Knockdown of UbcH10 Enhances the Chemosensitivity of Dual Drug Resistant Breast Cancer Cells to Epirubicin and Docetaxel
Abstract
:1. Introduction
2. Results and Discussion
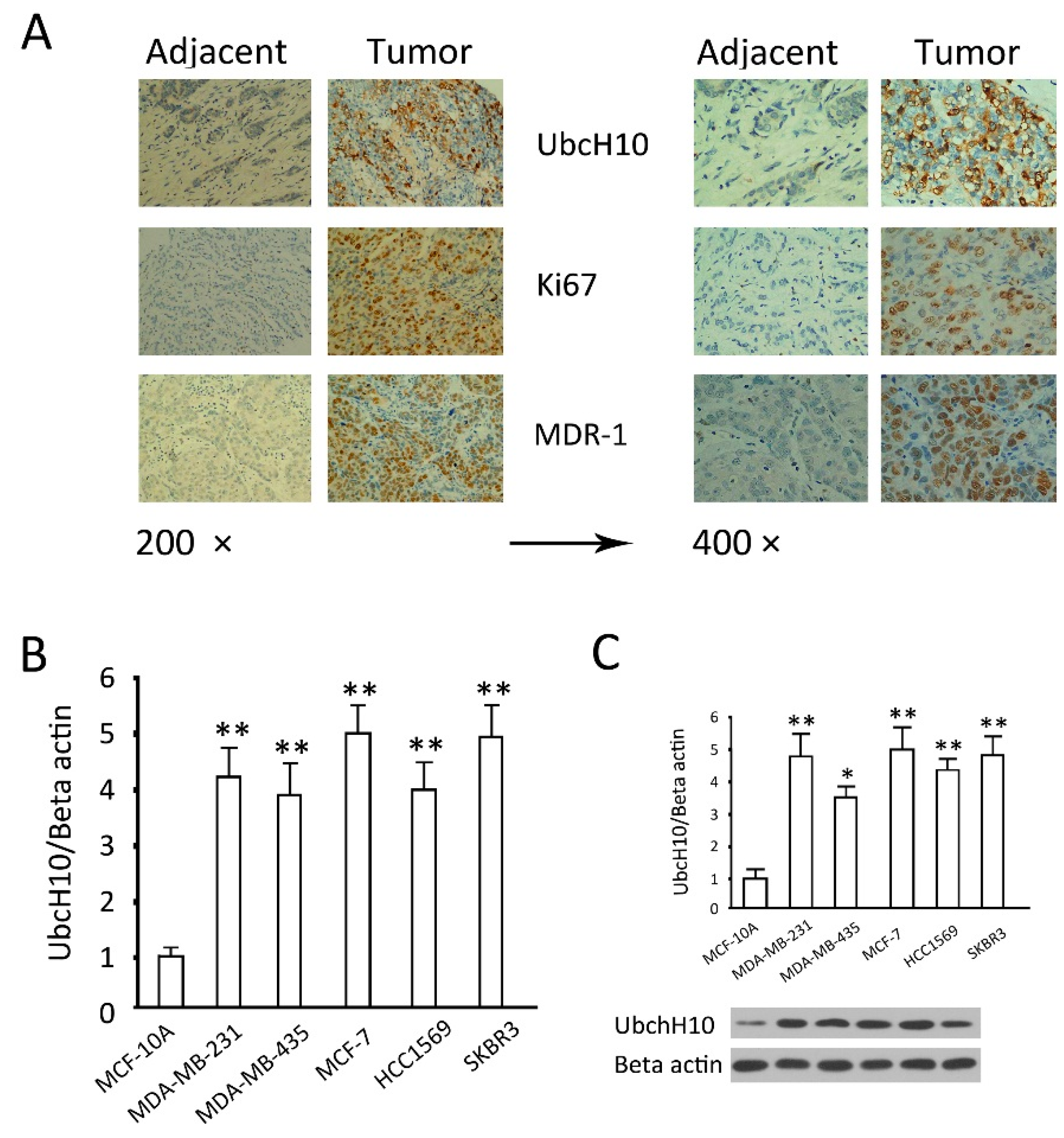
2.1. Analysis of Differential Expression of Ubiquitin-Conjugating Enzyme H10 (UbcH10) in Breast Cancer Tissues and Cell Lines
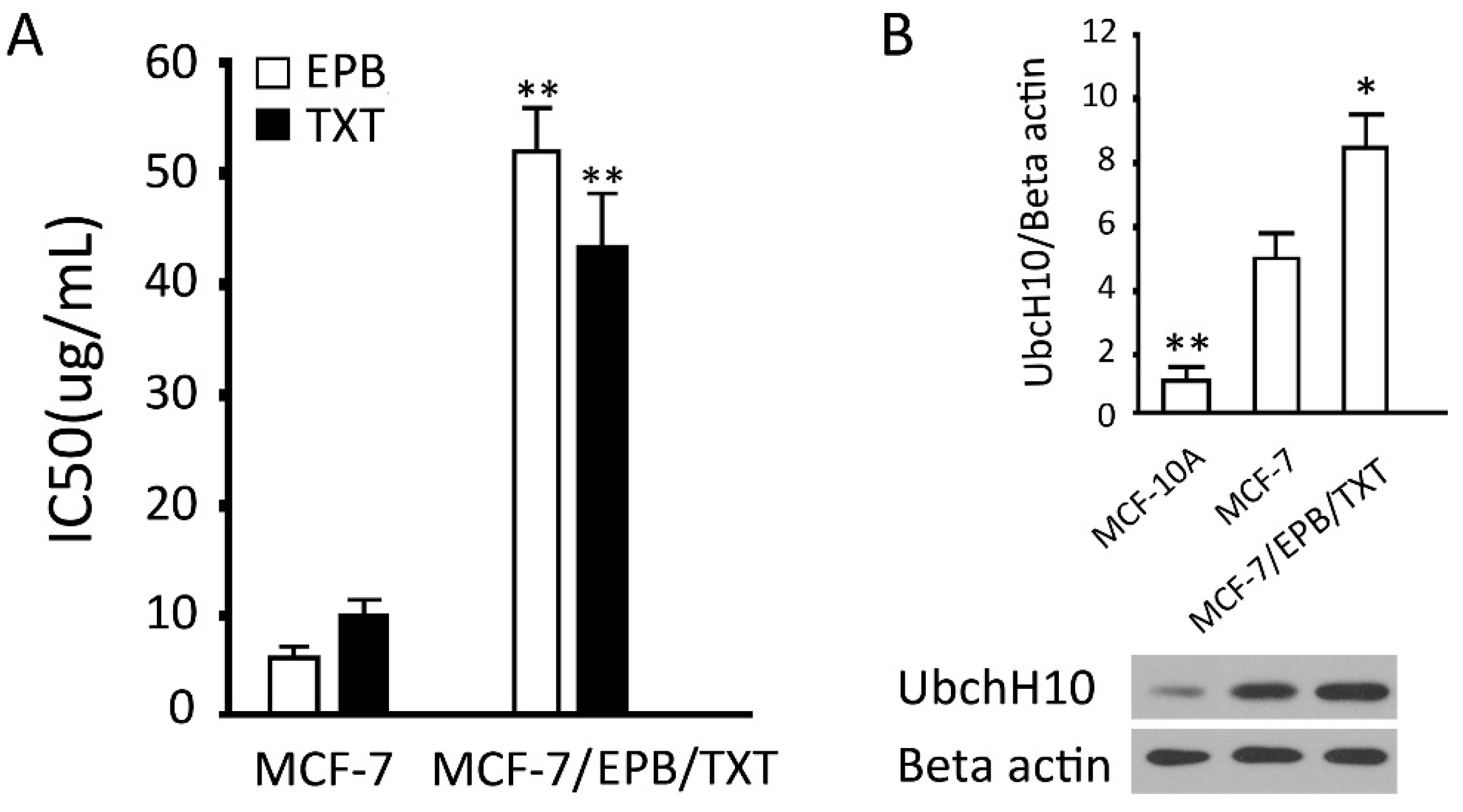
2.2. Establishment and Verification of the Dual Drug Resistant Cell Line (MCF-7/EPB/TXT)
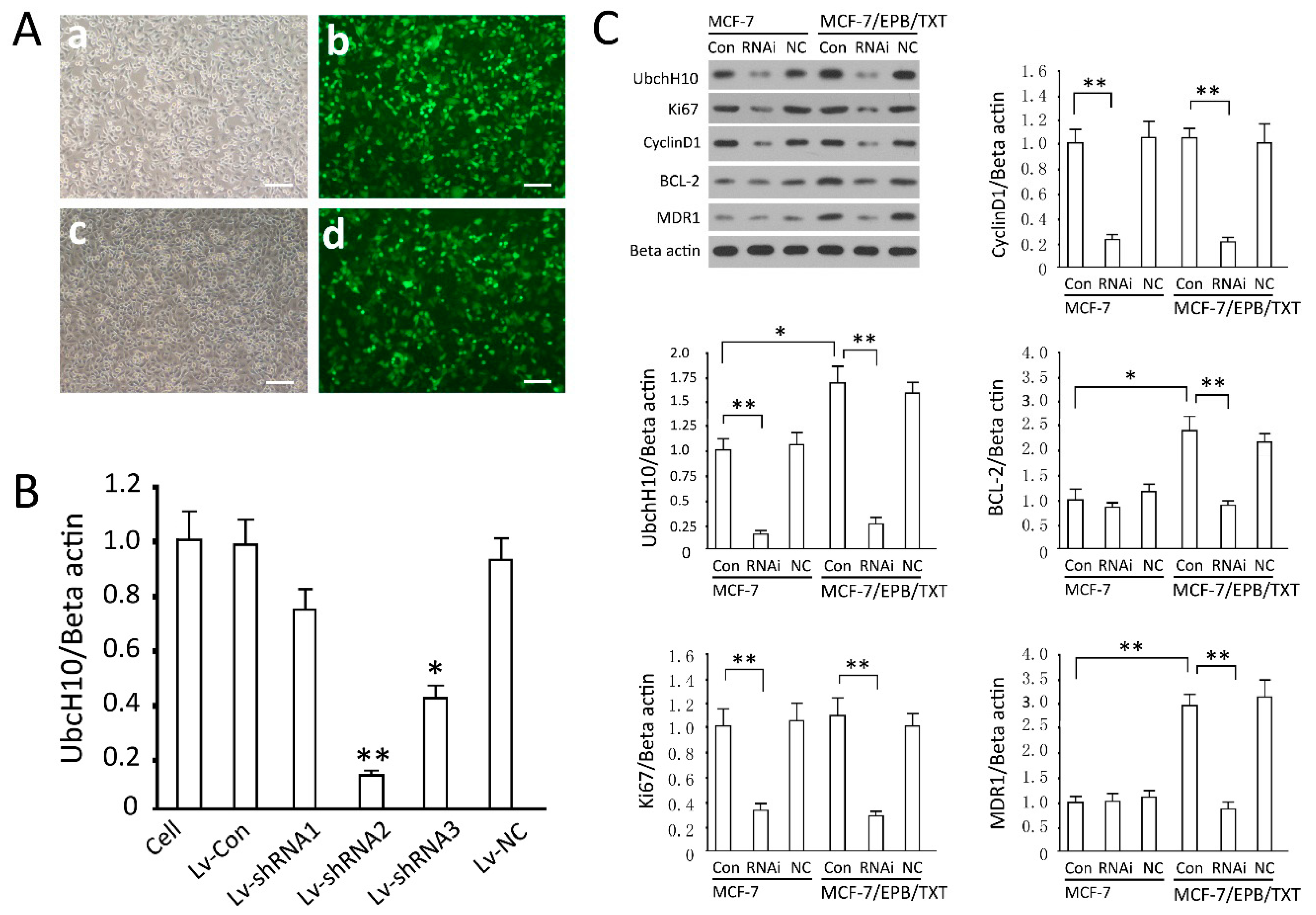
2.3. Detection of Gene Transfection Efficiency and Screening of Effective siRNA Sequence
2.4. Effects of UbcH10 Knockdown on Relevant Protein Contents
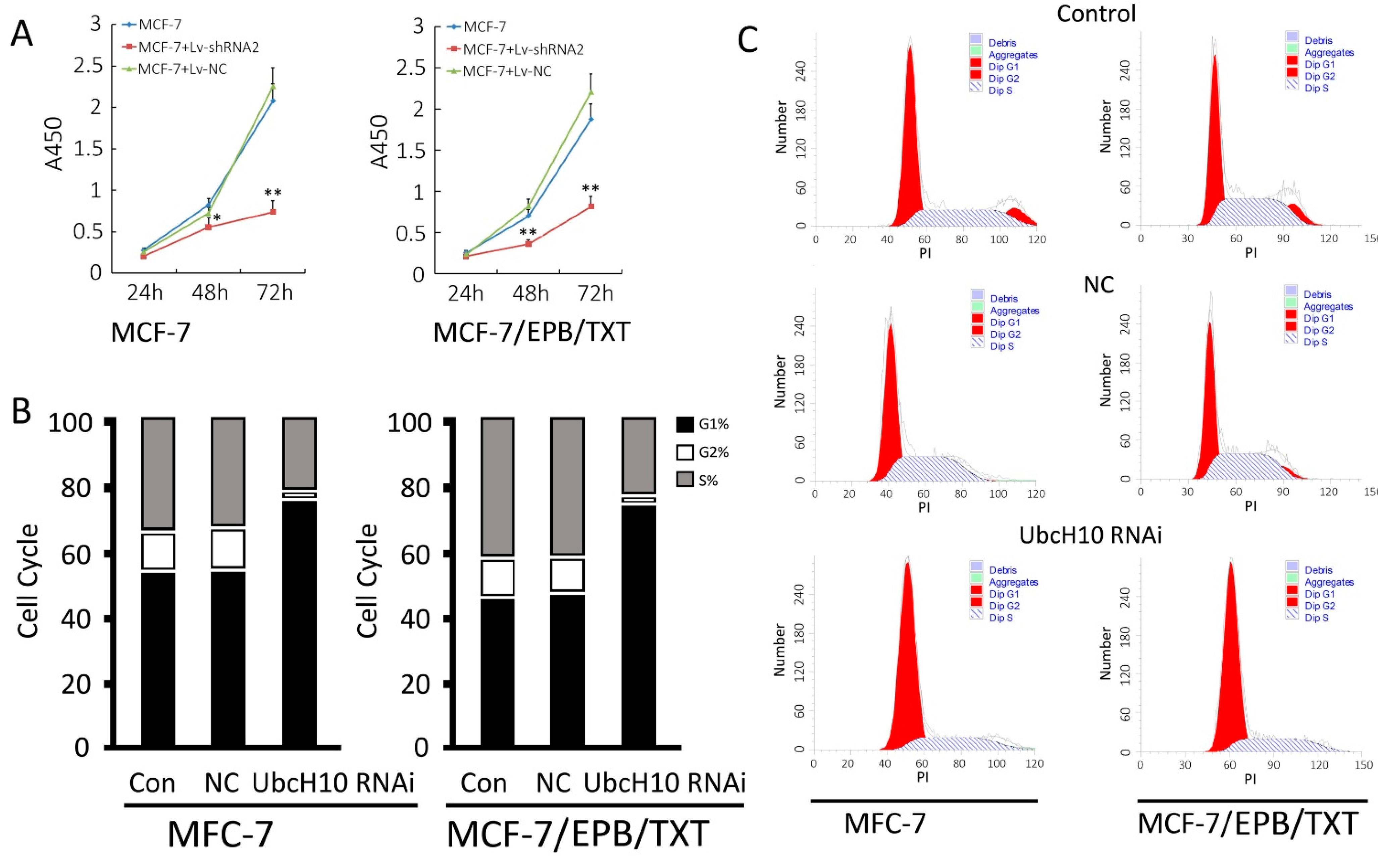
2.5. Effects of UbcH10 Knockdown on Proliferation and Cell Cycle in MCF-7 and MCF-7/EPB/TXT Cells
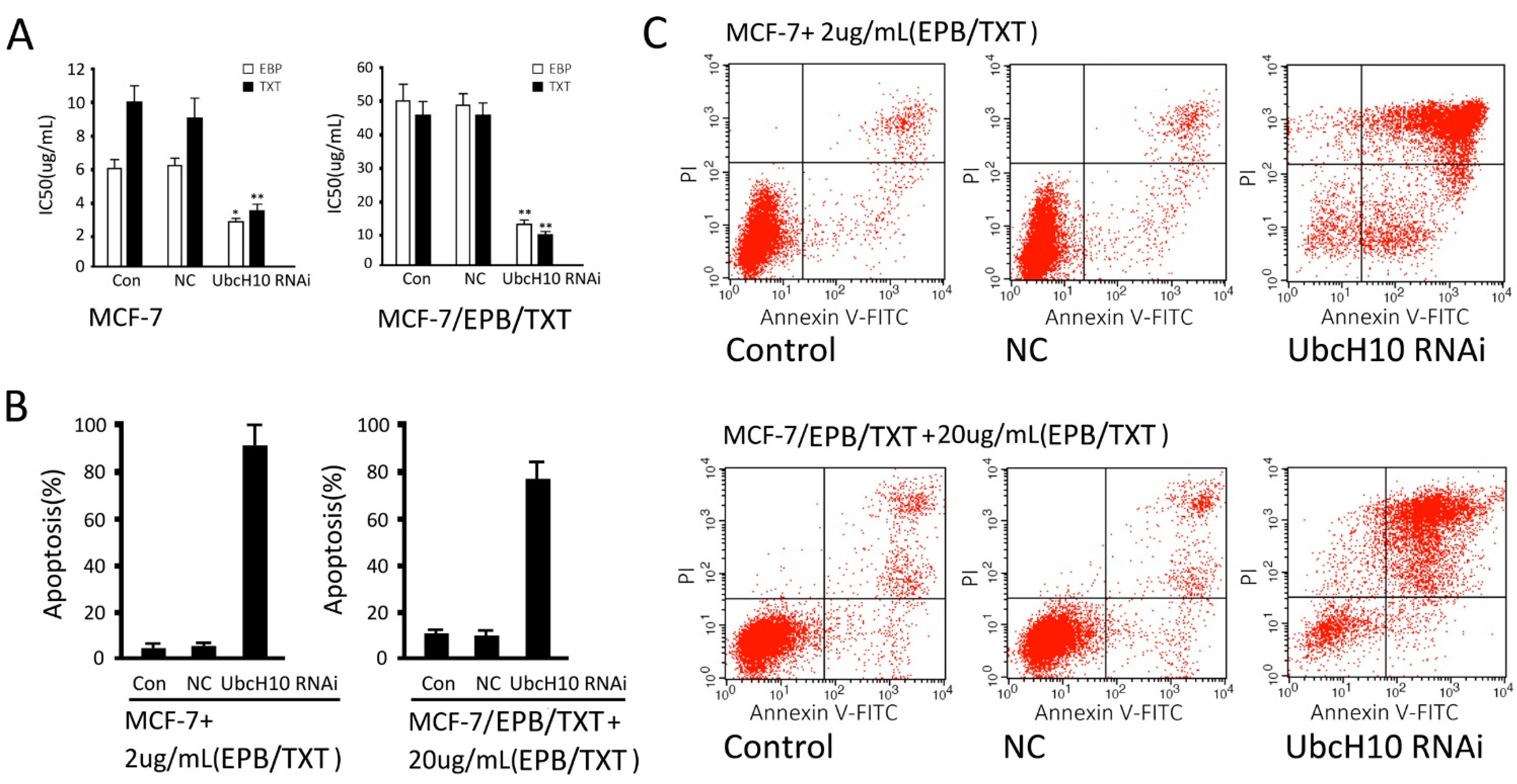
2.6. Effects of UbcH10 Knockdown on Sensitivity of Resistant Cells to Chemotherapeutics
3. Experimental Section
3.1. Analysis on Differential UbcH10 Expression in Tumor and Adjacent Tissues from Breast Cancer Patients
3.2. Examination of UbcH10 Expression in Breast Cancer Cell Lines
3.3. Establishment of EPB and TXT Resistant Cell Line
3.4. Screening of Effective siRNA Sequence and Intervention of UbcH10
3.5. Effects of UbcH10 Gene Silencing on Proliferation and Cell Cycle Distribution of MCF-7 and MCF-7/EPB/TXT Cells
3.6. Detection of Effects of UbcH10 Gene Interference on Sensitivity of Resistant Cells to Chemotherapeutics
3.7. Effects of UbcH10 Gene Silencing on Relevant Protein Contents
3.8. Detection of UbcH10 mRNA by Real-Time PCR
3.9. Detection of Relative Protein Content by Western Blotting
3.10. Detection of Cell Proliferation Using CCK-8
3.11. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Li, W.; Zang, W.; Liu, P.; Wang, Y.; Du, Y.; Chen, X.; Deng, M.; Sun, W.; Wang, L.; Zhao, G.; et al. MicroRNA-124 inhibits cellular proliferation and invasion by targeting ETS-1 in breast cancer. Tumour Biol. 2014, 35, 10897–10904. [Google Scholar]
- Zhao, X.; Rotenberg, S.A. Phosphorylation of Cdc42 effector protein-4 (CEP4) by protein kinase C promotes motility of human breast cells. J. Biol. Chem. 2014, 289, 25844–25854. [Google Scholar] [CrossRef] [PubMed]
- Harvey, S.L.; Khasraw, M. Alternative targeted therapy for early Her2 positive breast cancer. Gland Surg. 2013, 2, 42–45. [Google Scholar] [PubMed]
- Bi, X.; Wu, C.; Han, M.; Cai, J. Correlations of ALDH1 expression with molecular subtypes and ABCG2 in breast cancer. Gland Surg. 2012, 1, 12–19. [Google Scholar] [PubMed]
- Pratt, M.A. Paradoxical roles for FOXA1 in anti-estrogen resistance and as a luminal differentiation factor in breast cancer. Gland Surg. 2012, 1, 9–11. [Google Scholar] [PubMed]
- Vardhini, N.V.; Rao, P.J.; Murthy, P.B.; Sudhakar, G. GHOXD10 expression in human breast cancer. Tumour Biol. 2014, 35, 10855–10860. [Google Scholar] [CrossRef] [PubMed]
- Yi, W.; Peng, J.; Zhang, Y.; Fu, F.; Zou, Q.; Tang, Y. Differential protein expressions in breast cancer between drug sensitive tissues and drug resistant tissues. Gland Surg. 2013, 2, 62–68. [Google Scholar] [PubMed]
- Gaule, P.B.; Crown, J.; O’Donovan, N.; Duffy, M.J. cMET in triple-negative breast cancer: Is it a therapeutic target for this subset of breast cancer patients? Expert Opin. Ther. Targets 2014, 18, 999–1009. [Google Scholar] [CrossRef] [PubMed]
- Jerjees, D.A.; Alabdullah, M.; Alkaabi, M.; Abduljabbar, R.; Muftah, A.; Nolan, C.; Green, A.R.; Ellis, I.O.; Rakha, E.A. ERK1/2 is related to oestrogen receptor and predicts outcome in hormone-treated breast cancer. Breast Cancer Res. Treat. 2014, 147, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wang, N.; Li, W.; Liu, P.; Chen, Q.; Situ, H.; Zhong, S.; Guo, L.; Lin, Y.; Shen, J.; et al. MicroRNA-124 inhibits cellular proliferation and invasion by targeting ETS-1 in breast cancer. Carcinogenesis 2014, 35, 2346–2356. [Google Scholar]
- Xie, C.; Powell, C.; Yao, M.; Wu, J.; Dong, Q. Ubiquitin-conjugating enzyme E2C: A potential cancer biomarker. Int. J. Biochem. Cell Biol. 2014, 47, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Van Ree, J.H.; Jeganathan, K.B.; Malureanu, L.; van Deursen, J.M. Overexpression of the E2 ubiquitin-conjugating enzyme UbcH10 causes chromosome missegregation and tumor formation. J. Cell Biol. 2010, 188, 83–100. [Google Scholar] [CrossRef] [PubMed]
- Perrotta, I.; Bruno, L.; Maltese, L.; Russo, E.; Donato, A.; Donato, G. Immunohistochemical analysis of the ubiquitin-conjugating enzyme UbcH10 in lung cancer: A useful tool for diagnosis and therapy. J. Histochem. Cytochem. 2012, 60, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Morikawa, T.; Kawai, T.; Abe, H.; Kume, H.; Homma, Y.; Fukayama, M. UBE2C is a marker of unfavorable prognosis in bladder cancer after radical cystectomy. Int. J. Clin. Exp. Pathol. 2013, 6, 1367–1374. [Google Scholar] [PubMed]
- Chen, S.M.; Jiang, C.Y.; Wu, J.Y.; Liu, B.; Chen, Y.J.; Hu, C.J.; Liu, X.X. RNA interference-mediated silencing of UbcH10 gene inhibits colorectal cancer cell growth in vitro and in vivo. Clin. Exp. Pharmacol. Physiol. 2010, 37, 525–529. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Jiang, L.; Wang, L.; He, J.; Yu, H.; Sun, G.; Chen, J.; Xiu, Q.; Li, B. UbcH10 expression provides a useful tool for the prognosis and treatment of non-small cell lung cancer. J. Cancer Res. Clin. Oncol. 2012, 138, 1951–1961. [Google Scholar] [CrossRef] [PubMed]
- Berlingieri, MT.; Pallante, P.; Sboner, A.; Barbareschi, M.; Bianco, M.; Ferraro, A.; Mansueto, G.; Borbone, E.; Guerriero, E.; Troncone, G.; et al. UbcH10 is overexpressed in malignant breast carcinomas. Eur. J. Cancer 2007, 43, 2729–2735. [Google Scholar]
- Fujita, T.; Ikeda, H.; Kawasaki, K.; Taira, N.; Ogasawara, Y.; Nakagawara, A.; Doihara, H. Clinicopathological relevance of UbcH10 in breast cancer. Cancer Sci. 2009, 100, 238–248. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.H.; Guo, S.Y.; Wang, C.; Gu, Y.; Bao, J.L. Expression of UbcH10 correlates with prognosis of infiltrating ductal carcinoma of breast. J. Surg. Concept. Pract. 2014, 19, 47–52. [Google Scholar]
- Townsley, F.M.; Aristarkhov, A.; Beck, S.; Hershko, A.; Ruderman, J.V. Dominant-negative cyclin-selective ubiquitin carrier protein E2-C/UbcH10 blocks cells in metaphase. Proc. Natl. Acad. Sci. USA 1997, 94, 2362–2367. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, Y.; Ozaki, T.; Miyazaki, K.; Aoyama, M.; Miyazaki, M.; Nakagawara, A. UbcH10 is the cancer-related E2 ubiqutin-conjugating enzyme. Cancer Res. 2003, 63, 4167–4173. [Google Scholar] [PubMed]
- Steger, G.G.; Greil, R.; Lang, A.; Rudas, M.; Fitzal, F.; Mlineritsch, B.; Hartmann, B.L.; Bartsch, R.; Melbinger, E.; Hubalek, M.; et al. Epirubicin and docetaxel with or without capecitabine as neoadjuvant treatment for early breast cancer: Final results of a randomized phase III study (ABCSG-24). Ann. Oncol. 2014, 25, 366–371. [Google Scholar] [PubMed]
- Chen, X.; Ye, G.; Zhang, C.; Li, X.; Chen, Y.; Xie, X.; Zheng, H.; Cao, Y.; Wu, K.; Ni, D.; et al. Superior outcome after neoadjuvant chemotherapy with TXTetaxel, anthracycline, and cyclophosphamide versus docetaxel plus cyclophosphamide: Results from the NATT trial in triple negative or Her2 positive breast cancer. Breast Cancer Res. Treat. 2013, 142, 549–558. [Google Scholar] [PubMed]
- Mirzaei, H.R.; Rasekh, P.S.; Nasrollahi, F.; Rasekh, P.S.; Tirabad, Z.A.; Moein, H.R.; Ghaffari, P.; Hajian, P. Dose-dense epirubicin and cyclophosphamide followed by TXTetaxel as adjuvant chemotherapy in node-positive breast cancer. Int. J. Breast Cancer 2013, 2013, 404396. [Google Scholar] [CrossRef] [PubMed]
- Videira, M.; Reis, R.L.; Brito, M.A. Deconstructing breast cancer cell biology and the mechanisms of multidrug resistance. Biochim. Biophys. Acta 2014, 1846, 312–325. [Google Scholar] [PubMed]
- Pedrini, I.; Gazzano, E.; Chegaev, K.; Rolando, B.; Marengo, A.; Kopecka, J.; Fruttero, R.; Ghigo, D.; Arpicco, S.; Riganti, C. Liposomal nitrooxy-doxorubicin: One step over caelyx in drug-resistant human cancer cells. Mol. Pharm. 2014, 11, 3068–3079. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.D.; Wolf, C.R. Molecular mechanisms of drug resistance. Biochem. J. 1990, 272, 281–295. [Google Scholar] [PubMed]
- Shirakawa, K.; Takara, K.; Tanigawara, Y.; Aoyama, N.; Kasuga, M.; Komada, F.; Sakaeda, T.; Okumura, K. Interaction of docetaxel (“Taxotere”) with human P-glycoprotein. Jpn. J. Cancer Res. 1999, 90, 1380–1386. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, C.; Pan, Y.-H.; Shan, M.; Xu, M.; Bao, J.-L.; Zhao, L.-M. Knockdown of UbcH10 Enhances the Chemosensitivity of Dual Drug Resistant Breast Cancer Cells to Epirubicin and Docetaxel. Int. J. Mol. Sci. 2015, 16, 4698-4712. https://doi.org/10.3390/ijms16034698
Wang C, Pan Y-H, Shan M, Xu M, Bao J-L, Zhao L-M. Knockdown of UbcH10 Enhances the Chemosensitivity of Dual Drug Resistant Breast Cancer Cells to Epirubicin and Docetaxel. International Journal of Molecular Sciences. 2015; 16(3):4698-4712. https://doi.org/10.3390/ijms16034698
Chicago/Turabian StyleWang, Cheng, Yun-Hao Pan, Ming Shan, Ming Xu, Jia-Lin Bao, and Li-Ming Zhao. 2015. "Knockdown of UbcH10 Enhances the Chemosensitivity of Dual Drug Resistant Breast Cancer Cells to Epirubicin and Docetaxel" International Journal of Molecular Sciences 16, no. 3: 4698-4712. https://doi.org/10.3390/ijms16034698




