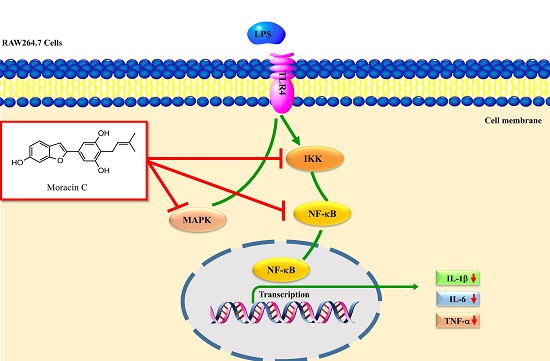
Moracin C, A Phenolic Compound Isolated from Artocarpus heterophyllus, Suppresses Lipopolysaccharide-Activated Inflammatory Responses in Murine Raw264.7 Macrophages
Abstract
:1. Introduction
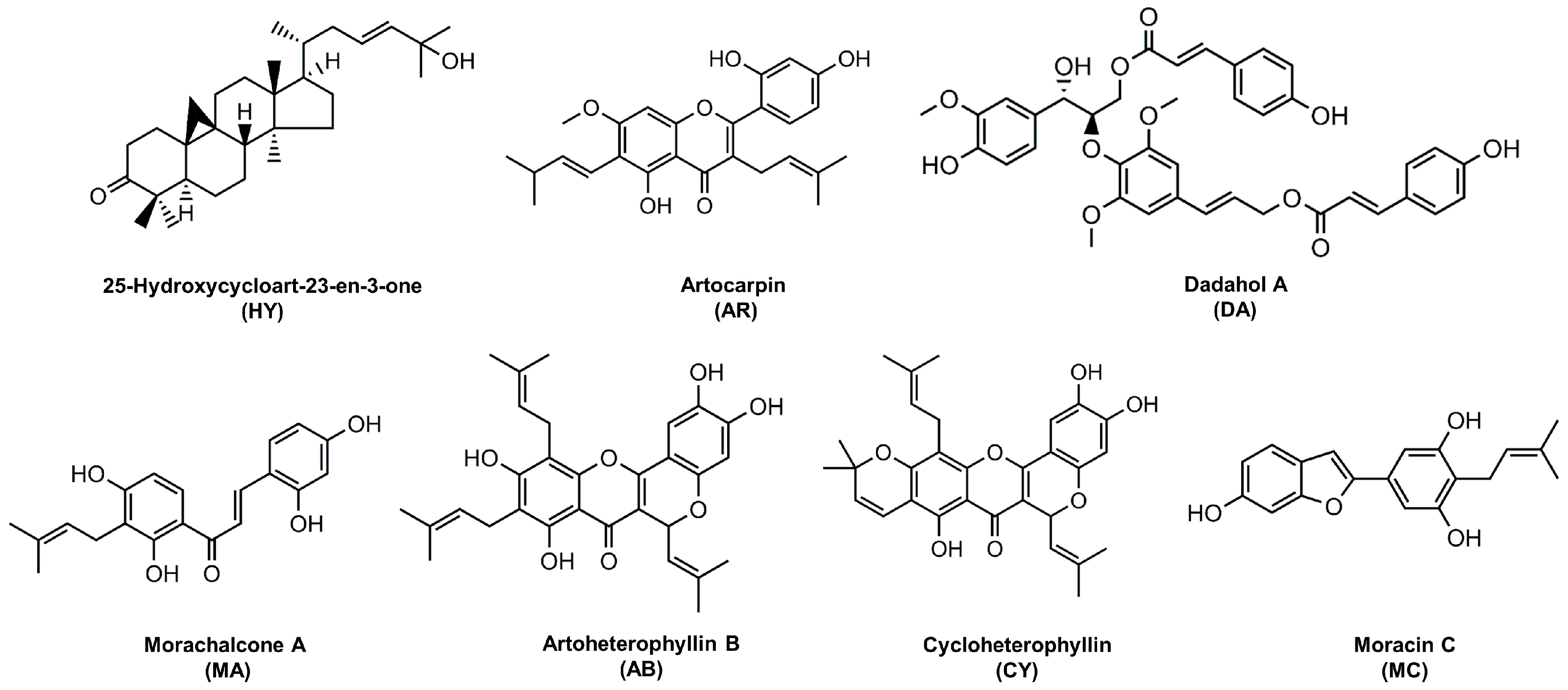
2. Results
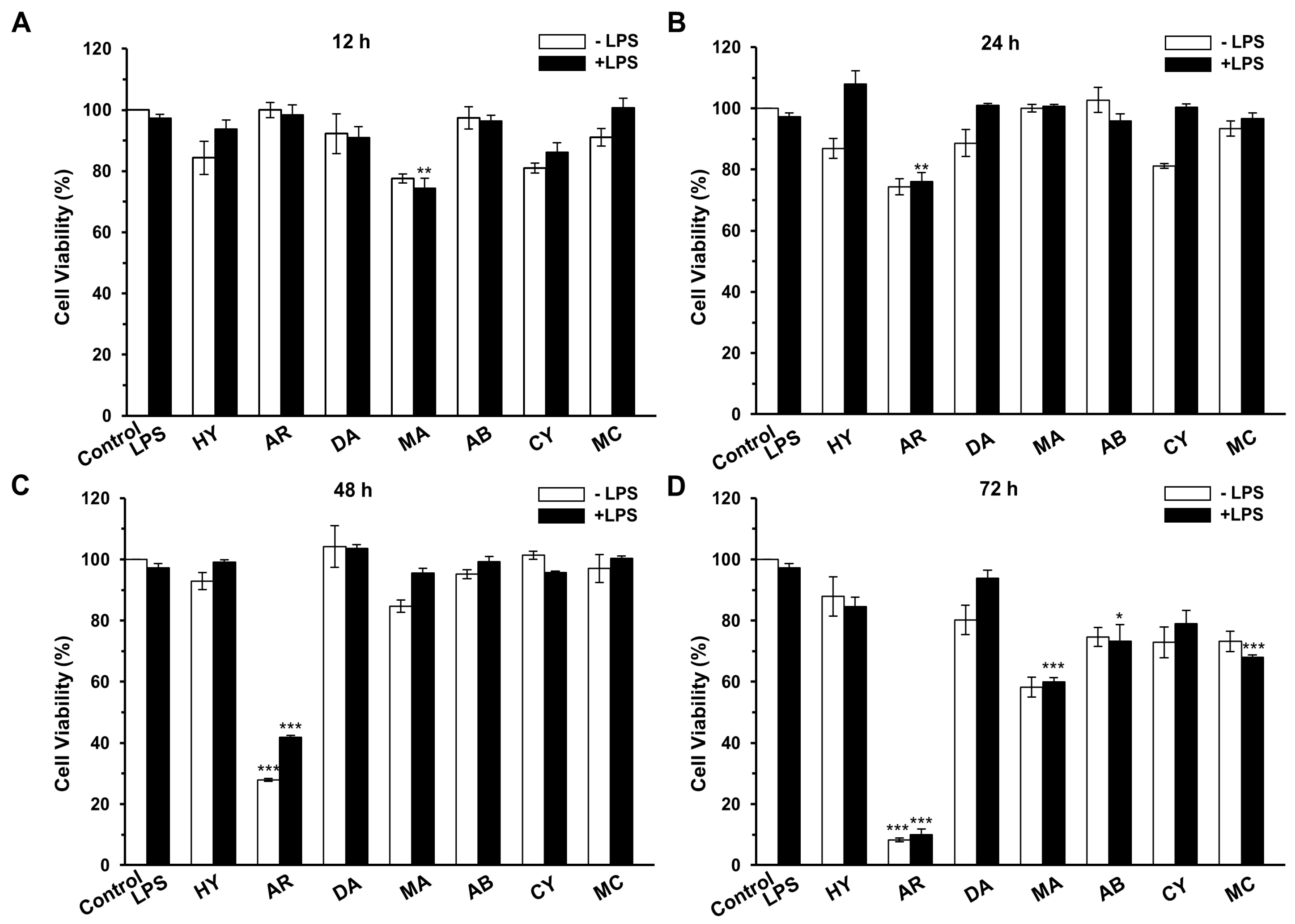
2.1. Cytotoxicity on Macrophage
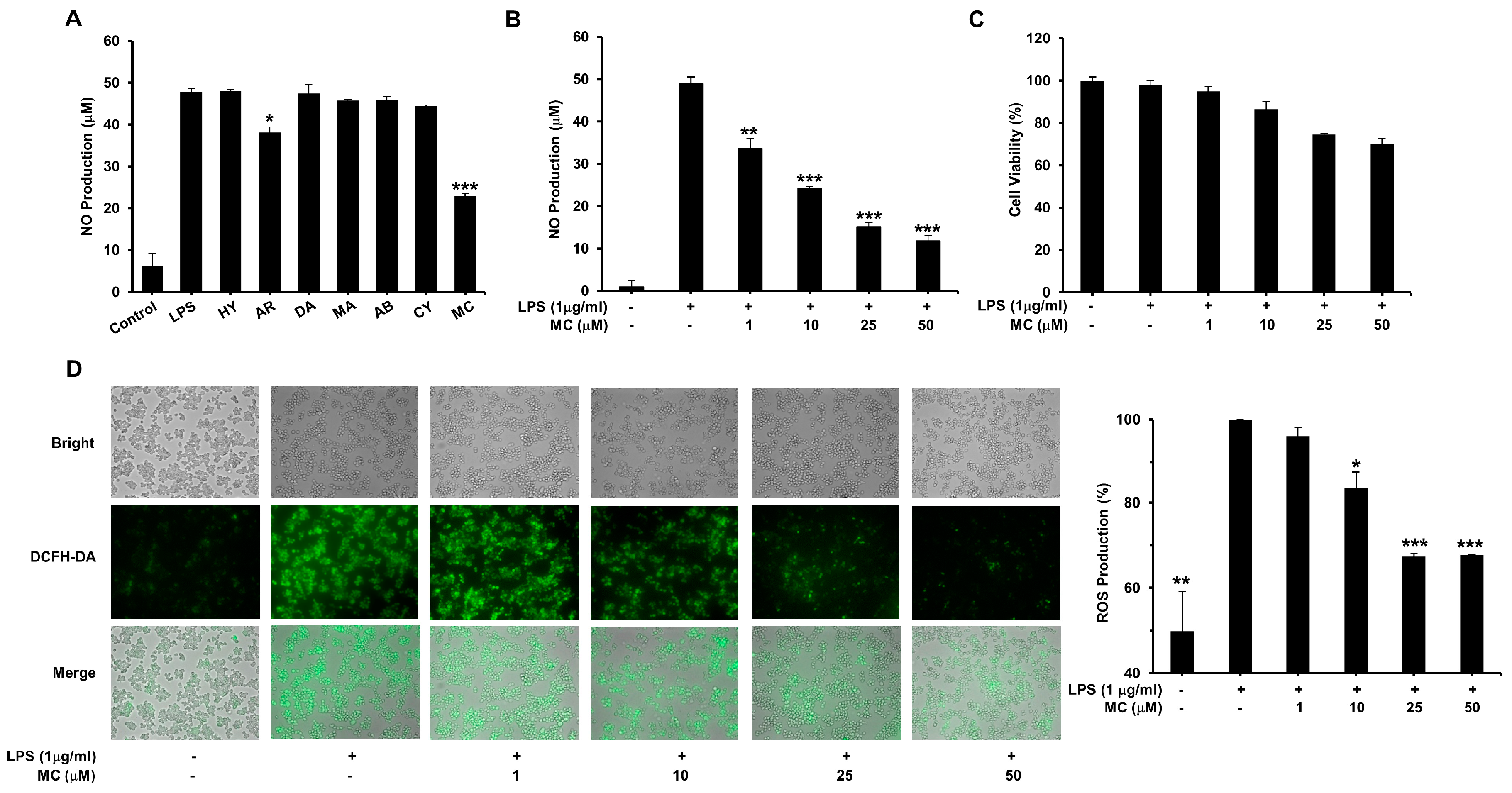
2.2. Effect of Natural Products on the Generation of NO
2.3. MC Reduces LPS-Induced ROS Production
2.4. MC Inhibits LPS-Induced mRNA and Protein Expression of iNOS and COX-2
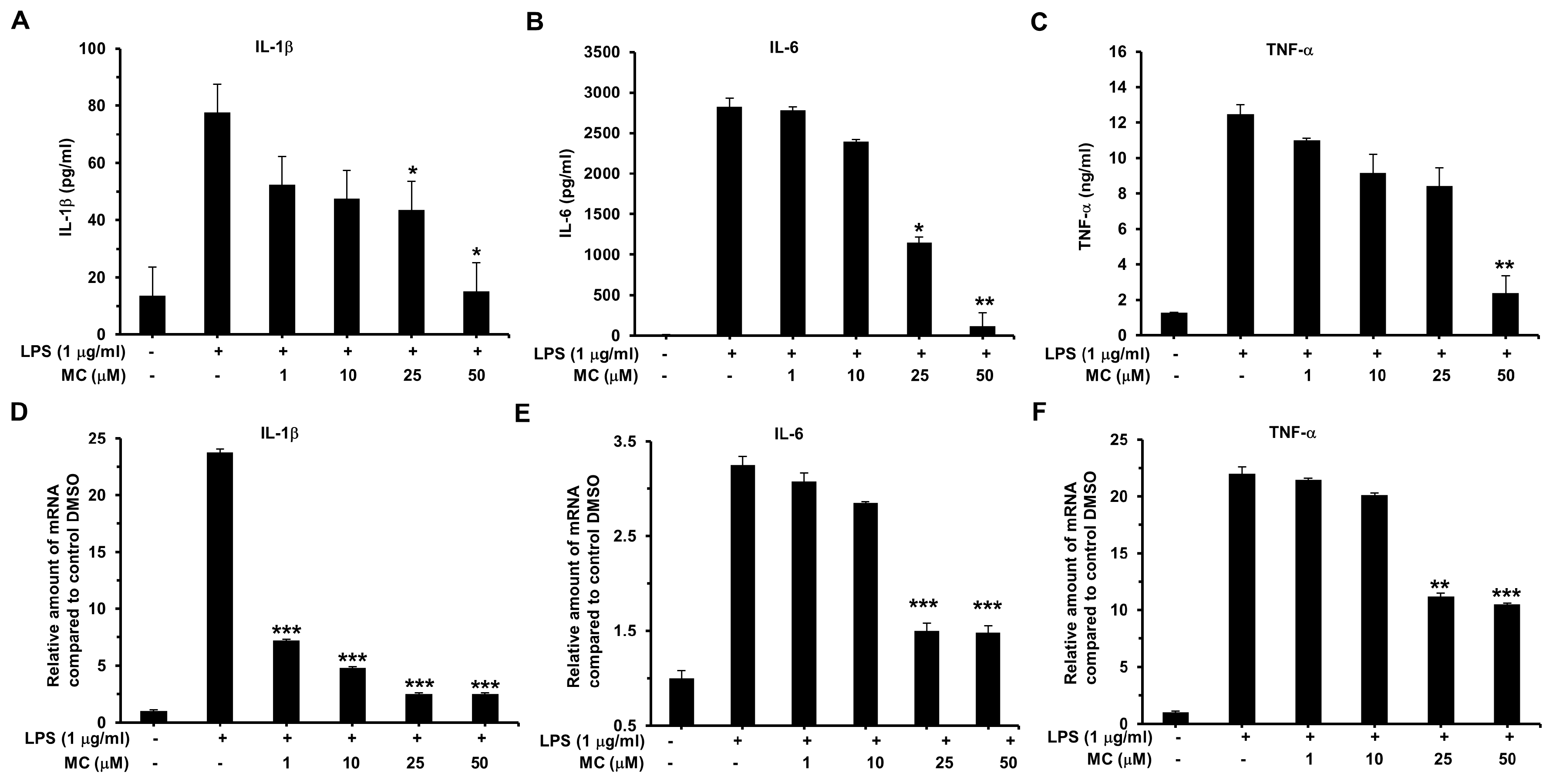
2.5. MC Suppresses LPS-Induced Pro-Inflammatory Cytokines Expression
2.6. MC Inhibits LPS-Induced TLR4 Expression and NF-κB Activation
2.7. MC Inhibits LPS-Induced MAPKs Pathways Activation
3. Discussion
4. Experimental Section
4.1. Materials and Reagents
4.2. Cell Culture
4.3. Nitrite Assay
4.4. Cell Viability Assay
4.5. Measurement of the Content of ROS
4.6. Cytokine Quantification
4.7. RNA Extraction and Reverse Transcriptase (RT-PCR)
4.8. Extraction of Total, Cytosol, and Nuclear Proteins
4.9. Western Blot Analysis
4.10. NF-κB Luciferase Reporter Assay
4.11. Immunofluorescence Analysis
4.12. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AB | Artoheterophyllin B |
| AR | Artocarpin |
| COX-2 | cyclooxygenase-2 |
| CY | Cycloheterophyllin |
| DA | Dadahol A |
| ERK | extracellular signal-regulated kinase |
| FBS | fetal bovine serum |
| HY | 25-Hydroxycycloart-23-en-3-one |
| IL-1β | interleukin-1β |
| IL-6 | interleukin-6 |
| iNOS | inducible nitric oxide synthase |
| JNK | c-Jun N-terminal kinase |
| LPS | lipopolysaccharide |
| MA | Morachalcone A |
| MAPKs | mitogen activated protein kinases |
| MC | Moracin C |
| NO | nitric oxide |
| NF-κB | Nuclear Factor-κB |
| ROS | reactive oxygen species |
| TLRs | Toll-like receptors |
| TNF-α | tumor necrosis factor α |
References
- Killeen, M.J.; Linder, M.; Pontoniere, P.; Crea, R. NF-κβ signaling and chronic inflammatory diseases: Exploring the potential of natural products to drive new therapeutic opportunities. Drug Discov. Today 2014, 19, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Jayasooriya, R.G.P.T.; Lee, K.-T.; Lee, H.-J.; Choi, Y.H.; Jeong, J.-W.; Kim, G.-Y. Anti-inflammatory effects of β-hydroxyisovalerylshikonin in BV2 microglia are mediated through suppression of the PI3K/Akt/NF-κB pathway and activation of the Nrf2/HO-1 pathway. Food Chem. Toxicol. 2014, 65, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Nathan, C. Points of control in inflammation. Nature 2002, 420, 846–852. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.-S.; Xiang, Y.; Cui, Y.-L.; Lin, K.-M.; Zhang, X.-F. Dietary blue pigments derived from genipin, attenuate inflammation by inhibiting LPS-induced iNOS and COX-2 expression via the NF-κB inactivation. PLoS ONE 2012, 7, e34122. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.H.; Hong, H.M.; Lin, C.L.; Jhang, A.Z.; Tsai, J.H.; Badmaev, V.; Nagabhushanam, K.; Ho, C.T.; Chen, W.J. Se-methylselenocysteine inhibits lipopolysaccharide-induced NF-κB activation and iNOS induction in RAW 264.7 murine macrophages. Mol. Nutr. Food Res. 2011, 55, 723–732. [Google Scholar] [CrossRef] [PubMed]
- McInturff, J.E.; Modlin, R.L.; Kim, J. The role of toll-like receptors in the pathogenesis and treatment of dermatological disease. J. Investig. Dermatol. 2005, 125, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Tak, P.P.; Firestein, G.S. NF-κB: A key role in inflammatory diseases. J. Clin. Investig. 2001, 107, 7. [Google Scholar] [CrossRef] [PubMed]
- Viatour, P.; Merville, M.-P.; Bours, V.; Chariot, A. Phosphorylation of NF-κB and IκB proteins: Implications in cancer and inflammation. Trends Biochem. Sci. 2005, 30, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, T.; Fong, C. The resolution of inflammation: Anti-inflammatory roles for NF-κB. Int. J. Biochem. Cell Biol. 2010, 42, 519–523. [Google Scholar] [CrossRef] [PubMed]
- Thalhamer, T.; McGrath, M.; Harnett, M. MAPKs and their relevance to arthritis and inflammation. Rheumatology 2008, 47, 409–414. [Google Scholar] [CrossRef] [PubMed]
- DebRoy, A.; Vogel, S.M.; Soni, D.; Sundivakkam, P.C.; Malik, A.B.; Tiruppathi, C. Endotoxin-induced STIM1 expression in endothelial cells requires cooperative signaling of transcription factors NF-κB and AP1/c-Fos and mediates lung vascular hyper-permeability. J. Biol. Chem. 2014, 289, 24188–24201. [Google Scholar] [CrossRef] [PubMed]
- Hwang, P.-A.; Chien, S.-Y.; Chan, Y.-L.; Lu, M.-K.; Wu, C.-H.; Kong, Z.-L.; Wu, C.-J. Inhibition of lipopolysaccharide (LPS)-induced inflammatory responses by sargassum hemiphyllum sulfated polysaccharide extract in RAW 264.7 macrophage cells. J. Agric. Food Chem. 2011, 59, 2062–2068. [Google Scholar] [CrossRef] [PubMed]
- Baker, D.D.; Chu, M.; Oza, U.; Rajgarhia, V. The value of natural products to future pharmaceutical discovery. Nat. Prod. Rep. 2007, 24, 1225–1244. [Google Scholar] [CrossRef] [PubMed]
- Li, J.W.-H.; Vederas, J.C. Drug discovery and natural products: End of an era or an endless frontier? Science 2009, 325, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, K.; Holmquist, L.; Steele, M.; Stuchbury, G.; Berbaum, K.; Schulz, O.; Benavente García, O.; Castillo, J.; Burnell, J.; Garcia Rivas, V. Plant-derived polyphenols attenuate lipopolysaccharide-induced nitric oxide and tumour necrosis factor production in murine microglia and macrophages. Mol. Nutr. Food Res. 2008, 52, 427–438. [Google Scholar] [CrossRef] [PubMed]
- Baliga, M.S.; Shivashankara, A.R.; Haniadka, R.; Dsouza, J.; Bhat, H.P. Phytochemistry, nutritional and pharmacological properties of Artocarpus heterophyllus Lam (jackfruit): A review. Food Res. Int. 2011, 44, 1800–1811. [Google Scholar] [CrossRef]
- Wei, B.-L.; Weng, J.-R.; Chiu, P.-H.; Hung, C.-F.; Wang, J.-P.; Lin, C.-N. Antiinflammatory flavonoids from artocarpus heterophyllus and artocarpus communis. J. Agric. Food Chem. 2005, 53, 3867–3871. [Google Scholar] [CrossRef] [PubMed]
- Likhitwitayawuid, K.; Sritularak, B.; Benchanak, K.; Lipipun, V.; Mathew, J.; Schinazi, R.F. Phenolics with antiviral activity from Millettia erythrocalyx and Artocarpus lakoocha. Nat. Prod. Res. 2005, 19, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Widyawaruyanti, A.; Kalauni, S.K.; Awale, S.; Nindatu, M.; Zaini, N.C.; Syafruddin, D.; Asih, P.B.S.; Tezuka, Y.; Kadota, S. New prenylated flavones from Artocarpus champeden, and their antimalarial activity in vitro. J. Nat. Med. 2007, 61, 410–413. [Google Scholar] [CrossRef]
- Furlan, M.; Roque, N.F.; Wolter Filho, W. Cycloartane derivatives from guarea trichilioides. Phytochemistry 1993, 32, 1519–1522. [Google Scholar] [CrossRef]
- Ko, F.N.; Cheng, Z.J.; Lin, C.N.; Teng, C.M. Scavenger and antioxidant properties of prenylflavones isolated from artocarpus heterophyllus. Free Radic. Biol. Med. 1998, 25, 160–168. [Google Scholar] [CrossRef]
- Zheng, Z.-P.; Chen, S.; Wang, S.; Wang, X.-C.; Cheng, K.-W.; Wu, J.-J.; Yang, D.; Wang, M. Chemical components and tyrosinase inhibitors from the twigs of artocarpus heterophyllus. J. Agric. Food Chem. 2009, 57, 6649–6655. [Google Scholar] [CrossRef] [PubMed]
- Han, A.-R.; Kang, Y.-J.; Windono, T.; Lee, S.K.; Seo, E.-K. Prenylated Flavonoids from the Heartwood of artocarpus c ommunis with Inhibitory Activity on Lipopolysaccharide-Induced Nitric Oxide Production. J. Nat. Prod. 2006, 69, 719–721. [Google Scholar] [CrossRef] [PubMed]
- Lucas, K.; Maes, M. Role of the Toll Like receptor (TLR) radical cycle in chronic inflammation: Possible treatments targeting the TLR4 pathway. Mol. Neurobiol. 2013, 48, 190–204. [Google Scholar] [CrossRef] [PubMed]
- Xie, Q.-W.; Nathan, C. The high-output nitric oxide pathway: Role and regulation. J. Leukoc. Biol. 1994, 56, 576–582. [Google Scholar] [PubMed]
- Ghosh, S.; Karin, M. Missing pieces in the NF-κB puzzle. Cell 2002, 109, 81–96. [Google Scholar] [CrossRef]
- Kim, K.-S.; Cui, X.; Lee, D.-S.; Ko, W.; Sohn, J.H.; Yim, J.H.; An, R.-B.; Kim, Y.-C.; Oh, H. Inhibitory effects of benzaldehyde derivatives from the marine fungus Eurotium sp. SF-5989 on inflammatory mediators via the induction of heme oxygenase-1 in lipopolysaccharide-stimulated RAW264. 7 macrophages. Int. J. Mol. Sci. 2014, 15, 23749–23765. [Google Scholar] [CrossRef] [PubMed]
- You, R.; Long, W.; Lai, Z.; Sha, L.; Wu, K.; Yu, X.; Lai, Y.; Ji, H.; Huang, Z.; Zhang, Y. Discovery of a potential anti-inflammatory agent: 3-oxo-29-noroleana-1, 9 (11), 12-trien-2, 20-dicarbonitrile. J. Med. Chem. 2013, 56, 1984–1995. [Google Scholar] [CrossRef] [PubMed]
- Carter, A.B.; Knudtson, K.L.; Monick, M.M.; Hunninghake, G.W. The p38 mitogen-activated protein kinase is required for NF-κB-dependent gene expression THE ROLE OF TATA-BINDING PROTEIN (TBP). J. Biol. Chem. 1999, 274, 30858–30863. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, C.; Campisi, J. Chronic inflammation (inflammaging) and its potential contribution to age-associated diseases. J. Gerontol. A Biol. Sci. Med. Sci. 2014, 69, S4–S9. [Google Scholar] [CrossRef] [PubMed]
- Jagtap, U.; Bapat, V. Artocarpus: A review of its traditional uses, phytochemistry and pharmacology. J. Ethnopharmacol. 2010, 129, 142–166. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.-G.; Matsuzaki, K.; Takamatsu, S.; Kitanaka, S. Inhibitory effects of constituents from morus alba var. multicaulis on differentiation of 3T3-L1 cells and nitric oxide production in RAW264. 7 cells. Molecules 2011, 16, 6010–6022. [Google Scholar] [CrossRef] [PubMed]
- Baeuerle, P.A.; Baltimore, D. NF-κB: Ten years after. Cell 1996, 87, 13–20. [Google Scholar] [CrossRef]
- Kaminska, B. MAPK signalling pathways as molecular targets for anti-inflammatory therapy—From molecular mechanisms to therapeutic benefits. BBA-Proteins Proteom. 2005, 1754, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Rajapakse, N.; Kim, M.M.; Mendis, E.; Kim, S.K. Inhibition of inducible nitric oxide synthase and cyclooxygenase-2 in lipopolysaccharide-stimulated RAW264. 7 cells by carboxybutyrylated glucosamine takes place via down-regulation of mitogen-activated protein kinase-mediated nuclear factor-κB signaling. Immunology 2008, 123, 348–357. [Google Scholar] [CrossRef] [PubMed]
- Bhaskar, S.; Shalini, V.; Helen, A. Quercetin regulates oxidized LDL induced inflammatory changes in human PBMCs by modulating the TLR-NF-κB signaling pathway. Immunobiology 2011, 216, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, A.A.; Guan, X.Q.; Reis, J.C.; Papasian, C.J.; Jabre, S.; Morrison, D.C.; Qureshi, N. Inhibition of nitric oxide and inflammatory cytokines in LPS-stimulated murine macrophages by resveratrol, a potent proteasome inhibitor. Lipids Health Dis. 2012, 11, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Bone, R.C. Toward a theory regarding the pathogenesis of the systemic inflammatory response syndrome: What we do and do not know about cytokine regulation. Crit. Care Med. 1996, 24, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Chow, C.-W.; Herrera Abreu, M.T.; Suzuki, T.; Downey, G.P. Oxidative stress and acute lung injury. Am. J. Respir. Cell Mol. Biol. 2003, 29, 427–431. [Google Scholar] [CrossRef] [PubMed]
- Seifert, U.; Bialy, L.P.; Ebstein, F.; Bech-Otschir, D.; Voigt, A.; Schröter, F.; Prozorovski, T.; Lange, N.; Steffen, J.; Rieger, M. Immunoproteasomes preserve protein homeostasis upon interferon-induced oxidative stress. Cell 2010, 142, 613–624. [Google Scholar] [CrossRef] [PubMed]
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Di, A.; Gao, X.-P.; Qian, F.; Kawamura, T.; Han, J.; Hecquet, C.; Richard, D.Y.; Vogel, S.M.; Malik, A.B. The redox-sensitive cation channel TRPM2 modulates phagocyte ROS production and inflammation. Nat. Immunol. 2012, 13, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Bayr, H. Reactive oxygen species. Crit. Care Med. 2005, 33, 498–501. [Google Scholar] [CrossRef]
- LeBel, C.P.; Ischiropoulos, H.; Bondy, S.C. Evaluation of the probe 2′,7′-dichlorofluorescin as an indicator of reactive oxygen species formation and oxidative stress. Chem. Res. Toxicol. 1992, 5, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Khor, T.O.; Saw, C.L.L.; Lin, W.; Wu, T.; Huang, Y.; Kong, A.-N.T. Role of Nrf2 in suppressing LPS-induced inflammation in mouse peritoneal macrophages by polyunsaturated fatty acids docosahexaenoic acid and eicosapentaenoic acid. Mol. Pharm. 2010, 7, 2185–2193. [Google Scholar] [CrossRef] [PubMed]
- Trompouki, E.; Hatzivassiliou, E.; Tsichritzis, T.; Farmer, H.; Ashworth, A.; Mosialos, G. CYLD is a deubiquitinating enzyme that negatively regulates NF-κB activation by TNFR family members. Nature 2003, 424, 793–796. [Google Scholar] [CrossRef] [PubMed]




© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yao, X.; Wu, D.; Dong, N.; Ouyang, P.; Pu, J.; Hu, Q.; Wang, J.; Lu, W.; Huang, J. Moracin C, A Phenolic Compound Isolated from Artocarpus heterophyllus, Suppresses Lipopolysaccharide-Activated Inflammatory Responses in Murine Raw264.7 Macrophages. Int. J. Mol. Sci. 2016, 17, 1199. https://doi.org/10.3390/ijms17081199
Yao X, Wu D, Dong N, Ouyang P, Pu J, Hu Q, Wang J, Lu W, Huang J. Moracin C, A Phenolic Compound Isolated from Artocarpus heterophyllus, Suppresses Lipopolysaccharide-Activated Inflammatory Responses in Murine Raw264.7 Macrophages. International Journal of Molecular Sciences. 2016; 17(8):1199. https://doi.org/10.3390/ijms17081199
Chicago/Turabian StyleYao, Xue, Dang Wu, Ningning Dong, Ping Ouyang, Jiaqian Pu, Qian Hu, Jingyuan Wang, Weiqiang Lu, and Jin Huang. 2016. "Moracin C, A Phenolic Compound Isolated from Artocarpus heterophyllus, Suppresses Lipopolysaccharide-Activated Inflammatory Responses in Murine Raw264.7 Macrophages" International Journal of Molecular Sciences 17, no. 8: 1199. https://doi.org/10.3390/ijms17081199