Intense Resistance Exercise Promotes the Acute and Transient Nuclear Translocation of Small Ubiquitin-Related Modifier (SUMO)-1 in Human Myofibres
Abstract
:1. Introduction
2. Results
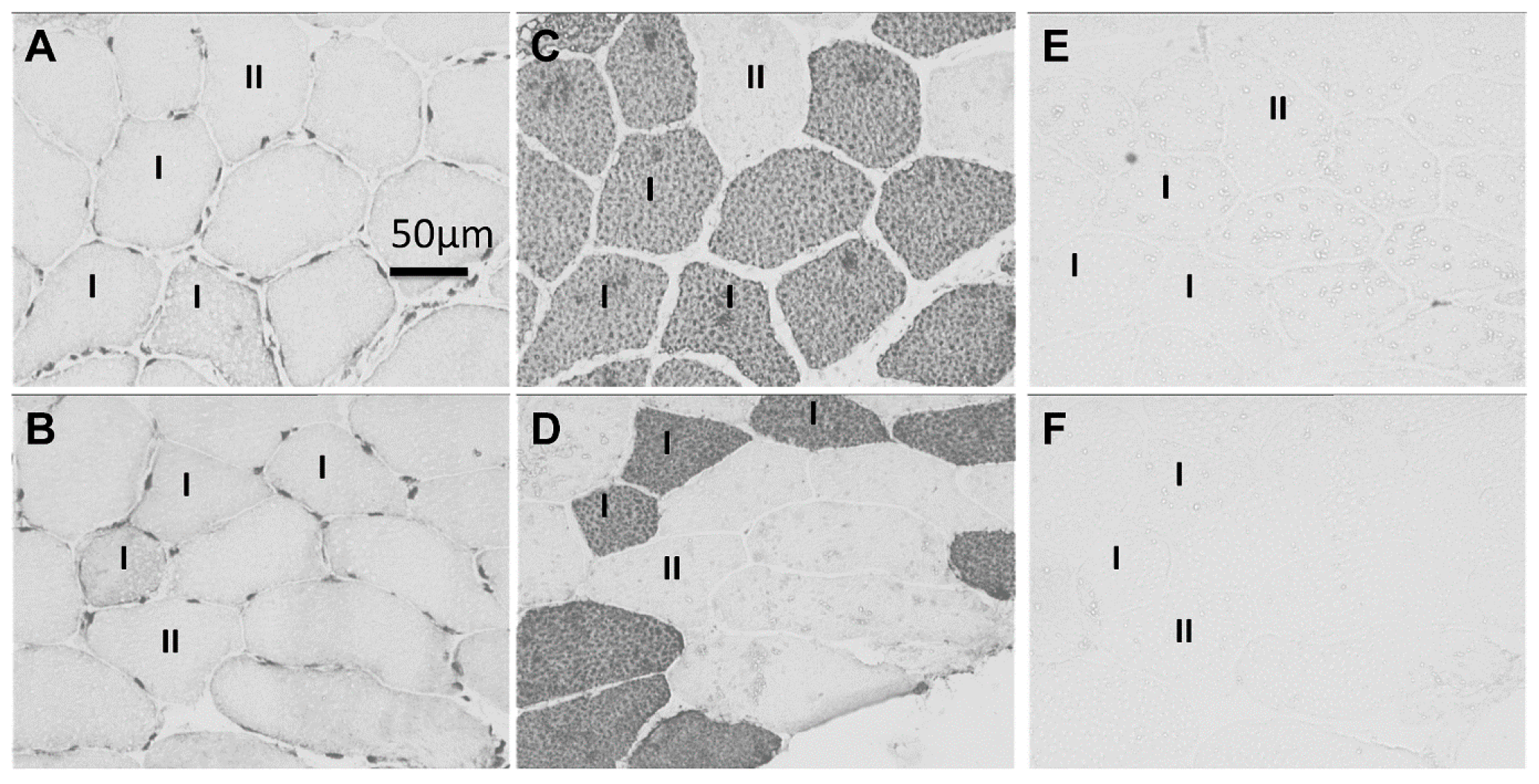
2.1. Immunohistochemistry
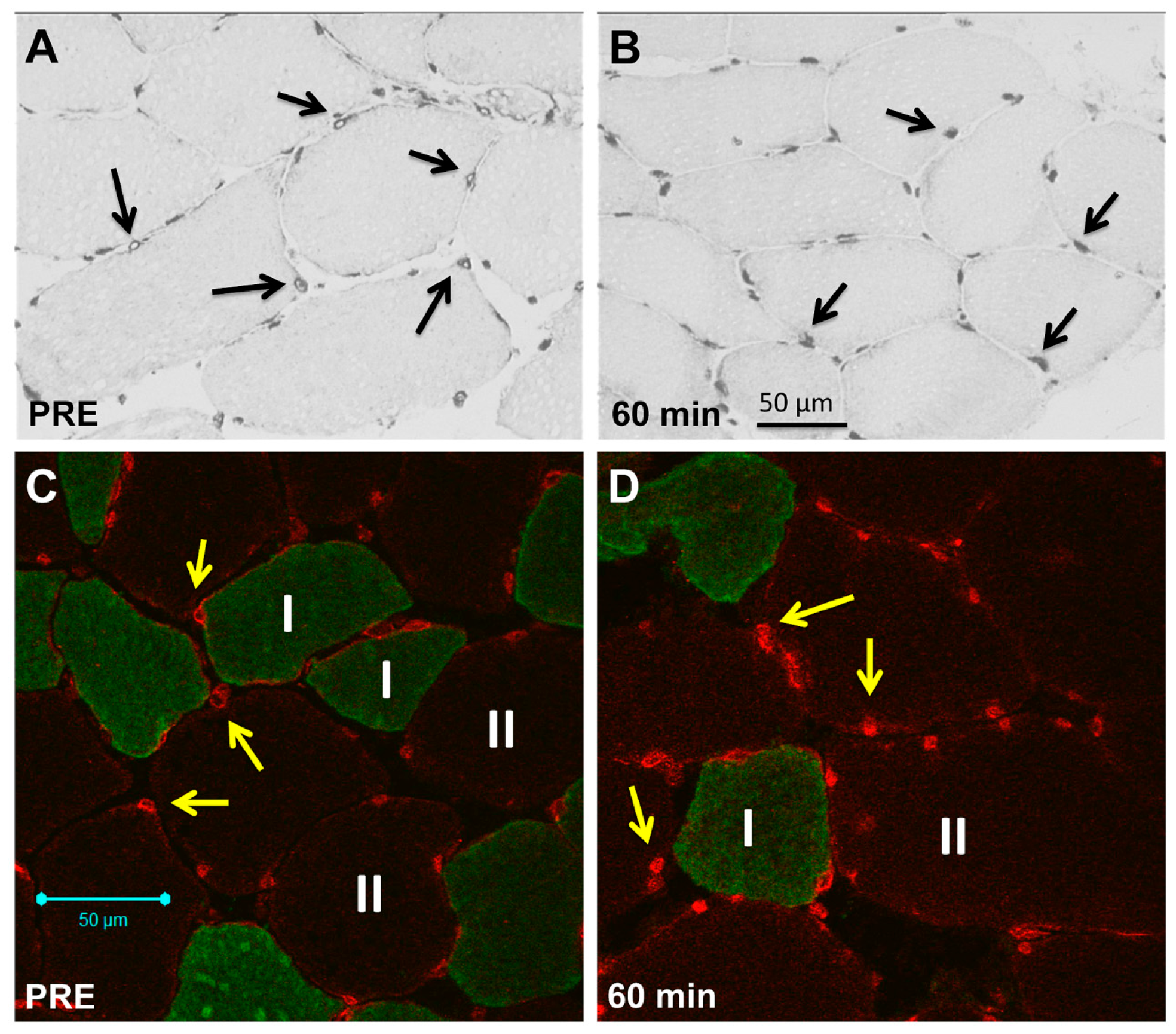
2.2. Sarcoplasmic Small Ubiquitin-Related Modifier (SUMO)-1-1 Localization
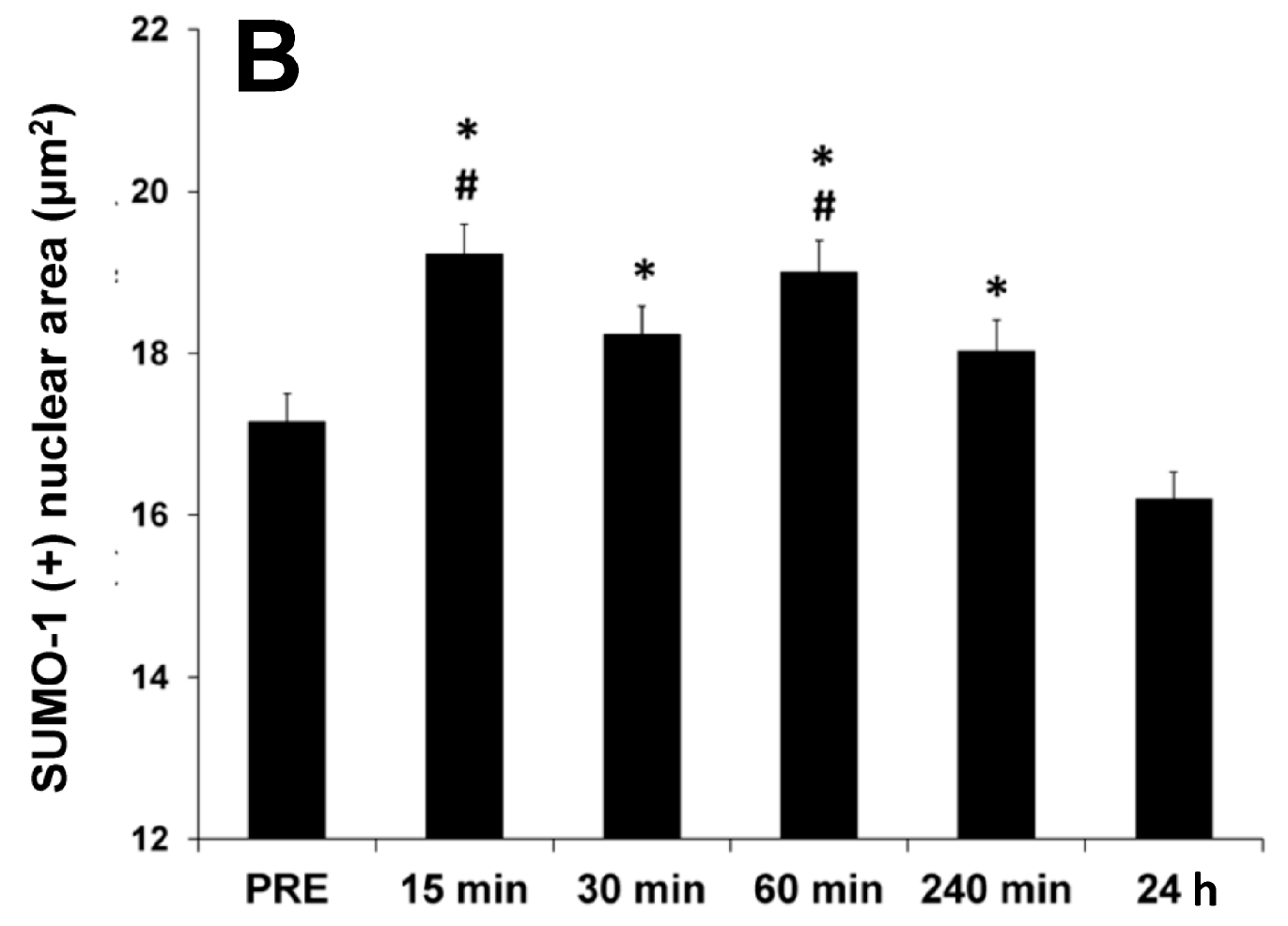
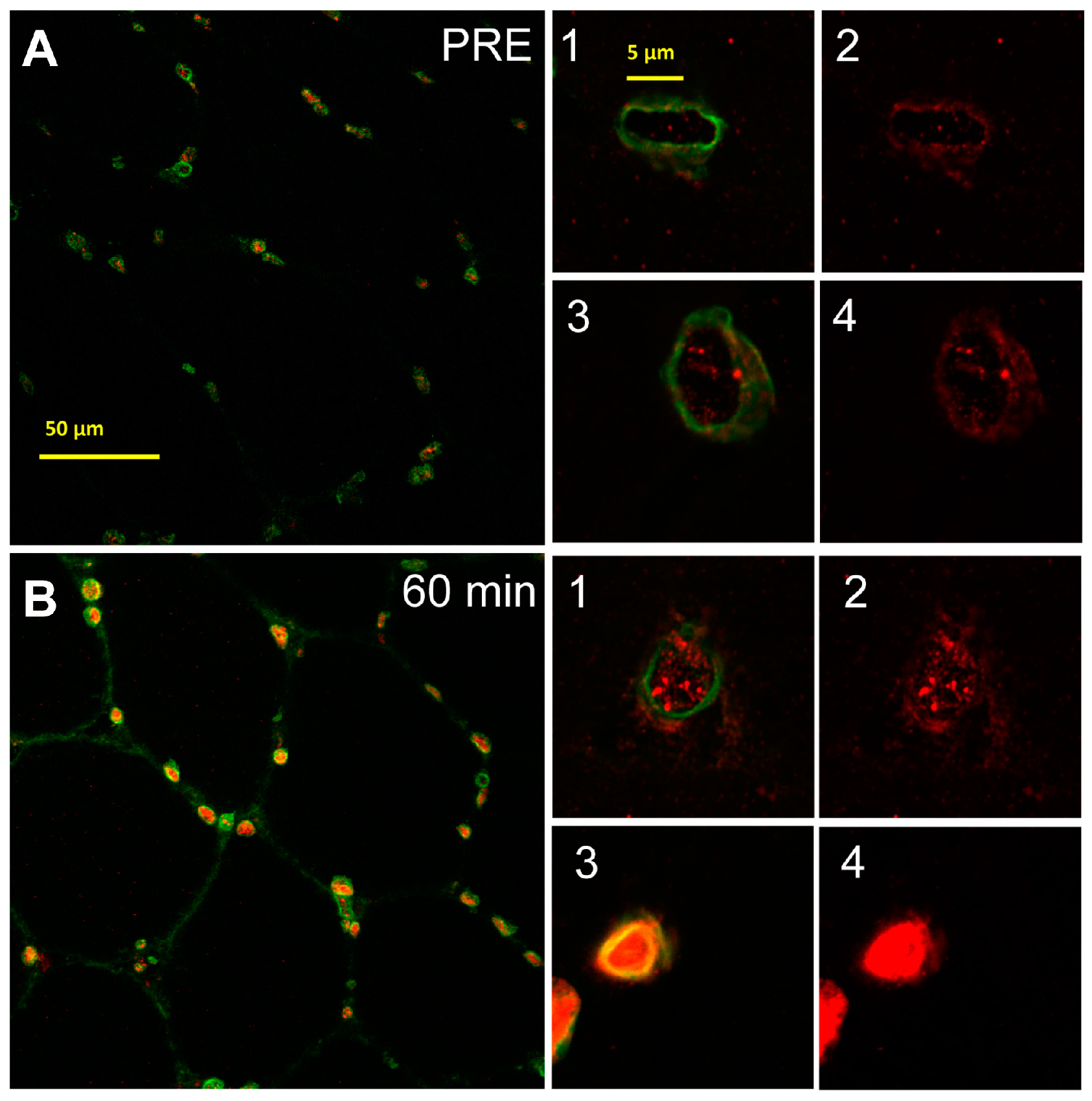
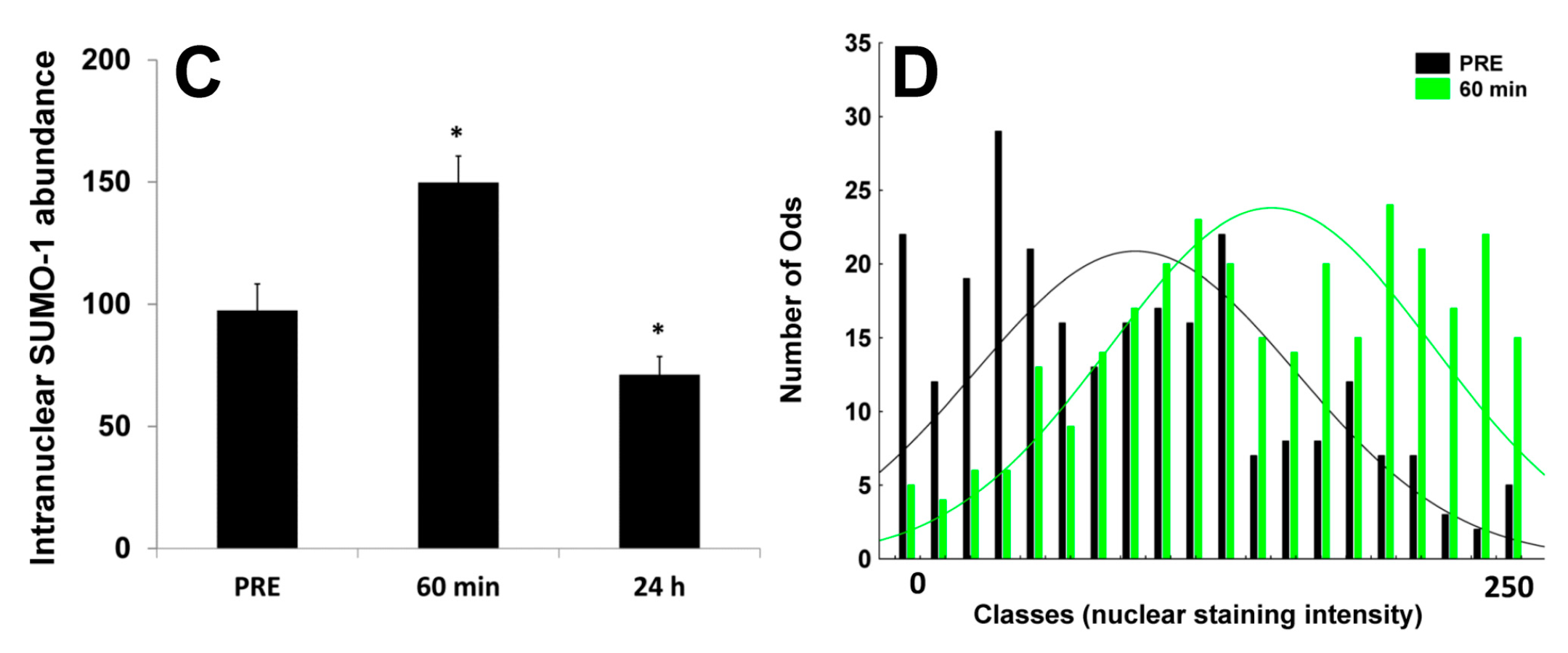
2.3. Nuclear SUMO-1 Localization
3. Discussion
4. Materials and Methods
4.1. Study Design
4.2. Subjects
4.3. Standardization of Diet and Activity before Exercise
4.4. Experimental Trials
4.5. Skeletal Muscle Biopsies
4.6. Tissue Processing and Staining
4.7. Analysis of Nuclear and Sarcoplasmic SUMO-1 Localization
4.8. Immunofluorescence Analysis
4.9. Statistics
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Anckar, J.; Sistonen, L. Sumo: Getting it on. Biochem. Soc. Trans. 2007, 35, 1409–1413. [Google Scholar] [PubMed]
- Seeler, J.S.; Dejean, A. Nuclear and unclear functions of SUMO. Nat. Rev. Mol. Cell Biol. 2003, 4, 690–699. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, K.A.; Henley, J.M. Mechanisms, regulation and consequences of protein sumoylation. Biochem. J. 2010, 428, 133–145. [Google Scholar] [CrossRef] [PubMed]
- Bekes, M.; Prudden, J.; Srikumar, T.; Raught, B.; Boddy, M.N.; Salvesen, G.S. The dynamics and mechanism of SUMO chain deconjugation by SUMO-specific proteases. J. Biol. Chem. 2011, 286, 10238–10247. [Google Scholar] [CrossRef] [PubMed]
- Kang, X.; Li, J.; Zou, Y.; Yi, J.; Zhang, H.; Cao, M.; Yeh, E.T.H.; Cheng, J. Piasy stimulates HIF1α sumoylation and negatively regulates HIF1α activity in response to hypoxia. Oncogene 2010, 29, 5568–5578. [Google Scholar] [CrossRef] [PubMed]
- Shao, R.; Zhang, F.P.; Tian, F.; Anders Friberg, P.; Wang, X.; Sjöland, H.; Billig, H. Increase of SUMO-1 expression in response to hypoxia: Direct interaction with HIF-1α in adult mouse brain and heart in vivo. FEBS Lett. 2004, 569, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Treuter, E.; Venteclef, N. Transcriptional control of metabolic and inflammatory pathways by nuclear receptor sumoylation. Biochim. Biophys. Acta Mol. Basis Dis. 2011, 1812, 909–918. [Google Scholar] [CrossRef] [PubMed]
- Nagai, S.; Davoodi, N.; Gasser, S.M. Nuclear organization in genome stability: Sumo connections. Cell Res. 2011, 21, 474–485. [Google Scholar] [CrossRef] [PubMed]
- Cai, Q.L.; Robertson, E.S. Ubiquitin/SUMO modification regulates VHL protein stability and nucleocytoplasmic localization. PLoS ONE 2010, 5. [Google Scholar] [CrossRef] [PubMed]
- Hannoun, Z.; Greenhough, S.; Jaffray, E.; Hay, R.T.; Hay, D.C. Post-translational modification by SUMO. Toxicology 2010, 278, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Matunis, M.J.; Coutavas, E.; Blobel, G. A novel ubiquitin-like modification modulates the partitioning of the Ran-GTPase-activating protein RanGAP1 between the cytosol and the nuclear pore complex. J. Cell Biol. 1996, 135, 1457–1470. [Google Scholar] [CrossRef] [PubMed]
- Matunis, M.J.; Wu, J.A.; Blobel, G. SUMO-1 modification and its role in targeting the Ran GTPase-activating protein, RanGAP1, to the nuclear pore complex. J. Cell Biol. 1998, 140, 499–509. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.S.; Ahn, B.Y.; Kim, M.; Kho, J.H.; Jung, H.S.; Park, K.S. SUMO modification selectively regulates transcriptional activity of peroxisome-proliferator-activated receptor γ in C2C12 myotubes. Biochem. J. 2011, 433, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Muller, S.; Berger, M.; Lehembre, F.; Seeler, J.S.; Haupt, Y.; Dejean, A. C-Jun and p53 activity is modulated by SUMO-1 modification. J. Biol. Chem. 2000, 275, 13321–13329. [Google Scholar] [CrossRef] [PubMed]
- Riquelme, C.; Barthel, K.K.B.; Liu, X. SUMO-1 modification of MEF2A regulates its transcriptional activity. J. Cell. Mol. Med. 2006, 10, 132–144. [Google Scholar] [CrossRef] [PubMed]
- Gill, G. Something about SUMO inhibits transcription. Curr. Opin. Genet. Dev. 2005, 15, 536–541. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, A.; Wang, Z.; Feng, X.; Olson, E.N.; Schwartz, R.J. Myocardin sumoylation transactivates cardiogenic genes in pluripotent 10T1/2 fibroblasts. Mol. Cell. Biol. 2007, 27, 622–632. [Google Scholar] [CrossRef] [PubMed]
- Matafora, V.; D’Amato, A.; Mori, S.; Blasi, F.; Bachi, A. Proteomics analysis of nucleolar SUMO-1 target proteins upon proteasome inhibition. Mol. Cell. Proteom. 2009, 8, 2243–2255. [Google Scholar] [CrossRef] [PubMed]
- Kho, C.; Lee, A.; Jeong, D.; Oh, J.G.; Chaanine, A.H.; Kizana, E.; Park, W.J.; Hajjar, R.J. SUMO1-dependent modulation of SERCA2A in heart failure. Nature 2011, 477, 601–605. [Google Scholar] [CrossRef] [PubMed]
- Uda, M.; Kawasaki, H.; Iizumi, K.; Shigenaga, A.; Baba, T.; Naito, H.; Yoshioka, T.; Yamakura, F. Sumoylated α-skeletal muscle actin in the skeletal muscle of adult rats. Mol. Cell. Biochem. 2015, 409, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Gehlert, S.; Suhr, F.; Gutsche, K.; Willkomm, L.; Kern, J.; Jacko, D.; Knicker, A.; Schiffer, T.; Wackerhage, H.; Bloch, W. High force development augments skeletal muscle signalling in resistance exercise modes equalized for time under tension. Pflugers Arch. 2014, 467. [Google Scholar] [CrossRef] [PubMed]
- Ulbricht, A.; Gehlert, S.; Leciejewski, B.; Schiffer, T.; Bloch, W.; Höhfeld, J. Induction and adaptation of chaperone-assisted selective autophagy casa in response to resistance exercise in human skeletal muscle. Autophagy 2015, 11, 538–546. [Google Scholar] [CrossRef] [PubMed]
- Bentzinger, C.F.; Lin, S.; Romanino, K.; Castets, P.; Guridi, M.; Summermatter, S.; Handschin, C.; Tintignac, L.A.; Hall, M.N.; Rüegg, M.A. Differential response of skeletal muscles to mTORC1 signaling during atrophy and hypertrophy. Skelet. Muscle 2013, 3, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Drummond, M.J.; Dreyer, H.C.; Fry, C.S.; Glynn, E.L.; Rasmussen, B.B. Nutritional and contractile regulation of human skeletal muscle protein synthesis and mTORC1 signaling. J. Appl. Physiol. 2009, 106, 1374–1384. [Google Scholar] [CrossRef] [PubMed]
- Klossner, S.; Durieux, A.C.; Freyssenet, D.; Flueck, M. Mechano-transduction to muscle protein synthesis is modulated by FAK. Eur. J. Appl. Physiol. 2009, 106, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Rockl, K.S.C.; Hirshman, M.F.; Brandauer, J.; Fujii, N.; Witters, L.A.; Goodyear, L.J. Skeletal muscle adaptation to exercise training—AMP-activated protein kinase mediates muscle fiber type shift. Diabetes 2007, 56, 2062–2069. [Google Scholar] [CrossRef] [PubMed]
- Atheron, P.J.; Babraj, J.A.; Smith, K.; Singh, J.; Rennie, M.J.; Wackerhage, H. Selective activation of AMPK-PGC-1 α or PKB-TSC2-mTOR signaling can explain specific adaptive responses to endurance or resistance training-like electrical muscle stimulation. FASEB J. 2005, 19, 786–788. [Google Scholar]
- Powers, S.K.; Jackson, M.J. Exercise-induced oxidative stress: Cellular mechanisms and impact on muscle force production. Physiol. Rev. 2008, 88, 1243–1276. [Google Scholar] [CrossRef] [PubMed]
- Matsakas, A.; Patel, K. Intracellular signalling pathways regulating the adaptation of skeletal muscle to exercise and nutritional changes. Histol. Histopathol. 2009, 24, 209–222. [Google Scholar] [PubMed]
- Potthoff, M.J.; Wu, H.; Arnold, M.A.; Shelton, J.M.; Backs, J.; McAnally, J.; Richardson, J.A.; Bassel-Duby, R.; Olson, E.N. Histone deacetylase degradation and MEF2 activation promote the formation of slow-twitch myofibers. J. Clin. Investig. 2007, 117, 2459–2467. [Google Scholar] [CrossRef] [PubMed]
- Chin, E.R.; Olson, E.N.; Richardson, J.A.; Yano, Q.; Humphries, C.; Shelton, J.M.; Wu, H.; Zhu, W.G.; Bassel-Duby, R.; Williams, R.S. A calcineurin-dependent transcriptional pathway controls skeletal muscle fiber type. Genes Dev. 1998, 12, 2499–2509. [Google Scholar] [CrossRef] [PubMed]
- Adams, G. The molecular response of skeletal muscle to resistance training. Dtsch. Z. Sportmed. 2010, 61, 61–67. [Google Scholar]
- Glass, D.J. Signalling pathways that mediate skeletal muscle hypertrophy and atrophy. Nat. Cell Biol. 2003, 5, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Urso, M.L.; Scrimgeour, A.G.; Chen, Y.-W.; Thompson, P.D.; Clarkson, P.M. Analysis of human skeletal muscle after 48 h immobilization reveals alterations in mRNA and protein for extracellular matrix components. J. Appl. Physiol. 2006, 101, 1136–1148. [Google Scholar] [CrossRef] [PubMed]
- McGee, S.L.; Howlett, K.F.; Starkie, R.L.; Cameron-Smith, D.; Kemp, B.E.; Hargreaves, M. Exercise increases nuclear ampk α2 in human skeletal muscle. Diabetes 2003, 52, 926–928. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.W.; Cseresnyes, Z.; Randall, W.R.; Schneider, M.F. Activity-dependent nuclear translocation and intranuclear distribution of NFATC in adult skeletal muscle fibers. J. Cell Biol. 2001, 155, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Creer, A.; Jemiolo, B.; Trappe, S. Time course of myogenic and metabolic gene expression in response to acute exercise in human skeletal muscle. J. Appl. Physiol. 2005, 98, 1745–1752. [Google Scholar] [CrossRef] [PubMed]
- Camera, D.M.; Edge, J.; Short, M.J.; Hawley, J.A.; Coffey, V.G. Early time course of AKT phosphorylation after endurance and resistance exercise. Med. Sci. Sports Exerc. 2010, 42, 1843–1852. [Google Scholar] [CrossRef] [PubMed]
- Tannerstedt, J.; Apro, W.; Blomstrand, E. Maximal lengthening contractions induce different signaling responses in the type I and type II fibers of human skeletal muscle. J. Appl. Physiol. 2009, 106, 1412–1418. [Google Scholar] [CrossRef] [PubMed]
- Koopman, R.; Zorenc, A.H.G.; Gransier, R.J.J.; Cameron-Smith, D.; van Loon, L.J.C. Increase in S6K1 phosphorylation in human skeletal muscle following resistance exercise occurs mainly in type II muscle fibers. Am. J. Physiol.-Endocrinol. Metab. 2006, 290, E1245–E1252. [Google Scholar] [CrossRef] [PubMed]
- Mayhew, D.L.; Kim, J.S.; Cross, J.M.; Ferrando, A.A.; Bamman, M.M. Translational signaling responses preceding resistance training-mediated myofiber hypertrophy in young and old humans. J. Appl. Physiol. 2009, 107, 1655–1662. [Google Scholar] [CrossRef] [PubMed]
- Agbor, T.A.; Taylor, C.T. SUMO, hypoxia and the regulation of metabolism. Biochem. Soc. Trans. 2008, 36, 445–448. [Google Scholar] [CrossRef] [PubMed]
- Agbor, T.A.; Cheong, A.; Comerford, K.M.; Scholz, C.C.; Bruning, U.; Clarke, A.; Cummins, E.P.; Cagney, G.; Taylor, C.T. Small ubiquitin-related modifier (SUMO)-1 promotes glycolysis in hypoxia. J. Biol. Chem. 2011, 286, 4718–4726. [Google Scholar] [CrossRef] [PubMed]
- Ameln, H.; Gustafsson, T.; Sundberg, C.J.; Okamoto, K.; Jansson, E.; Poellinger, L.; Makino, Y. Physiological activation of hypoxia inducible factor-1 in human skeletal muscle. FASEB J. 2005, 19, 1009–1011. [Google Scholar] [CrossRef] [PubMed]
- Baar, K. Involvement of PPARγ co-activator-1, nuclear respiratory factors 1 and 2, and PPARα in the adaptive response to endurance exercise. Proc. Nutr. Soc. 2004, 63, 269–273. [Google Scholar] [CrossRef] [PubMed]
- Boppart, M.D.; Aronson, D.; Gibson, L.; Roubenoff, R.; Abad, L.W.; Bean, J.; Goodyear, L.J.; Fielding, R.A. Eccentric exercise markedly increases c-Jun NH2-terminal kinase activity in human skeletal muscle. J. Appl. Physiol. 1999, 87, 1668–1673. [Google Scholar] [PubMed]
- Williamson, D.; Gallagher, P.; Harber, M.; Hollon, C.; Trappe, S. Mitogen-activated protein kinase (MAPK) pathway activation: Effects of age and acute exercise on human skeletal muscle. J. Physiol. 2003, 547, 977–987. [Google Scholar] [CrossRef] [PubMed]
- Rytinki, M.M.; Palvimo, J.J. Sumo wrestling in cell movement. Cell Res. 2011, 21, 3–5. [Google Scholar] [CrossRef] [PubMed]
- McGee, S.L.; Hargreaves, M. Exercise and myocyte enhancer factor 2 regulation in human skeletal muscle. Diabetes 2004, 53, 1208–1214. [Google Scholar] [CrossRef] [PubMed]
- Hood, D.A. Mechanisms of exercise-induced mitochondrial biogenesis in skeletal muscle. Appl. Physiol. Nutr. Metab. 2009, 34, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Martineau, L.C.; Gardiner, P.F. Insight into skeletal muscle mechanotransduction: MAPK activation is quantitatively related to tension. J. Appl. Physiol. 2001, 91, 693–702. [Google Scholar] [PubMed]
- Glass, D.J. Two tales concerning skeletal muscle. J. Clin. Investig. 2007, 117, 2388–2391. [Google Scholar] [CrossRef] [PubMed]
- Little, J.P.; Safdar, A.; Bishop, D.; Tarnopolsky, M.A.; Gibala, M.J. An acute bout of high-intensity interval training increases the nuclear abundance of PGC-1α and activates mitochondrial biogenesis in human skeletal muscle. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 300, R1303–R1310. [Google Scholar] [CrossRef] [PubMed]
- Gehlert, S.; Theis, C.; Weber, S.; Schiffer, T.; Hellmich, M.; Platen, P.; Bloch, W. Exercise-induced decline in the density of LYVE-1-positive lymphatic vessels in human skeletal muscle. Lymphat. Res. Biol. 2010, 8, 165–173. [Google Scholar] [CrossRef] [PubMed]



© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gehlert, S.; Klinz, F.J.; Willkomm, L.; Schiffer, T.; Suhr, F.; Bloch, W. Intense Resistance Exercise Promotes the Acute and Transient Nuclear Translocation of Small Ubiquitin-Related Modifier (SUMO)-1 in Human Myofibres. Int. J. Mol. Sci. 2016, 17, 646. https://doi.org/10.3390/ijms17050646
Gehlert S, Klinz FJ, Willkomm L, Schiffer T, Suhr F, Bloch W. Intense Resistance Exercise Promotes the Acute and Transient Nuclear Translocation of Small Ubiquitin-Related Modifier (SUMO)-1 in Human Myofibres. International Journal of Molecular Sciences. 2016; 17(5):646. https://doi.org/10.3390/ijms17050646
Chicago/Turabian StyleGehlert, Sebastian, Franz Josef Klinz, Lena Willkomm, Thorsten Schiffer, Frank Suhr, and Wilhelm Bloch. 2016. "Intense Resistance Exercise Promotes the Acute and Transient Nuclear Translocation of Small Ubiquitin-Related Modifier (SUMO)-1 in Human Myofibres" International Journal of Molecular Sciences 17, no. 5: 646. https://doi.org/10.3390/ijms17050646
APA StyleGehlert, S., Klinz, F. J., Willkomm, L., Schiffer, T., Suhr, F., & Bloch, W. (2016). Intense Resistance Exercise Promotes the Acute and Transient Nuclear Translocation of Small Ubiquitin-Related Modifier (SUMO)-1 in Human Myofibres. International Journal of Molecular Sciences, 17(5), 646. https://doi.org/10.3390/ijms17050646








