Toxicological Considerations, Toxicity Assessment, and Risk Management of Inhaled Nanoparticles
Abstract
:1. Introduction
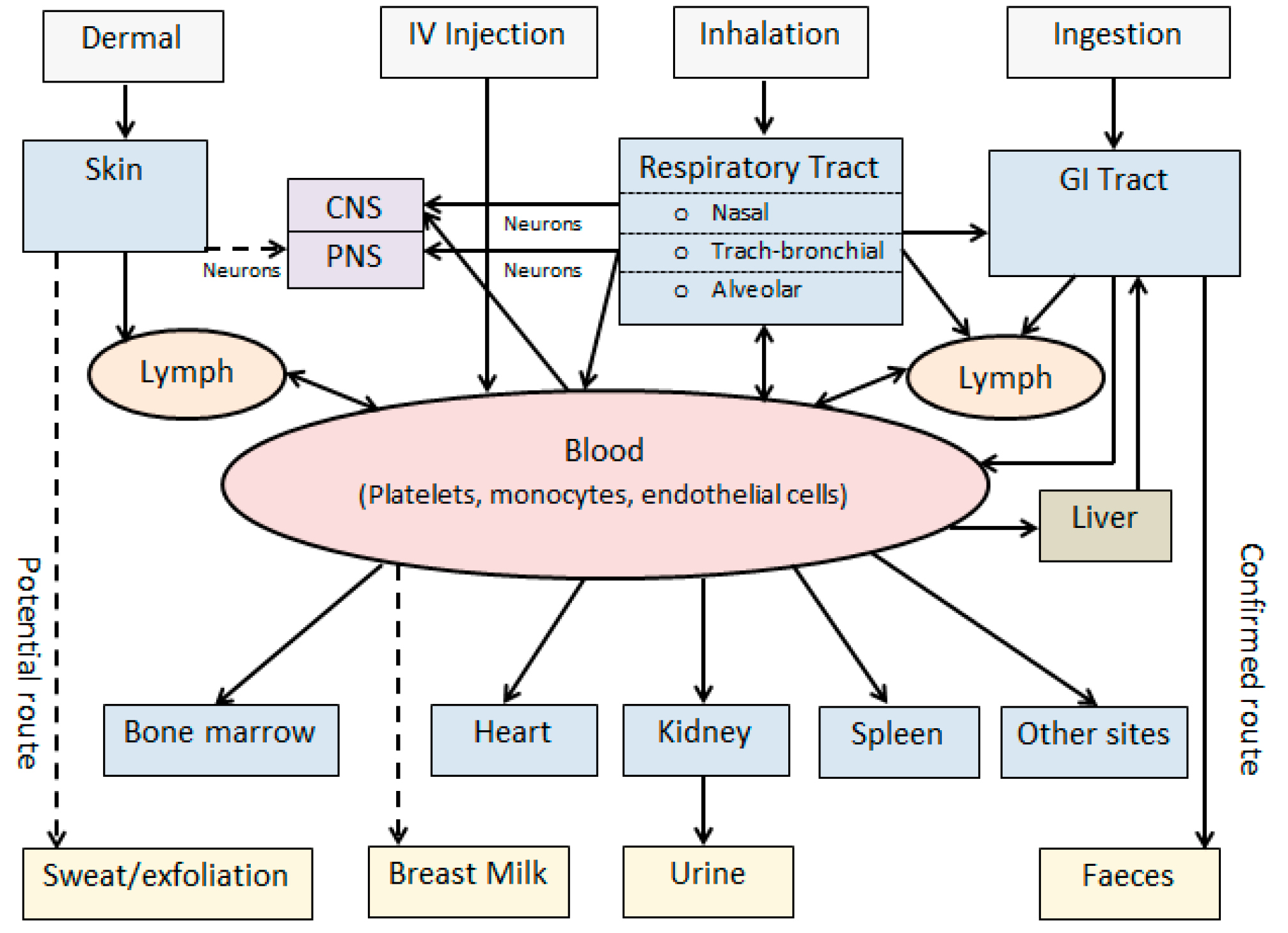
2. Nanoparticle Exposure and Biokinetic Pathways
3. Nanotoxicity and the Potential Mechanisms
4. Toxicity Assessment of NPs
5. Risk Management of NPs
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- ISO. ISO/TR 27687. Nanotechnologies—Terminology and Definitions for Nano-Objects, Nanoparticle, Nanofibre and Nanoplate, 1st ed.; the International Organization for Standardization: Geneva, Switzerland, 2008. [Google Scholar]
- ISO. ISO/TR 80004-2. Nanotechnologies—Vocabulary—Part 2: Nano-Objects, 1st ed.; the International Organisation for Standardization: Geneva, Switzerland, 2015. [Google Scholar]
- Oberdorster, G.; Kane, A.B.; Klaper, R.D.; Hurt, R.H. Nanotoxicology. In Casarett and Doull’s Toxicology—The Basic Science of Poisons; Klaassen, C.D., Ed.; McGraw Hill: New York, NY, USA, 2013; pp. 1189–1229. [Google Scholar]
- Renn, O.; Roco, M. White Paper on Nanotechnology Risk Governance; International Risk Governance Council: Geneva, Switzerland, 2006. [Google Scholar]
- Karn, B.; Masciangioli, T.; Zhang, W.; Colvin, V.; Alivisatos, P. Nanotechnology and the Environment; Applications and Implications; American Chemical Society: Washington, DC, USA, 2005. [Google Scholar]
- Hougaard, K.S.; Campagnolo, L.; Chavatte-Palmer, P.; Tarrade, A.; Rousseau-Ralliard, D.; Valentino, S.; Park, M.V.; de Jong, W.H.; Wolterink, G.; Piersma, A.H.; et al. A perspective on the developmental toxicity of inhaled nanoparticles. Reprod. Toxicol. 2015, 56, 118–140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Linkov, I.; Satterstrom, F.K.; Corey, L.M. Nanotoxicology and nanomedicine: Making hard decisions. Nanomedicine 2008, 4, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Beck-Broichsitter, M.; Merkel, O.M.; Kissel, T. Controlled pulmonary drug and gene delivery using polymeric nano-carriers. J. Control. Release 2012, 161, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Puri, A. Nanoparticles: Crossing barriers and membrane interactions. Mol. Membr. Biol. 2010, 27, 213–214. [Google Scholar] [CrossRef] [PubMed]
- Drobne, D. Nanotoxicology for safe and sustainable nanotechnology. Arh. Hig. Rada Toksikol. 2007, 58, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Mazaheri, M.; Eslahi, N.; Ordikhani, F.; Tamjid, E.; Simchi, A. Nanomedicine applications in orthopedic medicine: State of the art. Int. J. Nanomed. 2015, 10, 6039–6053. [Google Scholar]
- Khalili Fard, J.; Jafari, S.; Eghbal, M.A. A review of molecular mechanisms involved in toxicity of nanoparticles. Adv. Pharm. Bull. 2015, 5, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Accomasso, L.; Gallina, C.; Turinetto, V.; Giachino, C. Stem cell tracking with nanoparticles for regenerative medicine purposes: An overview. Stem Cells Int. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Oberdorster, G.; Oberdorster, E.; Oberdorster, J. Nanotoxicology: An emerging discipline evolving from studies of ultrafine particles. Environ. Health Perspect. 2005, 113, 823–839. [Google Scholar] [CrossRef] [PubMed]
- Warheit, D.B. Nanoparticles health impacts. Mater. Today 2004, 7, 32–35. [Google Scholar] [CrossRef]
- Dechsakulthorn, F.; Hayes, A.; Bakand, S.; Joeng, L.; Winder, C. In vitro cytotoxicity of selected nanoparticles using human skin fibroblasts. Altern. Anim. Test. Exp. 2008, 14, 397–400. [Google Scholar]
- Aufderheide, M.; Halter, B.; Mohle, N.; Hochrainer, D. The CULTEX RFS: A comprehensive technical approach for the in vitro exposure of airway epithelial cells to the particulate matter at the air-liquid interface. BioMed Res. Int. 2013, 2013, 734137. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Li, X.; Du, X. Exposure to nanoparticles is related to pleural effusion, pulmonary fibrosis and granuloma. Eur. Respir. J. 2009, 34, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Rinaldo, M.; Andujar, P.; Lacourt, A.; Martinon, L.; Canal Raffin, M.; Dumortier, P.; Pairon, J.C.; Brochard, P. Perspectives in biological monitoring of inhaled nanosized particles. Ann. Occup. Hyg. 2015, 59, 669–680. [Google Scholar] [CrossRef] [PubMed]
- Bakand, S.; Hayes, A.; Dechsakulthorn, F. Nanoparticles: A review of particle toxicology following inhalation exposure. Inhal. Toxicol. 2012, 24, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Hayes, A.J.; Bakand, S. Toxicological perspectives of inhaled therapeutics and nanoparticles. Expert Opin. Drug Metab. Toxicol. 2014, 10, 933–947. [Google Scholar] [CrossRef] [PubMed]
- Crosera, M.; Bovenzi, M.; Maina, G.; Adami, G.; Zanette, C.; Florio, C.; Filon Larese, F. Nanoparticle dermal absorption and toxicity: A review of the literature. Int. Arch. Occup. Environ. Health 2009, 82, 1043–1055. [Google Scholar] [CrossRef] [PubMed]
- Asgharian, B.; Price, O.; Oberdorster, G. A modeling study of the effect of gravity on airflow distribution and particle deposition in the lung. Inhal. Toxicol. 2006, 18, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Siegmann, K.; Scherrer, L.; Siegmann, H.C. Physical and chemical properties of airborne nanoscale particles and how to measure the impact on human health. J. Mol. Struct. 1999, 458, 191–201. [Google Scholar] [CrossRef]
- Rozman, K.K.; Klaassen, C.D. Absorption, distribution and excretion of toxicants. In Casarett and Doull's Toxicology: The Basic Science of Poisons, 6th ed.; Klaassen, C.D., Ed.; McGraw-Hill: New York, NY, USA, 2001; pp. 105–132. [Google Scholar]
- Witschi, H.P. Toxic responses of the respiratory system. In Casarett and Doull's Toxicology: The Basic Science of Poisons, 6th ed.; Klaassen, C.D., Ed.; McGraw-Hill: New York, NY, USA, 2001; pp. 515–534. [Google Scholar]
- Lambre, C.R.; Auftherheide, M.; Bolton, R.E.; Fubini, B.; Haagsman, H.P.; Hext, P.M.; Jorissen, M.; Landry, Y.; Morin, J.P.; Nemery, B.; et al. In vitro tests for respiratory toxicity, the report and recommendations of ECVAM workshop 18. Altern. Lab. Anim. 1996, 24, 671–681. [Google Scholar]
- Bakand, S.; Winder, C.; Khalil, C.; Hayes, A. Toxicity assessment of industrial chemicals and airborne contaminants: Transition from in vivo to in vitro test methods: A review. Inhal. Toxicol. 2005, 17, 775–787. [Google Scholar] [CrossRef] [PubMed]
- Blank, F.; Gehr, P.; Rutishauser, R.R. In Vitro Human Lung Cell Culture Models to Study the Toxic Potential of Nanoparticles; John Wily & Sons Ltd.: Chichester, UK, 2009. [Google Scholar]
- Lundborg, M.; Johard, U.; Lastbom, L.; Gerde, P.; Camner, P. Human alveolar macrophage phagocytic function is impaired by aggregates of ultrafine carbon particles. Environ. Res. 2001, 86, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Bergeron, S.; Archambault, D.P. Canadian Stewardship Practices for Environmental Nanotechnology; Science-Metrix: Montreal, QC, Canada, 2005. [Google Scholar]
- Brown, D.M.; Kinloch, J.A.; Bangert, U.; Windle, A.H.; Walter, D.M.; Walker, G.S.; Scotchford, C.A.; Donaldson, K.; Stone, V. An in vitro study of the potential of carbon nanotubes and nanofibres to induce inflammatory mediators and frustrated phagocytosis. Carbon 2007, 45, 1743–1756. [Google Scholar] [CrossRef]
- Mublfeld, C.; Gebr, P.; Rutishauser, B.R. Translocation and cellular entering mechanisms of nanoparticles in the respiratory tract. Swiss Med. Wkly. 2008, 138, 387–391. [Google Scholar]
- Kan, H.; London, S.J.; Chen, G.; Zhang, Y.; Song, G.; Zhao, N.; Jiang, L.; Chen, B. Season, sex, age, and education as modifiers of the effects of outdoor air pollution on daily mortality in shanghai, china: The public health and air pollution in Asia (PAPA) study. Environ. Health Perspect. 2008, 116, 1183–1188. [Google Scholar] [CrossRef] [PubMed]
- Bachler, G.; Losert, S.; Umehara, Y.; von Goetz, N.; Rodriguez-Lorenzo, L.; Petri-Fink, A.; Rothen-Rutishauser, B.; Hungerbuehler, K. Translocation of gold nanoparticles across the lung epithelial tissue barrier: Combining in vitro and in silico methods to substitute in vivo experiments. Part. Fibre Toxicol. 2015, 12, 18. [Google Scholar] [CrossRef] [PubMed]
- Dockery, D.W.; Pope, C.A.; Xu, X.; Spengler, J.D.; Ware, J.H.; Fay, M.E.; Ferris, B.G., Jr.; Speizer, F.E. An association between air pollution and mortality in six U.S. Cities. N. Engl. J. Med. 1993, 329, 1753–1759. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, A.J.; Johnston, S.L. Air pollution and infection in respiratory illness. Br. Med. Bull. 2003, 68, 95–112. [Google Scholar] [CrossRef] [PubMed]
- Borm, P.J.; Robbins, D.; Haubold, S.; Kuhlbusch, T.; Fissan, H.; Donaldson, K.; Schins, R.; Stone, V.; Kreyling, W.; Lademann, J.; et al. The potential risks of nanomaterials: A review carried out for ECETOC. Part. Fibre Toxicol. 2006, 3, 11. [Google Scholar] [CrossRef] [PubMed]
- Ruckerl, R.; Schneider, A.; Breitner, S.; Cyrys, J.; Peters, A. Health effects of particulate air pollution: A review of epidemiological evidence. Inhal. Toxicol. 2011, 23, 555–592. [Google Scholar] [CrossRef] [PubMed]
- Stone, V.; Johnston, H.; Clift, M.J. Air pollution, ultrafine and nanoparticle toxicology: Cellular and molecular interactions. IEEE Trans. Nanobiosci. 2007, 6, 331–340. [Google Scholar] [CrossRef]
- Lawson, G.; Wang, H. Public health impact of diesel exhast: Toxicity of nano-sized diesel exhaust particles—Part 1. Environ. Health 2006, 6, 17–21. [Google Scholar]
- Donaldson, K.; Tran, L.; Jimenez, L.A.; Duffin, R.; Newby, D.E.; Mills, N.; MacNee, W.; Stone, V. Combustion-derived nanoparticles: A review of their toxicology following inhalation exposure. Part. Fibre Toxicol. 2005, 2, 10. [Google Scholar] [CrossRef] [PubMed]
- Nel, A.; Xia, T.; Madler, L.; Li, N. Toxic potential of materials at the nanolevel. Science 2006, 311, 622–627. [Google Scholar] [CrossRef] [PubMed]
- Karlson, H.L.; Cronholm, P.; Gustafsson, J.; Moller, L. Copper oxide nanoparticles are highly toxic: A comparison between metal oxide nanoparticles and carbon nanotubes. Chem. Res. Toxicol. 2008, 21, 1726–1732. [Google Scholar] [CrossRef] [PubMed]
- David, A.; Wagner, G.R. Respiratory system. In Encyclopaedia of Occupational Health and Safety, 4th ed.; Stellman, J.M., Ed.; International Labour Office: Geneva, Switzerland, 1998. [Google Scholar]
- Winder, C. Toxicology of gases, vapours and particulates. In Occupational Toxicology, 2nd ed.; Winder, C., Stacey, N.H., Eds.; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Peters, K.; Unger, R.E.; Kirkpatrick, C.J.; Gatti, A.M.; Monari, E. Effects of nano-scaled particles on endothelial cell function in vitro: Studies on viability, proliferation and inflammation. J. Mater. Sci. Mater. Med. 2004, 15, 321–325. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S.M.; Hess, K.L.; Gearhart, J.M.; Geiss, K.T.; Schlager, J.J. In vitro toxicity of nanoparticles in BRL 3A rat liver cells. Toxicol. Vitr. 2005, 19, 975–983. [Google Scholar] [CrossRef] [PubMed]
- Gojova, A.; Guo, B.; Kota, R.S.; Rutledge, J.C.; Kennedy, I.M.; Barakat, A.I. Induction of inflammation in vascular endothelial cells by metal oxide nanoparticles: Effect of particle composition. Environ. Health Perspect. 2007, 115, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Geiser, M.; Rothen-Rutishauser, B.; Kapp, N.; Schurch, S.; Kreyling, W.; Schulz, H.; Semmler, M.; Im Hof, V.; Heyder, J.; Gehr, P. Ultrafine particles cross cellular membranes by nonphagocytic mechanisms in lungs and in cultured cells. Environ. Health Perspect. 2005, 113, 1555–1560. [Google Scholar] [CrossRef] [PubMed]
- Chithrani, D.B. Intracellular uptake, transport, and processing of gold nanostructures. Mol. Membr. Biol. 2010, 27, 299–311. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, K.; Stone, V.; Tran, C.L.; Kreyling, W.; Borm, P.J. Nanotoxicology. Occup. Environ. Med. 2004, 61, 727–728. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; von dem Bussche, A.; Hurt, R.H.; Kane, A.B.; Gao, H. Cell entry of one-dimensional nanomaterials occurs by tip recognition and rotation. Nat. Nanotechnol. 2011, 6, 714–719. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Xia, T.; Ntim, S.A.; Ji, Z.; Lin, S.; Meng, H.; Chung, C.H.; George, S.; Zhang, H.; Wang, M.; et al. Dispersal state of multiwalled carbon nanotubes elicits profibrogenic cellular responses that correlate with fibrogenesis biomarkers and fibrosis in the murine lung. ACS Nano 2011, 5, 9772–9787. [Google Scholar] [CrossRef] [PubMed]
- Fenoglio, I.; Fubini, B.; Ghibaudi, E.M.; Turci, F. Multiple aspects of the interaction of biomacromolecules with inorganic surfaces. Adv. Drug Deliv. Rev. 2011, 63, 1186–1209. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, K.; Stone, V.; Gilmour, P.S.; Brown, D.M.; MacNee, W. Ultrafine particles: Mechanisms of lung injury. Philos. Trans. R. Soc. A 2000, 358, 2741–2749. [Google Scholar] [CrossRef]
- Renwick, L.C.; Brown, D.; Clouter, A.; Donaldson, K. Increased inflammation and altered macrophage chemotactic responses caused by two ultrafine particle types. Occup. Environ. Med. 2004, 61, 442–447. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.Y.; Joachim, E.; Choi, H.; Kim, K. Toxicity of silica nanoparticles depends on size, dose, and cell type. Nanomedicine 2015, 11, 1407–1416. [Google Scholar] [CrossRef] [PubMed]
- Tolstoshev, A. Nanotechnology, Assessing the Environmental Risks for Australia; Earth Policy Centre, University of Melbourne: Melbourne, Australian, 2006. [Google Scholar]
- Donaldson, K.; Brown, D.; Clouter, A.; Duffin, R.; MacNee, W.; Renwick, L.; Tran, L.; Stone, V. The pulmonary toxicology of ultrafine particles. J. Aerosol Med. 2002, 15, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Dubey, P.; Matai, I.; Kumar, S.U.; Sachdev, A.; Bhushan, B.; Gopinath, P. Perturbation of cellular mechanistic system by silver nanoparticle toxicity: Cytotoxic, genotoxic and epigenetic potentials. Adv. Colloid Interface Sci. 2015, 221, 4–21. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Huang, Y.W.; Zhou, X.D.; Ma, Y. In vitro toxicity of silica nanoparticles in human lung cancer cells. Toxicol. Appl. Pharmacol. 2006, 217, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Thit, A.; Selck, H.; Bjerregaard, H.F. Toxic mechanisms of copper oxide nanoparticles in epithelial kidney cells. Toxicol. Vitr. 2015, 29, 1053–1059. [Google Scholar] [CrossRef] [PubMed]
- Sanganeria, P.; Sachar, S.; Chandra, S.; Bahadur, D.; Ray, P.; Khanna, A. Cellular internalization and detailed toxicity analysis of protein-immobilized iron oxide nanoparticles. J. Biomed. Mater. Res. B 2015, 103, 125–134. [Google Scholar] [CrossRef] [PubMed]
- McShan, D.; Ray, P.C.; Yu, H. Molecular toxicity mechanism of nanosilver. J. Food Drug Anal. 2014, 22, 116–127. [Google Scholar] [CrossRef] [PubMed]
- Boland, S.; Hussain, S.; Baeza-Squiban, A. Carbon black and titanium dioxide nanoparticles induce distinct molecular mechanisms of toxicity. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2014, 6, 641–652. [Google Scholar] [CrossRef] [PubMed]
- Mustajbegovic, J.; Zuskin, E.; Schachter, E.N.; Kern, J.; Vitale, K.; Ebling, Z.; Vrcic-Keglevic, M. Respiratory findings in chemical workers exposed to low concentrations of organic and inorganic air pollutants. Am. J. Ind. Med. 2000, 38, 431–440. [Google Scholar] [CrossRef]
- Khandoga, A.; Stoeger, T.; Khandoga, A.G.; Bihari, P.; Karg, E.; Ettehadieh, D.; Lakatos, S.; Fent, J.; Schulz, H.; Krombach, F. Platelet adhesion and fibrinogen deposition in murine microvessels upon inhalation of nanosized carbon particles. J. Thromb. Haemost. 2010, 8, 1632–1640. [Google Scholar] [CrossRef] [PubMed]
- Brunner, T.J.; Wick, P.; Manser, P.; Spohn, P.; Grass, R.N.; Limbach, L.K.; Bruinink, A.; Stark, W.J. In vitro cytotoxicity of oxides nanoparticles: Comparison to asbestos, silical, and the effects of particle solubility. Environ. Sci. Technol. 2006, 40, 4374–4381. [Google Scholar] [CrossRef] [PubMed]
- Seaton, A. Nanotechnology and the occupational physician. Occup. Med. 2006, 56, 312–316. [Google Scholar] [CrossRef] [PubMed]
- Poland, C.A.; Duffin, R.; Kinloch, I.; Maynard, A.; Wallace, W.A.; Seaton, A.; Stone, V.; Brown, S.; Macnee, W.; Donaldson, K. Carbon nanotubes introduced into the abdominal cavity of mice show asbestos-like pathogenicity in a pilot study. Nat. Nanotechnol. 2008, 3, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Kettler, K.; Veltman, K.; van de Meent, D.; van Wezel, A.; Hendriks, A.J. Cellular uptake of nanoparticles as determined by particle properties, experimental conditions, and cell type. Environ. Toxicol. Chem. 2014, 33, 481–492. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.Y.; Ramachandran, G.; Kandlikar, M. The impact of toxicity testing costs on nanomaterial regulation. Environ. Sci. Technol. 2009, 43, 3030–3034. [Google Scholar] [CrossRef] [PubMed]
- Hayes, A.; Bakand, S.; Winder, C. Novel in vitro exposure techniques for toxicity testing and biomonitoring of airborne contaminants. In Drug Testing in Vitro-Breakthroughs and Trends in Cell Culture Technology; Marx, U., Sandig, V., Eds.; Wiley-VCH: Berlin, Germany, 2007; pp. 103–124. [Google Scholar]
- Stone, V.; Johnston, H.; Schins, R.P. Development of in vitro systems for nanotoxicology: Methodological considerations. Crit. Rev. Toxicol. 2009, 39, 613–626. [Google Scholar] [CrossRef] [PubMed]
- ICCVAM. Report of the International Workshop on in Vitro Methods for Assessing Acute Systemic Toxicity; Interagency Coordinating Committee on the Validation of Alternative Methods: Research Triangle Park, NC, USA, 2001. [Google Scholar]
- Bakand, S.; Hayes, A. Troubleshooting methods for toxicity testing of airborne chemicals in vitro. J. Pharmacol. Toxicol. Methods 2010, 61, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Sayes, C.M.; Reed, K.L.; Warheit, D.B. Assessing toxicity of fine and nanoparticles: Comparing in vitro measurements to in vivo pulmonary toxicity profiles. Toxicol. Sci. 2007, 97, 163–180. [Google Scholar] [CrossRef] [PubMed]
- Allen, C.B. In vitro models for lung toxicology. In Toxicology of the Lung, 4th ed.; Gardner, D.E., Ed.; Taylor & Francis: Boca Raton, FL, USA, 2006; pp. 107–150. [Google Scholar]
- Mondrinos, M.J.; Koutzaki, S.; Jiwanmall, E.; Li, M.; Dechadarevian, J.P.; Lelkes, P.I.; Finck, C.M. Engineering three-dimensional pulmonary tissue constructs. Tissue Eng. 2006, 12, 717–728. [Google Scholar] [CrossRef] [PubMed]
- Lazarovici, P.; Li, M.; Perets, A.; Mondrinos, M.J.; Lecht, S.; Koharski, C.D.; Bidez, P.R., III; Fink, C.M.; Lelkes, P.I. Intelligent biomatrices and engineered tissue constructs: In-vitro models for drug discovery and toxicity testing. In Drug Testing in Vitro-Breakthroughs and Trends in Cell Culture Technology; Marx, U., Sandig, V., Eds.; Wiley-VCH.: Berlin, Germany, 2007; pp. 3–51. [Google Scholar]
- Mondrinos, M.J.; Koutzaki, S.; Lelkes, P.I.; Finck, C.M. A tissue-engineered model of fetal distal lung tissue. Am. J. Physiol. Lung Cell Mol. Physiol. 2007, 293, L639–L650. [Google Scholar] [CrossRef] [PubMed]
- Aufderheide, M. Direct exposure methods for testing native atmospheres. Exp. Toxicol. Pathol. 2005, 57, 213–226. [Google Scholar] [CrossRef] [PubMed]
- Bakand, S.; Winder, C.; Khalil, C.; Hayes, A. A novel in vitro exposure technique for toxicity testing of selected volatile organic compounds. J. Environ. Monit. 2006, 8, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Bakand, S.; Winder, C.; Khalil, C.; Hayes, A. An experimental in vitro model for dynamic direct exposure of human cells to airborne contaminants. Toxicol. Lett. 2006, 165, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Lestari, F.; Green, A.R.; Chattopadhyay, G.; Hayes, A.J. An alternative method for fire smoke toxicity assessment using human lung cells. Fire Saf. J. 2006, 41, 605–615. [Google Scholar] [CrossRef]
- Lestari, F.; Markovic, B.; Green, A.R.; Chattopadhyay, G.; Hayes, A.J. Comparative assessment of three in vitro exposure methods for combustion toxicity. J. Appl. Toxicol. 2006, 26, 99–114. [Google Scholar] [CrossRef] [PubMed]
- Bakand, S.; Hayes, A.; Winder, C. An integrated in vitro approach for toxicity testing of airborne contaminants. J. Toxicol. Environ. Health 2007, 70, 1604–1612. [Google Scholar] [CrossRef] [PubMed]
- Bakand, S.; Winder, C.; Hayes, A. Comparative in vitro cytotoxicity assessment of selected gaseous compounds in human alveolar epithelial cells. Toxicol. Vitr. 2007, 21, 1341–1347. [Google Scholar] [CrossRef] [PubMed]
- Potera, C. More human, more humane: A new approach for testing airborne pollutants. Environ. Health Perspect. 2007, 115, A148–A151. [Google Scholar] [CrossRef] [PubMed]
- Tippe, A.; Heinzmann, U.; Roth, C. Deposition of fine and ultrafine aerosol particles during exposure at the air/cell interface. Aerosol Sci. 2002, 33, 207–218. [Google Scholar] [CrossRef]
- Bitterle, E.; Karg, E.; Schroeppel, A.; Kreyling, W.G.; Tippe, A.; Ferron, G.A.; Schmid, O.; Heyder, J.; Maier, K.L.; Hofer, T. Dose-controlled exposure of A549 epithelial cells at the air-liquid interface to airborne ultrafine carbonaceous particles. Chemosphere 2006, 65, 1784–1790. [Google Scholar] [CrossRef] [PubMed]
- Paur, H.R.; Mulhopt, S.; Weiss, C.; Diabate, S. In vitro exposure systems and bioassays for the assessment of toxicity of nanoparticles to the human lung. J. Consum. Prot. Food Saf. 2008, 3, 319–329. [Google Scholar] [CrossRef]
- Mulhopt, S.; Diabate, S.; Krebs, T.; Weiss, C.; Paur, H.R. Lung toxicity determination by in vitro exposure at the air liquid interface with an integrated online dose measurement. J. Phys. Conf. Ser. 2009, 170, 1. [Google Scholar] [CrossRef]
- Aufderheide, M.; Mohr, U. A modified CULTEX® system for the direct exposure of bacteria to inhalable substances. Exp. Toxicol. Pathol. 2004, 55, 451–454. [Google Scholar] [CrossRef] [PubMed]
- ISO. ISO/TR 12885. Nanotechnologies—Health and Safety Practices in Occupational Settings Relevant to Nanotechnologies, 1st ed.; the International Organization for Standardization: Geneva, Switzerland, 2008. [Google Scholar]
- ISO. ISO/TR 13121. Nanotechnologies—Nanomaterial Risk Evaluation, 1st ed.; the International Organization for Standardization: Geneva, Switzerland, 2011. [Google Scholar]
- Australia, S.W. How to Manage Work, Health and Safety Risks: Code of Practice; Safe Work SA: Canberra, Australia, 2011. [Google Scholar]
- Monteiro-Riveiere, N.A.; Inman, A.O. Challenges for assessing carbon nanomaterial toxicity to the skin. Carbon 2006, 44, 1070–1078. [Google Scholar] [CrossRef]
- Casey, A.; Herzog, E.; Davoren, M.; Lyng, F.M.; Byrne, H.J.; Chambers, G. Spectroscopic analysis confirms the interactions between single walled carbon nanotubes and various dyes commonly used to assess cytotoxicty. Carbon 2007, 45, 1425–1432. [Google Scholar] [CrossRef]
- Casey, A.; Herzog, E.; Lyng, F.M.; Byrne, H.J.; Chambers, G.; Davoren, M. Single walled carbon nanotubes induce indirect cytotoxicity by medium depletion in A549 lung cells. Toxicol. Lett. 2008, 179, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; von Dem Bussche, A.; Buechner, M.; Yan, A.; Kane, A.B.; Hurt, R.H. Adsorption of essential micronutrients by carbon nanotubes and the implications for nanotoxicity testing. Small 2008, 4, 721–727. [Google Scholar] [CrossRef] [PubMed]
- Englert, B.C. Nanomaterials and the environment: Uses, methods and measurement. J. Environ. Monit. 2007, 9, 1154–1161. [Google Scholar] [CrossRef] [PubMed]
- Cohen, B.S.; McCammon, J. Air Sampling Instruments for Evaluation of Atmospheric Contaminants, 9th ed.; ACGIH: Cincinnati, OH, USA, 2001. [Google Scholar]
- Sayes, C.M.; Gobin, A.M.; Ausman, K.D.; Mendez, J.; West, J.L.; Colvin, V.L. Nano-C60 cytotoxicity is due to lipid peroxidation. Biomaterials 2005, 26, 7587–7595. [Google Scholar] [CrossRef] [PubMed]
- Wysokinska, E.; Cichos, J.; Ziolo, E.; Bednarkiewicz, A.; Strzadala, L.; Karbowiak, M.; Hreniak, D.; Kalas, W. Cytotoxic interactions of bare and coated NaGdF4:Yb3+:Er3+ nanoparticles with macrophage and fibroblast cells. Toxicol. Vitr. 2016, 32, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Schilling, K.; Bradford, B.; Castelli, D.; Dufour, E.; Nash, J.F.; Pape, W.; Schulte, S.; Tooley, I.; van den Bosch, J.; Schellauf, F. Human safety review of “nano” titanium dioxide and zinc oxide. Photochem. Photobiol. Sci. 2010, 9, 495–509. [Google Scholar] [CrossRef] [PubMed]
- Sayes, C.M.; Liang, F.; Hudson, J.L.; Mendez, J.; Guo, W.; Beach, J.M.; Moore, V.C.; Doyle, C.D.; West, J.L.; Billups, W.E.; et al. Functionalization density dependence of single-walled carbon nanotubes cytotoxicity in vitro. Toxicol. Lett. 2006, 161, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Sasidharan, A.; Panchakarla, L.S.; Chandran, P.; Menon, D.; Nair, S.; Rao, C.N.; Koyakutty, M. Differential nano-bio interactions and toxicity effects of pristine versus functionalized graphene. Nanoscale 2011, 3, 2461–2464. [Google Scholar] [CrossRef] [PubMed]
- Shenava, A.; Sharma, M.; Shetty, V.; Shenoy, S. Silver nanoparticles: A boon in clinical medicine. J. Oral Res. Rev. 2015, 7, 35–38. [Google Scholar] [CrossRef]
- Foldbjerg, R.; Dang, D.A.; Autrup, H. Cytotoxicity and genotoxicity of silver nanoparticles in the human lung cancer cell line, A549. Arch. Toxicol. 2011, 85, 743–750. [Google Scholar] [CrossRef] [PubMed]
- Alarcon, E.I.; Vulesevic, B.; Argawal, A.; Ross, A.; Bejjani, P.; Podrebarac, J.; Ravichandran, R.; Phopase, J.; Suuronen, E.J.; Griffith, M. Coloured cornea replacements with anti-infective properties: Expanding the safe use of silver nanoparticles in regenerative medicine. Nanoscale 2016, 8, 6484–6489. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.N.; Poon, W.; Tavares, A.J.; McGilvray, I.D.; Chan, W.C. Nanoparticle-liver interactions: Cellular uptake and hepatobiliary elimination. J. Control. Release 2016. [Google Scholar] [CrossRef] [PubMed]
- Zalk, D.M.; Paik, S.Y.; Paul Swuste, P. Evaluating the control banding nanotool: A qualitative risk assessment method for controlling nanoparticle exposures. J. Nanopart. Res. 2009, 11, 1685–1704. [Google Scholar] [CrossRef]
- Ramachandran, G.; Ostraat, M.; Evans, D.E.; Methner, M.M.; O’Shaughnessy, P.; D’Arcy, J.; Geraci, C.L.; Stevenson, E.; Maynard, A.; Rickabaugh, K. A strategy for assessing workplace exposures to nanomaterials. J. Occup. Environ. Hyg. 2011, 8, 673–685. [Google Scholar] [CrossRef] [PubMed]
- ISO. ISO/TS 12901-1. Nanotechnologies—Occupational Risk Management Applied to Engineered Nanomaterial—Part 1: Principles and Approaches; The International Organization for Standardization: Geneva, Switzerland, 2012. [Google Scholar]
- ISO. ISO/TS 12901–2. Nanotechnologies—Occupational Risk Management Applied to Engineered Nanomaterial—Part 2: Use of the Control Banding Approach; The International Organization for Standardization: Geneva, Switzerland, 2014. [Google Scholar]
- Schulte, P.A.; Salamanca-Buentello, F. Ethical and scientific issues of nanotechnology in the workplace. Environ. Health Perspect. 2007, 115, 5–12. [Google Scholar] [CrossRef] [PubMed]
| Nano-Objects | Nanoparticle | Nanofibre or Nanotube | Nanoplate |
|---|---|---|---|
| Geometrical Characteristics | Isometric 3 ext. dimensions in nanoscale | One-dimensional (1D) 2 ext. dimensions in nanoscale | Two-dimensional (2D) 1 ext. dimension in nanoscale |
| Example | Bucky ball | Carbon nanotubes (CNTs) | Graphene |
| Nanomaterial Properties | Potential Biological Effects |
|---|---|
| Size/size distribution (aerodynamic, hydrodynamic) | Crossing tissue and cell membranes |
| Cellular injury | |
| Phagocytosis impairment, breakdown in defense mechanisms | |
| Migration to other organs | |
| Transportation of other environmental pollutants | |
| Surface properties Surface area/mass ratio | Increased reactivity |
| Increased toxicity | |
| Chemical composition Surface characteristics | ROS generation |
| Oxidative stress | |
| Inflammation | |
| Cytokine production | |
| Glutathione depletion | |
| Mitochondrial exhaustion | |
| Cellular injury | |
| Protein and DNA damage | |
| Insolubility or low water solubility | Bioaccumulation inside living systems such as human cells, tissues and lungs |
| Potential long-term effects | |
| Agglomeration/aggregation | Interruption of cellular processes |
| Cellular injury |
| Risk Management | Details of Each Step Relevant to Nanomaterials |
|---|---|
| Identify hazard | Describe nanomaterial & applications |
| Determine nanomaterial profiles | |
| Physicochemical profiles | |
| Hazard profiles | |
| Exposure profiles | |
| Evaluate risk | Based on the combination of: Identified hazards, exposure, potential risks |
| Exposure patterns: including likelihood and severity | |
| Control risk | Level 1: Eliminate the hazard |
| Eliminating the nanomaterial | |
| Level 2: Substitute, Isolate and engineering controls | |
| Substitute to a safer material, product or process | |
| Apply process containment | |
| Use local exhausted ventilation systems equipped with efficient filters (e.g., HEPA) | |
| Level 3: Reduce exposure by | |
| Administrative controls (e.g., develop Safety data sheets and safe work procedures) | |
| Personal protective equipment (e.g., appropriate gloves, eye and respiratory protection) | |
| Decide, document & act | Decide: Whether or in what capacity to continue development and production of the nanomaterial |
| Sharing information with the stakeholders | |
| Further information to be collected | |
| Review & adapt | Update the risk assessment process through: Regular reviews |
| Reviews triggered by specific events |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bakand, S.; Hayes, A. Toxicological Considerations, Toxicity Assessment, and Risk Management of Inhaled Nanoparticles. Int. J. Mol. Sci. 2016, 17, 929. https://doi.org/10.3390/ijms17060929
Bakand S, Hayes A. Toxicological Considerations, Toxicity Assessment, and Risk Management of Inhaled Nanoparticles. International Journal of Molecular Sciences. 2016; 17(6):929. https://doi.org/10.3390/ijms17060929
Chicago/Turabian StyleBakand, Shahnaz, and Amanda Hayes. 2016. "Toxicological Considerations, Toxicity Assessment, and Risk Management of Inhaled Nanoparticles" International Journal of Molecular Sciences 17, no. 6: 929. https://doi.org/10.3390/ijms17060929





