Cellular Localization and Regulation of Expression of the PLET1 Gene in Porcine Placenta
Abstract
:1. Introduction
2. Results
2.1. PLET1 Gene Was Uniquely Expressed by Trophoblast Cells throughout Pig Placenta Development
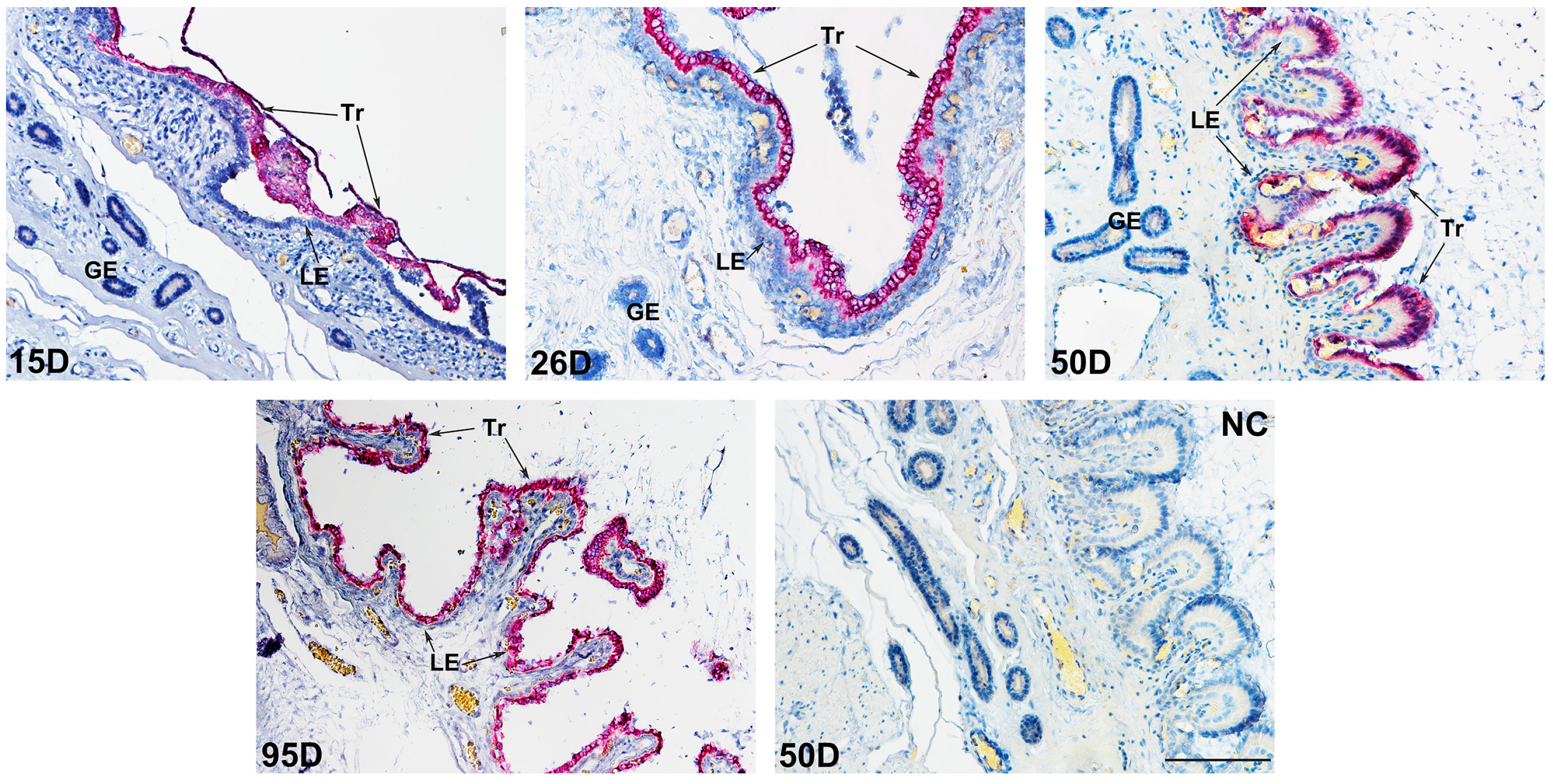
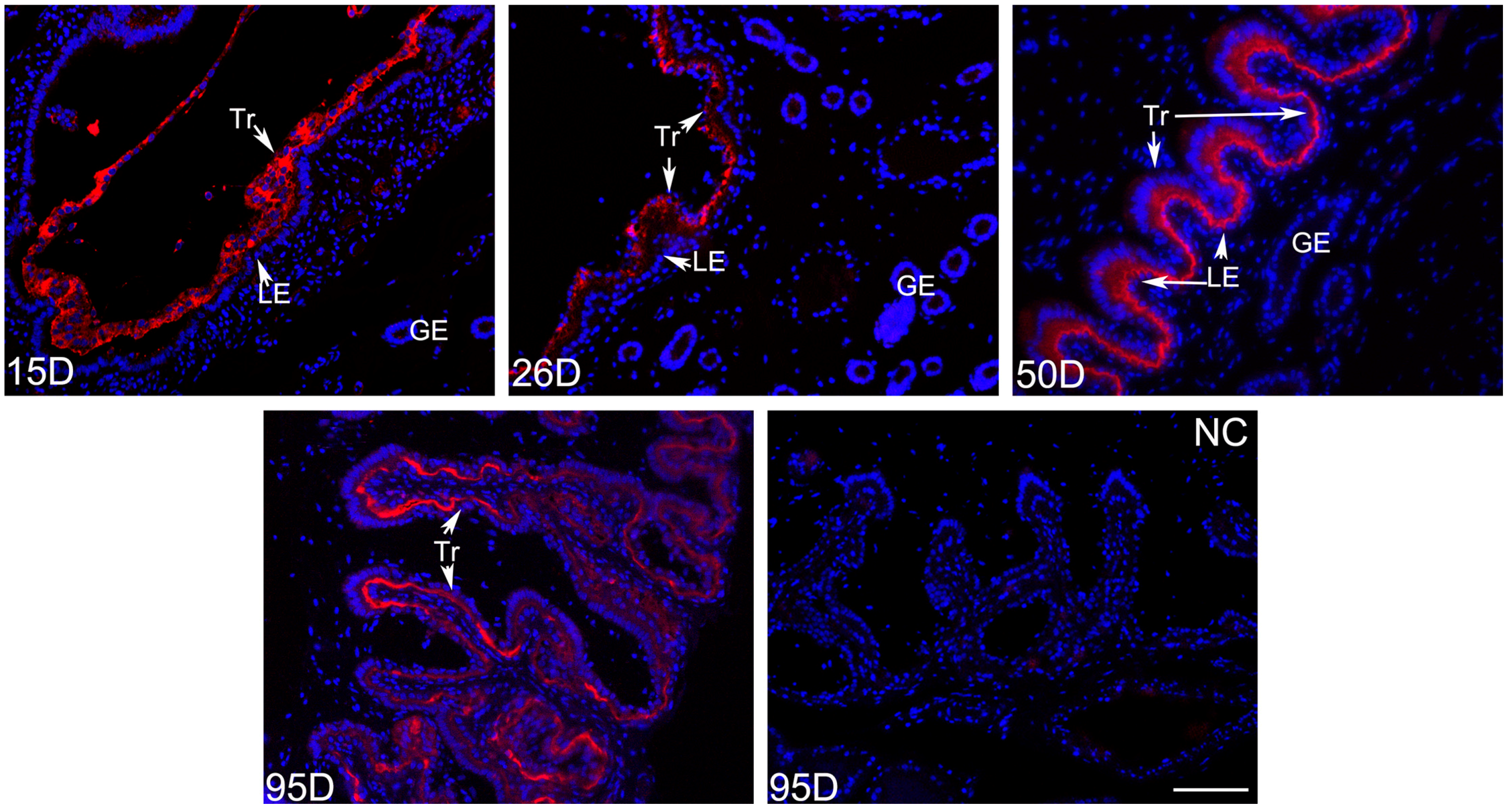
2.2. The Location Pattern of PLET1 in Pig Trophoblast Cells Changes as the Pregnancy Proceeds
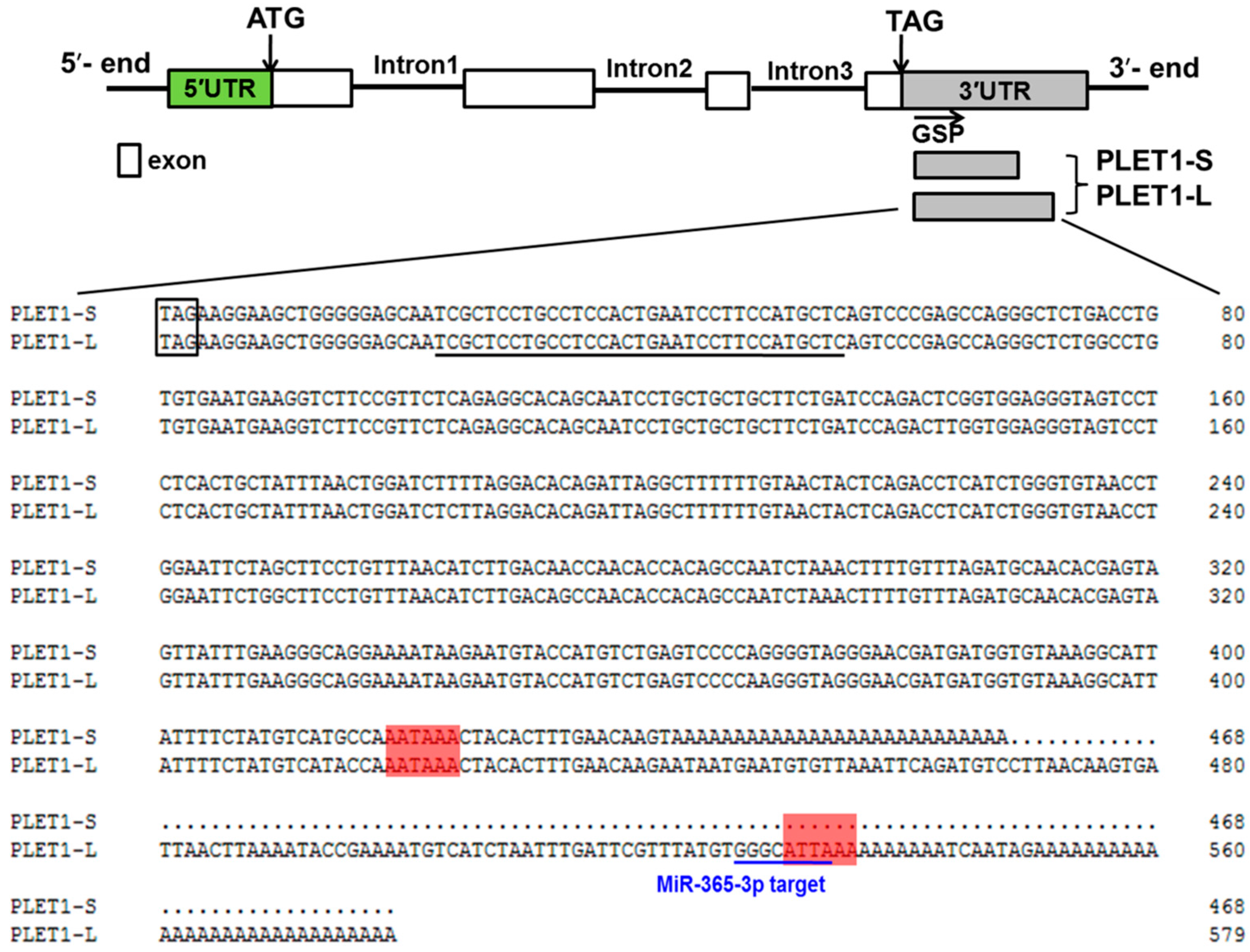
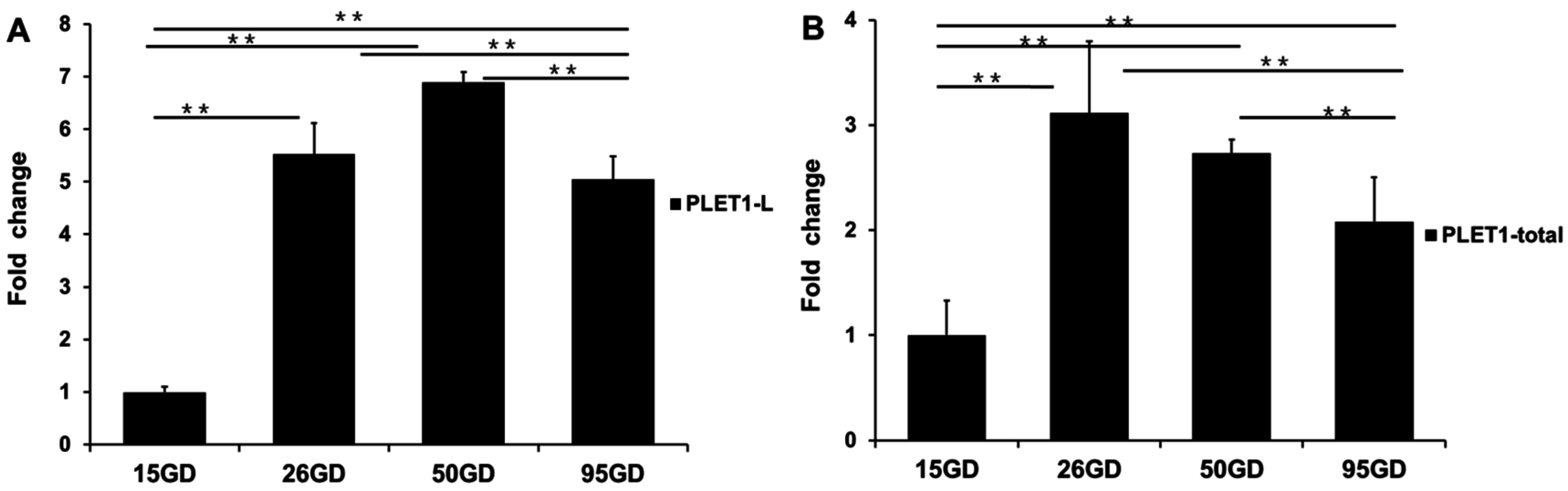
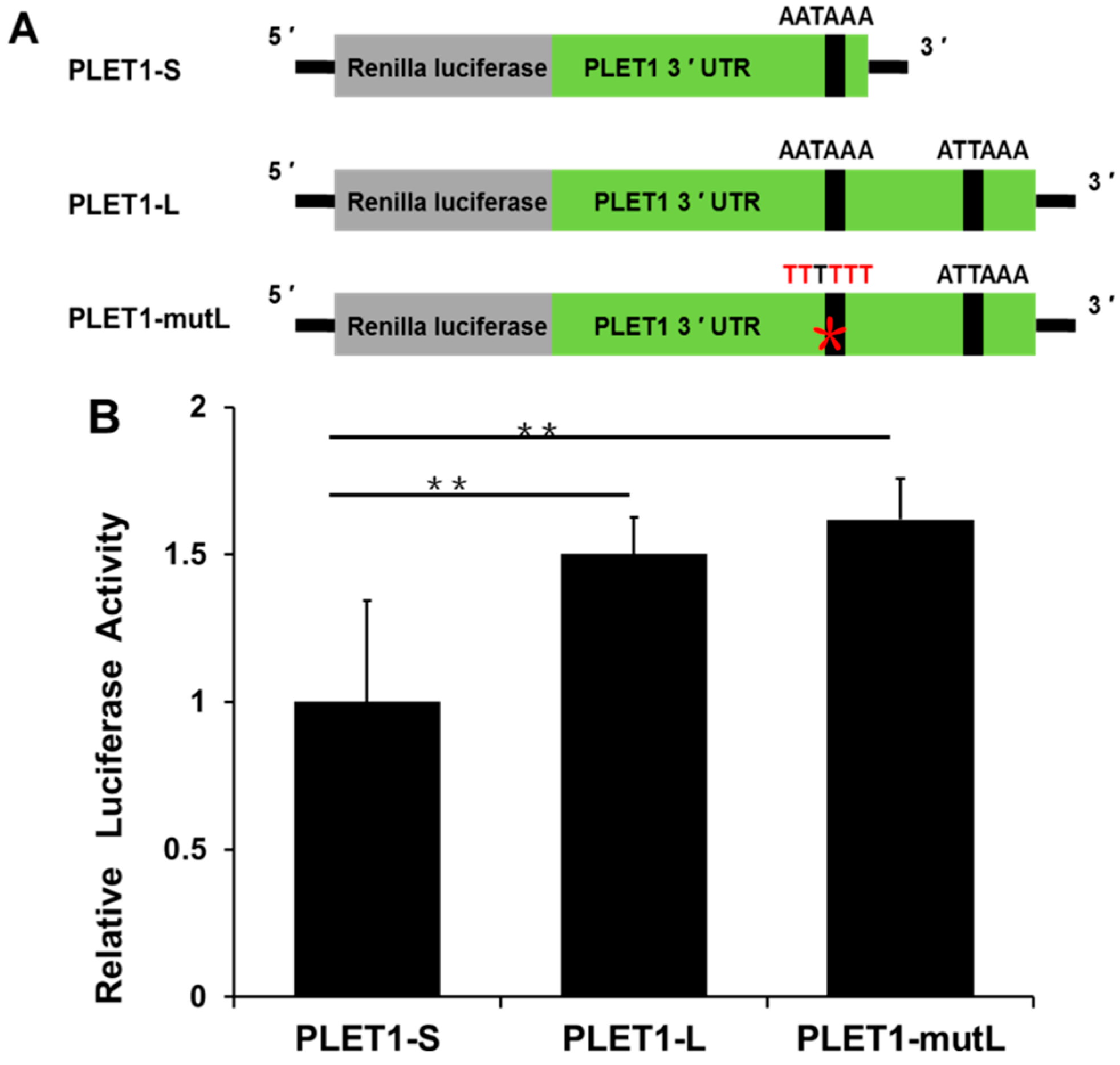
2.3. Two Different PLET1 Transcripts Were Identified to Be Expressed in Porcine Placenta by 3′RACE and Showed Different Expression Levels as the Placenta Developed
2.4. The Long and Short PLET1 3′ UTR Differentially Mediate the Luciferase Activity In Vitro
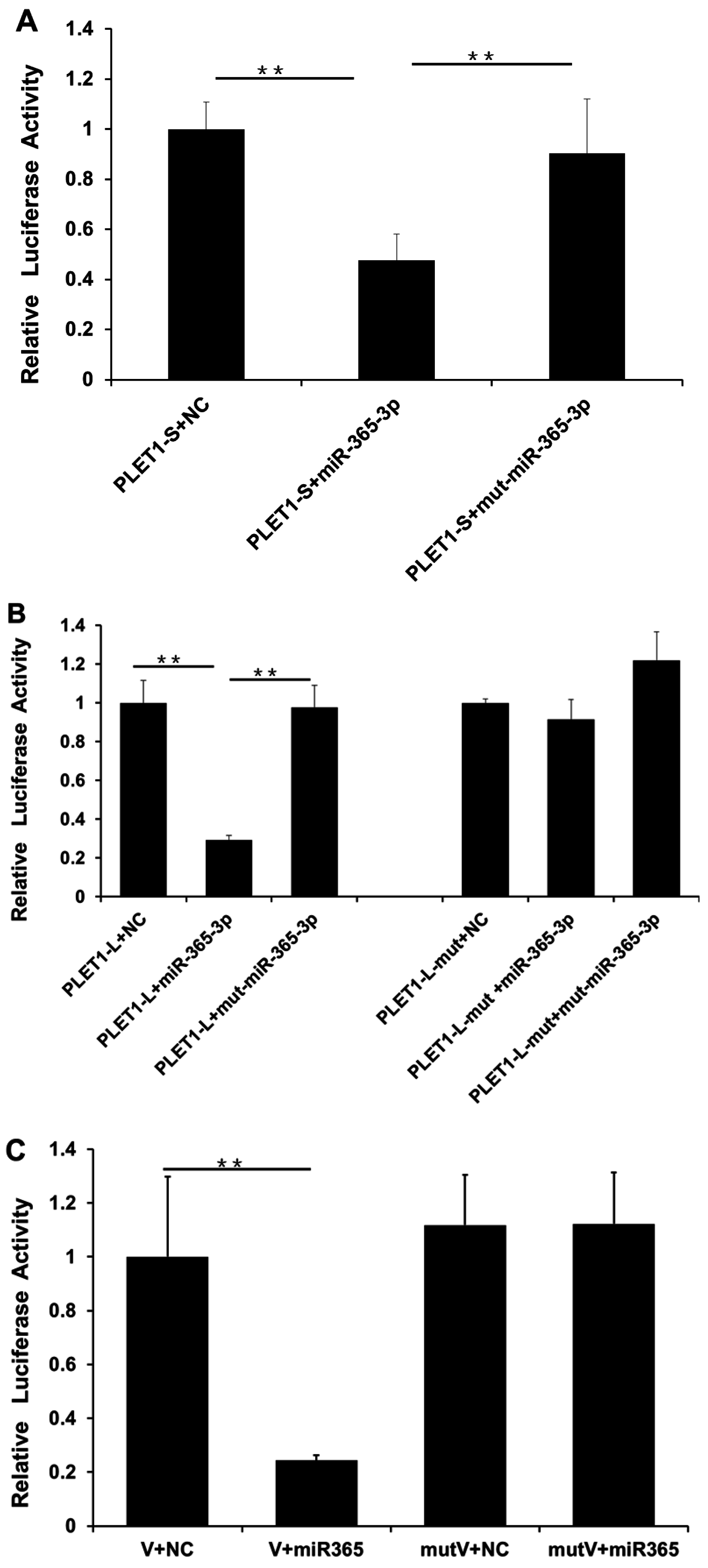
2.5. Investigation of the Interaction between Pig PLET1 and miR-365-3p In Vitro
2.6. miR-365-3p Was Differentially Expressed during Days 15, 26, 50, and 95 of Gestation in Pigs
2.7. PLET1 Is an N-Glycosylation Modified Protein
3. Discussion
4. Materials and Methods
4.1. Animals and Tissue Collections
4.2. Detection of the PLET1 mRNA Expression in Porcine Conceptus and Placenta by In Situ Hybridization
4.3. Investigation of PLET1 Protein Expression in Porcine Conceptus and Placenta by Immunohistochemistry
4.4. Investigation of the PLET1 Protein Expression in Porcine Conceptus and Placenta by Immunoflurescence
4.5. Cloning of the 5′and 3′ End of PLET1 Gene
4.6. Quantitative RT-PCR Analyses
4.7. Construction of Porcine PLET1 3′UTRs Luciferase Reporter Vector and Luciferase Reporter Assay
4.8. Validation of the Interaction between miR-365-3p and PLET1
4.9. Protein Enzymatic Deglycosylation by PNGase F and endoH
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rawn, S.M.; Cross, J.C. The evolution, regulation, and function of placenta-specific genes. Annu. Rev. Cell Dev. Biol. 2008, 24, 159–181. [Google Scholar] [CrossRef] [PubMed]
- Ono, R.; Nakamura, K.; Inoue, K.; Naruse, M.; Usami, T.; Wakisaka-Saito, N.; Hino, T.; Suzuki-Migishima, R.; Ogonuki, N.; Miki, H.; et al. Deletion of Peg10, an imprinted gene acquired from a retrotransposon, causes early embryonic lethality. Nat. Genet. 2006, 38, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.L.; Yang, Q.; Zhang, H.; Lin, H.Y.; Zhou, Z.; Lu, X.; Zhu, C.; Xue, L.Q.; Wang, H. Role of placenta-specific protein 1 in trophoblast invasion and migration. Reproduction 2014, 148, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Anson-Cartwright, L.; Dawson, K.; Holmyard, D.; Fisher, S.J.; Lazzarini, R.A.; Cross, J.C. The glial cells missing-1 protein is essential for branching morphogenesis in the chorioallantoic placenta. Nat. Genet. 2000, 25, 311–314. [Google Scholar] [PubMed]
- Whyte, A.; Bacon, M.; Ellis, S. A monoclonal antibody to an antigen present on the microvillous membrane of the trophectoderm of the preimplantation blastocyst of the pig. J. Reprod. Fertil. 1984, 71, 599–605. [Google Scholar] [CrossRef] [PubMed]
- Finch, A.M.; Yang, L.G.; Nwagwu, M.O.; Page, K.R.; McArdle, H.J.; Ashworth, C.J. Placental transport of leucine in a porcine model of low birth weight. Reproduction 2004, 128, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Dantzer, V. Electron microscopy of the initial stages of placentation in the pig. Anat. Embryol. 1985, 172, 281–293. [Google Scholar] [CrossRef] [PubMed]
- Bazer, F.W.; Johnson, G.A. Pig blastocyst-uterine interactions. Differentiation 2014, 87, 52–65. [Google Scholar] [CrossRef] [PubMed]
- Wright, E.C.; Miles, J.R.; Lents, C.A.; Rempel, L.A. Uterine and placenta characteristics during early vascular development in the pig from day 22 to 42 of gestation. Anim. Reprod. Sci. 2016, 164, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Vallet, J.L.; Miles, J.R.; Freking, B.A. Development of the pig placenta. Soc. Reprod. Fertil. Suppl. 2009, 66, 265–279. [Google Scholar] [PubMed]
- Vallet, J.L.; Freking, B.A. Differences in placental structure during gestation associated with large and small pig fetuses. J. Anim. Sci. 2007, 85, 3267–3275. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.Z.; Wang, M.; Su, L.J.; Li, X.P.; Zhao, S.H.; Yu, M. The Expression Pattern of MicroRNAs and the Associated Pathways Involved in the Development of Porcine Placental Folds That Contribute to the Expansion of the Exchange Surface Area. Biol. Reprod. 2015, 93, 62. [Google Scholar] [CrossRef] [PubMed]
- Du, Z.Q.; Eisley, C.J.; Onteru, S.K.; Madsen, O.; Groenen, M.A.M.; Ross, J.W.; Rothschild, M.F. Identification of species-specific novel transcripts in pig reproductive tissues using RNA-seq. Anim. Genet. 2014, 45, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Krawczynski, K.; Najmula, J.; Bauersachs, S.; Kaczmarek, M.M. MicroRNAome of porcine conceptuses and trophoblasts: Expression profile of micrornas and their potential to regulate genes crucial for establishment of pregnancy. Biol. Reprod. 2015, 92, 21. [Google Scholar] [CrossRef] [PubMed]
- Li, H.N.; Wu, B.; Geng, J.N.; Zhou, J.W.; Zheng, R.; Chai, J.; Li, F.; Peng, J.; Jiang, S.W. Integrated analysis of miRNA/mRNA network in placenta identifies key factors associated with labor onset of Large White and Qingping sows. Sci. Rep. 2015, 5, 13074. [Google Scholar] [CrossRef] [PubMed]
- Jeong, W.; Kim, J.; Bazer, F.W.; Song, G. Epidermal growth factor stimulates proliferation and migration of porcine trophectoderm cells through protooncogenic protein kinase 1 and extracellular-signal-regulated kinases 1/2 mitogen-activated protein kinase signal transduction cascades during early pregnancy. Mol. Cell. Endocrinol. 2013, 381, 302–311. [Google Scholar] [PubMed]
- Jeong, W.; Kim, J.; Bazer, F.W.; Song, G. Stimulatory Effect of Vascular Endothelial Growth Factor on Proliferation and Migration of Porcine Trophectoderm Cells and Their Regulation by the Phosphatidylinositol-3-Kinase-AKT and Mitogen-Activated Protein Kinase Cell Signaling Pathways. Biol. Reprod. 2014, 90, 50. [Google Scholar] [CrossRef] [PubMed]
- Jeong, W.; Song, G.; Bazer, F.W.; Kim, J. Insulin-like growth factor I induces proliferation and migration of porcine trophectoderm cells through multiple cell signaling pathways, including protooncogenic protein kinase 1 and mitogen-activated protein kinase. Mol. Cell. Endocrinol. 2014, 384, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Jaeger, L.A.; Spiegel, A.K.; Ing, N.H.; Johnson, G.A.; Bazer, F.W.; Burghardt, R.C. Functional effects of transforming growth factor beta on adhesive properties of porcine trophectoderm. Endocrinology 2005, 146, 3933–3942. [Google Scholar] [CrossRef] [PubMed]
- Massuto, D.A.; Kneese, E.C.; Johnson, G.A.; Burghardt, R.C.; Hooper, R.N.; Ing, N.H.; Jaeger, L.A. Transforming growth factor beta (TGFB) signaling is activated during porcine implantation: Proposed role for latency-associated peptide interactions with integrins at the conceptus-maternal interface. Reproduction 2010, 139, 465–478. [Google Scholar] [CrossRef] [PubMed]
- Johnson, G.A.; Burghardt, R.C.; Bazer, F.W.; Spencer, T.E. Osteopontin: Roles in implantation and placentation. Biol. Reprod. 2003, 69, 1458–1471. [Google Scholar] [CrossRef] [PubMed]
- Johnson, G.A.; Burghardt, R.C.; Bazer, F.W. Osteopontin a leading candidate adhesion molecule for implantation in pigs and sheep. J. Anim. Sci. Biotechnol. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Erikson, D.W.; Burghardt, R.C.; Bayless, K.J.; Johnson, G.A. Secreted phosphoprotein 1 (SPP1, osteopontin) binds to integrin αvβ6 on porcine trophectoderm cells and integrin αvβ3 on uterine luminal epithelial cells, and promotes trophectoderm cell adhesion and migration. Biol. Reprod. 2009, 81, 814–825. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Bailey, D.W.; Dunlap, K.A.; Burghardt, R.C.; Spencer, T.E.; Bazer, F.W.; Johnson, G.A. Cathepsin, B, Cathepsin L, Cystatin C in the Porcine Uterus and Placenta: Potential Roles in Endometrial/Placental Remodeling and in Fluid-Phase Transport of Proteins Secreted by Uterine Epithelia Across Placental Areolae. Biol. Reprod. 2010, 82, 854–864. [Google Scholar] [CrossRef] [PubMed]
- Miles, J.R.; Vallet, J.L.; Freking, B.A.; Nonneman, D.J. Molecular cloning and characterization of heparanase mRNA in the porcine placenta throughout gestation. Reprod. Fertil. Dev. 2009, 21, 757–772. [Google Scholar] [CrossRef] [PubMed]
- Hong, L.J.; Hou, C.Y.; Li, X.P.; Li, C.C.; Zhao, S.H.; Yu, M. Expression of heparanase is associated with breed-specific morphological characters of placental folded bilayer between Yorkshire and Meishan pigs. Biol. Reprod. 2014, 90, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Vallet, J.L.; McNeel, A.K.; Johnson, G.; Bazer, F.W. Triennial Reproduction Symposium: Limitations in uterine and conceptus physiology that lead to fetal losses. J. Anim. Sci. 2013, 91, 3030–3040. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.H.; Simmons, D.G.; Cross, J.C.; Scheetz, T.E.; Casavant, T.L.; Soares, M.B.; Tuggle, C.K. PLET1 (C11orf34), a highly expressed and processed novel gene in pig and mouse placenta, is transcribed but poorly spliced in human. Genomics 2004, 84, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.H.; Tuggle, C.K. Linkage mapping and expression analyses of a novel gene, placentally expressed transcript 1 (PLET1) in the pig. Anim. Genet. 2003, 35, 66–76. [Google Scholar] [CrossRef]
- Murray, A.; Sienerth, A.R.; Hemberger, M. Plet1 is an epigenetically regulated cell surface protein that provides essential cues to direct trophoblast stem cell differentiation. Sci. Rep. 2016, 6, 25112. [Google Scholar] [CrossRef] [PubMed]
- Ross, J.W.; Ashworth, M.D.; Stein, D.R.; Couture, O.P.; Tuggle, C.K.; Geisert, R.D. Identification of differential gene expression during porcine conceptus rapid trophoblastic elongation and attachment to uterine luminal epithelium. Physiol. Genom. 2009, 36, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Chikh, K.; Vey, S.; Simonot, C.; Vanier, M.T.; Millat, G. Niemann-Pick type C disease: Importance of N-glycosylation sites for function and cellular location of the NPC2 protein. Mol. Genet. Metab. 2004, 83, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Penuela, S.; Bhalla, R.; Nag, K.; Laird, D.W. Glycosylation Regulates Pannexin Intermixing and Cellular Localization. Mol. Biol. Cell 2009, 20, 4313–4323. [Google Scholar]
- Raymond, K.; Richter, A.; Kreft, M.; Frijns, E.; Janssen, H.; Slijper, M.; Praetzel-Wunder, S.; Langbein, L.; Sonnenberg, A. Expression of the orphan protein Plet-1 during trichilemmal differentiation of anagen hair follicles. J. Investig. Dermatol. 2010, 130, 1500–1513. [Google Scholar] [CrossRef] [PubMed]
- Depreter, M.G.; Blair, N.F.; Gaskell, T.L.; Nowell, C.S.; Davern, K.; Pagliocca, A.; Stenhouse, F.H.; Farley, A.M.; Fraser, A.; Vrana, J.; et al. Identification of Plet-1 as a specific marker of early thymic epithelial progenitor cells. Proc. Natl. Acad. Sci. USA 2008, 105, 961–966. [Google Scholar] [CrossRef] [PubMed]
- Mayr, C.; Bartel, D.P. Widespread shortening of 3′UTRs by alternative cleavage and polyadenylation activates oncogenes in cancer cells. Cell 2009, 138, 673–684. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moore, M.J. From Birth to Death: The Complex Lives of Eukaryotic mRNAs. Science 2005, 309, 1514–1518. [Google Scholar] [CrossRef] [PubMed]
- Lianoglou, S.; Garg, V.; Yang, J.L.; Leslie, C.S.; Mayr, C. Ubiquitously transcribed genes use alternative polyadenylation to achieve tissue-specific expression. Genes Dev. 2013, 27, 2380–2396. [Google Scholar] [CrossRef] [PubMed]
- Tian, B.; Hu, J.; Zhang, H.; Lutz, C.S. A large-scale analysis of mRNA polyadenylation of human and mouse genes. Nucleic Acids Res. 2005, 33, 201–212. [Google Scholar] [CrossRef] [PubMed]
- An, J.J.; Gharami, K.; Liao, G.Y.; Woo, N.H.; Lau, A.G.; Vanevski, F.; Torre, E.R.; Jones, K.R.; Feng, Y.; Lu, B.; et al. Distinct role of long 3′UTR BDNF mRNA in spine morphology and synaptic plasticity in hippocampal neurons. Cell 2008, 134, 175–187. [Google Scholar] [CrossRef] [PubMed]
- Berkovits, B.D.; Mayr, C. Alternative 3′UTRs act as scaffolds to regulate membrane protein localization. Nature 2015, 522, 363–367. [Google Scholar] [CrossRef] [PubMed]
- Engelmann, J.C.; Spang, R. A least angle regression model for the prediction of canonical and non-canonical miRNA-mRNA interactions. PLoS ONE 2012, 7, e40634. [Google Scholar] [CrossRef] [PubMed]
- Ghoshal, A.; Shankar, R.; Bagchi, S.; Grama, A.; Chaterji, S. MicroRNA target prediction using thermodynamic and sequence curves. BMC Genom. 2015, 16, 999. [Google Scholar] [CrossRef] [PubMed]
- Hong, L.J.; Hou, C.Y.; Li, X.P.; Li, C.; Yu, M. Expression pattern of CD34 at the maternal-foetal interface during pregnancy in pigs. Reprod. Domest. Anim. 2013, 48, 762–767. [Google Scholar] [CrossRef] [PubMed]
- Wäneskog, M.; Bjerling, P. Multi-fragment site-directed mutagenic overlap extension polymerase chain reaction as a competitive alternative to the enzymatic assembly method. Anal. Biochem. 2014, 444, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Su, L.J.; Liu, R.Z.; Cheng, W.; Zhu, M.J.; Li, X.P.; Zhao, S.H.; Yu, M. Expression patterns of microRNAs in porcine endometrium and their potential roles in embryo implantation and placentation. PLoS ONE 2014, 9, e87867. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; He, H.B.; Zhang, W.Y.; Zhang, H.X.; Bai, J.B.; Liu, H.Z.; Cao, J.H.; Chang, K.C.; Li, X.Y.; Zhao, S.H. miR-29 targets Akt3 to reduce proliferation and facilitate differentiation of myoblasts in skeletal muscle development. Cell Death Dis. 2013, 4, e668. [Google Scholar] [CrossRef] [PubMed]



© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teng, L.; Hong, L.; Liu, R.; Chen, R.; Li, X.; Yu, M. Cellular Localization and Regulation of Expression of the PLET1 Gene in Porcine Placenta. Int. J. Mol. Sci. 2016, 17, 2048. https://doi.org/10.3390/ijms17122048
Teng L, Hong L, Liu R, Chen R, Li X, Yu M. Cellular Localization and Regulation of Expression of the PLET1 Gene in Porcine Placenta. International Journal of Molecular Sciences. 2016; 17(12):2048. https://doi.org/10.3390/ijms17122048
Chicago/Turabian StyleTeng, Liu, Linjun Hong, Ruize Liu, Ran Chen, Xinyun Li, and Mei Yu. 2016. "Cellular Localization and Regulation of Expression of the PLET1 Gene in Porcine Placenta" International Journal of Molecular Sciences 17, no. 12: 2048. https://doi.org/10.3390/ijms17122048





