Induction of Nickel Accumulation in Response to Zinc Deficiency in Arabidopsis thaliana
Abstract
:1. Introduction
2. Results
2.1. Ni Accumulation under Zn Deficiency in Hydroponics
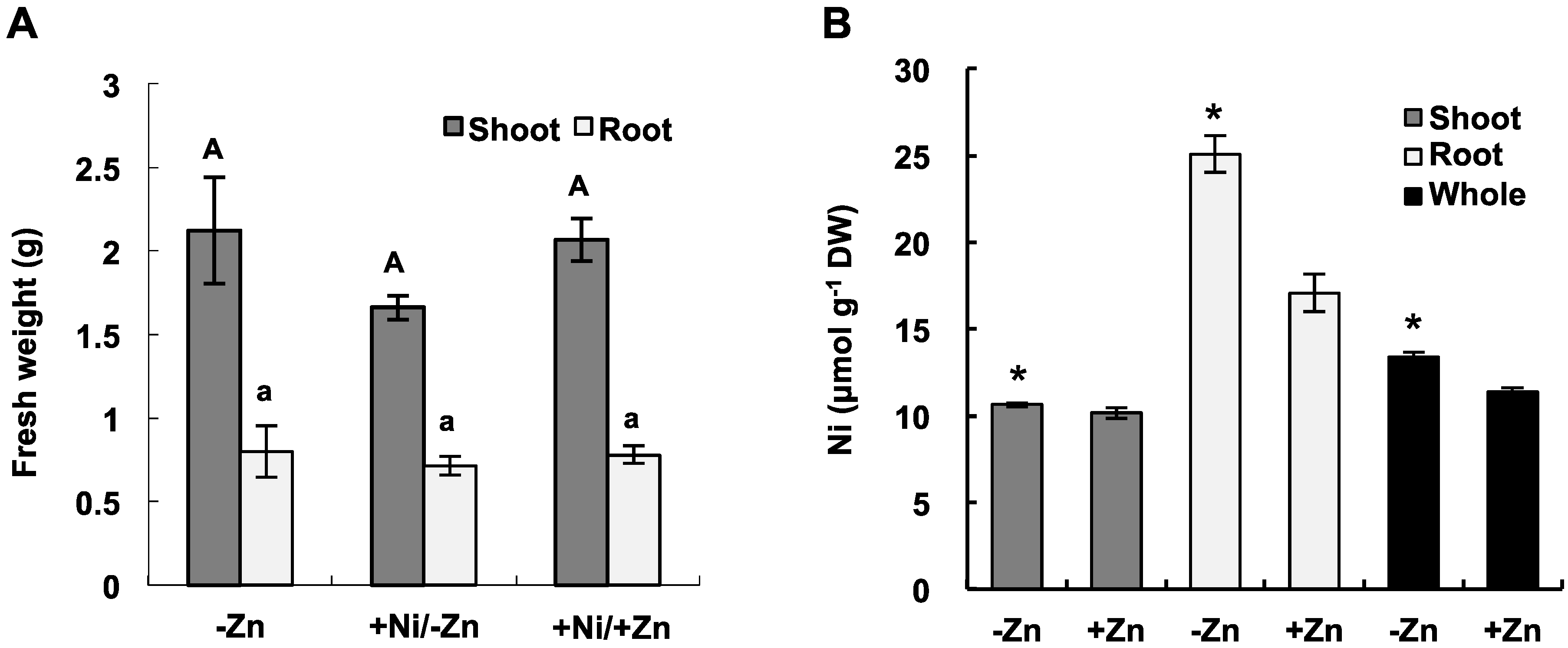
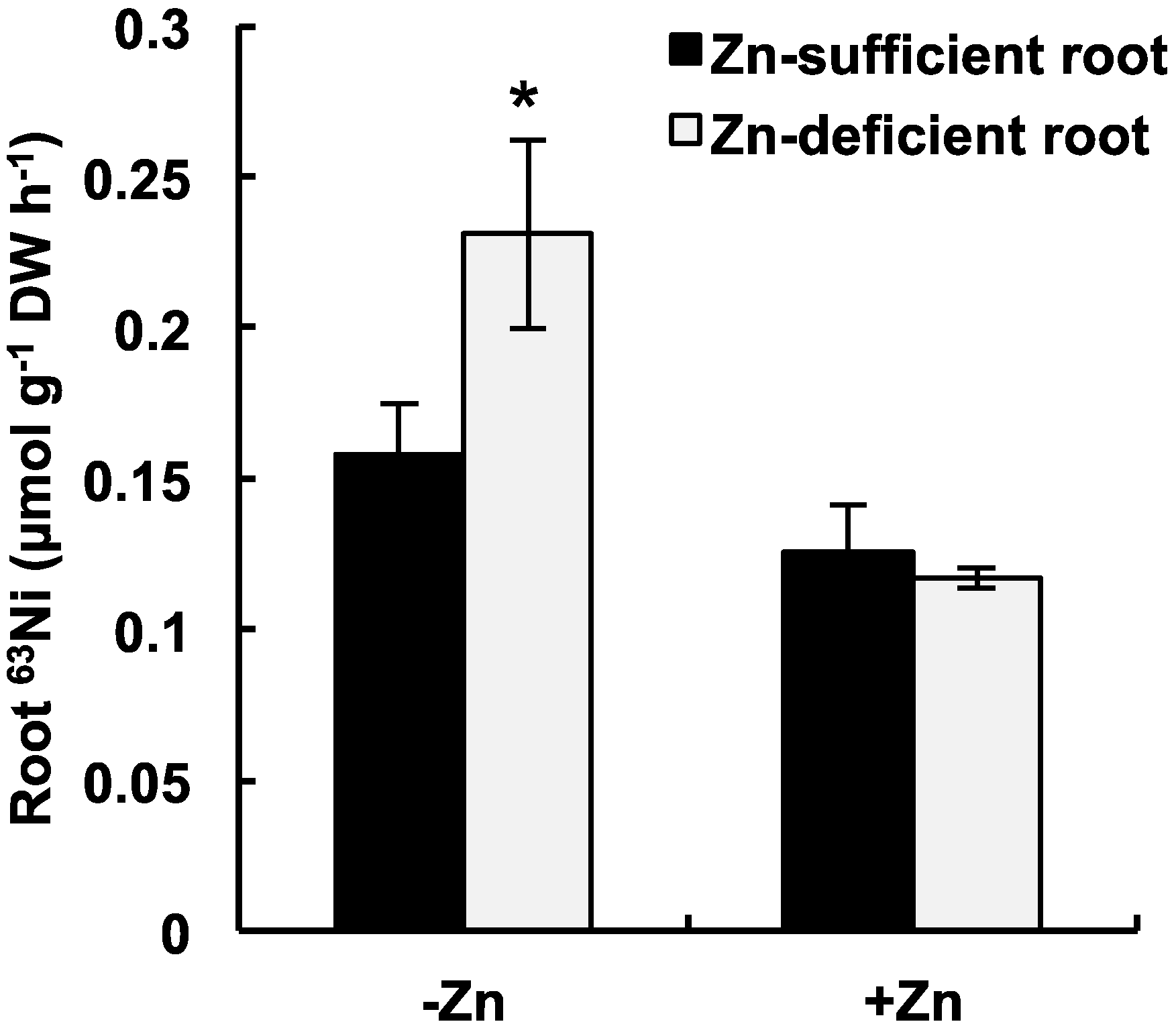
2.2. Short-Term 63Ni Absorption in Zn-Deficient/Sufficient Plants
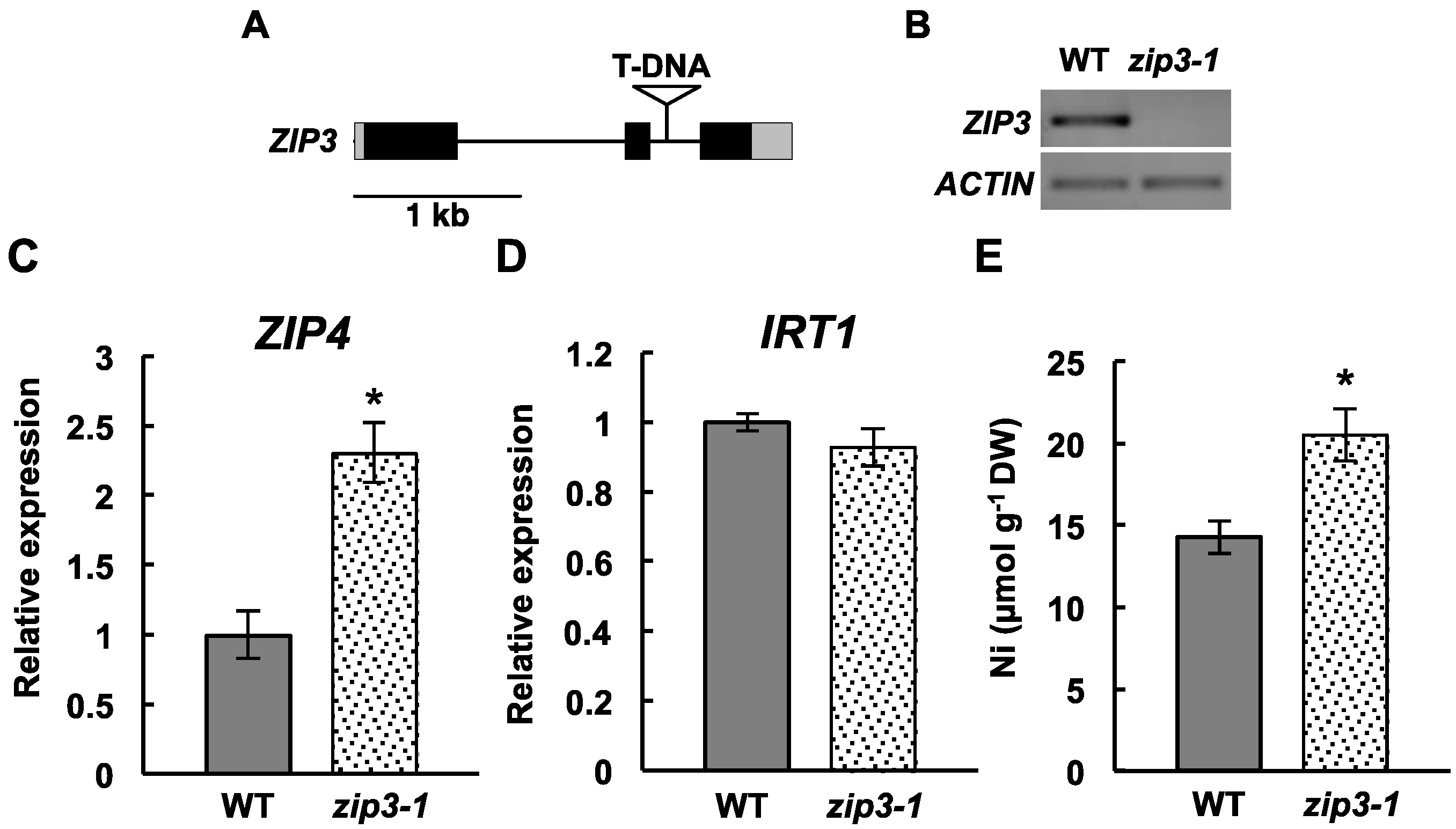
2.3. Ni Accumulation in a ZIP3 Mutant
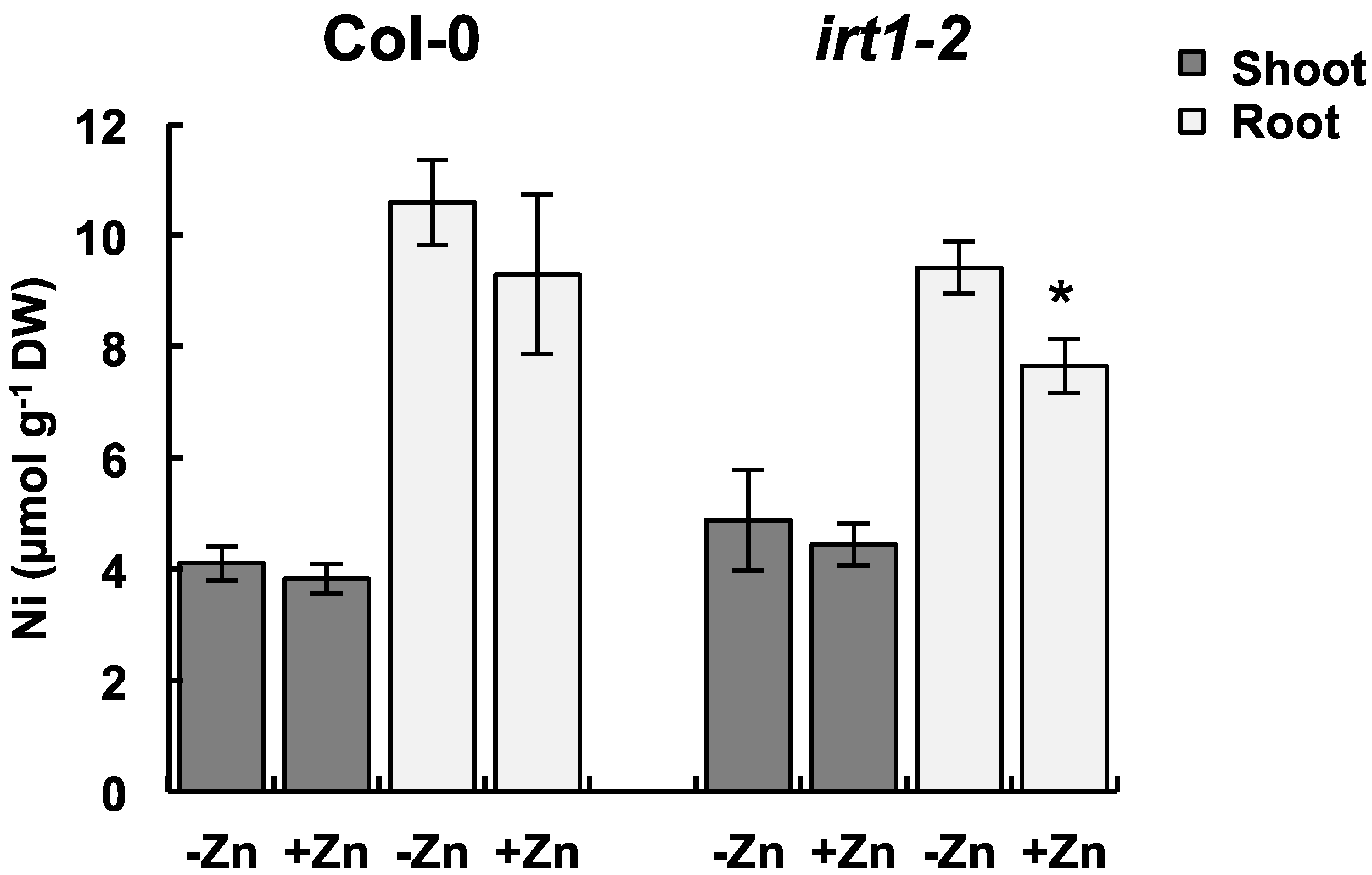
2.4. Ni Accumulation in the IRT1 Mutant
3. Discussion
4. Experimental Section
4.1. Analysis of Ni Accumulation in Plants Grown in Hydroponic Culture and Agar Plate Culture
4.2. Short-Term Ni2+ Uptake Assay Using 63Ni Isotope
4.3. Identification of the ZIP3-Defective Mutant and Gene Expression Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Brown, P.H.; Welch, R.M.; Cary, E.E. Nickel: A micronutrient essential for higher plants. Plant Physiol. 1987, 85, 801–803. [Google Scholar] [CrossRef] [PubMed]
- Brown, P.H.; Welch, R.M.; Madison, J.T. Effect of nickel deficiency on soluble amino acid, and nitrogen levels in barley. Plant Soil 1990, 125, 19–27. [Google Scholar] [CrossRef]
- Seregin, I.V.; Kozhevnikova, A.D. Physiological role of nickel and its toxic effects on higher plants. Russ. J. Plant Physiol. 2006, 53, 257–277. [Google Scholar] [CrossRef]
- Temple, P.J.; Bisessar, S. Uptake and toxicity of nickel and other metals in crops grown on soil contaminated by a nickel refinery. J. Plant Nutr. 1981, 3, 473–482. [Google Scholar] [CrossRef]
- Frank, R.; Stonefield, K.I.; Suda, P. Impact of nickel contamination on the production of vegetables on an organic soil, Ontario, Canada, 1980–1981. Sci. Total Environ. 1982, 26, 41–65. [Google Scholar] [CrossRef]
- Bisessar, S. Effects of lime on nickel uptake and toxicity in celery grown on muck soil contaminated by a nickel refinery. Sci. Total Environ. 1989, 84, 83–90. [Google Scholar] [CrossRef]
- Nishida, S.; Tsuzuki, C.; Kato, A.; Aisu, A.; Yoshida, J.; Mizuno, T. AtIRT1, the primary iron uptake transporter in the root, mediates excess nickel accumulation in Arabidopsis thaliana. Plant Cell Physiol. 2011, 52, 1433–1442. [Google Scholar] [CrossRef] [PubMed]
- Nishida, S.; Aisu, A.; Mizuno, T. Induction of IRT1 by the nickel-induced iron-deficient response in Arabidopsis. Plant Signal. Behav. 2012, 7, 329–331. [Google Scholar] [CrossRef] [PubMed]
- Worms, I.A.; Parthasarathy, N.; Wilkinson, K.J. Ni uptake by a green alga. 1. Validation of equilibrium models for complexation effects. Environ. Sci. Technol. 2007, 41, 4258–4263. [Google Scholar] [CrossRef] [PubMed]
- Lock, K.; van Eeckhout, H.; de Schamphelaere, K.A.; Criel, P.; Janssen, C.R. Development of a biotic ligand model (BLM) predicting nickel toxicity to barley (Hordeum vulgare). Chemosphere 2007, 66, 1346–1352. [Google Scholar] [CrossRef] [PubMed]
- Degryse, F.; Shahbazi, A.; Verheyen, L.; Smolders, E. Diffusion limitations in root uptake of cadmium and zinc, but not nickel, and resulting bias in the Michaelis constant. Plant Physiol. 2012, 160, 1097–1109. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Tutone, A.F.; Drummond, R.S.; Gardner, R.C.; Luan, S. A novel family of magnesium transport genes in Arabidopsis. Plant Cell 2001, 13, 2761–2775. [Google Scholar] [CrossRef] [PubMed]
- Li, L.G.; Sokolov, L.N.; Yang, Y.H.; Li, D.P.; Ting, J.; Pandy, G.K.; Luan, S. A mitochondrial magnesium transporter functions in Arabidopsis pollen development. Mol. Plant 2008, 1, 675–685. [Google Scholar] [CrossRef] [PubMed]
- Cataldo, D.A.; Garland, T.R.; Wildung, R.E. Nickel in plants: I. Uptake kinetics using intact soybean seedlings. Plant Physiol. 1978, 62, 563–565. [Google Scholar] [CrossRef] [PubMed]
- Grotz, N.; Fox, T.; Connolly, E.; Park, W.; Guerinot, M.L.; Eide, D. Identification of a family of Zinc transporter genes from Arabidopsis that respond to zinc deficiency. Proc. Natl. Acad. Sci. USA 1998, 95, 7220–7224. [Google Scholar] [CrossRef] [PubMed]
- Wintz, H.; Fox, T.; Wu, Y.Y.; Feng, V.; Chen, W.; Chang, H.S.; Zhu, T.; Vulpe, C. Expression profiles of Arabidopsis thaliana in mineral deficiencies reveal novel transporters involved in metal homeostasis. J. Biol. Chem. 2003, 278, 47644–47653. [Google Scholar] [CrossRef] [PubMed]
- Milner, M.J.; Seamon, J.; Craft, E.; Kochian, L.V. Transport properties of members of the ZIP family in plants and their role in Zn and Mn homeostasis. J. Exp. Bot. 2013, 64, 369–381. [Google Scholar] [CrossRef] [PubMed]
- Lasat, M.M.; Baker, A.J.; Kochian, L.V. Physiological characterization of root Zn2+ absorption and translocation to shoots in Zn hyperaccumulator and nonaccumulator species of Thlaspi. Plant Physiol. 1996, 112, 1715–1722. [Google Scholar] [PubMed]
- Krämer, U.; Smith, R.D.; Wenzel, W.W.; Raskin, I.; Salt, D.E. The role of metal transport and tolerance in nickel hyperaccumulation by Thlaspi goesingense Hálácsy. Plant Physiol. 1997, 115, 1641–1650. [Google Scholar] [PubMed]
- Van de Mortel, J.E.; Almar Villanueva, L.; Schat, H.; Kwekkeboom, J.; Coughlan, S.; Moerland, P.D.; Ver Loren van Themaat, E.; Koornneef, M.; Aarts, M.G. Large expression differences in genes for iron and zinc homeostasis, stress response, and lignin biosynthesis distinguish roots of Arabidopsis thaliana and the related metal hyperaccumulator Thlaspi caerulescens. Plant Physiol. 2006, 142, 1127–1147. [Google Scholar] [CrossRef] [PubMed]
- Assunção, A.G.; Herrero, E.; Lin, Y.F.; Huettel, B.; Talukdar, S.; Smaczniak, C.; Immink, R.G.; van Eldik, M.; Fiers, M.; Schat, H.; et al. Arabidopsis thaliana transcription factors bZIP19 and bZIP23 regulate the adaptation to zinc deficiency. Proc. Natl. Acad. Sci. USA 2010, 107, 10296–10301. [Google Scholar] [CrossRef] [PubMed]
- Korshunova, Y.O.; Eide, D.; Clark, W.G.; Guerinot, M.L.; Pakrasi, H.B. The IRT1 protein from Arabidopsis thaliana is a metal transporter with a broad substrate range. Plant Mol. Biol. 1999, 40, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Vert, G.; Grotz, N.; Dédaldéchamp, F.; Gaymard, F.; Guerinot, M.L.; Briat, J.F.; Curie, C. IRT1, an Arabidopsis transporter essential for iron uptake from the soil and for plant growth. Plant Cell 2002, 14, 1223–1233. [Google Scholar] [CrossRef] [PubMed]
- Shaff, J.E.; Schultz, B.A.; Craft, E.J.; Clark, R.T.; Kochian, L.V. GEOCHEM-EZ: A chemical speciation program with greater power and flexibility. Plant Soil 2010, 330, 207–214. [Google Scholar] [CrossRef]
- Suzuki, Y.; Kawazu, T.; Koyama, H. Arabidopsis RNA isolation. Biotechniques 2004, 37, 542–544. [Google Scholar] [PubMed]
- Takano, J.; Wada, M.; Ludewig, U.; Schaaf, G.; von Wirén, N.; Fujiwara, T. The Arabidopsis major intrinsic protein NIP5; 1 is essential for efficient boron uptake and plant development under boron limitation. Plant Cell 2006, 18, 1498–1509. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nishida, S.; Kato, A.; Tsuzuki, C.; Yoshida, J.; Mizuno, T. Induction of Nickel Accumulation in Response to Zinc Deficiency in Arabidopsis thaliana. Int. J. Mol. Sci. 2015, 16, 9420-9430. https://doi.org/10.3390/ijms16059420
Nishida S, Kato A, Tsuzuki C, Yoshida J, Mizuno T. Induction of Nickel Accumulation in Response to Zinc Deficiency in Arabidopsis thaliana. International Journal of Molecular Sciences. 2015; 16(5):9420-9430. https://doi.org/10.3390/ijms16059420
Chicago/Turabian StyleNishida, Sho, Aki Kato, Chisato Tsuzuki, Junko Yoshida, and Takafumi Mizuno. 2015. "Induction of Nickel Accumulation in Response to Zinc Deficiency in Arabidopsis thaliana" International Journal of Molecular Sciences 16, no. 5: 9420-9430. https://doi.org/10.3390/ijms16059420




