Cyclin B3 Deficiency Impairs Germline Stem Cell Maintenance and Its Overexpression Delays Cystoblast Differentiation in Drosophila Ovary
Abstract
:1. Introduction
2. Results
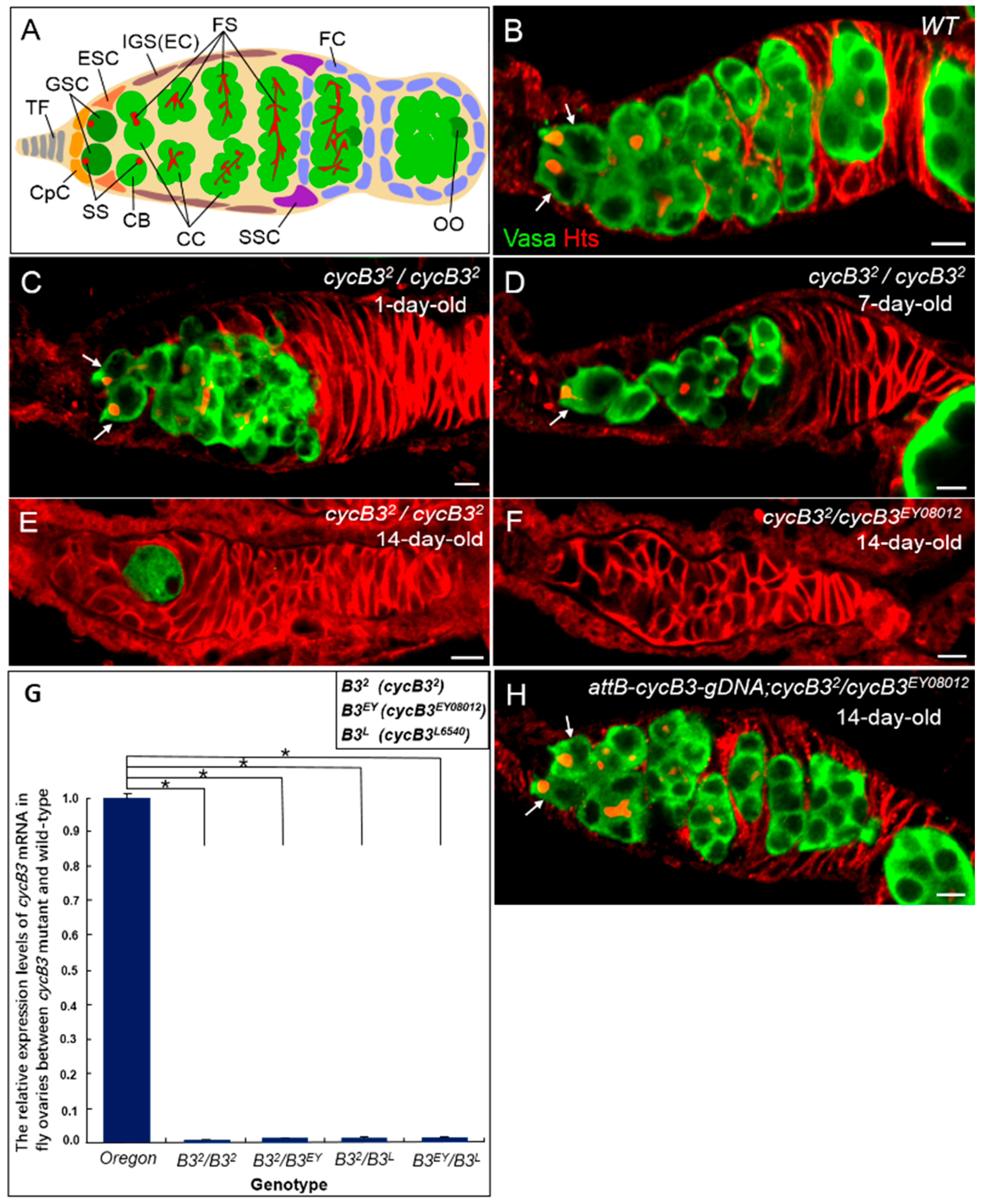
2.1. Deficiency of cycB3 Impairs GSCs Maintenance in Drosophila Ovary
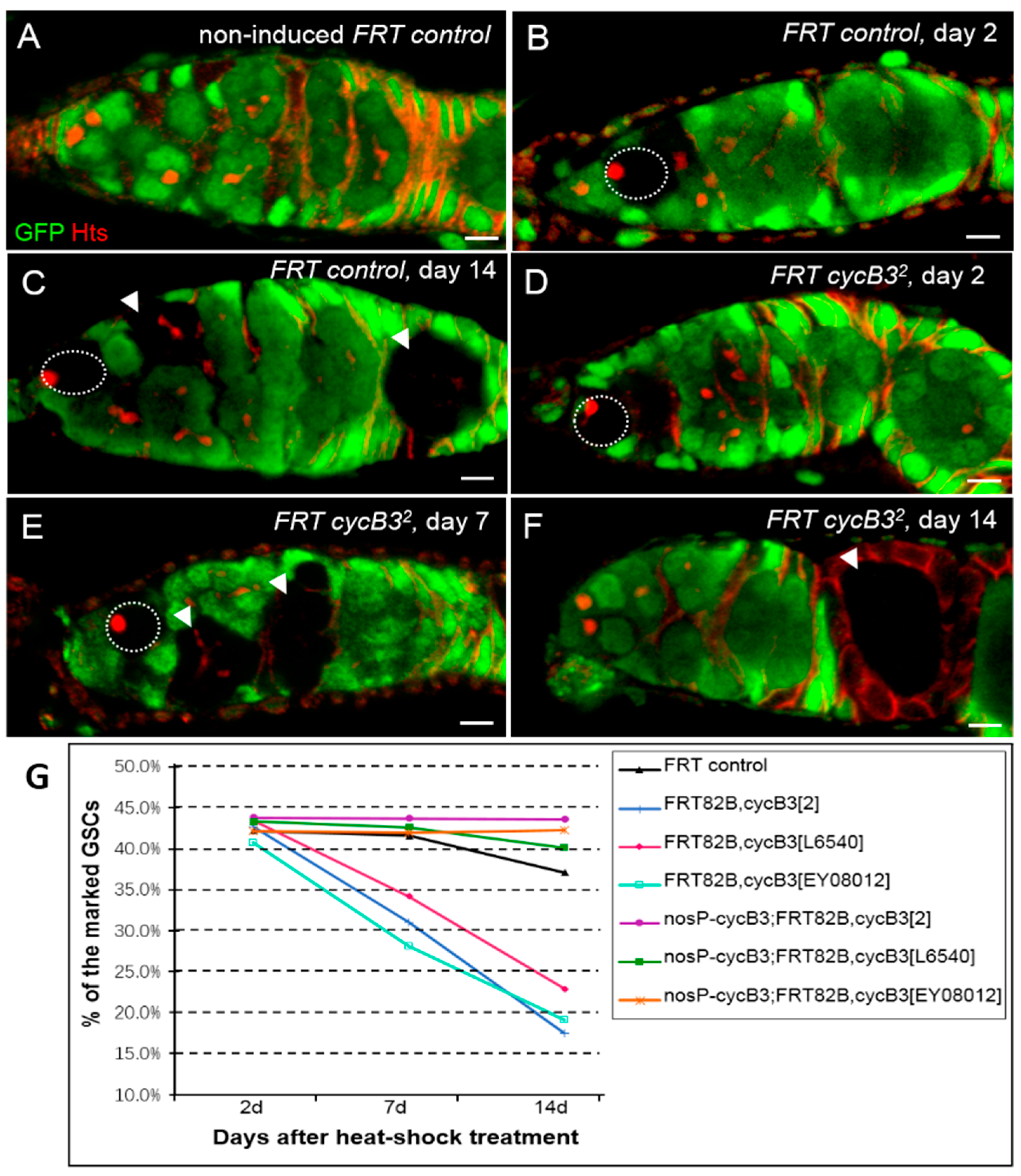
2.2. The Gene cycB3 Functions as an Intrinsic Factor in Controlling GSC Maintenance
2.3. cycB3 Does Not Influence Bmp/dpp-Mediated Dad Expression
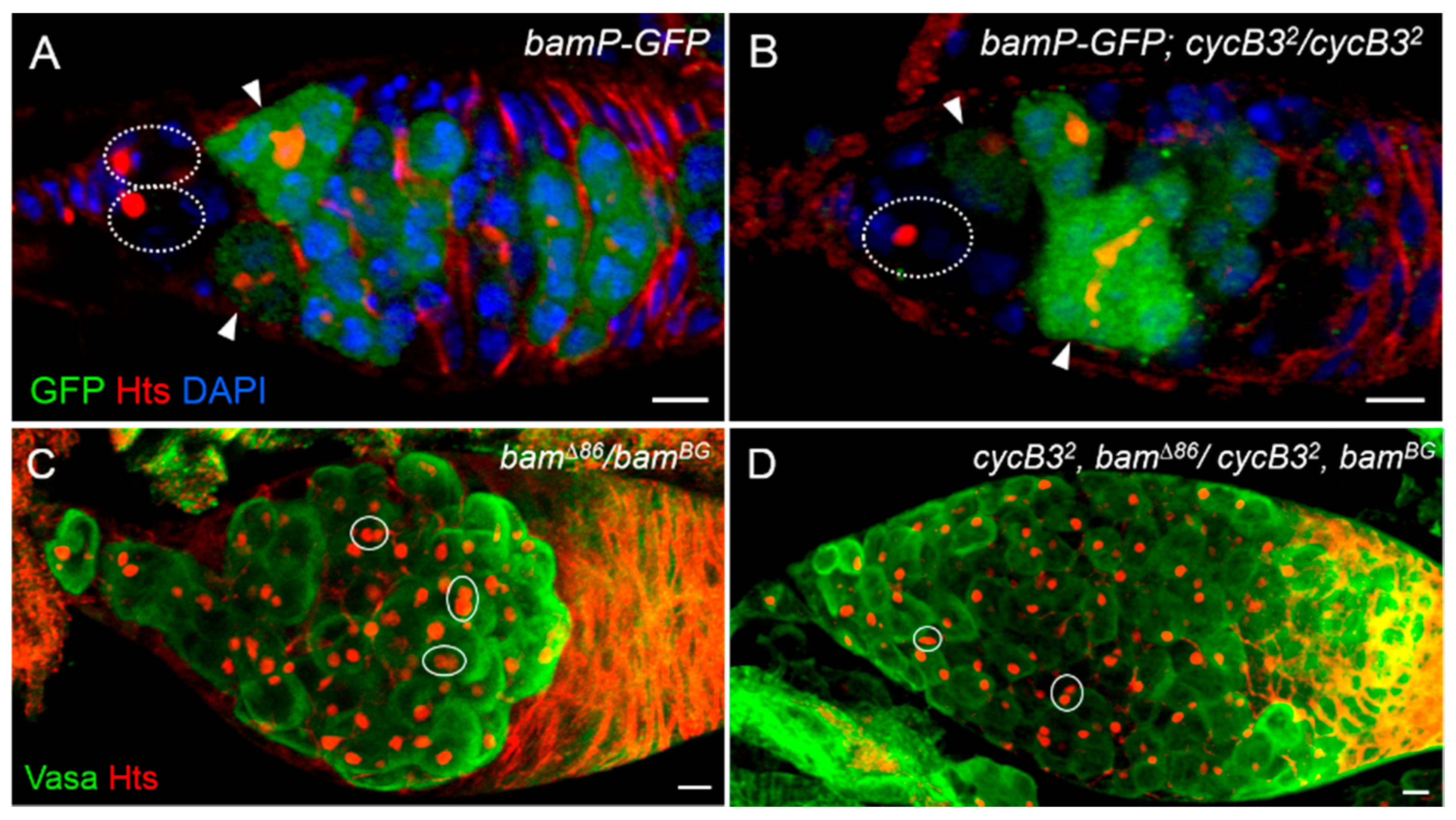
2.4. cycB3 Is Not Required for Bam Transcriptional Silencing and Acts Probably in a Bam-Dependent Manner, in Ovarian GSCs of Drosophila
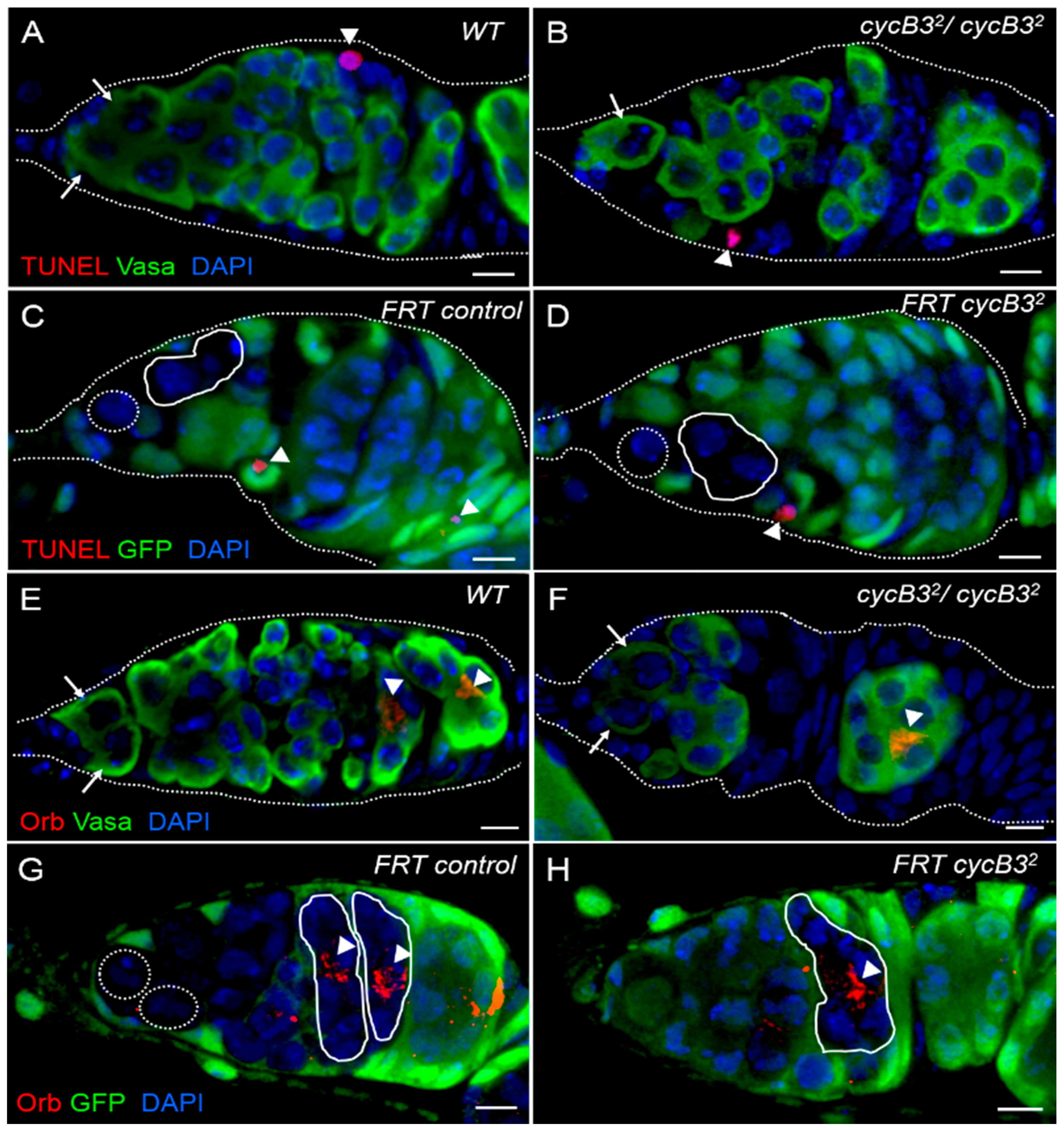
2.5. Deficiency of cycB3 Fails to Cause Apoptosis in Ovarian GSCs, Nor Influences CB Differentiation into Oocytes in Drosophila Ovary
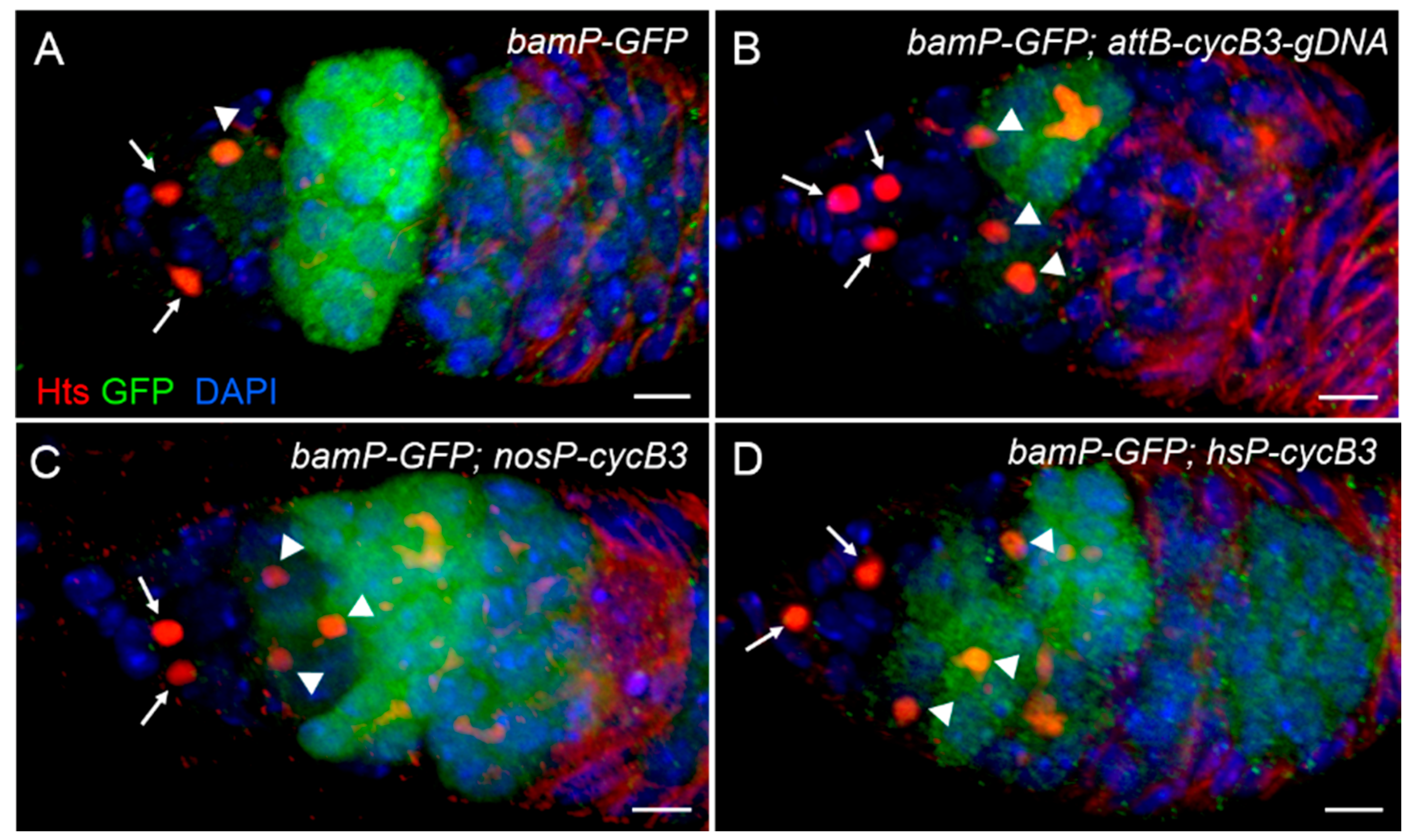
2.6. Over-Expression of cycB3 Fails to Increases the Number of GSCs, but Dramatically Enhances the Number of CBs
3. Discussion
4. Materials and Methods
4.1. Constructs
4.2. Drosophila Stocks
4.3. Immunohistochemistry and Microscopy for Drosophila Ovary
4.4. Quantitative Real-Time PCR (qPCR)
4.5. TUNEL Apoptotic Cell Detection
4.6. Generating and Analyzing Marked Germline Clones
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| cycB3 | cyclin B3 |
| GSC | germline stem cell |
| GFP | gfp fluorescent protein |
| CB | Cystoblast |
| Df | Deficiency strain of cycB3 gene |
| qPCR | quantitative real-time PCR |
| nosP-gvp | nanosP-gal4:vp16 |
| AHST | after heat-shock treatment |
| UASp | pattB-UAS |
| hsP | pattB-hsP |
| nosP | pattB-nosP |
| Dad | Daughters agaist Dpp |
| TUNEL | Terminal deoxynucleotidyl transferase(TdT)-mediated dUTP Nick End Labeling |
| WT | wild-type |
References
- Lin, H. The stem-cell niche theory: Lessons from flies. Nat. Rev. Genet. 2002, 3, 931–940. [Google Scholar] [CrossRef] [PubMed]
- Spradling, A.; Drummond-Barbosa, D.; Kai, T. Stem cells find their niche. Nature 2001, 414, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Decotto, E.; Spradling, A.C. The Drosophila ovarian and testis stem cell niches: Similar somatic stem cells and signals. Dev. Cell 2005, 9, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Kirilly, D.; Xie, T. The Drosophila ovary: An active stem cell community. Cell Res. 2007, 17, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Wong, M.D.; Kawase, E.; Xi, R.; Ding, B.C.; McCarthy, J.J.; Xie, T. Bmp signals from niche cells directly repress transcription of a differentiation-promoting gene, bag of marbles, in germline stem cells in the Drosophila ovary. Development 2004, 131, 1353–1364. [Google Scholar] [CrossRef] [PubMed]
- Xie, T.; Spradling, A.C. Decapentaplegic is essential for the maintenance and division of germline stem cells in the Drosophila ovary. Cell 1998, 94, 251–260. [Google Scholar] [CrossRef]
- Xia, L.; Jia, S.; Huang, S.; Wang, H.; Zhu, Y.; Mu, Y.; Kan, L.; Zheng, W.; Wu, D.; Li, X.; et al. The Fused/Smurf complex controls the fate of Drosophila germline stem cells by generating a gradient BMP response. Cell 2010, 143, 978–990. [Google Scholar] [CrossRef] [PubMed]
- Xia, L.; Zheng, X.; Zheng, W.; Zhang, G.; Wang, H.; Tao, Y.; Chen, D. The Niche-dependent feedback loop generates a BMP activity gradient to determine the germline stem cell fate. Curr. Biol. 2012, 22, 515–521. [Google Scholar] [CrossRef] [PubMed]
- Ji, S.; Li, C.; Hu, L.; Liu, K.; Mei, J.; Luo, Y.; Tao, Y.; Xia, Z.; Sun, Q.; Chen, D. Bam-dependent deubiquitinase complex can disrupt germ-line stem cell maintenance by targeting cyclin A. Proc. Natl. Acad. Sci. USA 2017, 114, 6316–6321. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; McKearin, D.M. A discrete transcriptional silencer in the bam gene determines asymmetric division of the Drosophila germline stem cell. Development 2003, 130, 1159–1170. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; McKearin, D. Gene circuitry controlling a stem cell niche. Curr. Biol. 2005, 15, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Spradling, A.C. A novel group of pumilio mutations affects the asymmetric division of germline stem cells in the Drosophila ovary. Development 1997, 124, 2463–2476. [Google Scholar] [PubMed]
- Forbes, A.; Lehmann, R. Nanos and Pumilio have critical roles in the development and function of Drosophila germline stem cells. Development 1998, 125, 679–690. [Google Scholar] [PubMed]
- Jiang, X.; Xia, L.; Chen, D.; Yang, Y.; Huang, H.; Yang, L.; Zhao, Q.; Shen, L.; Wang, J.; Chen, D. Otefin, a nuclear membrane protein, determines the fate of germline stem cells in Drosophila via interaction with Smad complexes. Dev. Cell 2008, 14, 494–506. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Wang, Q.; Huang, H.; Xia, L.; Jiang, X.; Kan, L.; Sun, Q.; Chen, D. Effete-mediated degradation of Cyclin A is essential for the maintenance of germline stem cells in Drosophila. Development 2009, 136, 4133–4142. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Lin, H. The division of Drosophila germline stem cells and their precursors requires a specific cyclin. Curr. Biol. 2005, 15, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Ables, E.T.; Drummond-Barbosa, D. Cyclin E controls Drosophila female germline stem cell maintenance independently of its role in proliferation by modulating responsiveness to niche signals. Development 2013, 140, 530–540. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Wang, Q.; Li, W.; Mao, F.; Yue, S.; Liu, S.; Liu, X.; Xiao, S.; Xia, L. Gcn5 determines the fate of Drosophila germline stem cells through degradation of Cyclin A. FASEB J. 2017, 31, 2185–2194. [Google Scholar] [CrossRef] [PubMed]
- Swenson, K.I.; Farrell, K.M.; Ruderman, J.V. The clam embryo protein cyclin A induces entry into M phase and the resumption of meiosis in Xenopus oocytes. Cell 1986, 47, 861–870. [Google Scholar] [CrossRef]
- Evans, T.; Rosenthal, E.T.; Youngblom, J.; Distel, D.; Hunt, T. Cyclin: A protein specified by maternal mRNA in sea urchin eggs that is destroyed at each cleavage division. Cell 1983, 33, 389–396. [Google Scholar] [CrossRef]
- Hoffmann, I.; Clarke, P.R.; Marcote, M.J.; Karsenti, E.; Draetta, G. Phosphorylation and activation of human cdc25-C by cdc2-cyclin B and its involvement in the self-amplification of MPF at mitosis. EMBO J. 1993, 12, 53–63. [Google Scholar] [PubMed]
- Tassan, J.P.; Jaquenoud, M.; Leopold, P.; Schultz, S.J.; Nigg, E.A. Identification of human cyclin-dependent kinase 8, a putative protein kinase partner for cyclin C. Proc. Natl. Acad. Sci. USA 1995, 92, 8871–8875. [Google Scholar] [CrossRef] [PubMed]
- Kato, J.; Matsushime, H.; Hiebert, S.W.; Ewen, M.E.; Sherr, C.J. Direct binding of cyclin D to the retinoblastoma gene product (pRb) and pRb phosphorylation by the cyclin D-dependent kinase CDK4. Genes Dev. 1993, 7, 331–342. [Google Scholar] [PubMed]
- Koff, A.; Giordano, A.; Desai, D.; Yamashita, K.; Harper, J.W.; Elledge, S.; Nishimoto, T.; Morgan, D.O.; Franza, B.R.; Roberts, J.M. Formation and activation of a cyclin E-cdk2 complex during the G1 phase of the human cell cycle. Science 1992, 257, 1689–1694. [Google Scholar] [CrossRef] [PubMed]
- Edwards, M.C.; Wong, C.; Elledge, S.J. Human cyclin K, a novel RNA polymerase II-associated cyclin possessing both carboxy-terminal domain kinase and Cdk-activating kinase activity. Mol. Cell. Biol. 1998, 18, 4291–4300. [Google Scholar] [CrossRef] [PubMed]
- Echard, A.; O’Farrell, P.H. The degradation of two mitotic cyclins contributes to the timing of cytokinesis. Curr. Biol. 2003, 13, 373–383. [Google Scholar] [CrossRef]
- Sigrist, S.; Jacobs, H.; Stratmann, R.; Lehner, C.F. Exit from mitosis is regulated by Drosophila fizzy and the sequential destruction of cyclins A, B and B3. EMBO J. 1995, 14, 4827–4838. [Google Scholar] [PubMed]
- Knoblich, J.A.; Lehner, C.F. Synergistic action of Drosophila cyclins A and B during the G2-M transition. EMBO J. 1993, 12, 65–74. [Google Scholar] [PubMed]
- Jacobs, H.W.; Knoblich, J.A.; Lehner, C.F. Drosophila Cyclin B3 is required for female fertility and is dispensable for mitosis like Cyclin B. Genes Dev. 1998, 12, 3741–3751. [Google Scholar] [CrossRef] [PubMed]
- Leismann, O.; Lehner, C.F. Drosophila securin destruction involves a D-box and a KEN-box and promotes anaphase in parallel with Cyclin A degradation. J. Cell Sci. 2003, 116 Pt 12, 2453–2460. [Google Scholar] [CrossRef] [PubMed]
- De Luca, G.; Ferretti, R.; Bruschi, M.; Mezzaroma, E.; Caruso, M. Cyclin D3 critically regulates the balance between self-renewal and differentiation in skeletal muscle stem cells. Stem Cells 2013, 31, 2478–2491. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.A.; Kalderon, D. Cyclin E-dependent protein kinase activity regulates niche retention of Drosophila ovarian follicle stem cells. Proc. Natl. Acad. Sci. USA 2009, 106, 21701–21706. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.A.; Simon, M.C. Functional analysis of the Cdk7.cyclin H.Mat1 complex in mouse embryonic stem cells and embryos. J. Biol. Chem. 2010, 285, 15587–15598. [Google Scholar] [CrossRef] [PubMed]
- Dai, Q.; Lei, T.; Zhao, C.; Zhong, J.; Tang, Y.Z.; Chen, B.; Yang, J.; Li, C.; Wang, S.; Song, X.; et al. Cyclin K-containing kinase complexes maintain self-renewal in murine embryonic stem cells. J. Biol. Chem. 2012, 287, 25344–25352. [Google Scholar] [CrossRef] [PubMed]
- Bellen, H.J.; Levis, R.W.; Liao, G.; He, Y.; Carlson, J.W.; Tsang, G.; Evans-Holm, M.; Hiesinger, P.R.; Schulze, K.L.; Rubin, G.M.; et al. The BDGP gene disruption project: Single transposon insertions associated with 40% of Drosophila genes. Genetics 2004, 167, 761–781. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Yue, L.; Spradling, A.C. The Drosophila fusome, a germline-specific organelle, contains membrane skeletal proteins and functions in cyst formation. Development 1994, 120, 947–956. [Google Scholar] [PubMed]
- Lasko, P.F.; Ashburner, M. The product of the Drosophila gene vasa is very similar to eukaryotic initiation factor-4A. Nature 1988, 335, 611–617. [Google Scholar] [CrossRef] [PubMed]
- Bustin, S.A. Quantification of mRNA using real-time reverse transcription PCR (RT-PCR): Trends and problems. J. Mol. Endocrinol. 2002, 29, 23–39. [Google Scholar] [CrossRef] [PubMed]
- Lhocine, N.; Ribeiro, P.S.; Buchon, N.; Wepf, A.; Wilson, R.; Tenev, T.; Lemaitre, B.; Gstaiger, M.; Meier, P.; Leulier, F. PIMS modulates immune tolerance by negatively regulating Drosophila innate immune signaling. Cell Host Microbe 2008, 4, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Bischof, J.; Maeda, R.K.; Hediger, M.; Karch, F.; Basler, K. An optimized transgenesis system for Drosophila using germ-line-specific phiC31 integrases. Proc. Natl. Acad. Sci. USA 2007, 104, 3312–3317. [Google Scholar] [CrossRef] [PubMed]
- Ferrandon, D.; Jung, A.C.; Criqui, M.; Lemaitre, B.; Uttenweiler-Joseph, S.; Michaut, L.; Reichhart, J.; Hoffmann, J.A. A drosomycin-GFP reporter transgene reveals a local immune response in Drosophila that is not dependent on the Toll pathway. EMBO J. 1998, 17, 1217–1227. [Google Scholar] [CrossRef] [PubMed]
- Gallant, P.; Nigg, E.A. Identification of a novel vertebrate cyclin: Cyclin B3 shares properties with both A- and B-type cyclins. EMBO J. 1994, 13, 595–605. [Google Scholar] [PubMed]
- Rorth, P. Gal4 in the Drosophila female germline. Mech. Dev. 1998, 78, 113–118. [Google Scholar] [CrossRef]
- Brand, A.H.; Manoukian, A.S.; Perrimon, N. Ectopic expression in Drosophila. Methods Cell Biol. 1994, 44, 635–654. [Google Scholar] [PubMed]
- Sadowski, I.; Ma, J.; Triezenberg, S.; Ptashne, M. GAL4-VP16 is an unusually potent transcriptional activator. Nature 1988, 335, 563–564. [Google Scholar] [CrossRef] [PubMed]
- Van Doren, M.; Williamson, A.L.; Lehmann, R. Regulation of zygotic gene expression in Drosophila primordial germ cells. Curr. Biol. 1998, 8, 243–246. [Google Scholar] [CrossRef]
- Xu, T.; Rubin, G.M. Analysis of genetic mosaics in developing and adult Drosophila tissues. Development 1993, 117, 1223–1237. [Google Scholar] [PubMed]
- Tsuneizumi, K.; Nakayama, T.; Kamoshida, Y.; Kornberg, T.B.; Christian, J.L.; Tabata, T. Daughters against dpp modulates dpp organizing activity in Drosophila wing development. Nature 1997, 389, 627–631. [Google Scholar] [PubMed]
- Inoue, H.; Imamura, T.; Ishidou, Y.; Takase, M.; Udagawa, Y.; Oka, Y.; Tsuneizumi, K.; Tabata, T.; Miyazono, K.; Kawabata, M. Interplay of signal mediators of decapentaplegic (Dpp): Molecular characterization of mothers against dpp, Medea, and daughters against dpp. Mol. Biol. Cell. 1998, 9, 2145–2156. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Kan, L.; Chen, Y.; Zheng, X.; Li, W.; Zhang, W.; Cao, L.; Lin, X.; Ji, S.; Huang, S.; et al. Ci antagonizes Hippo signaling in the somatic cells of the ovary to drive germline stem cell differentiation. Cell Res. 2015, 25, 1152–1170. [Google Scholar] [CrossRef] [PubMed]
- Kai, T.; Spradling, A. An empty Drosophila stem cell niche reactivates the proliferation of ectopic cells. Proc. Natl. Acad. Sci. USA 2003, 100, 4633–4638. [Google Scholar] [CrossRef] [PubMed]
- Lantz, V.; Chang, J.S.; Horabin, J.I.; Bopp, D.; Schedl, P. The Drosophila orb RNA-binding protein is required for the formation of the egg chamber and establishment of polarity. Genes Dev. 1994, 8, 598–613. [Google Scholar] [CrossRef] [PubMed]
- Xi, R.; Doan, C.; Liu, D.; Xie, T. Pelota controls self-renewal of germline stem cells by repressing a Bam-independent differentiation pathway. Development 2005, 132, 5365–5374. [Google Scholar] [CrossRef] [PubMed]
- Ohlstein, B.; McKearin, D. Ectopic expression of the Drosophila Bam protein eliminates oogenic germline stem cells. Development 1997, 124, 3651–3662. [Google Scholar] [PubMed]
- Kreutzer, M.A.; Richards, J.P.; De Silva-Udawatta, M.N.; Temenak, J.J.; Knoblich, J.A.; Lehner, C.F.; Bennett, K.L. Caenorhabditis elegans cyclin A- and B-type genes: A cyclin A multigene family, an ancestral cyclin B3 and differential germline expression. J. Cell Sci. 1995, 108 Pt 6, 2415–2424. [Google Scholar] [PubMed]
- Nguyen, T.B.; Manova, K.; Capodieci, P.; Lindon, C.; Bottega, S.; Wang, X.Y.; Refik-Rogers, J.; Pines, J.; Wolgemuth, D.J.; Koff, A. Characterization and expression of mammalian cyclin b3, a prepachytene meiotic cyclin. J. Biol. Chem. 2002, 277, 41960–41969. [Google Scholar] [CrossRef] [PubMed]
- Yuan, K.; O’Farrell, P.H. Cyclin B3 is a mitotic cyclin that promotes the metaphase-anaphase transition. Curr. Biol. 2015, 25, 811–816. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Tao, X.; Zhou, L.; Sun, F.; Sun, M.; Fang, X. Spaghetti, a homolog of human RPAP3 (RNA polymerase II-associated protein 3), determines the fate of germline stem cells in Drosophila ovary. Cell Biol. Int. 2017. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Zhu, X.; Zhou, L.; Wang, J.; Tao, X.; Wang, S.; Sun, F.; Kan, X.; Han, Z.; Gu, Y. Gilgamesh is required for the maintenance of germline stem cells in Drosophila testis. Sci. Rep. 2017, 7, 5737. [Google Scholar] [CrossRef] [PubMed]



| Genotype | Age 1 | Germaria | ||||
|---|---|---|---|---|---|---|
| Empty | 0 Germline Stem Cell (GSC) (Cysts Only) | 1 GSC | 2–3 GSCs | Total | ||
| Oregon | Day 1 | 0 | 0 | 1.4% | 98.6% | 146 |
| Day 7 | 0 | 0.6% | 3.9% | 95.5% | 178 | |
| Day 14 | 0.7% | 1.4% | 7.4% | 90.5% | 148 | |
| cycB32/cycB32 | Day 1 | 2.9% | 2.2% | 5.8% | 89.1% | 274 |
| Day 7 | 26.6% | 4.4% | 29.6% | 39.4% | 203 | |
| Day 14 | 42.1% | 10.5% | 24.8% | 22.6% | 420 * | |
| cycB32/cycB3EY08012 | Day 1 | 1.9% | 0.5% | 3.3% | 94.3% | 209 |
| Day 7 | 22.5% | 6.0% | 25.8% | 45.7% | 267 | |
| Day 14 | 38.8% | 5.6% | 28.0% | 27.6% | 340 * | |
| cycB32/cycB3L6540 | Day 1 | 3.1% | 3.6% | 6.7% | 86.6% | 194 |
| Day 7 | 8.0% | 1.9% | 26.5% | 63.6% | 162 | |
| Day 14 | 29.5% | 15.8% | 26.5% | 28.2% | 444 * | |
| cycB3EY08012/cycB3L6540 | Day 1 | 6.9% | 0.9% | 11.4% | 80.8% | 219 |
| Day 7 | 15.9% | 6.0% | 47.5% | 30.6% | 183 | |
| Day 14 | 35.2% | 13.5% | 32.9% | 18.4% | 207 * | |
| cycB32/Df | Day 1 | 4.3% | 3.8% | 6.9% | 85.0% | 160 |
| Day 7 | 5.6% | 11.7% | 24.8% | 57.9% | 214 | |
| Day 14 | 32.9% | 20.8% | 25.9% | 20.4% | 216 * | |
| Genotype | The Average Number of GSCs (Mean ± SD) | p Value | The Average Number of CBs (Mean ± SD) | p-Value |
|---|---|---|---|---|
| bamP-GFP (WT) | 2.1 ± 0.4 (n = 87) | - | 1.1 ± 0.4 (n = 87) | - |
| bamP-GFP; attB-cycB3-gDNA | 2.6 ± 0.9 (n = 83) | p < 0.05 | 2.8 ± 1.3 (n = 83) | p < 0.01 |
| bamP-GFP; nosP-cycB3 | 3.0 ± 1.4 (n = 113) | p < 0.05 | 3.1 ± 1.7 (n = 113) | p < 0.01 |
| bamP-GFP; hsP-cycB3 (RT) | 2.1 ± 0.5 (n = 89) | p > 0.05 | 1.1 ± 0.5 (n = 89) | p < 0.01 |
| bamP-GFP; hsP-cycB3 (HS) | 2.2 ± 0.8 (n = 98) | 3.2 ± 1.4 (n = 98) |
| Primer Names | Sequences of Primers |
|---|---|
| cycB3 | 5’-CAGTGCCCGAGGAAGTAGAG-3′ (sense) |
| 5’-GCATATAGTCGGCAATGGGG-3′ (antisense) | |
| rp49 | 5’-CACTTCATCCGCCACCAGTC-3′ (sense) |
| 5’-CGCTTGTTCGATCCGTAACC-3′ (antisense) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, D.; Zhou, L.; Sun, F.; Sun, M.; Tao, X. Cyclin B3 Deficiency Impairs Germline Stem Cell Maintenance and Its Overexpression Delays Cystoblast Differentiation in Drosophila Ovary. Int. J. Mol. Sci. 2018, 19, 298. https://doi.org/10.3390/ijms19010298
Chen D, Zhou L, Sun F, Sun M, Tao X. Cyclin B3 Deficiency Impairs Germline Stem Cell Maintenance and Its Overexpression Delays Cystoblast Differentiation in Drosophila Ovary. International Journal of Molecular Sciences. 2018; 19(1):298. https://doi.org/10.3390/ijms19010298
Chicago/Turabian StyleChen, Dongsheng, Lijuan Zhou, Fuling Sun, Mingzhong Sun, and Xiaoqian Tao. 2018. "Cyclin B3 Deficiency Impairs Germline Stem Cell Maintenance and Its Overexpression Delays Cystoblast Differentiation in Drosophila Ovary" International Journal of Molecular Sciences 19, no. 1: 298. https://doi.org/10.3390/ijms19010298






