2. Results
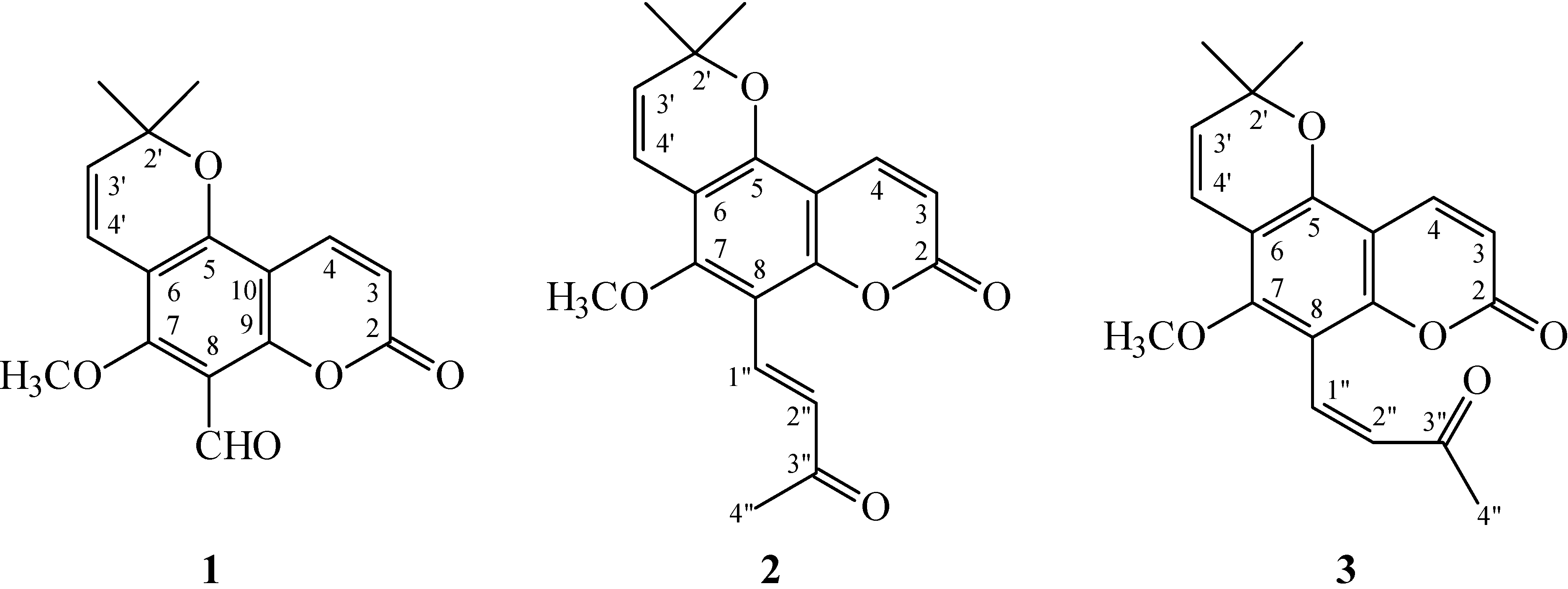
Chromatographic purification of the EtOAc-soluble fraction of a MeOH extract of stem bark of Z. avicennae on a silica gel column and preparative thin-layer chromatography (TLC) afforded three new (1–3) and fifteen known compounds (4–18).
8-Formylalloxanthoxyletin (
1) was isolated as amorphous powder with molecular formula C
16H
14O
5 as determined by positive-ion high-resolution electrospray ionization (HR-ESI-MS), showing an [M + Na]
+ ion at
m/
z 309.0739 (calculated for C
16H
14O
5Na, 309.0739). The presence of carbonyl groups was revealed by the bands at 1693, 1729 cm
−1 in the Infrared (IR) spectrum, and was confirmed by the resonances at δ 159.4, 185.7 in the
13C NMR spectrum. The
1H NMR spectrum of
1 showed the presence of a 2,2-dimethyl-2
H-pyran moiety [δ 1.53 (6H, s, Me-2' × 2), 5.70, 6.64 (each 1H, each d,
J = 10.5 Hz, H-3' and H-4')], a methoxy group [δ 3.92 (3H, s, OMe-7)], a formyl group [δ 10.53 (1H, s, CHO-8)], and the typical H
-3 and H
-4 protons of the coumarin nucleus [δ 6.32, 8.00 (each 1H, each d,
J = 10.0 Hz, H
-3 and H-4)]. The
1H NMR spectrum of
1 was similar to those of alloxanthoxyletin (
4) [
7], except that the 8-formyl group [δ 10.53 (1H, s)] of
1 replaced H-8 of alloxanthoxyletin (
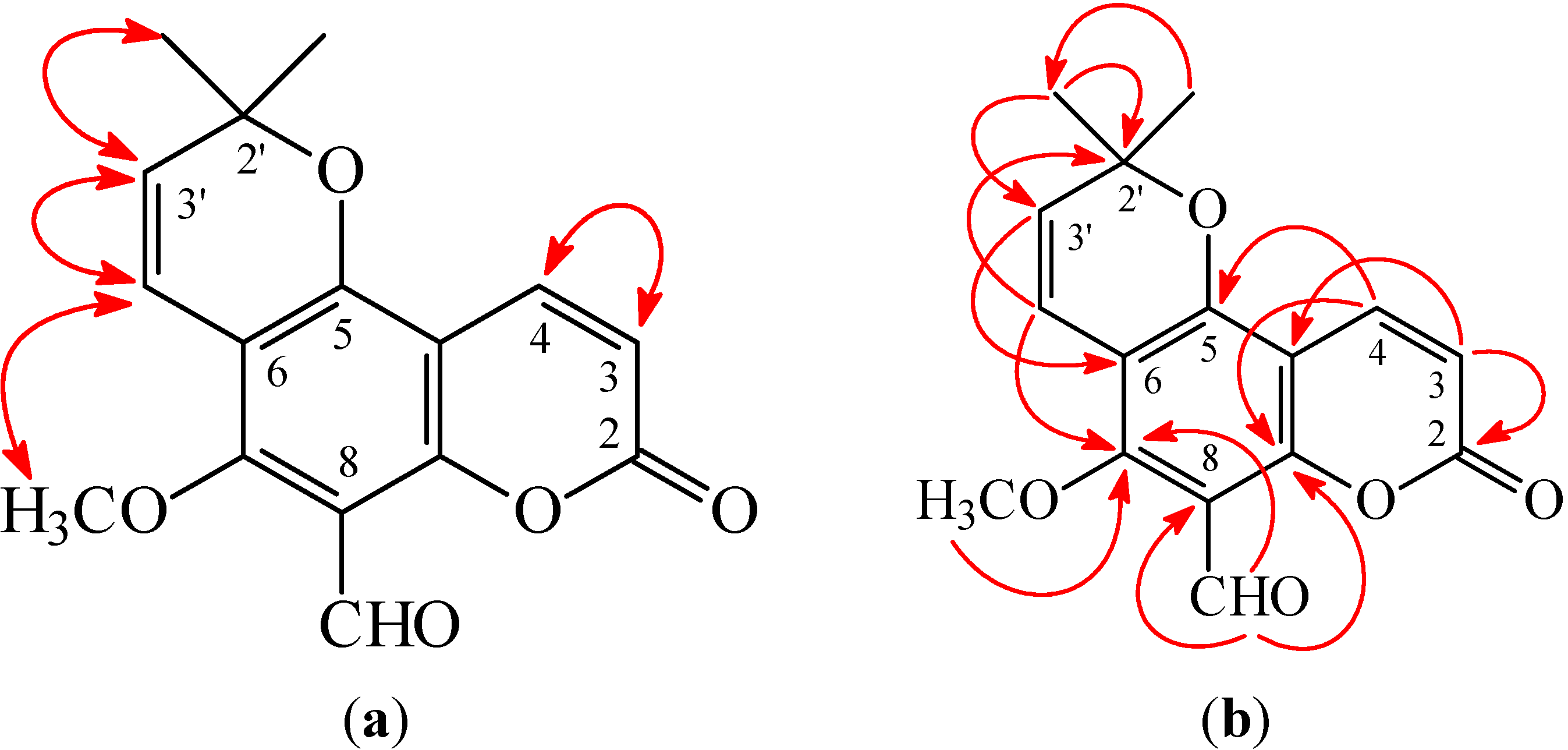
4). This was supported by HMBC correlation observed between CHO-8 (δ 10.53) and C-7 (δ 160.2), C-8 (δ 110.7), and C-9 (δ 157.5). The full assignment of
1H and
13C NMR resonances was confirmed by
1H–
1H correlation spectroscopy (COSY), nuclear Overhauser effect spectrometry (NOESY) (
Figure 3), Distortionless Enhancement by Polazization Transfer (DEPT), eteronuclear single-quantum coherence (HSQC), and heteronuclear multiple-bond correlation (HMBC) (
Figure 3) techniques. According to the evidence above, the structure of
1 was elucidated as 5-methoxy-2,2-dimethyl-8-oxo-2,8-dihydropyrano [2,3-
f]chromene-6-carbaldehyde, named 8-formylalloxanthoxyletin.
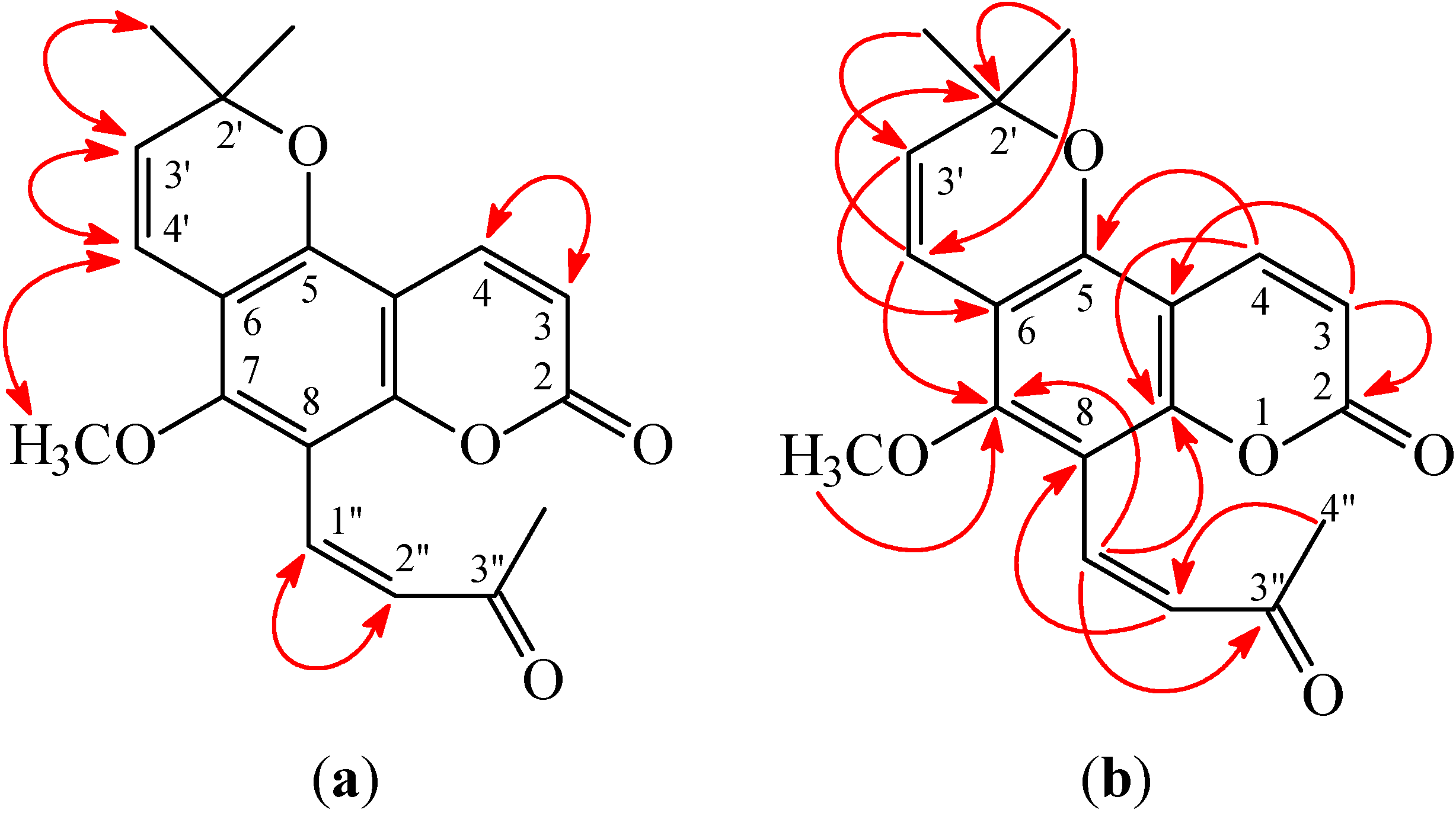
Figure 3.
Key nuclear Overhauser effect spectrometry (NOESY) (a) and heteronuclear multiple-bond correlation (HMBC) (b) correlations of 1.
Figure 3.
Key nuclear Overhauser effect spectrometry (NOESY) (a) and heteronuclear multiple-bond correlation (HMBC) (b) correlations of 1.
(
E)-Avicennone (
2) was obtained as amorphous powder. Its molecular formula, C
19H
18O
5, was determined on the basis of the positive HRESIMS at
m/
z 349.1056 [M + Na]
+ (calcd 349.1052) and supported by the
1H,
13C, and DEPT NMR data. The presence of carbonyl groups was revealed by the bands at 1695 and 1734 cm
−1 in the IR spectrum, and was confirmed by the resonances at δ 160.0 and 199.8 in the
13C NMR spectrum. The
1H NMR spectrum of
2 displayed the presence of a 2,2-dimethyl-2
H-pyran moiety [δ 1.51 (6H, s, Me-2' × 2), 5.70, 6.59 (each 1H, each d,
J = 10.2 Hz, H-3' and H-4')], a methoxy group [δ 3.81 (3H, s, OMe-7)], an (
E)-3-oxobut-1-enyl group [δ 2.42 (3H, s, Me-3''), 7.32, 7.83 (each 1H, each d,
J = 16.5 Hz, H-2'' and H-1'')], and the typical H
-3 and H
-4 protons of the coumarin nucleus [δ 6.30, 8.02 (each 1H, each d,
J = 9.6 Hz, H
-3 and H-4)]. The
1H NMR data of
2 were similar to 8-formylalloxanthoxyletin (
1), except that the (
E)-3-oxobut-1-enyl group at C-8 of
2 replaced 8-formyl group of 8-formylalloxanthoxyletin (
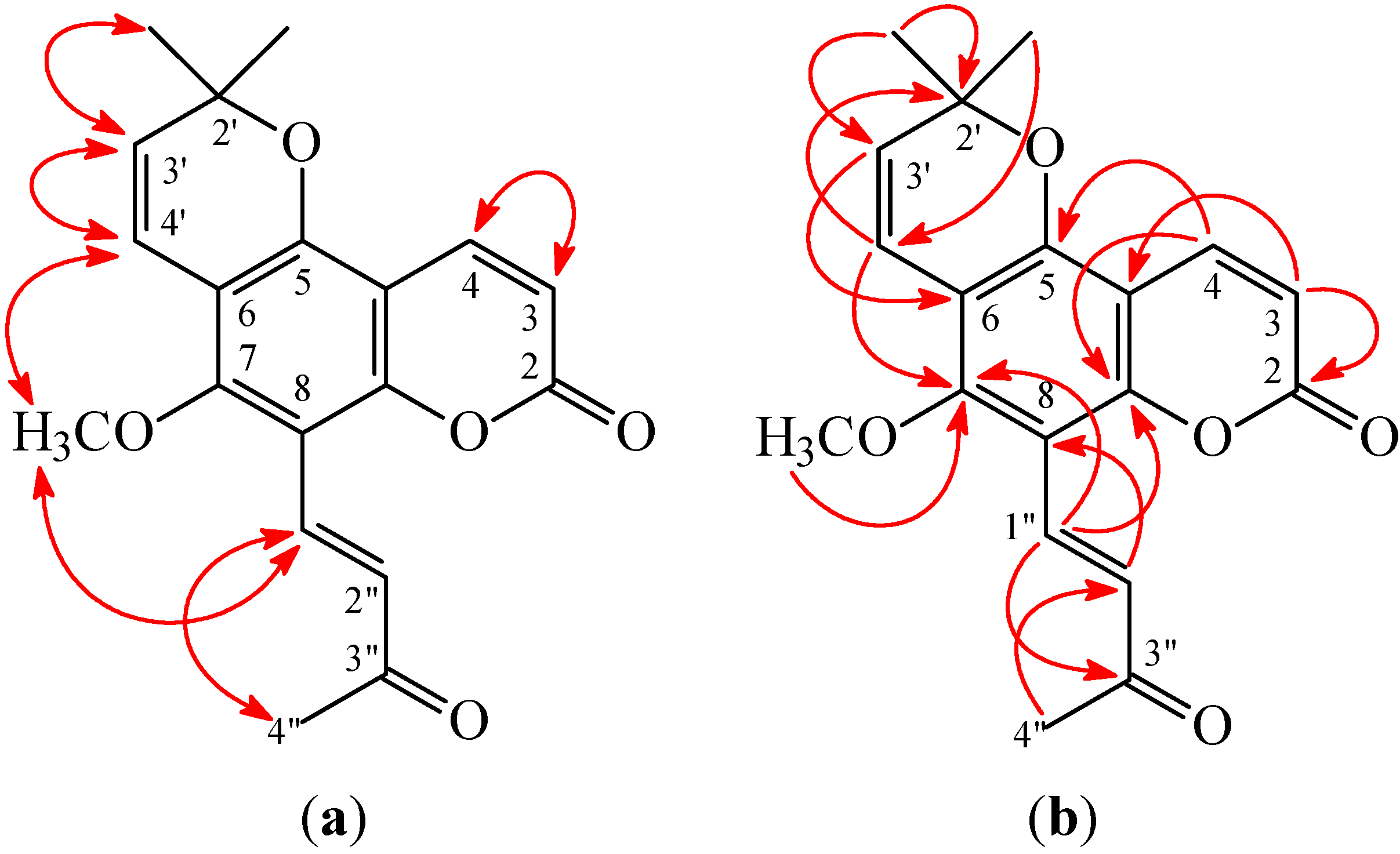
1). This was supported by the NOESY correlations between H-1'' (δ 7.83) and H-4'' (δ 2.42) and OMe-7 (δ 3.81) and by the HMBC correlation between H-1'' (δ 7.83) and C-7 (δ 159.2), C-8 (δ 107.5), C-9 (δ 154.1), and C-3'' (δ 199.8) of
2. In addition, the (
E)-configuration of
2 was established by the following evidences: (a) The larger coupling constant (
J = 16.5 Hz) between H-1'' and H-2'' of
2; (b) The NOESY correlations were observed between H-1'' (δ 7.83) and H-4'' (δ 2.42) (
Figure 3). Thus, (
E)-avicennone was elucidated as structure
2. This structure was confirmed by the
1H-
1H COSY, NOESY (
Figure 4), DEPT, HSQC, and HMBC techniques (
Figure 4).
Figure 4.
Key NOESY (a) and HMBC (b) correlations of 2.
Figure 4.
Key NOESY (a) and HMBC (b) correlations of 2.
(
Z)-Avicennone (
3) was isolated as amorphous powder. The molecular formula C
19H
18O
5 was deduced from a sodium adduct ion at
m/
z 349.1054 [M + Na]
+ (calcd 349.1052) in the HR-ESI mass spectrum. The presence of carbonyl groups was revealed by the bands at 1694, 1734 cm
−1 in the IR spectrum. The
1H NMR spectrum of
3 illustrated the presence of a 2,2-dimethyl-2
H-pyran moiety [δ 1.49 (6H, s, Me-2' × 2), 5.64, 6.56 (each 1H, each d,
J = 10.2 Hz, H-3' and H-4')], a methoxy group [δ 3.72 (3H, s, OMe-7)], an (
Z)-3-oxobut-1-enyl group [δ 2.23 (3H, s, Me-3''), 6.40, 6.79 (each 1H, each d,
J = 12.6 Hz, H-2'' and H-1'')], and the typical H
-3 and H
-4 protons of the coumarin nucleus [δ 6.23, 7.99 (each 1H, each d,
J = 9.6 Hz, H
-3 and H-4)]. The
1H and
13C NMR data of
3 were similar to those of (
E)-avicennone (
2), except that the (
Z)-3-oxobut-1-enyl group [δ
H 2.23 (3H, s, Me-3''), 6.40, 6.79 (each 1H, each d,
J = 12.6 Hz, H-2'' and H-1''); δ
C 29.7 (C-4''), 128.6 (C-1''), 132.3 (C-2''), 200.6 (C-3'')] at C-8 of
3 replaced the (
E)-3-oxobut-1-enyl group at C-8 of (
E)-avicennone (
2). This was supported by the coupling constant (
J = 12.6 Hz) between H-1'' and H-2'' of
3 and by the NOESY correlations between H-1'' (δ 6.79) and H-2'' (δ 6.40). According to the above data, the structure of
3 was elucidated as (
Z)-avicennone. This was supported by
1H–
1H COSY and NOESY (
Figure 5) experiments, and
13C NMR assignments were confirmed by DEPT, HSQC, and HMBC (
Figure 5) techniques.
Figure 5.
Key NOESY (a) and HMBC (b) correlations of 3.
Figure 5.
Key NOESY (a) and HMBC (b) correlations of 3.
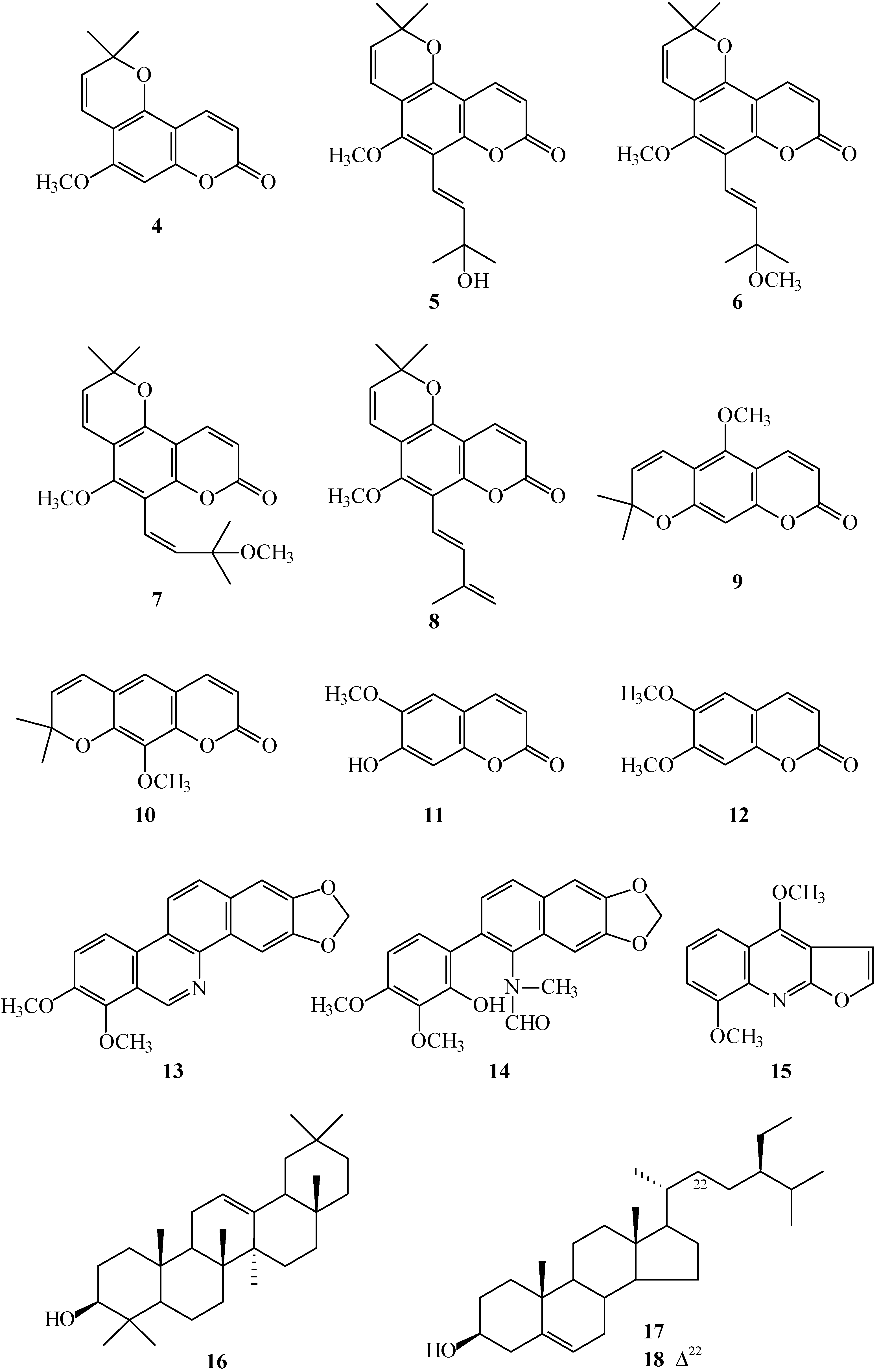
The known isolates were readily identified by a comparison of physical and spectroscopic data (UV, IR,
1H NMR, [α]
D, and MS) with corresponding authentic samples or literature values, and this included nine coumarins, alloxanthoxyletin (
4) [
7], avicennol (
5) [
8], avicennol methyl ether (
6) [
9],
cis-avicennol methyl ether (
7) [
9], avicennin (
8) [
8] [
9], xanthoxyletin (
9) [
7], luvangetin (
10) [
10], scopoletin (
11) [
11], and aesculetin dimethyl ether (
12) [
12]; two benzo[
c]phenanthridine derivatives, norchelerythrine (
13) [
13] and arnottianamide (
14) [
14]; a furoquinoline, γ-fagarine (
15) [
14]; a triterpene, β-amyrin (
16) [
15]; and two steroids, β-sitosterol (
17) [
16] and stigmasterol (
18) [
16].
Granule proteases (e.g., elastase, cathepsin G, and proteinase-3) and reactive oxygen species (ROS) (e.g., superoxide anion (O
2•−) and hydrogen peroxide) produced by human neutrophils are involved in the pathogenesis of a variety of inflammatory diseases. The effects on neutrophil pro-inflammatory responses of compounds isolated from the stem bark of
Z. avicennae were evaluated by suppressing fMet-Leu-Phe/cytochalasin B (fMLP/CB)-induced superoxide anion (O
2•−) generation and elastase release by human neutrophils. The inhibitory activity data on neutrophil pro-inflammatory responses are summarized in
Table 1. Diphenyleneiodonium and phenylmethylsulfonyl fluoride were used as positive controls for O
2•− generation and elastase release, respectively. From the results of our biological tests, the following conclusions can be drawn: (a) 8-Formylalloxanthoxyletin (
1), alloxanthoxyletin (
4), xanthoxyletin (
9), aesculetin dimethyl ether (
12), and γ-fagarine (
15) exhibited potent inhibition (IC
50 ≤ 7.65 µg/mL) of superoxide anion (O
2•−) generation by human neutrophils in response to fMLP/CB; (b) 8-Formylalloxanthoxyletin (
1), (
E)-avicennone (
2), alloxanthoxyletin (
4), avicennin (
8), and xanthoxyletin (
9) exhibited potent inhibition (IC
50 ≤ 8.17 µg/mL) against fMLP-induced elastase release; (c) Among the coumarins with a 2,2-dimethyl-2
H-pyran moiety at C-6,7, xanthoxyletin (
9) (with a 5-methoxy group) exhibited more effective inhibition than its analogue, luvangetin (
10) (with a 8-methoxy group) against fMLP-induced O
2•− generation and elastase release; (d) Among the coumarins (
1–
8) with a 7-methoxy group and a 2,2-dimethyl-2
H-pyran moiety at C-5,6, 8-formylalloxanthoxyletin (
1) (with a 8-formyl group) and alloxanthoxyletin (
4) (without any substituted group at C-8) exhibited more effective inhibition than its analogues (
2,
3, and
5–
8) against fMLP-induced O
2•− generation and elastase release; (e) Alloxanthoxyletin (
4) and xanthoxyletin (
9) were the most effective among these compounds, with IC
50 values of 1.47 ± 0.53 and 1.47 ± 0.41 µg/mL, respectively, against fMLP-induced superoxide anion generation; and (f) 8-Formylalloxanthoxyletin (
1) exhibited the most effective among the isolates, with IC
50 value of 2.59 ± 0.52 µg/mL, against fMLP-induced elastase release.
Table 1.
Inhibitory effects of compounds 1–18 from the stem bark of Z. avicennae on superoxide radical anion generation and elastase release by human neutrophils in response to fMet-Leu-Phe/cytochalasin B a.
Table 1.
Inhibitory effects of compounds 1–18 from the stem bark of Z. avicennae on superoxide radical anion generation and elastase release by human neutrophils in response to fMet-Leu-Phe/cytochalasin B a.
| Compounds | Superoxide Anion | Elastase |
|---|
| IC50 [µg/mL] b or (Inh %) c |
|---|
| 8-Formylalloxanthoxyletin (1) | 4.60 ± 0.83 d | 2.59 ± 0.52 d |
| (E)-Avicennone (2) | (24.4 ± 5.1) e | 8.17 ± 1.61 d |
| (Z)-Avicennone (3) | (18.0 ± 3.1) | (40.2 ± 4.5) f |
| Alloxanthoxyletin (4) | 1.47 ± 0.53 d | 3.43 ± 0.63 d |
| Avicennol (5) | (27.6 ± 4.8) e | 10.9 ± 1.4 d |
| Avicennol methyl ether (6) | (28.6 ± 1.8) d | (6.31 ± 3.50) |
| cis-Avicennol methyl ether (7) | (23.2 ± 2.5) e | (6.51 ± 1.22) f |
| Avicennin (8) | (38.5 ± 4.2) d | 6.21 ± 1.60 d |
| Xanthoxyletin (9) | 1.47 ± 0.41 d | 4.18 ± 0.73 d |
| Luvangetin (10) | (29.0 ± 3.7) e | (5.28 ± 1.30) f |
| Aesculetin dimethyl ether (11) | 7.65 ± 1.62 | (45.8 ± 5.1) |
| Scopoletin (12) | (25.6 ± 4.8) | (43.7 ± 3.2) |
| Norchelerythrine (13) | (12.1 ± 2.2) e | (23.8 ± 4.5) e |
| Arnottianamide (14) | (22.8 ± 4.2) | (16.5 ± 5.1) |
| γ-Fagarine (15) | 6.85 ± 0.46 f | (21.3 ± 3.5) |
| β-Amyrin (16) | (8.75 ± 4.14) | (2.62 ± 1.61) |
| Mixture of β-Sitosterol (17) and Stigmasterol (18) | (1.25 ± 1.03) | (2.24 ± 1.14) |
| Diphenyleneiodonium g | 0.53 ± 0.22 d | – |
| Phenylmethylsulfonyl fluoride g | – | 34.2 ± 5.6 d |
4. Experimental Section
4.1. Ethics Statement
Blood was taken from healthy human donors (20–30 years old) by venipuncture, using a protocol (No. 102-1595A3) approved by the Institutional Review Board at Chang Gung Memorial Hospital. All donors gave written consent. The Medical Ethics Committee of Chang Gung Memorial Hospital approved this consent procedure.
4.2. General Experimental Procedures
Melting points were determined on a Yanaco micro-melting point apparatus and were uncorrected. Optical rotations were measured using a Jasco DIP-370 polarimeter (Japan Spectroscopic Corporation, Tokyo, Japan) in CHCl3. Ultraviolet (UV) spectra were obtained on a Jasco UV-240 spectrophotometer (Japan Spectroscopic Corporation). Infrared (IR) spectra (neat or KBr) were recorded on a Perkin Elmer 2000 FT-IR spectrometer (Perkin Elmer Corporation, Norwalk, CT, USA). Nuclear magnetic resonance (NMR) spectra, including correlation spectroscopy (COSY), nuclear Overhauser effect spectrometry (NOESY), heteronuclear multiple-bond correlation (HMBC), and heteronuclear single-quantum coherence (HSQC) experiments, were acquired using Varian INOVA-500 or Varian VNMRS-600 spectrometer (Varian Inc., Palo Alto, CA, USA) operating at 500 or 600 MHz (1H) and 125 or 150 MHz (13C), respectively, with chemical shifts given in ppm (δ) using tetramethylsilane (TMS) as an internal standard. Electrospray ionisation (ESI) and high-resolution electrospray ionization (HRESI)-mass spectra were recorded on a Bruker APEX II (Bruker, Bremen, Germany) or a VG Platform Electrospray ESI/MS mass spectrometer (Fison, Villeurbanne, France). Silica gel (70–230, 230–400 mesh, Merck, Darmstadt, Germany) was used for column chromatography (CC). Silica gel 60 F-254 (Merck, Darmstadt, Germany) was used for thin-layer chromatography (TLC) and preparative thin-layer chromatography (PTLC).
4.3. Plant Material
The stem bark of Z. avicennae was collected from Yanpu, Pingtung County, Taiwan, in June 2006 and identified by Jih-Jung Chen. A voucher specimen (ZA-200606) was deposited in the Department of Pharmacy, Tajen University, Pingtung, Taiwan.
4.4. Extraction and Isolation
The dried stem bark (8.75 kg) of Z. avicennae was pulverized and extracted three times with MeOH (40 L each) for 3 days. The MeOH extracts were concentrated under reduced pressure at 35 °C, and the residue (550 g) was partitioned between EtOAc and H2O (1:1). The EtOAc layer was concentrated to give a residue (fraction A, 302.8 g). The water layer was further extracted with n-BuOH, and the n-BuOH-soluble part (fraction B, 91.1 g) and the water-solubles (fraction C, 127.3 g) were separated. Fraction A (115 g) was chromatographed on silica gel (70–230 mesh, 5.2 kg), eluting with CH2Cl2, gradually increasing the polarity with MeOH to give 14 fractions: A1 (10 L, CH2Cl2), A2 (4 L, CH2Cl2/MeOH, 99:1), A3 (5 L, CH2Cl2/MeOH, 95:1), A4 (5 L, CH2Cl2/MeOH, 90:1), A5 (2 L, CH2Cl2/MeOH, 80:1), A6 (3 L, CH2Cl2/MeOH, 70:1), A7 (4 L, CH2Cl2/MeOH, 50:1), A8 (7.5 L, CH2Cl2/MeOH, 30:1), A9 (2 L, CH2Cl2/MeOH, 20:1), A10 (4 L, CH2Cl2/MeOH, 10:1), A11 (4 L, CH2Cl2/MeOH, 5:1), A12 (3 L, CH2Cl2/MeOH, 3:1), A13 (2 L, CH2Cl2/MeOH, 1:1), A14 (2 L, MeOH). Fraction A2 (11.8 g) was chromatographed further on silica gel (230–400 mesh, 535 g) eluting with n-hexane/EtOAc (3:1–0:1) to give 14 fractions (each 500 mL, A2-1–A2-14). Part (112 mg) of fraction A2-13 was purified by preparative TLC (silica gel, CH2Cl2/acetone, 30:1) to afford 12 (3.8 mg) (Rf = 0.70). Part (96 mg) of fraction A2-14 was purified by preparative TLC (silica gel, CH2Cl2/acetone, 25:1) to obtain 15 (3.3 mg) (Rf = 0.60). Fraction A4 (9.1 g) was chromatographed further on silica gel (230–400 mesh, 425 g) eluting with n-hexane/EtOAc (3:1–0:1) to give 13 fractions (each 1 L, A4-1–A4-13). Fraction A4-1 (273 mg) was purified by CC (silica gel, n-hexane/acetone, 2:1–0:1) to afford 11 subfractions (each 200 mL, A4-1-1–A4-1-11). Fraction A4-1-9 (33 mg) was purified by preparative TLC (silica gel, CHCl3/EtOAc, 5:1) to give 11 (3.8 mg) (Rf = 0.50). Part (140 mg) of fraction A4-7 was purified by preparative TLC (silica gel, CHCl3/acetone, 40:1) to obtain 7 (3.7 mg) (Rf = 0.47). Part (148 mg) of fraction A4-9 was purified by preparative TLC (silica gel, n-hexane/EtOAc, 5:1) to yield 1 (3.5 mg) (Rf = 0.24). Fraction A5 (8.8 g) was chromatographed further on silica gel (230–400 mesh, 405 g) eluting with n-hexane/EtOAc (10:1–0:1) to give 21 fractions (each 1 L, A5-1–A5-21). Part (81 mg) of fraction A5-8 was purified by preparative TLC (silica gel, n-hexane/EtOAc, 3:1) to afford 16 (6.0 mg) (Rf = 0.71). Part (72 mg) of fraction A5-11 was purified by preparative TLC (silica gel, n-hexane/CHCl3, 1:3) to yield 9 (20.1 mg) (Rf = 0.33). Fraction A5-14 (225 mg) was purified by CC (silica gel, CH2Cl2/acetone, 45:1) to afford 5 subfractions (each 200 mL, A5-14-1–A5-14-5). Fraction A5-14-2 (28 mg) was purified by preparative TLC (silica gel, n-hexane/EtOAc, 5:1) to give 4 (10.2 mg) (Rf = 0.35). Fraction A5-14-3 (67 mg) was purified by preparative TLC (silica gel, n-hexane/EtOAc, 5:1) to obtain 6 (40.1 mg) (Rf = 0.30). Part (118 mg) of fraction A5-17 was purified by preparative TLC (silica gel, n-hexane/EtOAc, 2:1) to afford 10 (77 mg) (Rf = 0.40). Fraction A6 (10.5 g) was chromatographed further on silica gel (230–400 mesh, 478 g) eluting with n-hexane/acetone (2:1–0:1) to give 11 fractions (each 500 mL, A6-1–A6-11). Part (125 mg) of fraction A6-1 was purified by preparative TLC (silica gel, CHCl3/acetone, 80:1) to obtain 2 (3.9 mg) (Rf = 0.87) and 3 (2.8 mg) (Rf = 0.88). Fraction A7 (9.2 g) was chromatographed further on silica gel (230–400 mesh, 420 g) eluting with n-hexane/EtOAc (6:1–0:1) to give 17 fractions (each 500 mL, A7-1–A7-17). Part (136 mg) of fraction A7-7 was purified by preparative TLC (silica gel, n-hexane/EtOAc, 4:1) to obtain mixture of 17 and 18 (14.1 mg) (Rf = 0.50). Part (135 mg) of fraction A7-8 was purified by preparative TLC (silica gel, n-hexane/EtOAc, 3:1) to yield 8 (10.2 mg) (Rf = 0.46). Part (125 mg) of fraction A7-9 was purified by preparative TLC (silica gel, CHCl3/EtOAc, 15:1) to give 5 (3.5 mg) (Rf = 0.79). Part (95 mg) of fraction A7-13 was purified by preparative TLC (silica gel, n-hexane/EtOAc, 2:1) to yield 13 (4.3 mg) (Rf = 0.48). Part (118 mg) of fraction A7-15 was purified by preparative TLC (silica gel, n-hexane/EtOAc, 2:3) to give 14 (5.3 mg) (Rf = 0.62).
4.4.2. (E)-Avicennone (2)
Amorphous powder. UV (MeOH): λ
max (log ε) = 227 (4.00), 281 (4.16) nm. IR (neat): υ
max = 1734 (C=O), 1695 (C=O) cm
−1.
1H NMR (CDCl
3, 600 MHz): δ = 1.51 (6H, s, Me-2' × 2), 2.42 (3H, s, Me-3''), 3.81 (3H, s, OMe-7), 5.70 (1H, d,
J = 10.2 Hz, H-3'), 6.30 (1H, d,
J = 9.6 Hz, H-3), 6.59 (1H, d,
J = 10.2 Hz, H-4'), 7.32 (1H, d,
J = 16.5 Hz, H-2''), 7.83 (1H, d,
J = 16.5 Hz, H-1''), 8.02 (1H, d,
J = 9.6 Hz, H-4) (
Figure S5).
13C NMR (CDCl
3, 150 MHz): δ = 28.2 (Me-2' × 2), 29.7 (C-4''), 62.4 (OMe), 78.7 (C-2'), 106.3 (C-10), 107.5 (C-8), 110.8 (C-6), 113.2 (C-3), 116.1 (C-4'), 129.5 (C-3'), 130.3 (C-2''), 131.6 (C-1''), 138.2 (C-4), 149.7 (C-5), 154.1 (C-9), 159.2 (C-7), 160.0 (C-2), 199.8 (C-3'') (
Figure S6). ESI-MS:
m/
z = 349 [M + Na]
+ (
Figure S7). HR-ESI-MS:
m/
z = 349.1056 [M + Na]
+ (calcd for C
19H
18O
5Na, 349.1052) (
Figure S8).
4.4.3. (Z)-Avicennone (3)
Amorphous powder. UV (MeOH): λ
max (log ε) = 226 (4.01), 280 (4.14) nm. IR (KBr): υ
max = 1734 (C=O), 1694 (C=O) cm
−1.
1H NMR (CDCl
3, 600 MHz): δ = 1.49 (6H, s, Me-2' × 2), 2.23 (3H, s, Me-3''), 3.72 (3H, s, OMe-7), 5.64 (1H, d,
J = 10.2 Hz, H-3'), 6.23 (1H, d,
J = 9.6 Hz, H-3), 6.40 (1H, d,
J = 12.6 Hz, H-2''), 6.56 (1H, d,
J = 10.2 Hz, H-4'), 6.79 (1H, d,
J = 12.6 Hz, H-1''), 7.99 (1H, d,
J = 9.6 Hz, H-4) (
Figure S9).
13C NMR (CDCl
3, 150 MHz): δ = 28.2 (Me-2' × 2), 29.7 (C-4''), 62.4 (OMe), 77.8 (C-2'), 105.9 (C-10), 107.9 (C-8), 110.3 (C-6), 113.0 (C-3), 116.2 (C-4'), 128.6 (C-1''), 129.1 (C-3'), 132.3 (C-2''), 138.2 (C-4), 148.9 (C-5), 152.6 (C-9), 156.9 (C-7), 159.8 (C-2), 200.6 (C-3'') (
Figure S10). ESI-MS:
m/
z = 349 [M + Na]
+ (
Figure S11). HR-ESI-MS:
m/
z = 349.1054 [M + Na]
+ (calcd for C
19H
18O
5Na, 349.1052) (
Figure S12).
4.5. Biological Assay
The effect of the isolated compounds on neutrophil pro-inflammatory response was evaluated by monitoring the inhibition of superoxide anion generation and elastase release in fMLP/CB-activated human neutrophils in a concentration-dependent manner. The purity of the tested compounds was >98% as identified by NMR and MS.
4.5.1. Preparation of Human Neutrophils
Human neutrophils from venous blood of healthy, adult volunteers (20–28 years old) were isolated using a standard method of dextran sedimentation prior to centrifugation in a Ficoll Hypaque gradient and hypotonic lysis of erythrocytes [
17]. Purified neutrophils containing > 98% viable cells, as determined by the trypan blue exclusion method [
18], were re-suspended in a calcium (Ca
2+)-free HBSS buffer at pH 7.4 and were maintained at 4 °C prior to use.
4.5.2. Measurement of Superoxide Anion Generation
The assay for measurement of superoxide anion generation was based on the SOD-inhibitable reduction of ferricytochrome
c [
19,
20]. In brief, after supplementation with 0.5 mg/mL ferricytochrome
c and 1 mM Ca
2+, neutrophils (6 × 10
5/mL) were equilibrated at 37 °C for 2 min and incubated with different concentrations (10–0.01 μg/mL) of compounds or DMSO (as control) for 5 min. Cells were incubated with cytochalasin B (1 μg/mL) for 3 min prior to the activation with 100 nM formyl-
l-methionyl-
l-leucyl-
lphenylalanine for 10 min. Changes in absorbance with the reduction of ferricytochrome
c at 550 nm were continuously monitored in a double-beam, six-cell positioner spectrophotometer with constant stirring (Hitachi U-3010, Tokyo, Japan). Calculations were based on differences in the reactions with and without SOD (100 U/mL) divided by the extinction coefficient for the reduction of ferricytochrome
c (ε = 21.1/mM/10 mm).
4.5.3. Measurement of Elastase Release
Degranulation of azurophilic granules was determined by measuring elastase release as described previously [
19,
20]. Experiments were performed using MeO-Suc-Ala-Ala-Pro-Val-
p-nitroanilide as the elastase substrate. Briefly, after supplementation with MeO-Suc-Ala-Ala-Pro-Val-
p-nitroanilide (100 μM), neutrophils (6 × 10
5/mL) were equilibrated at 37 °C for 2 min and incubated with compounds for 5 min. Cells were stimulated with fMLP (100 nM)/CB (0.5 μg/mL), and changes in absorbance at 405 nm were monitored continuously in order to assay elastase release. The results were expressed as the percent of elastase release in the fMLP/CB-activated, drug-free control system.
4.5.4. Statistical Analysis
Results are expressed as the mean ± standard error of the mean (SEM), and comparisons were made using Student’s t-test. A probability of 0.05 or less was considered significant. The software SigmaPlot was used for the statistical analysis.