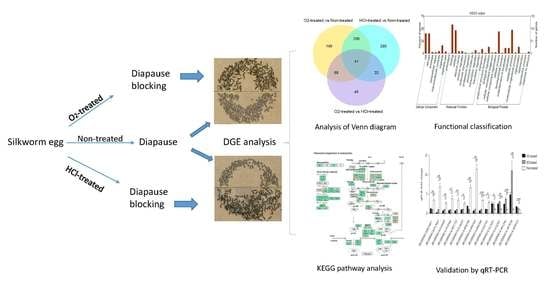
Transcriptional Response of Silkworm (Bombyx mori) Eggs to O2 or HCl Treatment
Abstract
:1. Introduction
2. Results
2.1. RNA-Sequencing and General Transcription Patterns
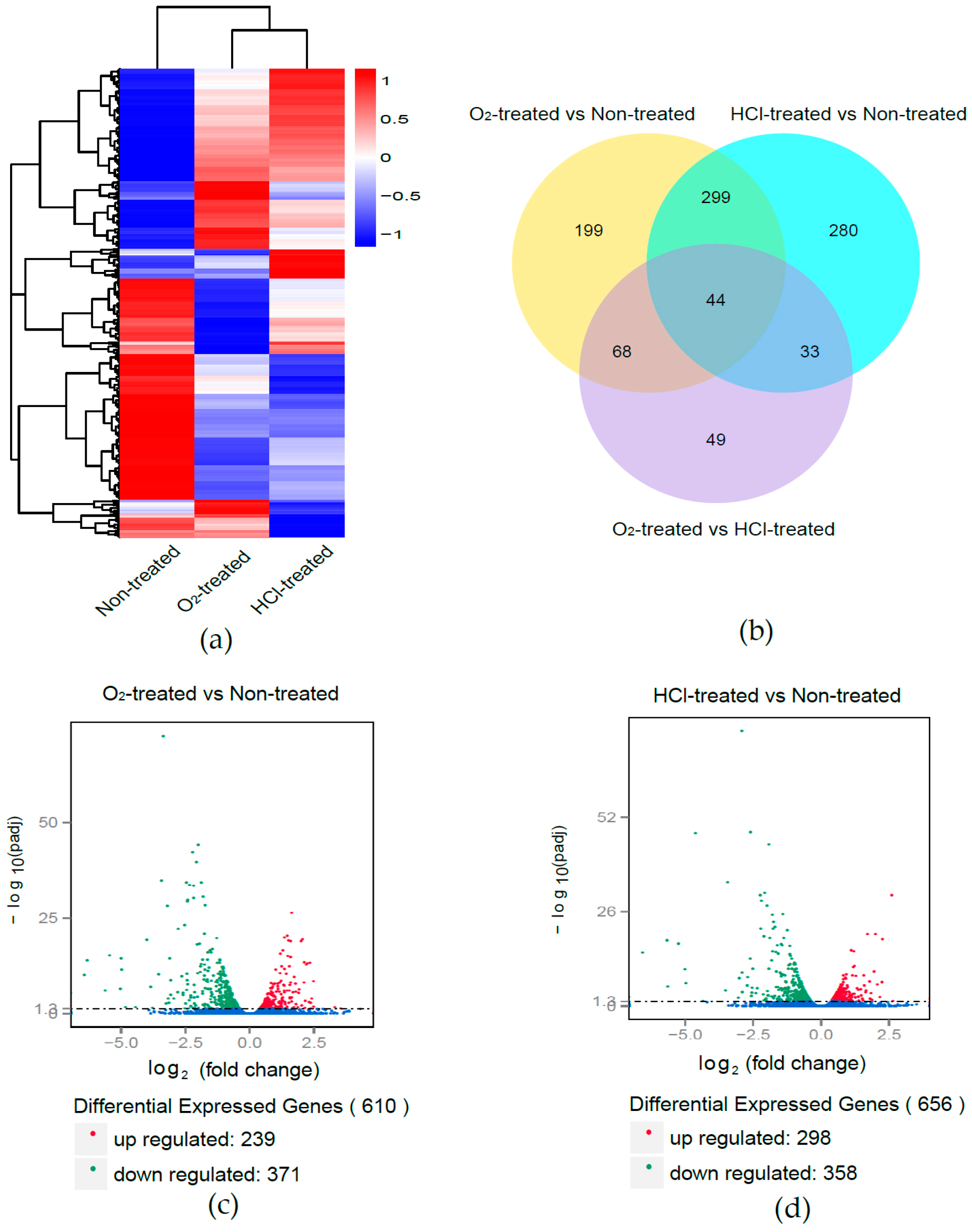
2.2. Differentially Expressed Genes (DEGs) in O2-Treated and HCl-Treated Eggs
2.3. Common DEGs between O2-Treated and HCl-Treated Eggs
2.4. GO and KEGG Enrichment Analysis of DEGs
2.5. Validation of RNA-seq Data by qRT-PCR
3. Discussion
3.1. Trehalose Synthesis and Transportation
3.2. Proteins Involved in Polyols
3.3. Heat Shock Proteins (HSPs)
3.4. Cytochrome P450 (CYP)
3.5. Growth-Arrest and DNA Damage Inducible 45 (GADD45)
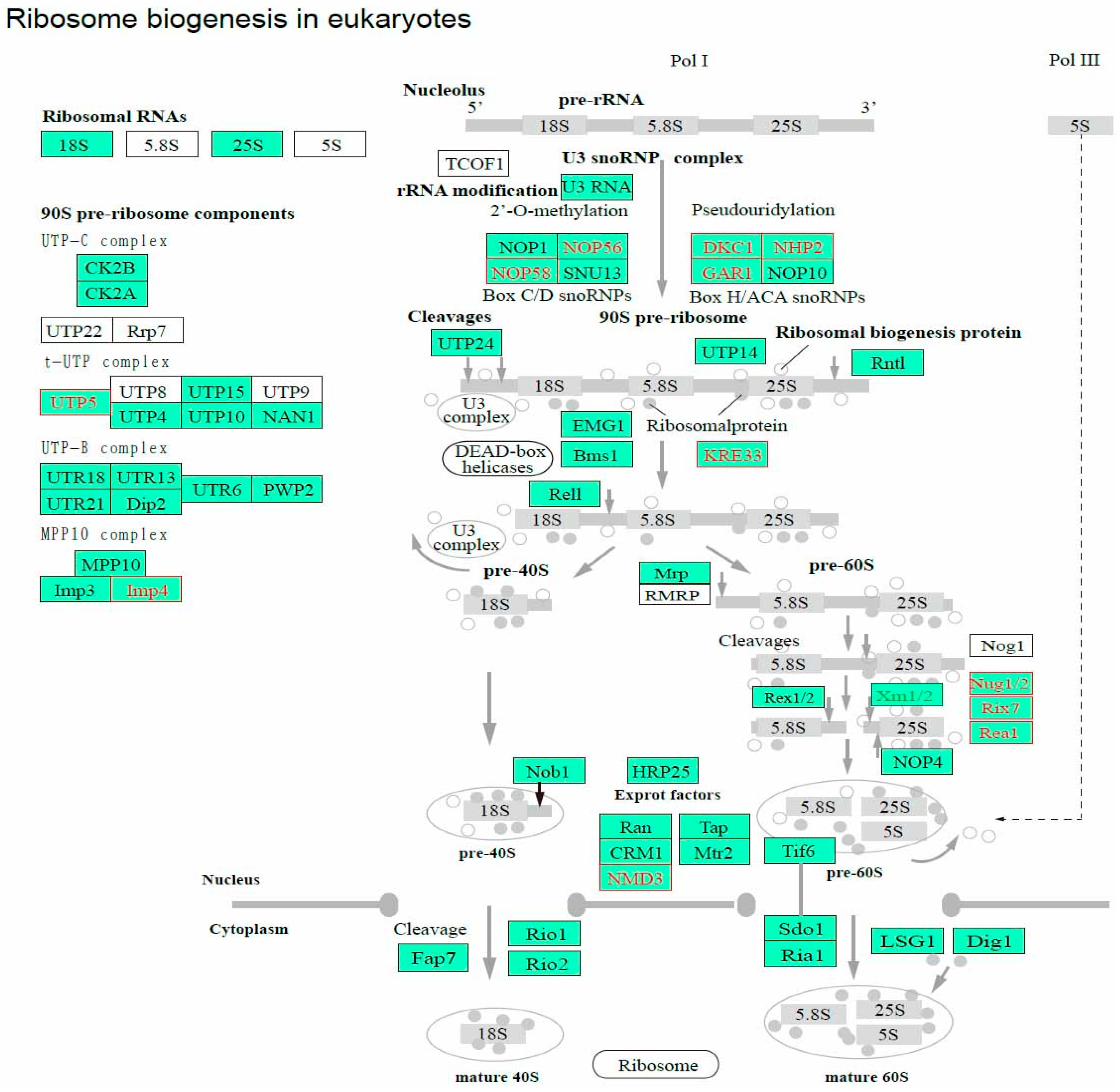
3.6. Ribosome Biogenesis and Spliceosome
4. Materials and Methods
4.1. Sample preparation
4.2. RNA Sequencing
4.3. Data Analysis
4.4. GO Annotation and Enrichment Analysis
4.5. Validation of DEGs by Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| HSP | Heat shock protein |
| DGE | Digital gene expression |
| DEGs | Differentially expressed genes |
References
- Nijhout, H. Insect Hormones; Princeton University Press: Princeton, NJ, USA, 1994; pp. 160–165. [Google Scholar]
- Podrabsky, J.E.; Hand, S.C. Physiological strategies during animal diapause: lessons from brine shrimp and annual killifish. J. Exp. Biol. 2015, 218, 1897–1906. [Google Scholar] [CrossRef] [PubMed]
- Guo, F. Insect Hormones; Science Press of China: Beijing, China, 1979; pp. 230–233. [Google Scholar]
- Yamashita, O.; Hasegawa, K. Embryonic diapause. In Comprehensive Insect Physiology, Biochemistry and Pharmacology; Pergamon Press: Oxford, UK, 1985; Volume 1, pp. 407–434. [Google Scholar]
- Nakagaki, M.; Takei, R.; Nagashima, E.; Yaginuma, T. Cell cycles in embryos of the silkworm, Bombyx mori: G2-arrest at diapause stage. Roux's Arch. Dev. Biol. 1991, 200, 223–229. [Google Scholar] [CrossRef]
- Huang, J.; Zhu, W.; Xia, J.; Xiang, Z. Complete Works of Sericultural Technology in China; Sichuan Publishing House of Science & Technology Press: Chengdu, China, 1996; pp. 519–521. [Google Scholar]
- Meng, G.; Yao, J.M.; Wang, L.; Zhao, L.C. Variation in glutathione status associated with induction and initiation of diapause in eggs of the bivoltine strain of the silkworm Bombyx mori. Physiol. Entomol. 2011, 36, 173–179. [Google Scholar] [CrossRef]
- Yamashita, O. Diapause hormone of the silkworm, Bombyx mori: Structure, gene expression and function. J. Insect Physiol. 1996, 42, 669–679. [Google Scholar] [CrossRef]
- Chino, H. Carbohydrate metabolism in the diapause egg of the silkworm, Bombyx-mori. 2. conversion of glycogen into sorbitol and glycerol during diapause. J. Insect Physiol. 1958, 2, 1. [Google Scholar] [CrossRef]
- Sonobe, H.; Matsumoto, A.; Fukuzaki, Y.; Fujiwara, S. Carbohydrate-Metabolism and restricted oxygen-supply in the eggs of the silkworm, Bombyx-mori. J. Insect Physiol. 1979, 25, 381–388. [Google Scholar] [CrossRef]
- Shen, A.; Zhao, L.; Liu, H. Study on the Activation of Silkworm Diapausing Eggs With Hydrogen Peroxide. Acta Sericol. Sin. 2003, 29, 311–313. [Google Scholar]
- Yamamoto, T.; Mase, K.; Sawada, H. Diapause prevention effect of Bombyx mori by dimethyl sulfoxide. PLoS ONE 2013, 8, e64124. [Google Scholar] [CrossRef]
- Sasibhushan, S.; Ponnuvel, K.M.; Vijayaprakash, N.B. Changes in diapause related gene expression pattern during early embryonic development in HCl-treated eggs of bivoltine silkworm Bombyx mori (Lepidoptera: Bombycidae). Braz. Arch. Biol. Technol. 2013, 56, 1–10. [Google Scholar] [CrossRef]
- Fan, L.; Lin, J.; Zhong, Y.; Liu, J. Shotgun proteomic analysis on the diapause and non-diapause eggs of domesticated silkworm Bombyx mori. PLoS ONE 2013, 8, e60386. [Google Scholar] [CrossRef] [PubMed]
- Akitomo, S.; Egi, Y.; Nakamura, Y.; Suetsugu, Y.; Oishi, K.; Sakamoto, K. Genome-wide microarray screening for Bombyx mori genes related to transmitting the determination outcome of whether to produce diapause or non-diapause eggs. Insect Sci. 2015. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, O.; Yaginuma, T.; Hasegawa, K. Hormonal and Metabolic Control of Egg Diapause of the Silkworm, Bombyx-mori (Lepidoptera, Bombycidae). Entomol. Gen. 1981, 7, 195–211. [Google Scholar]
- Mitsumasu, K.; Azuma, M.; Niimi, T.; Yamashita, O.; Yaginuma, T. Membrane-penetrating trehalase from silkworm Bombyx mori. Molecular cloning and localization in larval midgut. Insect Mol. Biol. 2005, 14, 501–508. [Google Scholar] [CrossRef] [PubMed]
- Kamei, Y.; Hasegawa, Y.; Niimi, T.; Yamashita, O.; Yaginuma, T. Trehalase-2 protein contributes to trehalase activity enhanced by diapause hormone in developing ovaries of the silkworm, Bombyx mori. J. Insect Physiol. 2011, 57, 608–613. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, O.; Hasegawa, K. Diapause Hormone Action in Silkworm Ovaries Incubated in vitro 14c-Trehalose Incorporation into Glycogen. J. Insect Physiol. 1976, 22, 409–414. [Google Scholar] [CrossRef]
- Chen, Q.; Haddad, G.G. Role of trehalose phosphate synthase and trehalose during hypoxia: From flies to mammals. J. Exp. Biol. 2004, 207, 3125–3129. [Google Scholar] [CrossRef] [PubMed]
- Kanamori, Y.; Saito, A.; Hagiwara-Komoda, Y.; Tanaka, D.; Mitsumasu, K.; Kikuta, S.; Watanabe, M.; Cornette, R.; Kikawada, T.; Okuda, T. The trehalose transporter 1 gene sequence is conserved in insects and encodes proteins with different kinetic properties involved in trehalose import into peripheral tissues. Insect Biochem. Mol. 2010, 40, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Kikawada, T.; Saito, A.; Kanamori, Y.; Nakahara, Y.; Iwata, K.; Tanaka, D.; Watanabe, M.; Okuda, T. Trehalose transporter 1, a facilitated and high-capacity trehalose transporter, allows exogenous trehalose uptake into cells. Proc. Natl. Acad. Sci. USA 2007, 104, 11585–11590. [Google Scholar] [CrossRef] [PubMed]
- Chino, H. Conversion of Glycogen to Sorbitol and Glycerol in the Diapause Egg of the Bombyx Silkworm. Nature 1957, 180, 606–607. [Google Scholar] [CrossRef]
- Yaginuma, T.; Yamashita, O. Nad-Dependent Sorbitol Dehydrogenase-Activity in Relation to the Termination of Diapause in Eggs of Bombyx-mori. Insect Biochem. 1979, 9, 547–553. [Google Scholar] [CrossRef]
- Wang, T.; Hou, M.; Zhao, N.; Chen, Y.F.; Lv, Y.; Li, Z.R.; Zhang, R.; Zou, X.Y.; Hou, L. Cloning and expression of the sorbitol dehydrogenase gene during embryonic development and temperature stress in Artemia sinica. Gene 2013, 521, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Khodayari, S.; Moharramipour, S.; Larvor, V.; Hidalgo, K.; Renault, D. Deciphering the Metabolic Changes Associated with Diapause Syndrome and Cold Acclimation in the Two-Spotted Spider Mite Tetranychus urticae. PLoS ONE 2013, 8, e54025. [Google Scholar] [CrossRef]
- Hayward, S.A.; Pavlides, S.C.; Tammariello, S.P.; Rinehart, J.P.; Denlinger, D.L. Temporal expression patterns of diapause-associated genes in flesh fly pupae from the onset of diapause through post-diapause quiescence. J. Insect Physiol. 2005, 51, 631–640. [Google Scholar] [CrossRef] [PubMed]
- Yocum, G.D.; Joplin, K.H.; Denlinger, D.L. Expression of Heat-Shock Proteins in Response to High and Low-Temperature Extremes in Diapausing Pharate Larvae of the Gypsy-Moth, Lymantria-Dispar. Arch. Insect Biochem. 1991, 18, 239–249. [Google Scholar] [CrossRef]
- King, A.M.; MacRae, T.H. Insect heat shock proteins during stress and diapause. Annu. Rev. Entomol. 2015, 60, 59–75. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.W.; Li, X.; Yu, Q.Y.; Xiang, Z.H.; Kishino, H.; Zhang, Z. The small heat shock protein (sHSP) genes in the silkworm, Bombyx mori, and comparative analysis with other insect sHSP genes. BMC Evol. Biol. 2009, 9, 215. [Google Scholar] [CrossRef] [PubMed]
- King, A.M.; MacRae, T.H. The small heat shock protein p26 aids development of encysting Artemia embryos, prevents spontaneous diapause termination and protects against stress. PLoS ONE 2012, 7, e43723. [Google Scholar] [CrossRef] [PubMed]
- Feyereisen, R. Insect P450 enzymes. Annu. Rev. Entomol. 1999, 44, 507–533. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.G. Cytochromes P450 and insecticide resistance. Insect Biochem. Mol. 1999, 29, 757–777. [Google Scholar] [CrossRef]
- Yang, P.; Tanaka, H.; Kuwano, E.; Suzuki, K. A novel cytochrome P450 gene (CYP4G25) of the silkmoth Antheraea yamamai: Cloning and expression pattern in pharate first instar larvae in relation to diapause. J. Insect Physiol. 2008, 54, 636–643. [Google Scholar] [CrossRef] [PubMed]
- Feyereisen, R. Evolution of insect P450. Biochem. Soc. Trans. 2006, 34, 1252–1255. [Google Scholar] [CrossRef] [PubMed]
- Hannemann, F.; Bichet, A.; Ewen, K.M.; Bernhardt, R. Cytochrome P450 systems-biological variations of electron transport chains. BBA-Gen. Subj. 2007, 1770, 330–344. [Google Scholar] [CrossRef] [PubMed]
- Moskalev, A.A.; Smit-McBride, Z.; Shaposhnikov, M.V.; Plyusnina, E.N.; Zhavoronkov, A.; Budovsky, A.; Tacutu, R.; Fraifeld, V.E. Gadd45 proteins: Relevance to aging, longevity and age-related pathologies. Ageing Res. Rev. 2012, 11, 51–66. [Google Scholar] [CrossRef] [PubMed]
- Moskalev, A.; Plyusnina, E.; Shaposhnikov, M.; Shilova, L.; Kazachenok, A.; Zhavoronkov, A. The role of D-GADD45 in oxidative, thermal and genotoxic stress resistance. Cell Cycle 2012, 11, 4222–4241. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Poelchau, M.F.; Armbruster, P.A. Global transcriptional dynamics of diapause induction in non-blood-fed and blood-fed Aedes albopictus. PLoS Negl. Trop. Dis. 2015, 9, e0003724. [Google Scholar] [CrossRef] [PubMed]
- Thomson, E.; Ferreira-Cerca, S.; Hurt, E. Eukaryotic ribosome biogenesis at a glance. J. Cell Sci. 2013, 126, 4815–4821. [Google Scholar] [CrossRef] [PubMed]
- Kurata, S.; Koga, K.; Sakaguchi, B. Nucleolar size in parallel with ribosomal RNA synthesis at diapause termination in the eggs of Bombyx mori. Chromosoma 1978, 68, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Denlinger, D.L. A potential role for ribosomal protein S2 in the gene network regulating reproductive diapause in the mosquito Culex pipiens. J. Comp. Physiol. B 2010, 180, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Sim, C.; Denlinger, D.L. RNA interference directed against ribosomal protein S3a suggests a link between this gene and arrested ovarian development during adult diapause in Culex pipiens. Insect Mol. Biol. 2010, 19, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Will, C.L.; Luhrmann, R. Spliceosome structure and function. Cold Spring Harb. Perspect. Biol. 2011, 3, a003707. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Desneux, N.; Lei, C.; Niu, C. Transcriptome characterization analysis of Bactrocera minax and new insights into its pupal diapause development with gene expression analysis. Int. J. Biol. Sci. 2014, 10, 1051–1063. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Zhang, L.; Han, Y.; Ren, X.; Huang, J.; Chen, H. De novo transcriptome sequencing and analysis of Coccinella septempunctata L. in non-diapause, diapause and diapause-terminated states to identify diapause-associated genes. BMC Genom. 2015, 16, 1086. [Google Scholar] [CrossRef] [PubMed]
- Langmead, B.; Trapnell, C.; Pop, M.; Salzberg, S.L. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol. 2009, 10, R25. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Pertea, G.; Trapnell, C.; Pimentel, H.; Kelley, R.; Salzberg, S.L. TopHat2: Accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol. 2013, 14, R36. [Google Scholar] [CrossRef] [PubMed]
- Utturkar, S.M.; Klingeman, D.M.; Land, M.L.; Schadt, C.W.; Doktycz, M.J.; Pelletier, D.A.; Brown, S.D. Evaluation and validation of de novo and hybrid assembly techniques to derive high-quality genome sequences. Bioinformatics 2014, 30, 2709–2716. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Feng, Z.; Wang, X.; Wang, X.; Zhang, X. DEGseq: An R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics 2010, 26, 136–138. [Google Scholar] [CrossRef] [PubMed]
- Young, M.D.; Wakefield, M.J.; Smyth, G.K.; Oshlack, A. Gene ontology analysis for RNA-seq: Accounting for selection bias. Genome Biol. 2010, 11, R14. [Google Scholar] [CrossRef] [PubMed]
- Shaffer, J.P. Controlling the false discovery rate with constraints: The Newman-Keuls test revisited. Biom. J. Biom. Z. 2007, 49, 136–143. [Google Scholar] [CrossRef]




| Sample Name | Raw Reads | Clean Reads | Clean Bases | Total Mapped | Error Rate (%) | Q20 (%) | Q30 (%) | GC Content (%) |
|---|---|---|---|---|---|---|---|---|
| O2-treated_1 | 12,984,364 | 12,920,255 | 0.65 G | 12,360,505 (95.67%) | 0.01 | 97.9 | 94.07 | 43.13 |
| O2-treated_2 | 11,192,761 | 11,131,963 | 0.56 G | 10,740,908 (96.49%) | 0.01 | 98.84 | 96.07 | 43.38 |
| HCl-treated_1 | 13,670,896 | 13,598,605 | 0.68 G | 13,068,581 (96.1%) | 0.01 | 97.92 | 94.13 | 43.44 |
| HCl-treated_2 | 13,595,227 | 13,514,169 | 0.68 G | 12,993,548 (96.15%) | 0.01 | 98.64 | 95.87 | 45.03 |
| Non-treated_1 | 12,361,440 | 12,313,326 | 0.62 G | 11,827,559 (96.05%) | 0.01 | 97.91 | 94.12 | 43.15 |
| Non-treated_2 | 12,559,734 | 12,506,248 | 0.63 G | 11,993,516 (95.9%) | 0.01 | 98.16 | 94.66 | 43.5 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gong, J.; Tian, S.; Zhou, X.; Yang, H.; Zhu, Y.; Hou, Y. Transcriptional Response of Silkworm (Bombyx mori) Eggs to O2 or HCl Treatment. Int. J. Mol. Sci. 2016, 17, 1838. https://doi.org/10.3390/ijms17121838
Gong J, Tian S, Zhou X, Yang H, Zhu Y, Hou Y. Transcriptional Response of Silkworm (Bombyx mori) Eggs to O2 or HCl Treatment. International Journal of Molecular Sciences. 2016; 17(12):1838. https://doi.org/10.3390/ijms17121838
Chicago/Turabian StyleGong, Jing, Sha Tian, Xia Zhou, Huan Yang, Yong Zhu, and Yong Hou. 2016. "Transcriptional Response of Silkworm (Bombyx mori) Eggs to O2 or HCl Treatment" International Journal of Molecular Sciences 17, no. 12: 1838. https://doi.org/10.3390/ijms17121838