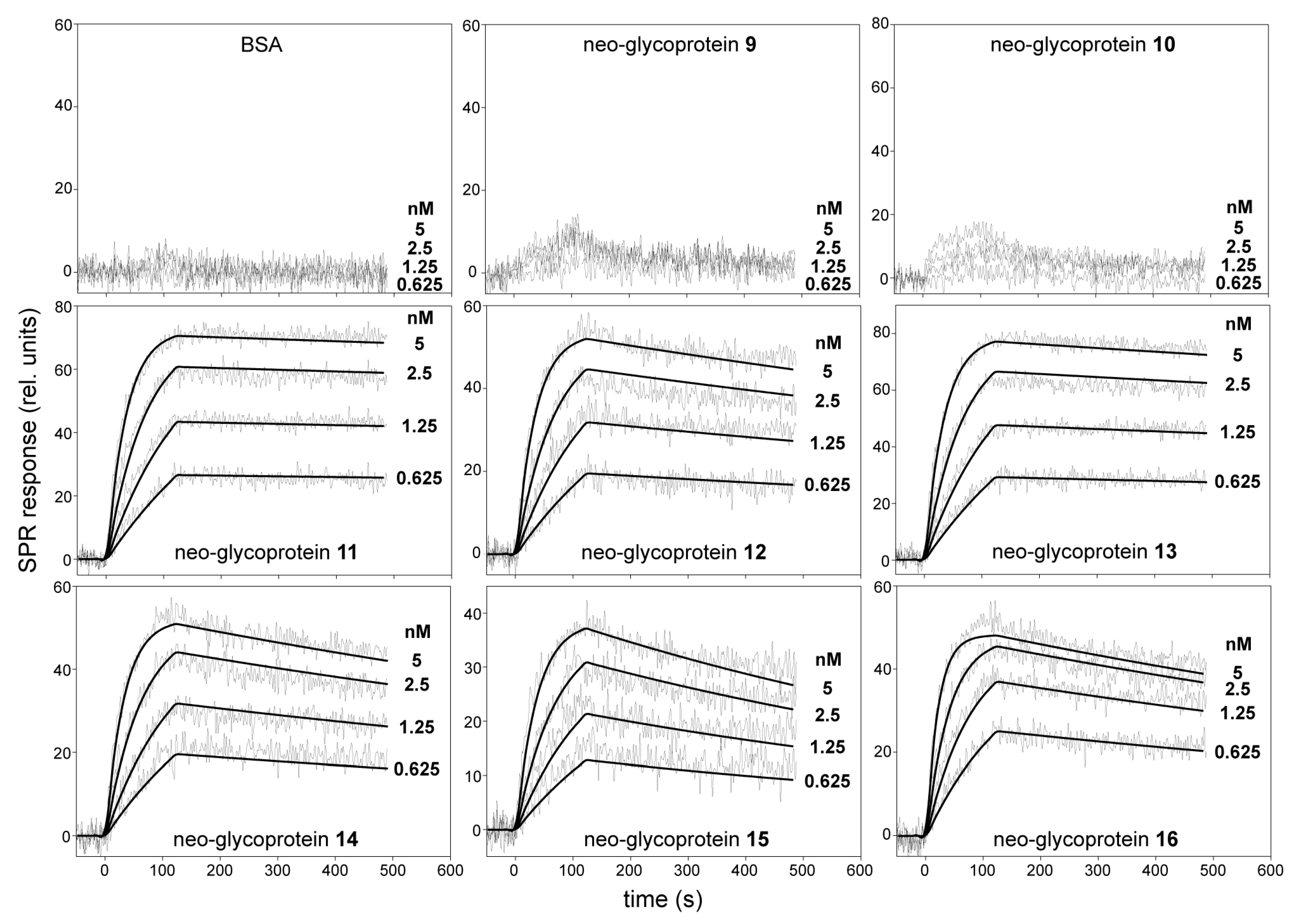
2.3. Binding Kinetics of Neo-Glycoproteins 9–16 with Gal-3 Determined by Surface Plasmon Resonance
2.4. Molecular Dynamics of Disaccharide (1,2) and Tetrasaccharide (3,4) Glycans in Gal-3 CRD
3. Discussion
4. Materials and Methods
4.1. Synthesis of Glycans 1–8 and Neo-Glycoproteins 9–16
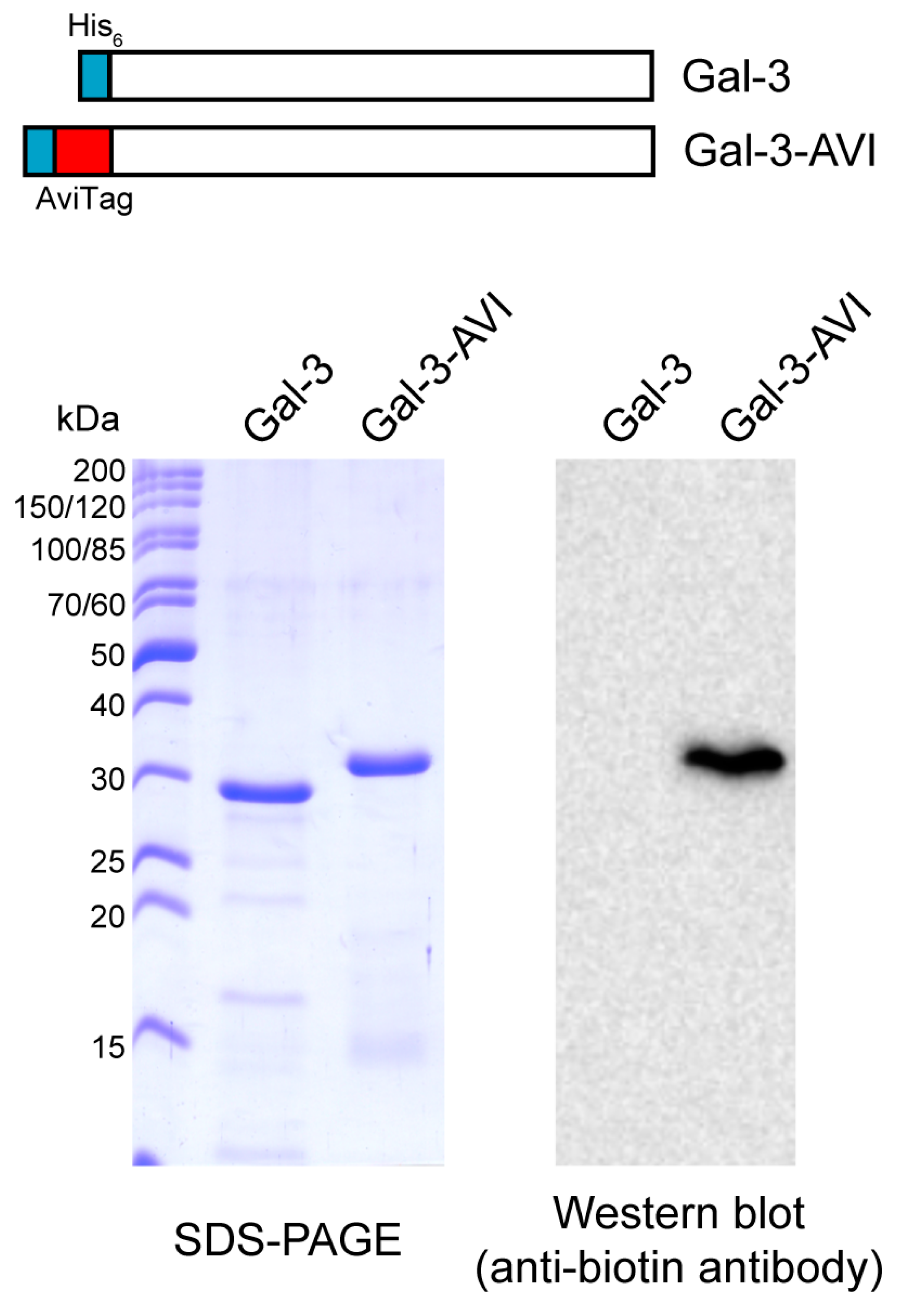
4.2. Protein Production
4.3. ELISA Assays with Gal-3
4.4. Surface Plasmon Resonance Measurements
4.5. Molecular Dynamics Simulations
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ASF | Asialofetuin |
| BSA | Bovine serum albumin |
| CRD | Carbohydrate recognition domain |
| ELISA | Enzyme-linked immunosorbent assay |
| ESI-MS | Electrospray ionization mass spectrometry |
| Gal-3 | Galectin-3 |
| Gal-3-AVI | Galectin-3 containing AviTag peptide sequence |
| HPLC | High-performance liquid chromatography |
| IC50 | Half maximal inhibitory concentration |
| LacNAc | N-Acetyllactosamine (β-d-Gal-(1→4)-d-GlcNAc) |
| LacdiNAc | N,N′-Diacetyllactosamine (β-d-GalNAc-(1→4)-d-GlcNAc) |
| NMR | Nuclear magnetic resonance |
| SDS-PAGE | Sodium dodecyl sulfate polyacrylamide gel electrophoresis |
| SPR | Surface plasmon resonance |
| TMB | 3,3′,5,5′-tetramethylbenzidine |
References
- Hirabayashi, J.; Kasai, K. The family of metazoan metal-independent β-galactoside-binding lectins: Structure, function and molecular evolution. Glycobiology 1993, 3, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.T.; Rabinovich, G.A. Galectins as modulators of tumour progression. Nat. Rev. Cancer 2005, 5, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Iurisci, I.; Cumashi, A.; Sherman, A.A.; Tsvetkov, Y.E.; Tinari, N.; Piccolo, E. Synthetic inhibitors of galectin-1 and -3 selectively modulate homotypic cell aggregation and tumor cell apoptosis. Anticancer Res. 2009, 29, 403–410. [Google Scholar] [PubMed]
- Ebrahim, A.H.; Alalawi, Z.; Mirandola, L.; Rakhshanda, R.; Dahlbeck, S.; Nguyen, D.; Jenkins, M.; Grizzi, F.; Cobos, E.; Figueroa, J.A.; et al. Galectins in cancer: Carcinogenesis, diagnosis and therapy. Ann. Transl. Med. 2014, 2, 88. [Google Scholar] [CrossRef] [PubMed]
- Tellez-Sanz, R.; Garcia-Fuentes, L.; Vargas-Berenguel, A. Human galectin-3 selective and high affinity inhibitors. Present state and future perspectives. Curr. Med. Chem. 2013, 20, 2979–2990. [Google Scholar] [CrossRef] [PubMed]
- Šimonová, A.; Kupper, C.E.; Böcker, S.; Müller, A.; Hofbauerová, K.; Pelantová, H.; Elling, L.; Křen, V.; Bojarová, P. Chemo-enzymatic synthesis of LacdiNAc dimers of varying length as novel galectin ligands. J. Mol. Catal. B Enzym. 2014, 101, 47–55. [Google Scholar] [CrossRef]
- Laaf, D.; Bojarová, P.; Mikulová, B.; Pelantová, H.; Křen, V.; Elling, L. Two-step enzymatic synthesis of β-d-N-acetylgalactosamine-(1→4)-d-N-acetylglucosamine (LacdiNAc) chitooligomers for deciphering galectin binding behavior. Adv. Synth. Catal. 2017, 359, 2101–2108. [Google Scholar] [CrossRef]
- Breloy, I.; Söte, S.; Ottis, P.; Bonar, D.; Grahn, A.; Hanisch, F.-G. O-Linked LacdiNAc-modified glycans in extracellular matrix glycoproteins are specifically phosphorylated at the subterminal GlcNAc. J. Biol. Chem. 2012, 287, 18275–18286. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.; Kenny, D.T.; Skoog, E.C.; Padra, M.; Adamczyk, B.; Vitizeva, V.; Thorell, A.; Venkatakrishnan, V.; Lindén, S.K.; Karlsson, N.G. Structural diversity of human gastric mucin glycans. Mol. Cell. Proteom. 2017, 16, 743–758. [Google Scholar] [CrossRef] [PubMed]
- Hirano, K.; Matsuda, A.; Shirai, T.; Furukawa, K. Expression of LacdiNAc groups on N-glycans among human tumors is complex. Biomed. Res. Int. 2014, 2014, 981627. [Google Scholar] [CrossRef] [PubMed]
- Haji-Ghassemi, O.; Gilbert, M.; Spence, J.; Schur, M.J.; Parker, M.J.; Jenkins, M.L.; Burke, J.E.; van Faassen, H.; Young, N.M.; Evans, S.V. Molecular basis for recognition of the cancer glycobiomarker, LacdiNAc (GalNAc[β1→4]GlcNAc), by Wisteria floribunda agglutinin. J. Biol. Chem. 2016, 291, 24085–24095. [Google Scholar] [CrossRef] [PubMed]
- Anugraham, M.; Jacob, F.; Everest-Dass, A.V.; Schoetzau, A.; Nixdorf, S.; Hacker, N.; Fink, D.; Heinzelmann-Schwarz, V.; Packer, N.H. Tissue glycomics distinguish tumour sites in women with advanced serous adenocarcinoma. Mol. Oncol. 2017, 11, 1595–1615. [Google Scholar] [CrossRef] [PubMed]
- Wuhrer, M.; Koeleman, C.A.M.; Deelder, A.M.; Hokke, C.H. Repeats of LacdiNAc and fucosylated LacdiNAc on N-glycans of the human parasite Schistosoma mansoni. FEBS J. 2006, 273, 347–361. [Google Scholar] [CrossRef] [PubMed]
- Hanzawa, K.; Suzuki, N.; Natsuka, S. Structures and developmental alterations of N-glycans of zebrafish embryos. Glycobiology 2017, 27, 228–245. [Google Scholar] [CrossRef] [PubMed]
- Böcker, S.; Laaf, D.; Elling, L. Galectin binding to neo-glycoproteins: LacDiNAc conjugated BSA as ligand for human galectin-3. Biomolecules 2015, 5, 1671–1696. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, H.; AlSadek, D.M. Galectin-3 as a potential target to prevent cancer metastasis. Clin. Med. Insights Oncol. 2015, 25, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Collins, P.M.; Bum-Erdene, K.; Yu, X.; Blanchard, H. Galectin-3 interactions with glycosphingolipids. J. Mol. Biol. 2014, 426, 1439–1451. [Google Scholar] [CrossRef] [PubMed]
- Leffler, H.; Carlsson, S.; Hedlund, M.; Qian, Y.; Poirier, F. Introduction to galectins. Glycoconj. J. 2004, 19, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, T.-J.; Lin, H.-Y.; Tu, Z.; Lin, T.-C.; Wu, S.-C.; Tseng, Y.-Y.; Liu, F.-T.; Hsu, S.-T.D.; Lin, C.-H. Dual thio-digalactoside-binding modes of human galectins as the structural basis for the design of potent and selective inhibitors. Sci. Rep. 2016, 6, 29457. [Google Scholar] [CrossRef] [PubMed]
- Rapoport, E.M.; Kurmyshkina, O.V.; Bovin, N.V. Mammalian galectins: Structure, carbohydrate specificity, and functions. Biochemistry 2008, 73, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Morris, S.; Ahmad, N.; André, S.; Kaltner, H.; Gabius, H.-J.; Brenowitz, M.; Brewer, F. Quaternary solution structures of galectins-1, -3, and -7. Glycobiology 2004, 14, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Nieminen, J.; Kuno, A.; Hirabayashi, J.; Sato, S. Visualization of galectin-3 oligomerization on the surface of neutrophils and endothelial cells using fluorescence resonance energy transfer. J. Biol. Chem. 2007, 282, 1374–1383. [Google Scholar] [CrossRef] [PubMed]
- Massa, S.M.; Cooper, D.N.W.; Leffler, H.; Barondes, S.H. L-29, an endogenous lectin, binds to glycoconjugate ligands with positive cooperativity. Biochemistry 1993, 32, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Lepur, A.; Salomonsson, E.; Nilsson, U.J.; Leffler, H. Ligand induced galectin-3 protein self-association. J. Biol. Chem. 2012, 287, 21751–21756. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, N.; Gabius, H.J.; André, S.; Kaltner, H.; Sabesan, S.; Roy, R.; Liu, B.; Macaluso, F.; Brewer, C.F. Galectin-3 precipitates as a pentamer with synthetic multivalent carbohydrates and forms heterogeneous cross-linked complexes. J. Biol. Chem. 2004, 279, 10841–10847. [Google Scholar] [CrossRef] [PubMed]
- Halimi, H.; Rigato, A.; Byrne, D.; Ferracci, G.; Sebban-Kreuzer, C.; ElAntak, L.; Guerlesquin, F. Glycan dependence of galectin-3 self-association properties. PLoS ONE 2014, 9, e111836. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Laaf, D.; Bojarová, P.; Pelantová, H.; Křen, V.; Elling, L. Tailored multivalent neo-glycoproteins: Synthesis, evaluation, and application of a library of galectin-3-binding glycan ligands. Bioconjug. Chem. 2017, 28, 2832–2840. [Google Scholar] [CrossRef] [PubMed]
- Lundquist, J.J.; Toone, E.J. The cluster glycoside effect. Chem. Rev. 2002, 102, 555–578. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Huang, W.; Orwenyo, J.; Banerjee, A.; Vasta, G.R.; Wang, L.-X. Design and synthesis of glycoprotein-based multivalent glyco-ligands for influenza hemagglutinin and human galectin-3. Bioorg. Med. Chem. 2013, 21, 2037–2044. [Google Scholar] [CrossRef] [PubMed]
- Böcker, S.; Elling, L. Biotinylated N-acetyllactosamine- and N,N-diacetyllactosamine-based oligosaccharides as novel ligands for human galectin-3. Bioengineering 2017, 4, 31. [Google Scholar] [CrossRef] [PubMed]
- Dam, T.K.; Gabius, H.J.; André, S.; Kaltner, H.; Lensch, M.; Brewer, C.F. Galectins bind to the multivalent glycoprotein asialofetuin with enhanced affinities and a gradient of decreasing binding constants. Biochemistry 2005, 44, 12564–12571. [Google Scholar] [CrossRef] [PubMed]
- Abraham, M.J.; Murtola, T.; Schulz, R.; Páll, S.; Smith, J.C.; Hess, B.; Lindahl, E. GROMACS: High performance molecular simulations through multi-level parallelism from laptops to supercomputers. SoftwareX 2015, 1, 19–25. [Google Scholar] [CrossRef]
- Lindorff-Larsen, K.; Piana, S.; Palmo, K.; Maragakis, P.; Klepeis, J.L.; O’Dror, R.; Shaw, D.E. Improved side-chain torsion potentials for the Amber ff99SB protein force field. Proteins 2010, 78, 1950–1958. [Google Scholar] [CrossRef] [PubMed]
- Kirschner, K.N.; Yongye, A.B.; Tschampel, S.M.; Daniels, C.R.; Foley, B.L.; Woods, R.J. GLYCAM06: A generalizable Biomolecular force field. Carbohydrates. J. Comput. Chem. 2008, 29, 622–655. [Google Scholar] [CrossRef] [PubMed]
- Sousa da Silva, A.W.; Vranken, W.F. ACPYPE—AnteChamber PYthon Parser interfacE. BMC Res. Notes 2012, 5, 367. [Google Scholar] [CrossRef] [PubMed]
- Korb, O.; Stützle, T.; Exner, T.E. Empirical scoring functions for advanced protein-ligand docking with PLANTS. J. Chem. Inf. Model. 2009, 49, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Saraboji, K.; Hakansson, M.; Genheden, S.; Diehl, C.; Qvist, J.; Weininger, U.; Nilsson, U.J.; Leffler, H.; Ryde, U.; Akke, M.; et al. The carbohydrate-binding site in galectin-3 is pre-organized to recognize a sugar-like framework of oxygens: Ultra-high resolution structures and water dynamics. Biochemistry 2012, 51, 296–306. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, H.; Yu, X.; Collins, P.M.; Bum-Erdene, K. Galectin-3 inhibitors: A patent review (2008–present). Expert Opin. Ther. Pat. 2014, 24, 1053–1065. [Google Scholar] [CrossRef] [PubMed]
- Öberg, C.T.; Leffler, H.; Nilsson, U.J. Inhibition of galectins with small molecules. CHIMIA Int. J. Chem. 2011, 65, 18–23. [Google Scholar] [CrossRef]
- Laaf, D.; Steffens, H.; Pelantová, H.; Bojarová, P.; Křen, V.; Elling, L. Chemo-enzymatic synthesis of branched N-acetyllactosamine glycan oligomers for galectin-3 inhibition. Adv. Synth. Catal. 2017, 359, 4015–4024. [Google Scholar] [CrossRef]
- Kamili, N.A.; Arthur, C.M.; Gerner-Smidt, C.; Tafesse, E.; Blenda, A.; Dias-Baruffi, M.; Stowell, S.R. Key regulators of galectin–glycan interactions. Proteomics 2016, 16, 3111–3125. [Google Scholar] [CrossRef] [PubMed]
- Stowell, S.R.; Arthur, C.M.; Mehta, P.; Slanina, K.A.; Blixt, O.; Leffler, H.; Smith, D.F.; Cummings, R.D. Galectin-1, -2, and -3 exhibit differential recognition of sialylated glycans and blood group antigens. J. Biol. Chem. 2008, 283, 10109–10123. [Google Scholar] [CrossRef] [PubMed]
- Barboni, E.A.M.; Bawumia, S.; Colin Hughes, R. Kinetic measurements of binding of galectin 3 to a laminin substratum. Glycoconjug. J. 1999, 16, 365–373. [Google Scholar] [CrossRef]
- Jin, F.; Chammas, R.; Engel, J.; Reinhold, V. Structure and function of laminin 1 glycans; glycan profiling. Glycobiology 1995, 5, 157–158. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, K.; Sato, Y.; Murakami, T.; Tanaka, M.; Niwa, O. One-step detection of galectins on hybrid monolayer surface with protruding lactoside. Anal. Chem. 2010, 82, 1175–1178. [Google Scholar] [CrossRef] [PubMed]
- Javier Muñoz, F.; Ignacio Santos, J.; Ardá, A.; André, S.; Gabius, H.-J.; Sinisterra, J.V.; Jiménez-Barbero, J.; Hernáiz, M.J. Binding studies of adhesion/growth-regulatory galectins with glycoconjugates monitored by surface plasmon resonance and NMR spectroscopy. Org. Biomol. Chem. 2010, 8, 2986–2992. [Google Scholar] [CrossRef] [PubMed]
- Sauerzapfe, B.; Namdjou, D.J.; Schumacher, T.; Linden, N.; Křenek, K.; Křen, V.; Elling, L. Characterization of recombinant fusion constructs of human beta 1,4-galactosyltransferase 1 and the lipase pre-propeptide from Staphylococcus hyicus. J. Mol. Catal. B Enzym. 2008, 50, 128–140. [Google Scholar] [CrossRef]
- Kupper, C.E.; Rosencrantz, R.R.; Henssen, B.; Pelantová, H.; Thönes, S.; Drozdová, A.; Křen, V.; Elling, L. Chemo-enzymatic modification of poly-N-acetyllactosamine (LacNAc) oligomers and N,N-diacetyllactosamine (LacDINAc) based on galactose oxidase treatment. Beilstein J. Org. Chem. 2012, 8, 712–725. [Google Scholar] [CrossRef] [PubMed]
- Fischöder, T.; Laaf, D.; Dey, C.; Elling, L. Enzymatic synthesis of N-acetyllactosamine (LacNAc) type 1 oligomers and characterization as multivalent galectin ligands. Molecules 2017, 22, 1320. [Google Scholar] [CrossRef] [PubMed]
- Sauerzapfe, B.; Křenek, K.; Schmiedel, J.; Wakarchuk, W.W.; Pelantová, H.; Křen, V.; Elling, L. Chemo-enzymatic synthesis of poly-N-acetyllactosamine (poly-LacNAc) structures and their characterization for CGL2-galectin-mediated binding of ECM glycoproteins to biomaterial surfaces. Glycoconj. J. 2009, 26, 141–159. [Google Scholar] [CrossRef] [PubMed]
- Rech, C.; Rosencrantz, R.R.; Křenek, K.; Pelantová, H.; Bojarová, P.; Römer, C.E.; Hanisch, F.G.; Křen, V.; Elling, L. Combinatorial one-pot synthesis of poly-N-acetyllactosamine oligosaccharides with Leloir-glycosyltransferases. Adv. Synth. Catal. 2011, 353, 2492–2500. [Google Scholar] [CrossRef]
- Bojarová, P.; Chytil, P.; Mikulová, B.; Bumba, L.; Konefał, R.; Pelantová, H.; Krejzová, J.; Slámová, K.; Petrásková, L.; Kotrchová, L.; et al. Glycan-decorated HPMA copolymers as high-affinity lectin ligands. Polym. Chem. 2017, 8, 2647–2658. [Google Scholar] [CrossRef]
- Essmann, U.; Perera, L.; Berkowitz, M.L.; Darden, T.; Lee, H.; Pedersen, L.G. A smooth particle mesh Ewald method. J. Chem. Phys. 1995, 103, 8577–8593. [Google Scholar] [CrossRef]
- Bussi, G.; Donadio, D.; Parrinello, M. Canonical sampling through velocity-rescaling. J. Chem. Phys. 2007, 126, 014101. [Google Scholar] [CrossRef] [PubMed]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef] [PubMed]
- Hansson, T.; Marelius, J.; Åqvist, J. Ligand binding affinity prediction by linear interaction energy methods. J. Comput. Aided Mol. Des. 1998, 12, 27–35. [Google Scholar] [CrossRef] [PubMed]


| Neo-Glycoprotein (Respective Glycan) a | Mw (kDa) | m b | IC50 Glycan (µM) c | IC50 Neo-Glycoprotein (nM) | rp d | rp/me | Kd Neo-Glycoprotein (nM) |
|---|---|---|---|---|---|---|---|
| 9 (glycan 1) | 76.4 | 17 | 42 ± 2 | 2026 ± 147 | 20.7 | 1.2 | 6290 ± 530 |
| 10 (glycan 2) | 78.2 | 18 | 36 ± 1 | 344 ± 38 | 104.7 | 5.8 | 4780 ± 1240 |
| 11 (glycan 3) | 87.4 | 21 | 7 ± 1 f | 11 ± 2 | 642.9 | 30.6 | 30 ± 4 f |
| 12 (glycan 4) | 84.7 | 19 | 13 ± 3 f | 31 ± 1 | 419.4 | 22.1 | 300 ± 60 f |
| 13 (glycan 5) | 86.6 | 16 | 6.20 ± 0.02 g | 37 ± 7 g | 169.9 | 10.6 | 76 ± 19 g |
| 14 (glycan 6) | 86.6 | 17 | 20.1 ± 0.1 g | 76 ± 8 g | 263.4 | 15.5 | 350 ± 110 g |
| 15 (glycan 7) | 85.9 | 17 | 12.5 ± 0.1 g | 212 ± 22 g | 58.9 | 3.5 | 700 ± 100 g |
| 16 (glycan 8) | 89.6 | 18 | 8.4 ± 0.1 g | 65 ± 4 g | 128.2 | 7.1 | 290 ± 90 g |
| Compound | Attached Glycan a | ka (M−1·s−1) | kd (s−1) | KD (M) |
|---|---|---|---|---|
| 9 | LacdiNAc | N.D. | N.D. | N.D. |
| 10 | LacNAc | N.D. | N.D. | N.D. |
| 11 | LacdiNAc-LacNAc | (5.9 ± 1.3) × 106 | (8.5 ± 2.5) × 10−5 | (1.4 ± 0.4) × 10−11 |
| 12 | LacNAc-LacNAc | (6.2 ± 2.1) × 106 | (4.2 ± 3.1) × 10−4 | (6.8 ± 3.9) × 10−11 |
| 13 | LacdiNAc-LacNAc-LacNAc | (5.8 ± 2.2) × 106 | (1.5 ± 0.4) × 10−4 | (2.6 ± 0.9) × 10−11 |
| 14 | LacNAc-LacNAc-LacNAc | (7.8 ± 2.9) × 106 | (5.1 ± 2.8) × 10−4 | (6.5 ± 2.6) × 10−11 |
| 15 | LacNAc type 1-LacNAc-LacNAc | (4.9 ± 1.8) × 106 | (1.3 ± 0.3) × 10−3 | (2.7 ± 1.0) × 10−10 |
| 16 | Galili-LacNAc-LacNAc | (9.3 ± 3.1) × 106 | (6.3 ± 2.0) × 10−4 | (6.8 ± 3.5) × 10−11 |
| ASF | Positive control standard | (4.8 ± 1.9) × 104 | (4.0 ± 1.7) × 10−4 | (8.3 ± 3.1) × 10−9 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bumba, L.; Laaf, D.; Spiwok, V.; Elling, L.; Křen, V.; Bojarová, P. Poly-N-Acetyllactosamine Neo-Glycoproteins as Nanomolar Ligands of Human Galectin-3: Binding Kinetics and Modeling. Int. J. Mol. Sci. 2018, 19, 372. https://doi.org/10.3390/ijms19020372
Bumba L, Laaf D, Spiwok V, Elling L, Křen V, Bojarová P. Poly-N-Acetyllactosamine Neo-Glycoproteins as Nanomolar Ligands of Human Galectin-3: Binding Kinetics and Modeling. International Journal of Molecular Sciences. 2018; 19(2):372. https://doi.org/10.3390/ijms19020372
Chicago/Turabian StyleBumba, Ladislav, Dominic Laaf, Vojtěch Spiwok, Lothar Elling, Vladimír Křen, and Pavla Bojarová. 2018. "Poly-N-Acetyllactosamine Neo-Glycoproteins as Nanomolar Ligands of Human Galectin-3: Binding Kinetics and Modeling" International Journal of Molecular Sciences 19, no. 2: 372. https://doi.org/10.3390/ijms19020372