Abortive Infection of Snakehead Fish Vesiculovirus in ZF4 Cells Was Associated with the RLRs Pathway Activation by Viral Replicative Intermediates
Abstract
:1. Introduction
2. Results
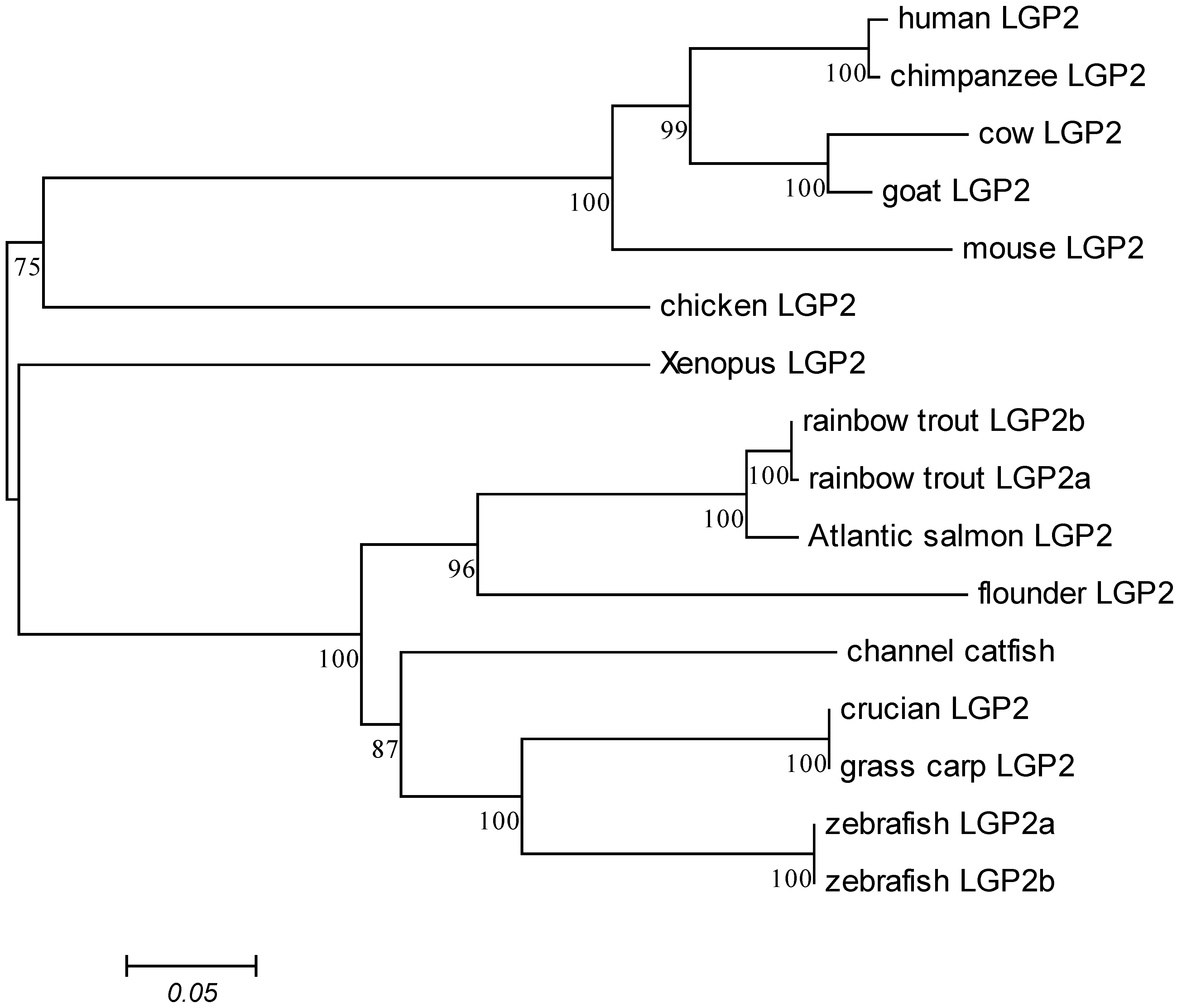
2.1. Characterization of DrLGP2a and DrLGP2b cDNAs
2.2. The mRNA and cRNA Expressions of the Viral N and G Genes in ZF4 Cells Infected with SHVV
2.3. Temporal Expression of IFN and Mx in ZF4 after Infection with SHVV


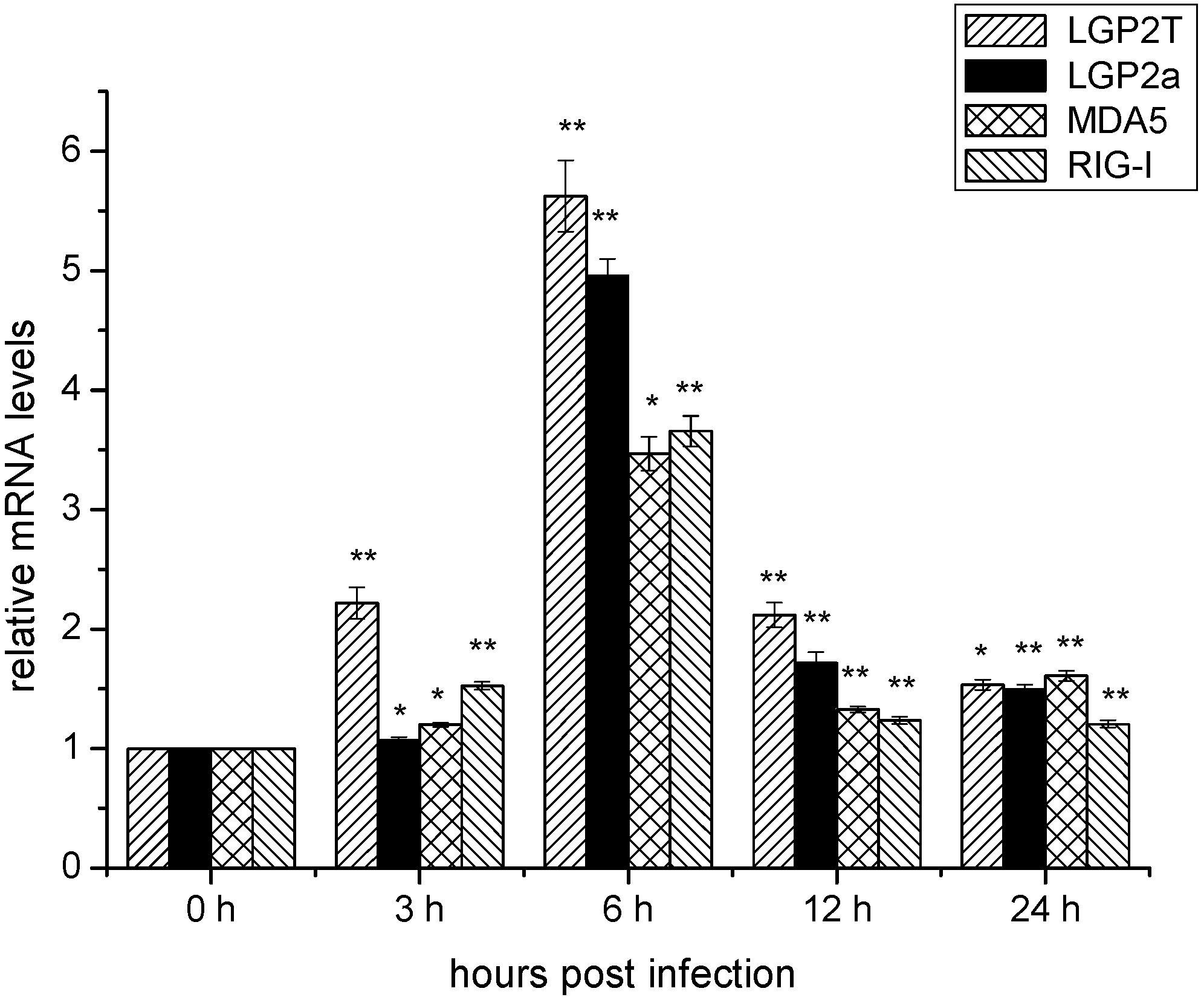
2.4. Temporal mRNA Expressions of RIG-I, MDA5, DrLGP2T and DrLGP2a in ZF4 Cells after Infection with SHVV
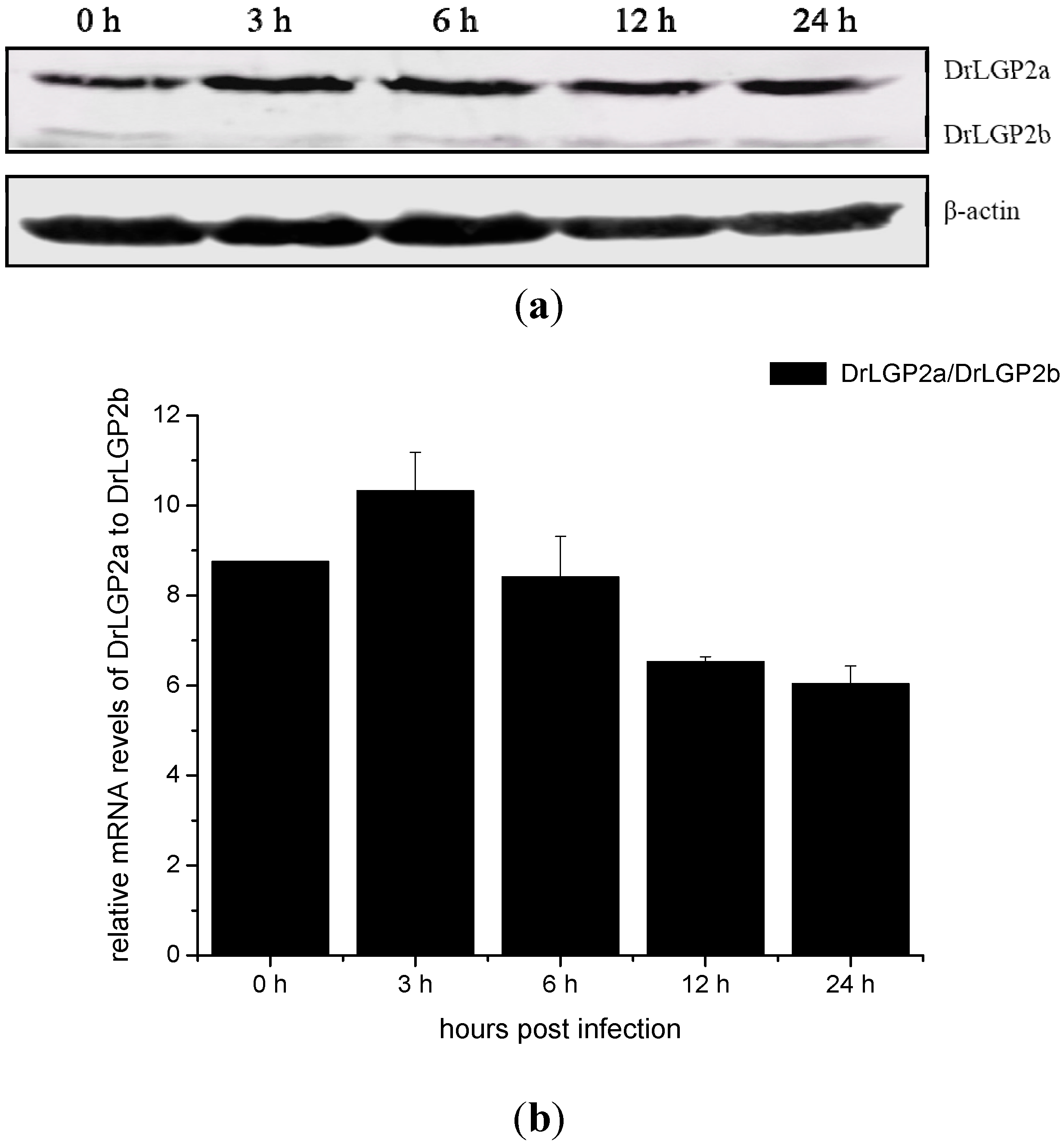
2.5. Temporal Expressions of DrLGP2a and DrLGP2b Proteins in ZF4 after the Infection with SHVV
| Names | Sequence (5'→3') | Application |
|---|---|---|
| LGP2-FW | CATTAATGAGAAGGGCATCGTT | DrLGP2 core fragment |
| LGP2-BW | GGCTCAGATTCTGGAAAACGC | DrLGP2 core fragment |
| 5' RACE Outer Primer | CATGGCTACATGCTGACAGCCTA | DrLGP2 5' RACE |
| 5' RACE Inner Primer | CGCGGATCCACAGCCTACTGATGATCAGTCGATG | DrLGP2 5' RACE |
| 3' RACE Outer Primer | TACCGTCGTTCCACTAGTGATTT | DrLGP2 3' RACE |
| 3' RACE Inner Primer | CGCGGATCCTCCACTAGTGATTTCACTATAGG | DrLGP2 3' RACE |
| LGP2-DEXDc-FW | ATCGAATTCATGGAGATCACTCTTAGATCAT | DrLGP2 antibody |
| LGP2-DEXDc-BW | ATCGCGGCCGCGTGCTGGTCTACCAGGT | DrLGP2 antibody |
| LGP2T-FW | ATGTGGCGTTACGTGGAGAA | DrLGP2T qRT-PCR |
| LGP2T-BW | TCTGCAGCATCGGTGTGTAA | DrLGP2T qRT-PCR |
| LGP2a-FW | GGCTGAACGGAAGAGGGAT | DrLGP2a qRT-PCR |
| LGP2a-BW | TTCCTGATGTCTCCTCCGC | DrLGP2a qRT-PCR |
| RIG-I-FW | TAAGGTGCTGAAGATGGCTC | RIG-I qRT-PCR |
| RIG-I-BW | TGATGGTGTTCTGTCCGTTG | RIG-I qRT-PCR |
| MDA5-FW | AGCCAGTTATCTGATCGGCG | MDA5 qRT-PCR |
| MDA5-BW | TCAGCGATGTCCAAACCCTC | MDA5 qRT-PCR |
| IFN-FW | TGCGTCTACTTGCGAATG | IFN qRT-PCR |
| IFN-BW | GGCTTGGAAATGGTGTCT | IFN qRT-PCR |
| Mx-FW | CTTGCGTGAGATGAGGTTGC | Mx qRT-PCR |
| Mx-BW | TGTCTGGCGGCTCAGTAAGT | Mx qRT-PCR |
| SHVV-N-FW | CCGCATCGGAAATCAAGCAG | N gene mRNA qRT-PCR |
| SHVV-N-BW | GTTGACCGCTTGCCCAATTT | N gene mRNA qRT-PCR |
| SHVV-G-FW | ACACCATACATGCCAGAGGC | G gene mRNA qRT-PCR |
| SHVV-G-BW | GCCTCGCTGGGTATCCAAAT | G gene mRNA qRT-PCR |
| SHVV-N-FW | CCGCATCGGAAATCAAGCAG | N gene cRNA qRT-PCR |
| SHVV-N-BW | GTTGACCGCTTGCCCAATTT | N gene cRNA qRT-PCR |
| SHVV-G-FW | ACACCATACATGCCAGAGGC | G gene cRNA qRT-PCR |
| SHVV-G-BW | GCCTCGCTGGGTATCCAAAT | G gene cRNA qRT-PCR |
| β-actin-FW | CACTGTGCCCATCTACGAG | β-actin qRT-PCR |
| β-actin-BW | CCATCTCCTGCTCGAAGTC | β-actin qRT-PCR |

2.6. The Activation of the IFN-α mRNA Expression Was Associated with the Viral Replicative Intermediates

3. Discussion
4. Experimental Section
4.1. Cell, Virus and Infection
4.2. RNA Isolation and cDNA Synthesis
4.3. Amplification of Full-Length cDNAs of DrLGP2a and LGP2b by RACE
4.4. Sequence Analysis
4.5. Generation of Polyclonal DrLGP2-DEXDc Specific Antibody
4.6. Quantitative Real Time PCR (qRT-PCR) Assay for mRNA Expressions of Target Genes
4.7. Detection Protein Level of DrLGP2 by Western Blot
4.8. IFN-α Expression in ZF4 Cells Transfected with Total Cellular RNAs
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yoneyama, M.; Kikuchi, M.; Matsumoto, K.; Imaizumi, T.; Miyagishi, M.; Taira, K.; Foy, E.; Loo, Y.M.; Gale, M., Jr.; Akira, S.; et al. Shared and unique functions of the DEXD/H-box helicases RIG-I, MDA5, and LGP2 in antiviral innate immunity. J. Immunol. 2005, 175, 2851–2858. [Google Scholar] [CrossRef] [PubMed]
- Seth, R.; Sun, L.; Ea, C.; Chen, Z. Identification and characterization of MAVS, a mitochondrial antiviral signaling protein that activates NF-κB and IRF 3. Cell 2005, 122, 669–682. [Google Scholar] [CrossRef] [PubMed]
- Kato, H.; Takeuchi, O.; Sato, S.; Yoneyama, M.; Yamamoto, M.; Matsui, K.; Uematsu, S.; Jung, A.; Kawai, T.; Ishii, K.J.; et al. Differential roles of MDA5 and RIG-I helicases in the recognition of RNA viruses. Nature 2006, 441, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Takahashi, K.; Sato, S.; Coban, C.; Kumar, H.; Kato, H.; Ishii, K.J.; Takeuchi, O.; Akira, S. IPS-1, an adaptor triggering RIG-I- and MDA5-mediated type I interferon induction. Nat. Immunol. 2005, 6, 981–988. [Google Scholar] [CrossRef] [PubMed]
- Kumar, H.; Kawai, T.; Kato, H.; Sato, S.; Takahashi, K.; Coban, C.; Yamamoto, M.; Uematsu, S.; Ishii, K.J.; Takeuchi, O.; et al. Essential role of IPS-1 in innate immune responses against RNA viruses. J. Exp. Med. 2006, 203, 1795–1803. [Google Scholar] [CrossRef] [PubMed]
- Myong, S.; Cui, S.; Cornish, P.; Kirchhofer, A.; Gack, M.; Jung, J.; Hopfner, K.; Ha, T. Cytosolic viral sensor RIG-I is a 5'-triphosphate-dependent translocase on double-stranded RNA. Science 2009, 323, 1070–1074. [Google Scholar] [CrossRef] [PubMed]
- Meylan, E.; Curran, J.; Hofmann, K.; Moradpour, D.; Binder, M.; Bartenschlager, R. Cardif is an adaptor protein in the RIG-I antiviral pathway and is targeted by hepatitis C virus. Nature 2005, 437, 1167–1172. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Hu, Y.; Zou, P.; Ren, S.; Nie, P.; Chang, M. MAVS splicing variants contribute to the induction of interferon and interferon-stimulated genes mediated by RIG-I-like receptors. Dev. Comp. Immunol. 2015, 49, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Xiong, R.; Nie, L.; Xiang, L.; Shao, J. Characterization of a PIAS4 homologue from zebrafish: Insights into its conserved negative regulatory mechanism in the TRIF, MAVS, and IFN signaling pathways during vertebrate evolution. J. Immunol. 2012, 188, 2653–2668. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Chang, M.; Nie, P.; Chris, J. Origin and evolution of the RIG-I like RNA helicase gene family. BMC Evol. Biol. 2009, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zou, P.; Chang, M.; Li, Y.; Huan Zhang, S.; Fu, J.; Chen, S.; Nie, P. Higher antiviral response of RIG-I through enhancing RIG-I/MAVS-mediated signaling by its long insertion variant in zebrafish. Fish Shellfish Immunol. 2014, 43, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Zou, P.; Chang, M.; Xue, N.; Liu, X.; Li, J.; Fu, J.; Chen, S.N.; Nie, P. Melanoma differentiation-associated gene 5 in zebrafish provoking higher interferon promoter activity through signaling enhancing of its shorter splicing variant. Immunology 2014, 141, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.; Collet, B.; Nie, P.; Lester, K.; Campbell, S.; Secombes, C.J.; Zou, J. Expression and functional characterization of the RIG-I-like receptors MDA5 and LGP2 in Rainbow trout (Oncorhynchus mykiss). J. Virol. 2011, 85, 8403–8412. [Google Scholar] [CrossRef] [PubMed]
- Lauksund, S.; Svingerud, T.; Bergan, V.; Robertsen, B. Atlantic salmon IPS-1 mediates induction of IFNα1 and activation of NF-κB and localizes to mitochondria. Dev. Comp. Immunol. 2009, 33, 1196–1204. [Google Scholar] [CrossRef] [PubMed]
- Su, J.; Huang, T.; Yang, C.; Zhang, R. Molecular cloning, characterization and expression analysis of interferon-β promoter stimulator 1 (IPS-1) gene from grass carp Ctenopharyngodon idella. Fish Shellfish Immunol. 2011, 30, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Wan, Q.; Su, J.; Chen, X.; Yang, C. Gene-based polymorphisms, genomic organization of interferon-β promoter stimulator 1 (IPS-1) gene and association study with the natural resistance to grass carp reovirus in grass carp Ctenopharyngodon idella. Dev. Comp. Immunol. 2013, 41, 756–765. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Su, J.; Heng, J.; Dong, J.; Zhang, R.; Zhu, H. Identification and expression profiling analysis of grass carp Ctenopharyngodon idella LGP2 cDNA. Fish Shellfish Immunol. 2010, 29, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Simora, R.; Ohtani, M.; Hikima, J.; Kondo, H.; Hirono, I.; Jung, T.; Aoki, T. Molecular cloning and antiviral activity of IFN-β promoter stimulator-1 (IPS-1) gene in Japanese flounder, Paralichthys olivaceus. Fish Shellfish Immunol. 2010, 29, 979–986. [Google Scholar] [CrossRef] [PubMed]
- Zeng, W.W.; Wang, Q.; Wang, Y.Y.; Liu, C.; Liang, H.R.; Fang, X.; Wu, S.Q. Genomic characterization and taxonomic position of a rhabdovirus from a hybrid snakehead. Arch. Virol. 2014, 159, 2469–2473. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wen, Y.; Hu, X.; Wang, W.; Liang, X.; Li, J.; Vakharia, V.; Lin, L. Breaking the host range: Mandarin fish was susceptible to a vesiculovirus derived from snakehead fish. J. Gen. Virol. 2015, 96, 775–781. [Google Scholar] [CrossRef]
- Driever, W.; Rangini, Z. Characterization of a cell-line derived from zebrafish (Brachydanio rerio) embryros. In Vitro Cell Dev. Biol. Anim. 1993, 29, 749–754. [Google Scholar] [CrossRef]
- Rothenfusser, S.; Goutagny, N.; di Perna, G.; Gong, M.; Monks, B.; Schoenemeyer, A.; Yamamoto, M.; Akira, S.; Fitzgerald, K. The RNA helicase LGP2 inhibits TLR-independent sensing of viral replication by retinoic acid-inducible gene-I. J. Immunol. 2005, 175, 5260–5268. [Google Scholar] [CrossRef] [PubMed]
- Satoh, T.; Kato, H.; Kumagai, Y.; Yoneyama, M.; Sato, S.; Matsushita, K.; Tsujimura, T.; Fujita, T.; Akira, S.; Takeuchi, O. LGP2 is a positive regulator of RIG-I- and MDA5-mediated antiviral responses. Proc. Natl. Acad. Sci. USA 2010, 107, 1512–1517. [Google Scholar] [CrossRef] [PubMed]
- Schlee, M.; Roth, A.; Hornung, V.; Hagmann, C.; Wimmenauer, V.; Barchet, W.; Coch, C.; Janke, M.; Mihailovic, A.; Wardle, G.; et al. Recognition of 5'-triphosphate by RIG-I helicase requires short blunt double-stranded RNA as contained in panhandle of negative-strand virus. Immunity 2009, 31, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.; Schwerd, T.; Hamm, W.; Hellmuth, J.; Cui, S.; Wenzel, M.; Hoffmann, F.; Michallet, M.; Besch, R.; Hopfner, K.; Endres, S.; Rothenfusser, S. 5'-Triphosphate RNA requires base-paired structures to activate antiviral signaling via RIG-I. Proc. Natl. Acad. Sci. USA. 2009, 106, 12067–12072. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Liu, Q.; Berube, N.; Zhou, Y. 5'-Triphosphate-siRNA: Potent inhibition of influenza A virus infection by gene silencing and RIG-I activation. J. Virol. 2012, 86, 10359–10369. [Google Scholar] [CrossRef] [PubMed]
- Lyles, S.D.; Kuzmin, I.; Rupprecht, E.C. Rhabdoviridae. In Field’s Virology, 6th ed.; Knipe, M.D., Howley, M.P., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013; pp. 885–922. [Google Scholar]
- Pham, P.H.; Lumsden, J.S.; Tafalla, C.; Dixon, B.; Bols, N.C. Differential effects of viral hemorrhagic septicemia virus (VHSV) genotypes IVa and IVb on gill epithelial and spleen macrophage cell lines from rainbow trout (Onchorhynchus mykiss). Fish Shellfish Immunol. 2013, 34, 632–640. [Google Scholar] [CrossRef] [PubMed]
- Larkin, M.A.; Blackshields, G.; Brown, N.P.; Chenna, R.; McGettigan, P.A.; McWilliam, H.; Valentin, F.; Wallace, I.M.; Wilm, A.; Lopez, R.; et al. Clustal W and Clustal X version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar] [CrossRef] [PubMed]
- Hall, B.G. Building phylogenetic trees from molecular data with MEGA. Mol. Biol. Evol. 2013, 30, 1229–1235. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, W.; Asim, M.; Yi, L.; Hegazy, A.M.; Hu, X.; Zhou, Y.; Ai, T.; Lin, L. Abortive Infection of Snakehead Fish Vesiculovirus in ZF4 Cells Was Associated with the RLRs Pathway Activation by Viral Replicative Intermediates. Int. J. Mol. Sci. 2015, 16, 6235-6250. https://doi.org/10.3390/ijms16036235
Wang W, Asim M, Yi L, Hegazy AM, Hu X, Zhou Y, Ai T, Lin L. Abortive Infection of Snakehead Fish Vesiculovirus in ZF4 Cells Was Associated with the RLRs Pathway Activation by Viral Replicative Intermediates. International Journal of Molecular Sciences. 2015; 16(3):6235-6250. https://doi.org/10.3390/ijms16036235
Chicago/Turabian StyleWang, Wenwen, Muhammad Asim, Lizhu Yi, Abeer M. Hegazy, Xianqin Hu, Yang Zhou, Taoshan Ai, and Li Lin. 2015. "Abortive Infection of Snakehead Fish Vesiculovirus in ZF4 Cells Was Associated with the RLRs Pathway Activation by Viral Replicative Intermediates" International Journal of Molecular Sciences 16, no. 3: 6235-6250. https://doi.org/10.3390/ijms16036235






