Current Knowledge on the Use of Computational Toxicology in Hazard Assessment of Metallic Engineered Nanomaterials
Abstract
:1. Introduction
2. State-of-the-Art of In Silico Models Serving Hazard Assessment of ENMs
2.1. Development of (Q)SARs and Read-Across Models for Metallic ENMs
- (i)
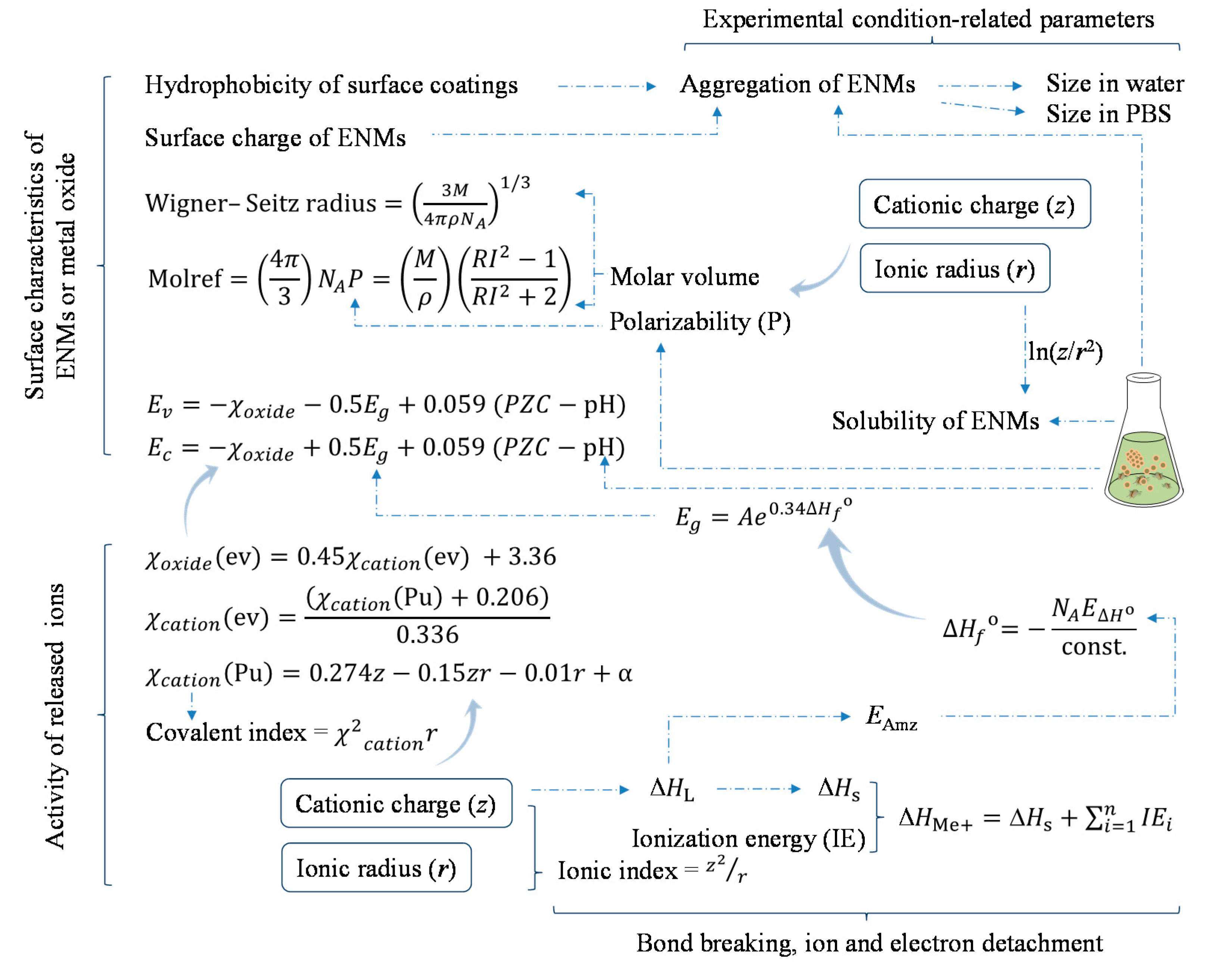
- Descriptors regarding the intrinsic properties of metal (oxide):
- Surface catalytic properties and redox modifications related factors include: Wigner–Seitz radius, mass density, band gap energy, overlap of conduction band energy levels with the cellular redox potential, conduction band energy, average of the alpha and beta LUMO (lowest unoccupied molecular orbital) energies of the metal oxide, accessible surface area, absolute electronegativity of the metal and the metal oxide, aligned electronegativity, electronegativity, Mulliken’s electronegativity of the cluster, S2 (SiRMS-derived number of oxygen’s atoms in a molecule, which was described by their electronegativity), S3 (tri-atomic fragments[Me]-[O]-[Me], which were encoded by SiRMS-derived descriptors, encoding electronegativity), and metal electronegativity.
- Characteristics related to the capability of ion and electron detachment and the activity of ions include: covalent index, cation polarizing power, atomization energy, metal oxide ionization energy, ionic index of metal cation, enthalpy of formation of metal oxide nanocluster representing a fragment of the surface, cationic charge, enthalpy of formation of a gaseous cation, charge of the metal cation corresponding to a given oxide, solubility, polarizability, molar refractivity, and polarization force.
- (ii)
- The nano-specific descriptors employed in the developed models include:
- The size of ENMs; and
- Parameters characterizing the surface chemistry of ENMs, e.g., hydrophobicity of surface coating chemicals, surface-area-to-volume ratio, surface coating and charge, surface area, polar surface area.
- (iii)
- The parameters indicating the dynamic changes of ENMs in media include:
- Zeta potential;
- Concentration of ENMs; and
- Descriptors representing the dispersion and aggregation of ENMs in media, e.g., aggregation parameter, size in DMEM (Dulbecco’s Modified Eagle’s Medium), relaxivity (representing ENM magnetic properties), size in phosphate buffered saline, size in water, aggregation size.
2.2. Development of SSDs for Metal-Based ENMs
3. The Struggle of Data Availability
- (i)
- Details of the tested organisms, e.g., taxonomic categorization, name of species, exposure route, life-stage or bacterial strain (for bacteria);
- (ii)
- Conditions of the performed experiments, e.g., test guideline used (if available) and possible modifications of the test guideline, preparation of test medium, composition of the exposure medium, media pH, light condition, and time-dependent medium stability;
- (iii)
- Information on the specific toxicity endpoints, e.g., observed biological effects, type of endpoint, experimental value of toxicity endpoint, and unit in which the endpoint is expressed; and
- (iv)
- Characteristics of the ENMs tested, e.g., type of ENMs, composition of core, distribution of particle size, surface coating, purity, crystallinity, surface area, surface charge, shape, agglomerate size and material zeta potential in media, stability in test medium.
4. Profiling Nanotoxicity on the Basis of In Silico Models
5. Outlook
6. Conclusions
- (i)
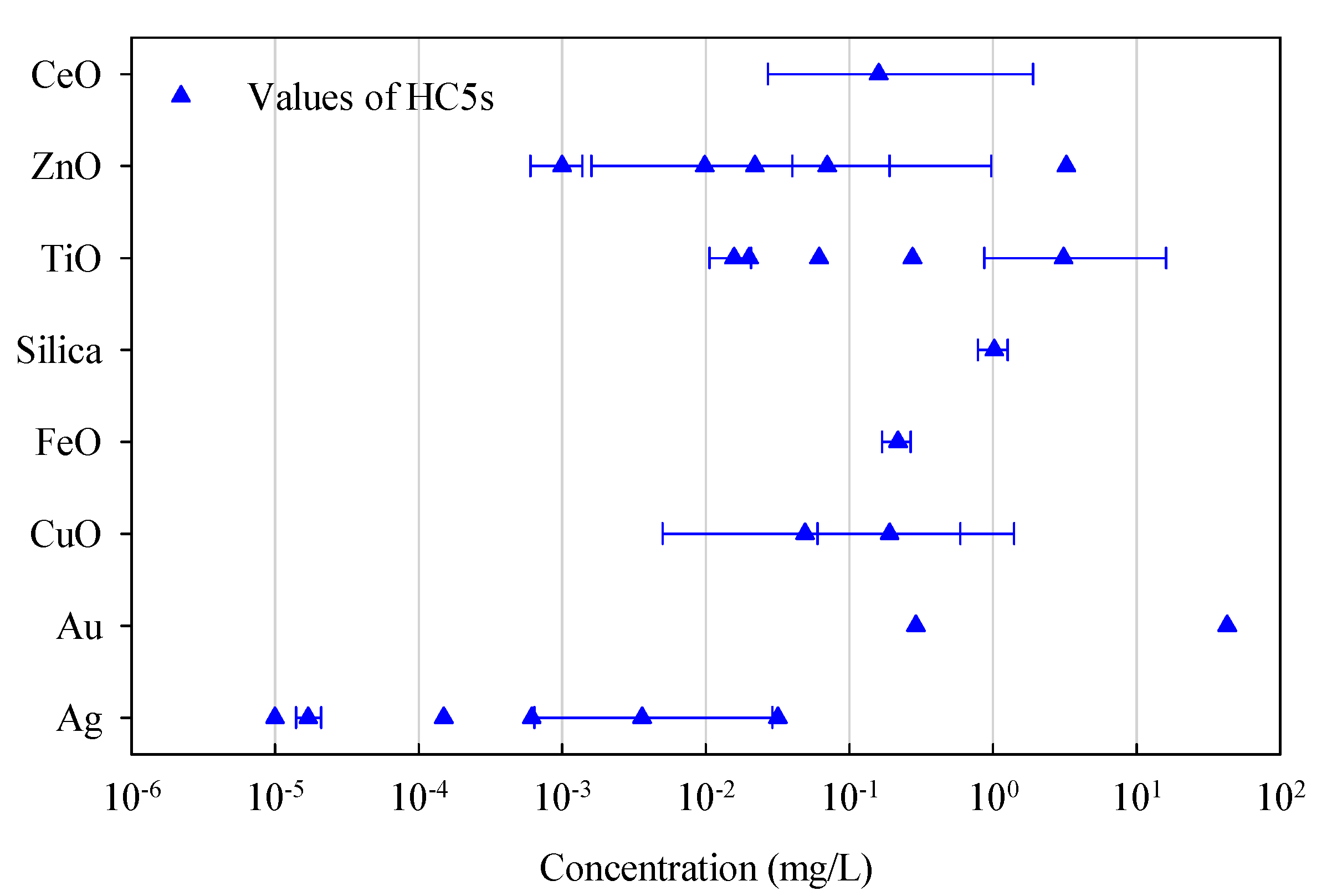
- An overview is provided of the current advances towards the development of in silico predictive models and SSDs for metallic ENMs. Based on reported models, factors such as solubility, hydrophobicity of ENM surface coating, and polarizability were concluded as enhancing the toxicity elicited by metallic ENMs. Meanwhile factors such as conduction band energy, ionization energy, and cationic charge were shown to play an opposite role in this respect. The studies on SSDs for ENMs showed that marginal risks are associated with the presence of Ag, TiO2, and ZnO ENMs in surface water, whereas high environmental risks are foreseen for those ENMs in sewage treatment effluents.
- (ii)
- A proposal is presented for preparation of a thoroughly curated dataset related to reporting of future results of laboratory studies, in light of enclosing sufficient information to allow for optimal ENM-related modeling based on laboratory assays.
- (iii)
- The mechanism of biological activities of metal-based ENMs is profiled based on employed descriptors. The intrinsic properties of ENMs such as cationic charge and ionic radius are considered pivotal in affecting nanotoxicity. However, surface chemistry of ENMs is shown to also be able to significantly modify the toxicity or bioavailability of metallic ENMs.
- (iv)
- Several suggestions for further studies are provided in the outlook, with regard to the use of existing nanotoxicity data for modeling, computation of nano-specific descriptors, and consideration of the transformation of ENMs in media into modeling. Finally, a roadmap is depicted to optimize the use of computational toxicology in hazard assessment of ENMs and to further advance the broader field of ENM-related modeling.
Acknowledgments
Conflicts of Interest
References
- European Commission. Key Enabling Technologies. Available online: https://ec.europa.eu/programmes/horizon2020/en/area/key-enabling-technologies (accessed on 8 March 2017).
- Nel, A.; Xia, T.; Mädler, L.; Li, N. Toxic potential of materials at the nanolevel. Science 2006, 311, 622–627. [Google Scholar] [CrossRef] [PubMed]
- Bondarenko, O.; Juganson, K.; Ivask, A.; Kasemets, K.; Mortimer, M.; Kahru, A. Toxicity of Ag, CuO and ZnO nanoparticles to selected environmentally relevant test organisms and mammalian cells in vitro: A critical review. Arch. Toxicol. 2013, 87, 1181–1200. [Google Scholar] [CrossRef] [PubMed]
- Juganson, K.; Ivask, A.; Blinova, I.; Mortimer, M.; Kahru, A. NanoE-Tox: New and in-depth database concerning ecotoxicity of nanomaterials. Beilstein J. Nanotechnol. 2015, 6, 1788–1804. [Google Scholar] [CrossRef] [PubMed]
- Savolainen, K.; Backman, U.; Brouwer, D.; Fadeel, B.; Fernandes, T.; Kuhlbusch, T.; Landsiedel, R.; Lynch, I.; Pylkkänen, L. Nanosafety in Europe 2015–2025: Towards Safe and Sustainable Nanomaterials and Nanotechnology Innovations; Finnish Institute of Occupational Health: Helsinki, Finland, 2013. [Google Scholar]
- Raies, A.B.; Bajic, V.B. In silico toxicology: Computational methods for the prediction of chemical toxicity. Wiley Interdiscip. Rev. Comput. Mol. Sci. 2016, 6, 147–172. [Google Scholar] [CrossRef] [PubMed]
- Reisfeld, B.; Mayeno, A.N. What is computational toxicology? Methods Mol. Biol. 2012, 929, 3–7. [Google Scholar] [PubMed]
- Kavlock, R.J.; Ankley, G.; Blancato, J.; Breen, M.; Conolly, R.; Dix, D.; Houck, K.; Hubal, E.; Judson, R.; Rabinowitz, J.; et al. Computational toxicology—A state of the science mini review. Toxicol. Sci. 2008, 103, 14–27. [Google Scholar] [CrossRef] [PubMed]
- United States Environmental Protection Agency (U.S. EPA). A Framework for a Computational Toxicology Research Program; United States Environmental Protection Agency (U.S. EPA): Washington, DC, USA, 2003.
- Chen, G.; Li, X.; Chen, J.; Zhang, Y.N.; Peijnenburg, W.J.G.M. Comparative study of biodegradability prediction of chemicals using decision trees, functional trees, and logistic regression. Environ. Toxicol. Chem. 2014, 33, 2688–2693. [Google Scholar] [CrossRef] [PubMed]
- Pavan, M.; Worth, A.P.; Netzeva, T.I. Review of QSAR Models for Bioconcentration; European Commission, Joint Research Centre: Ispra, Italy, 2006.
- Tunkel, J.; Howard, P.H.; Boethling, R.S.; Stiteler, W.; Loonen, H. Predicting ready biodegradability in the Japanese ministry of international trade and industry test. Environ. Toxicol. Chem. 2000, 19, 2478–2485. [Google Scholar] [CrossRef]
- Posthuma, L.; Traas, T.P.; Suter, G.W. General introduction to species sensitivity distributions. In Species Sensitivity Distroibution in Ecotoxicology; Posthuma, L., Suter, G.W., Traas, T.P., Eds.; Lewis: Boca Raton, FL, USA, 2002; pp. 3–10. [Google Scholar]
- European Chemicals Agency (ECHA). Guidance on Information Requirements and Chemical Safety Assessment, Part B: Hazard Assessment, Version 2.1; European Chemicals Agency: Helsinki, Finland, 2011.
- Chen, G.; Peijnenburg, W.J.G.M.; Kovalishyn, V.; Vijver, M.G. Development of nanostructure–activity relationships assisting the nanomaterial hazard categorization for risk assessment and regulatory decision-making. RSC Adv. 2016, 6, 52227–52235. [Google Scholar] [CrossRef]
- Kleandrova, V.V.; Luan, F.; González-Díaz, H.; Ruso, J.M.; Speck-Planche, A.; Cordeiro, M.N. Computational tool for risk assessment of nanomaterials: Novel QSTR-perturbation model for simultaneous prediction of ecotoxicity and cytotoxicity of uncoated and coated nanoparticles under multiple experimental conditions. Environ. Sci. Technol. 2014, 48, 14686–14694. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Tang, K.; Harper, S.; Harper, B.; Steevens, J.A.; Xu, R. Predictive modeling of nanomaterial exposure effects in biological systems. Int. J. Nanomedicine 2013, 8, 31–43. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Rallo, R.; George, S.; Ji, Z.; Nair, S.; Nel, A.E.; Cohen, Y. Classification NanoSAR development for cytotoxicity of metal oxide nanoparticles. Small 2011, 7, 1118–1126. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Ji, Z.; Xia, T.; Meng, H.; Low-Kam, C.; Liu, R.; Pokhrel, S.; Lin, S.; Wang, X.; Liao, Y.P.; et al. Use of metal oxide nanoparticle band gap to develop a predictive paradigm for oxidative stress and acute pulmonary inflammation. ACS Nano 2012, 6, 4349–4368. [Google Scholar] [CrossRef] [PubMed]
- Sizochenko, N.; Rasulev, B.; Gajewicz, A.; Mokshyna, E.; Kuz’min, V.E.; Leszczynski, J.; Puzyn, T. Causal inference methods to assist in mechanistic interpretation of classification nano-SAR models. RSC Adv. 2015, 5, 77739–77745. [Google Scholar] [CrossRef]
- Liu, R.; Zhang, H.Y.; Ji, Z.X.; Rallo, R.; Xia, T.; Chang, C.H.; Nel, A.; Cohen, Y. Development of structure-activity relationship for metal oxide nanoparticles. Nanoscale 2013, 5, 5644–5653. [Google Scholar] [CrossRef] [PubMed]
- Gajewicz, A.; Schaeublin, N.; Rasulev, B.; Hussain, S.; Leszczynska, D.; Puzyn, T.; Leszczynski, J. Towards understanding mechanisms governing cytotoxicity of metal oxides nanoparticles: Hints from nano-QSAR studies. Nanotoxicology 2015, 9, 313–325. [Google Scholar] [CrossRef] [PubMed]
- Luan, F.; Kleandrova, V.V.; González-Díaz, H.; Ruso, J.M.; Melo, A.; Speck-Planche, A.; Cordeiro, M.N. Computer-aided nanotoxicology: Assessing cytotoxicity of nanoparticles under diverse experimental conditions by using a novel QSTR-perturbation approach. Nanoscale 2014, 6, 10623–10630. [Google Scholar] [CrossRef] [PubMed]
- Fourches, D.; Pu, D.; Tassa, C.; Weissleder, R.; Shaw, S.Y.; Mumper, R.J.; Tropsha, A. Quantitative nanostructure-activity relationship modeling. ACS Nano 2010, 4, 5703–5712. [Google Scholar] [CrossRef] [PubMed]
- Shaw, S.Y.; Westly, E.C.; Pittet, M.J.; Subramanian, A.; Schreiber, S.L.; Weissleder, R. Perturbational profiling of nanomaterial biologic activity. Proc. Natl. Acad. Sci. USA 2008, 105, 7387–7392. [Google Scholar] [CrossRef] [PubMed]
- Epa, V.C.; Burden, F.R.; Tassa, C.; Weissleder, R.; Shaw, S.; Winkler, D.A. Modeling biological activities of nanoparticles. Nano Lett. 2012, 12, 5808–5812. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.P.; Gupta, S. Nano-QSAR modeling for predicting biological activity of diverse nanomaterials. RSC Adv. 2014, 4, 13215–13230. [Google Scholar] [CrossRef]
- Puzyn, T.; Rasulev, B.; Gajewicz, A.; Hu, X.; Dasari, T.P.; Michalkova, A.; Hwang, H.M.; Toropov, A.; Leszczynska, D.; Leszczynski, J. Using nano-QSAR to predict the cytotoxicity of metal oxide nanoparticles. Nat. Nanotechnol. 2011, 6, 175–178. [Google Scholar] [CrossRef] [PubMed]
- Sayes, C.; Ivanov, I. Comparative Study of Predictive Computational Models for Nanoparticle-Induced Cytotoxicity. Risk Anal. 2010, 30, 1723–1734. [Google Scholar] [CrossRef] [PubMed]
- Papa, E.; Doucet, J.P.; Doucet-Panaye, A. Linear and non-linear modelling of the cytotoxicity of TiO2 and ZnO nanoparticles by empirical descriptors. SAR QSAR Environ. Res. 2015, 26, 647–665. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Li, T.; Cheng, J.; Telesca, D.; Zink, J.I.; Jiang, J. Nano-QSAR modeling for predicting the cytotoxicity of metal oxide nanoparticles using novel descriptors. RSC Adv. 2016, 6, 25766–25775. [Google Scholar] [CrossRef]
- Gajewicz, A.; Cronin, M.T.; Rasulev, B.; Leszczynski, J.; Puzyn, T. Novel approach for efficient predictions properties of large pool of nanomaterials based on limited set of species: Nano-read-across. Nanotechnology 2015, 26, 015701. [Google Scholar] [CrossRef] [PubMed]
- Sizochenko, N.; Rasulev, B.; Gajewicz, A.; Kuz’min, V.; Puzyn, T.; Leszczynski, J. From basic physics to mechanisms of toxicity: The “liquid drop” approach applied to develop predictive classification models for toxicity of metal oxide nanoparticles. Nanoscale 2014, 6, 13986–13993. [Google Scholar] [CrossRef] [PubMed]
- Gajewicz, A.; Jagiello, K.; Cronin, M.T.D.; Leszczynski, J.; Puzyn, T. Addressing a bottle neck for regulation of nanomaterials: Quantitative read-across (Nano-QRA) algorithm for cases when only limited data is available. Environ. Sci. Nano 2017, 4, 346–358. [Google Scholar] [CrossRef]
- Mu, Y.; Wu, F.; Zhao, Q.; Ji, R.; Qie, Y.; Zhou, Y.; Hu, Y.; Pang, C.; Hristozov, D.; Giesy, J.P.; Xing, B. Predicting toxic potencies of metal oxide nanoparticles by means of nano-QSARs. Nanotoxicology 2016, 10, 1207–1214. [Google Scholar] [CrossRef] [PubMed]
- Kar, S.; Gajewicz, A.; Puzyn, T.; Roy, K.; Leszczynski, J. Periodic table-based descriptors to encode cytotoxicity profile of metal oxide nanoparticles: A mechanistic QSTR approach. Ecotoxicol. Environ. Saf. 2014, 107, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Pathakoti, K.; Huang, M.J.; Watts, J.D.; He, X.; Hwang, H.M. Using experimental data of Escherichia coli to develop a QSAR model for predicting the photo-induced cytotoxicity of metal oxide nanoparticles. J. Photochem. Photobiol. B 2014, 130, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Vijver, M.G.; Peijnenburg, W.J.G.M. Summary and analysis of the currently existing literature data on metal-based nanoparticles published for selected aquatic organisms: Applicability for toxicity prediction by (Q)SARs. Altern. Lab. Anim. 2015, 43, 221–240. [Google Scholar] [PubMed]
- Sushko, I.; Novotarskyi, S.; Körner, R.; Pandey, A.K.; Rupp, M.; Teetz, W.; Brandmaier, S.; Abdelaziz, A.; Prokopenko, V.V.; Tanchuk, V.Y.; et al. Online chemical modeling environment (OCHEM): Web platform for data storage, model development and publishing of chemical information. J. Comput. Aided Mol. Des. 2011, 25, 533–554. [Google Scholar] [CrossRef] [PubMed]
- Mahan, G.D.; Subbaswamy, K.R. Local Density Theory of Polarizability; Plenum Press: New York, NY, USA, 1990. [Google Scholar]
- Bendary, E.; Francis, R.R.; Ali, H.M.G.; Sarwat, M.I.; El Hady, S. Antioxidant and structure–activity relationships (SARs) of some phenolic and anilines compounds. Ann. Agric. Sci. 2013, 58, 173–181. [Google Scholar] [CrossRef]
- Asati, A.; Santra, S.; Kaittanis, C.; Perez, J.M. Surface-charge-dependent cell localization and cytotoxicity of cerium oxide nanoparticles. ACS Nano 2010, 4, 5321–5331. [Google Scholar] [CrossRef] [PubMed]
- El Badawy, A.M.; Silva, R.G.; Morris, B.; Scheckel, K.G.; Suidan, M.T.; Tolaymat, T.M. Surface charge-dependent toxicity of silver nanoparticles. Environ. Sci. Technol. 2011, 45, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Schaeublin, N.M.; Braydich-Stolle, L.K.; Schrand, A.M.; Miller, J.M.; Hutchison, J.; Schlager, J.J.; Hussain, S.M. Surface charge of gold nanoparticles mediates mechanism of toxicity. Nanoscale 2011, 3, 410–420. [Google Scholar] [CrossRef] [PubMed]
- Garner, K.L.; Suh, S.; Lenihan, H.S.; Keller, A.A. Species Sensitivity Distributions for Engineered Nanomaterials. Environ. Sci. Technol. 2015, 49, 5753–5759. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, R.; Meesters, J.A.; Ter Braak, C.J.; van de Meent, D.; van der Voet, H. Combining exposure and effect modeling into an integrated probabilistic environmental risk assessment for nanoparticles. Environ. Toxicol. Chem. 2016, 35, 2958–2967. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Deng, L.; Caballero-Guzman, A.; Nowack, B. Are engineered nano iron oxide particles safe? An environmental risk assessment by probabilistic exposure, effects and risk modeling. Nanotoxicology 2016, 10, 1545–1554. [Google Scholar] [CrossRef] [PubMed]
- Kwak, J.I.; Cui, R.; Nam, S.H.; Kim, S.W.; Chae, Y.; An, Y.J. Multispecies toxicity test for silver nanoparticles to derive hazardous concentration based on species sensitivity distribution for the protection of aquatic ecosystems. Nanotoxicology 2016, 10, 521–530. [Google Scholar] [CrossRef] [PubMed]
- Coll, C.; Notter, D.; Gottschalk, F.; Sun, T.; Som, C.; Nowack, B. Probabilistic environmental risk assessment of five nanomaterials (nano-TiO2, nano-Ag, nano-ZnO, CNT, and fullerenes). Nanotoxicology 2016, 10, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Kalinina, A.; Sun, T.; Nowack, B. Probabilistic modeling of the flows and environmental risks of nano-silica. Sci. Total Environ. 2016, 545–546, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Mahapatra, I.; Sun, T.Y.; Clark, J.R.; Dobson, P.J.; Hungerbuehler, K.; Owen, R.; Nowack, B.; Lead, J. Probabilistic modelling of prospective environmental concentrations of gold nanoparticles from medical applications as a basis for risk assessment. J. Nanobiotechnology 2015, 13, 93. [Google Scholar] [CrossRef] [PubMed]
- Semenzin, E.; Lanzellotto, E.; Hristozov, D.; Critto, A.; Zabeo, A.; Giubilato, E.; Marcomini, A. Species sensitivity weighted distribution for ecological risk assessment of engineered nanomaterials: The n-TiO2 case study. Environ. Toxicol. Chem. 2015, 34, 2644–2659. [Google Scholar] [CrossRef] [PubMed]
- Adam, N.; Schmitt, C.; De Bruyn, L.; Knapen, D.; Blust, R. Aquatic acute species sensitivity distributions of ZnO and CuO nanoparticles. Sci. Total Environ. 2015, 526, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Nam, S.H.; Shin, Y.J.; Lee, W.M.; Kim, S.W.; Kwak, J.I.; Yoon, S.J.; An, Y.J. Conducting a battery of bioassays for gold nanoparticles to derive guideline value for the protection of aquatic ecosystems. Nanotoxicology 2015, 9, 326–335. [Google Scholar] [CrossRef] [PubMed]
- Botha, T.L.; James, T.E.; Wepener, V. Comparative aquatic toxicity of gold nanoparticles and ionic gold using a species sensitivity distribution approach. J. Nanomater. 2015, 2015, 986902. [Google Scholar] [CrossRef]
- Haulik, B.; Balla, S.; Palfi, O.; Szekeres, L.; Jurikova, T.; Saly, P.; Bakonyi, G. Comparative ecotoxicity of the nano Ag, TiO2, and ZnO to aquatic species assemblages. Appl. Ecol. Env. Res. 2015, 13, 325–338. [Google Scholar]
- Gottschalk, F.; Kost, E.; Nowack, B. Engineered nanomaterials in water and soils: A risk quantification based on probabilistic exposure and effect modeling. Environ. Toxicol. Chem. 2013, 32, 1278–1287. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Peijnenburg, W.J.G.M.; Xiao, Y.; Vijver, M.G. Developing species sensitivity distributions for metallic nanomaterials considering the characteristics of nanomaterials, experimental conditions, and different types of endpoints. Food Chem. Toxicol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Keller, A.A.; Lazareva, A. Predicted Releases of Engineered Nanomaterials: From Global to Regional to Local. Environ. Sci. Technol. Lett. 2014, 1, 65–70. [Google Scholar] [CrossRef]
- Toropov, A.A.; Toropova, A.P.; Benfenati, E.; Gini, G.; Puzyn, T.; Leszczynska, D.; Leszczynski, J. Novel application of the CORAL software to model cytotoxicity of metal oxide nanoparticles to bacteria Escherichia coli. Chemosphere 2012, 89, 1098–1102. [Google Scholar] [CrossRef] [PubMed]
- Ahrens, L.H. The use of ionization potentials Part 1. Ionic radii of the elements. Geochim. Cosmochim. Acta 1952, 2, 155–169. [Google Scholar] [CrossRef]
- Lide, D.R. CRC Handbook of Chemistry and Physics; CRC Press: Boca Raton, FL, USA, 1998. [Google Scholar]
- Burello, E. Computational design of safer nanomaterials. Environ. Sci. Nano 2015, 2, 454–462. [Google Scholar] [CrossRef]
- Stohs, S.J.; Bagchi, D. Oxidative mechanisms in the toxicity of metal ions. Free Radic. Biol. Med. 1995, 18, 321–336. [Google Scholar] [CrossRef]
- Beer, C.; Foldbjerg, R.; Hayashi, Y.; Sutherland, D.S.; Autrup, H. Toxicity of silver nanoparticles—Nanoparticle or silver ion? Toxicol. Lett. 2012, 208, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Visnapuu, M.; Joost, U.; Juganson, K.; Künnis-Beres, K.; Kahru, A.; Kisand, V.; Ivask, A. Dissolution of silver nanowires and nanospheres dictates their toxicity to Escherichia coli. BioMed Res. Int. 2013, 2013, 819252. [Google Scholar] [CrossRef] [PubMed]
- Xiu, Z.M.; Zhang, Q.B.; Puppala, H.L.; Colvin, V.L.; Alvarez, P.J. Negligible particle-specific antibacterial activity of silver nanoparticles. Nano Lett. 2012, 12, 4271–4275. [Google Scholar] [CrossRef] [PubMed]
- Hua, J.; Vijver, M.G.; Richardson, M.K.; Ahmad, F.; Peijnenburg, W.J. Particle-specific toxic effects of differently shaped zinc oxide nanoparticles to zebrafish embryos (Danio rerio). Environ. Toxicol. Chem. 2014, 33, 2859–2868. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Palma, S.; Fisher, N.S.; Wong, S.S. Effect of morphology of ZnO nanostructures on their toxicity to marine algae. Aquat. Toxicol. 2011, 102, 186–196. [Google Scholar] [CrossRef] [PubMed]
- Arts, J.H.; Hadi, M.; Irfan, M.A.; Keene, A.M.; Kreiling, R.; Lyon, D.; Maier, M.; Michel, K.; Petry, T.; Sauer, U.G.; et al. A decision-making framework for the grouping and testing of nanomaterials (DF4nanoGrouping). Regul. Toxicol. Pharmacol. 2015, 71, S1–S27. [Google Scholar] [CrossRef] [PubMed]
- Arts, J.H.; Irfan, M.A.; Keene, A.M.; Kreiling, R.; Lyon, D.; Maier, M.; Michel, K.; Neubauer, N.; Petry, T.; Sauer, U.G.; et al. Case studies putting the decision-making framework for the grouping and testing of nanomaterials (DF4nanoGrouping) into practice. Regul. Toxicol. Pharmacol. 2016, 76, 234–261. [Google Scholar] [CrossRef] [PubMed]
- Tämm, K.; Sikk, L.; Burk, J.; Rallo, R.; Pokhrel, S.; Mädler, L.; Scott-Fordsmand, J.J.; Burk, P.; Tamm, T. Parametrization of nanoparticles: Development of full-particle nanodescriptors. Nanoscale 2016, 8, 16243–16250. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, H.-J. Fuzzy set theory. Wiley Interdiscip. Rev. Comput. Stat. 2010, 2, 317–332. [Google Scholar] [CrossRef]
- Hua, J.; Vijver, M.G.; Chen, G.; Richardson, M.K.; Peijnenburg, W.J.G.M. Dose metrics assessment for differently shaped and sized metal-based nanoparticles. Environ. Toxicol. Chem. 2016, 35, 2466–2473. [Google Scholar] [CrossRef] [PubMed]
- Oberdörster, G.; Oberdörster, E.; Oberdörster, J. Concepts of nanoparticle dose metric and response metric. Environ. Health Perspect. 2007, 115, A290. [Google Scholar] [CrossRef] [PubMed]


| Reference | Indicated ENM Characteristics in Models | Theoretical Descriptor | Experimental Descriptor | ENMs | Tested Organism | Data Retrieved from |
|---|---|---|---|---|---|---|
| [18] * | Number of metal and oxygen atoms, molecular weight, atomization energy, group and period in the periodic table, size, isoelectric point, zeta potential, concentration | √ | √ | 9 metal oxide ENMs | BEAS-2B cells | N/A |
| [19] | Band gap energy, overlap of conduction band energy levels with the cellular redox potential (−4.12 to −4.84 eV), solubility | √ | √ | 24 metal oxide ENMs | BEAS-2B cells, RAW 264.7 cells | N/A |
| [20] | Mass density, molecular weight, aligned electronegativity, covalent index, cation polarizing power, Wigner–Seitz radius, surface area, surface-area-to-volume ratio, aggregation parameter, two-atomic descriptor of van der Waals interactions, tri-atomic descriptor of atomic charges, tetra-atomic descriptor of atomic charges, size in DMEM | √ | √ | [19] | ||
| [21] * | Atomization energy, atomic mass, size, conduction band energy, metal oxide ionization energy, electronegativity, ionic index of metal cation | √ | ||||
| [22] | Enthalpy of formation of metal oxide nanocluster representing a fragment of the surface, Mulliken’s electronegativity of the cluster | √ | 18 metal oxide ENMs | HaCaT cells | N/A | |
| [23] * | Molar volume, polarizability, size | √ | √ | 41 metallic ENMs | Mammalian cells | Multiple resources |
| [24] * | Size, relaxivities, zeta potential | √ | √ | 50 metallic ENMs | Endothelial cells, vascular smooth muscle cells, human HepG2 cells, RAW 264.7 cells | [25] |
| [26] | Indicator variables of core material, surface coating, and surface charge | √ | ||||
| [27] | (i) Size, relaxivities, zeta potential; (ii) Oxygen percent, molar refractivity, polar surface area | √ | √ | (i) 44; (ii) 17 metallic ENMs | (i) Endothelial cells, vascular smooth muscle cells, human HepG2 cells, RAW 264.7 cells; (ii) E. coli | [25,28] |
| [29] * | Size, concentration, size in phosphate buffered saline, size in water, zeta potential | √ | √ | 24 TiO2, 18 ZnO ENMs | Rat L2 lung epithelial cells, rat lung alveolar macrophages | N/A |
| [30] | Size, concentration, size in phosphate buffered saline, size in water | √ | √ | [29] | ||
| [31] | Molecular weight, cationic charge, mass percentage of metal elements, size, aggregation size | √ | √ | (i) 17; (ii) 18 metal oxide ENMs | (i) E. coli; (ii) HaCaT cells | [22,28] |
| [32] * | Enthalpy of formation of a gaseous cation, Mulliken’s electronegativity of the cluster | √ | ||||
| [33] | (i) S1, Wigner–Seitz radius, mass density, cation polarizing power, S2, S3, proportion of surface molecules to molecules in volume; (ii) S1, Wigner–Seitz radius of oxide’s molecule, mass density, covalent index of the metal ion, S2, aggregation parameter | √ | √ | |||
| [34] | Enthalpy of formation of a gaseous cation, enthalpy of formation of metal oxide nanocluster representing a fragment of the surface, Mulliken’s electronegativity of the cluster | √ | ||||
| [28] | Enthalpy of formation of a gaseous cation | √ | 17 metal oxide ENMs | E. coli | N/A | |
| [35] | Polarization force, enthalpy of formation of a gaseous cation | √ | [28] | |||
| [36] | Charge of the metal cation corresponding to a given oxide, metal electronegativity | √ | ||||
| [37] | Dark: absolute electronegativity of the metal and the metal oxide; Light: molar heat capacity, average of the alpha and beta LUMO (lowest unoccupied molecular orbital) energies of the metal oxide | √ | N/A | |||
| [15] * | Molecular polarizability, accessible surface area, solubility | √ | 400; 450; 166 metallic ENMs | Various species | [38]; OCHEM | |
| [16] * | Molar volume, electronegativity, polarizability, size, hydrophobicity, polar surface area | √ | √ | 229 metallic ENMs | Various species | Multiple resources |
| [17] | Concentration, shell composition, surface functional groups, purity, core structure, and surface charge | √ | √ | 82 ENMs including metal and metal oxide ENMs, dendrimer, polymeric etc. | Zebrafish embryo | NBI knowledgebase |
| Reference | Type of ENMs | Reported HC5s | Number of Species in SSDs | Environmental Compartment |
|---|---|---|---|---|
| Jacobs et al., 2016 [46] | TiO2 | N/A | 31 | Water |
| Wang et al., 2016 [47] | FeOx | 0.218 (0.169–0.267) mg/L, 15–85% percentiles | 12 | Water |
| Kwak et al., 2016 [48] | Ag | 0.03173 mg/L (acute toxicity); 0.000614 mg/L (chronic toxicity) | 8 (acute toxicity); 5 (chronic toxicity) | Water |
| Coll et al., 2016 [49] | (i) Ag; (ii) TiO2; (iii) ZnO | (i) 0.000017 (0.000014–0.000021) mg/L in freshwater, 8.2 (4.3–12.5) mg/kg in soil; (ii) 0.0157 (0.0106–0.0207) mg/L in fresh water, 91.1 (47.6–134.9) mg/kg in soil; (iii) 0.001 (0.0006–0.00138) mg/L in freshwater, 1.1 (0.6–1.6) mg/kg in soil, 95% confidence intervals | (i) 33 (water), 4 (soil); (ii) 31 (water), 2 (soil); (iii) 21 (water), 7 (soil) | Water, soil |
| Wang et al., 2016 [50] | Silica | 1.023 (0.787–1.265) mg/L, 15–85% percentiles | 8 | Water |
| Mahapatra et al., 2015 [51] | Au | N/A | 8 (water) | Water, soil |
| Semenzin et al., 2015 [52] | TiO2 | 0.02 mg/L | 34 | Water |
| Adam et al., 2015 [53] | (i) ZnO; (ii) CuO | (i) 0.07 (0.04–0.19) mg/L; (ii) 0.19 (0.06–0.59) mg/L, 90% confidence intervals | (i) 12; (ii) 13 | Water |
| Garner et al., 2015 [45] | (i) Ag; (ii) Cu; (iii) CuO; (iv) ZnO; (v) Al2O3; (vi) CeO2; (vii) TiO2 | N/A | (i) Uncoated-Ag: 8, PVP-Ag: 6; (ii) 4; (iii) 5; (iv) 7; (v) 9; (vi) 7; (vii) 8 | Water |
| Nam et al., 2015 [54] | Au | 0.29 mg/L | 7 | Water |
| Botha et al., 2015 [55] | Au | 42.78 mg/L | 4 | Water |
| Haulik et al., 2015 [56] | (i) Ag; (ii) TiO2; (iii) ZnO | (i) 0.00015; (ii) 0.275; (iii) 3.246 mg/L | (i) 14; (ii) 11; (iii) 10 | Water |
| Gottschalk et al., 2013 [57] | (i) Ag; (ii) TiO2; (iii) ZnO | (i) 0.00001; (ii) 0.06151; (iii) 0.00985 mg/L | (i) 12; (ii) 18; (iii) 17 | Water |
| Chen et al., 2017 [58] | (i) Ag; (ii) CuO; (iii) ZnO; (iv) CeO2; (v) TiO2 | HC5s were calculated for various SSDs | Different hierarchies of species were used | Water |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, G.; Peijnenburg, W.; Xiao, Y.; Vijver, M.G. Current Knowledge on the Use of Computational Toxicology in Hazard Assessment of Metallic Engineered Nanomaterials. Int. J. Mol. Sci. 2017, 18, 1504. https://doi.org/10.3390/ijms18071504
Chen G, Peijnenburg W, Xiao Y, Vijver MG. Current Knowledge on the Use of Computational Toxicology in Hazard Assessment of Metallic Engineered Nanomaterials. International Journal of Molecular Sciences. 2017; 18(7):1504. https://doi.org/10.3390/ijms18071504
Chicago/Turabian StyleChen, Guangchao, Willie Peijnenburg, Yinlong Xiao, and Martina G. Vijver. 2017. "Current Knowledge on the Use of Computational Toxicology in Hazard Assessment of Metallic Engineered Nanomaterials" International Journal of Molecular Sciences 18, no. 7: 1504. https://doi.org/10.3390/ijms18071504






