The Inflammation-Related Gene S100A12 Is Positively Regulated by C/EBPβ and AP-1 in Pigs
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
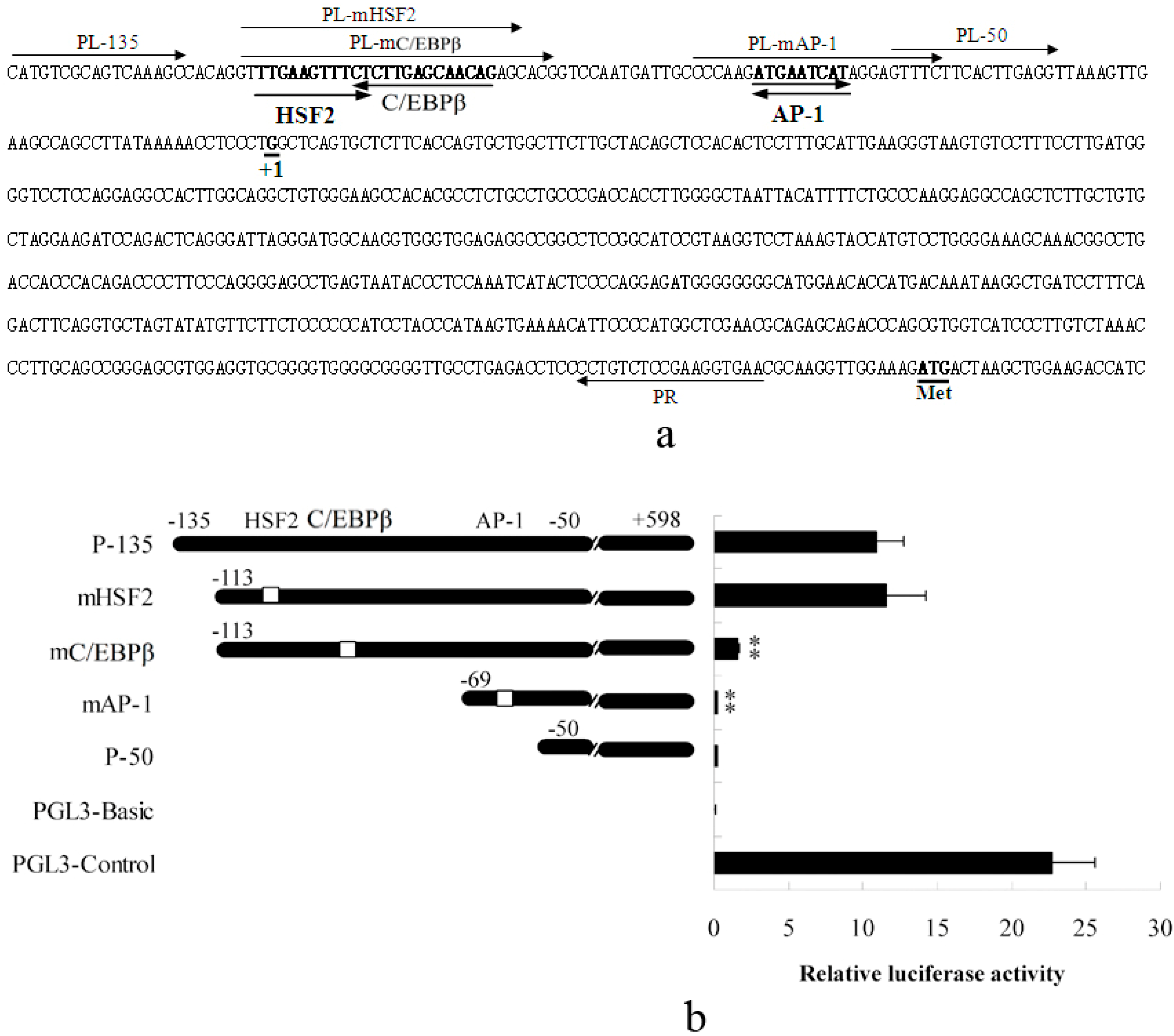
2.1.1. Abolished AP-1 and C/EBPβ Binding Sites Decrease Porcine S100A12 Gene Promoter Activity
2.1.2. Promoter Activity and Expression of the S100A12 Gene Were Increased When AP-1 or C/EBPβ Were Over-Expressed

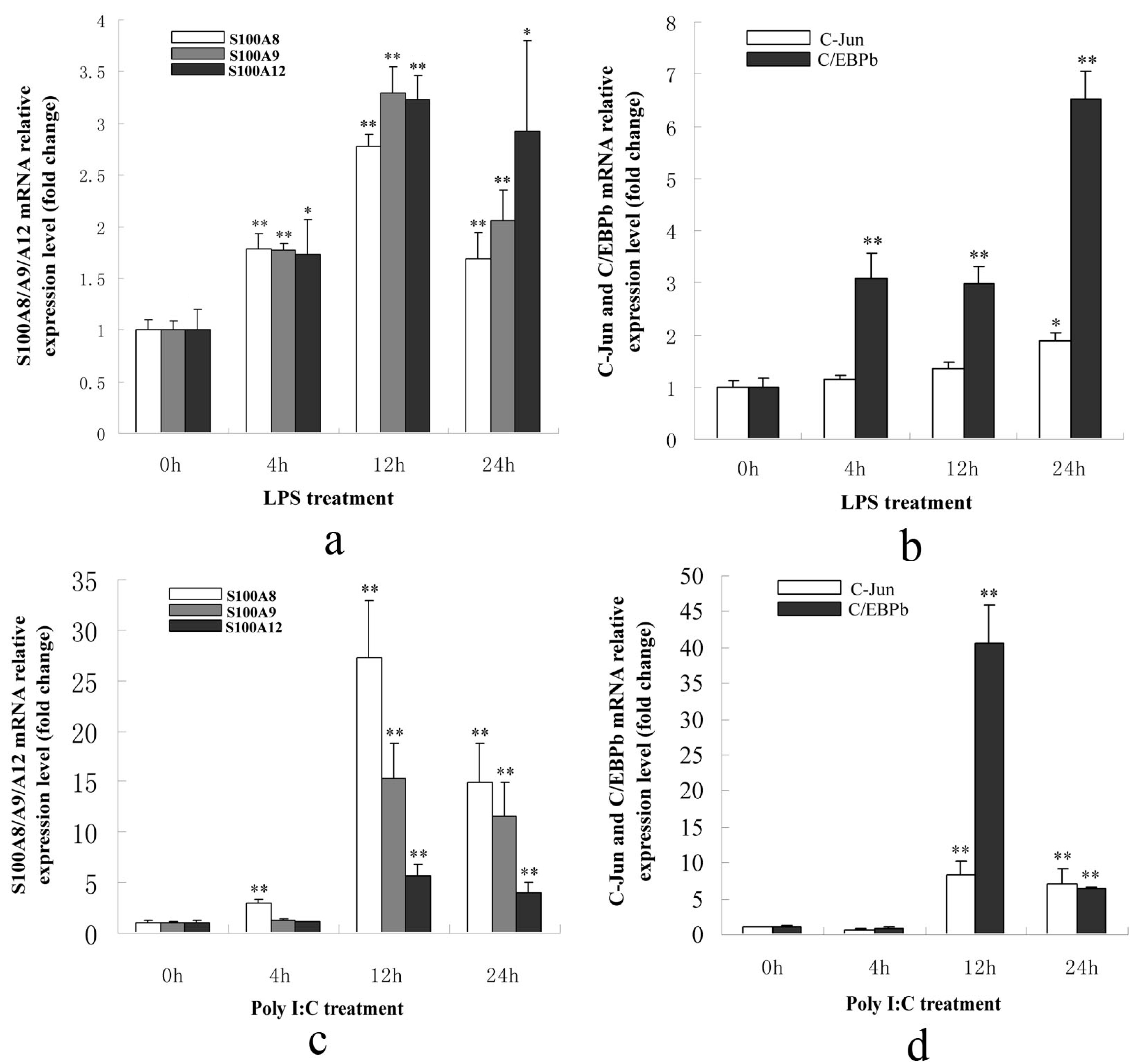
2.1.3. S100A8/A9/A12, AP-1 and C/EBPβ Genes Were Up-Regulated in PK-15 Cells When Treated with LPS or Poly I: C
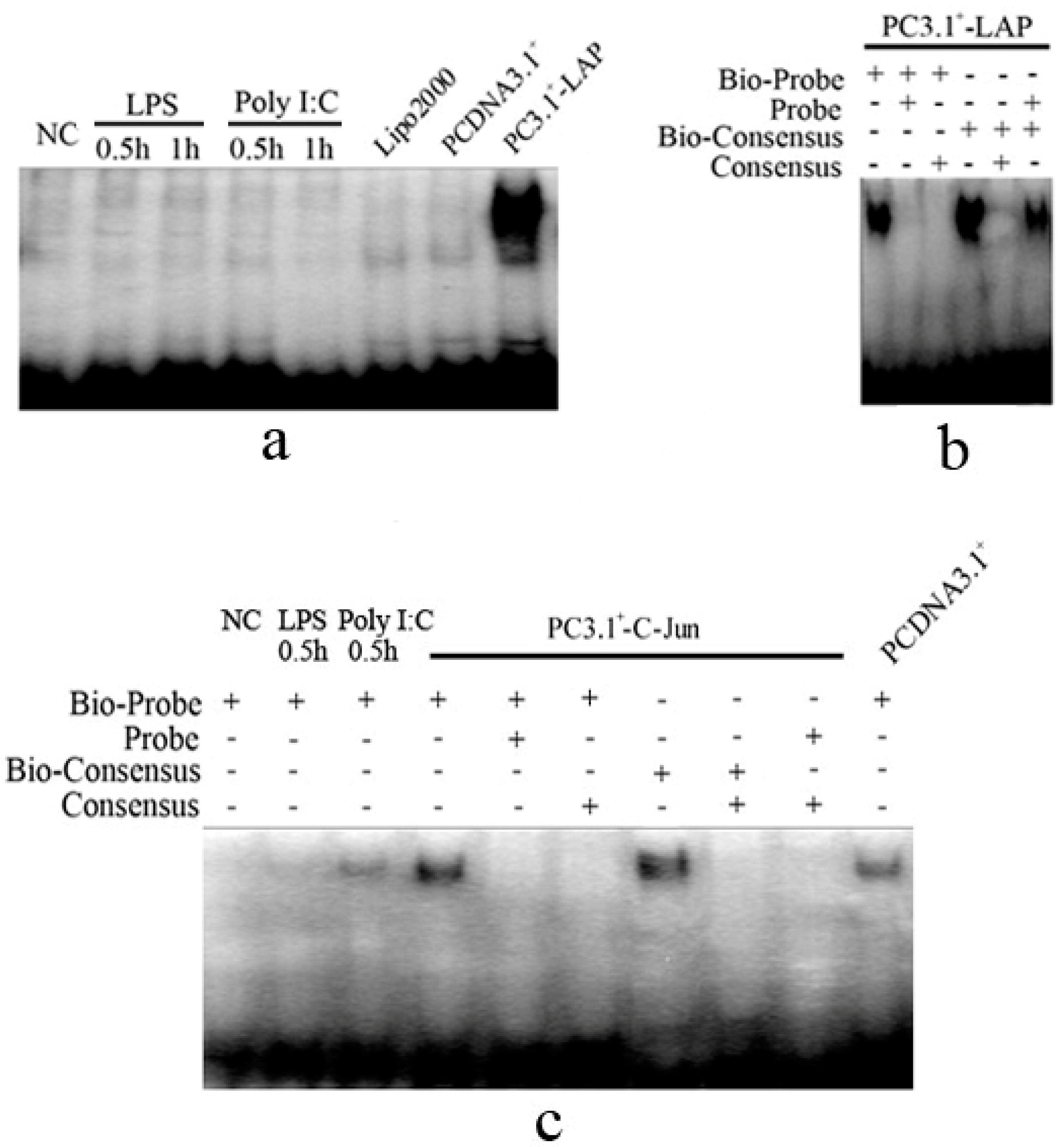
2.1.4. C/EBPβ and AP-1 Directly Bind the Promoter of the Porcine S100A12 Gene
2.1.5. C/EBPβ and AP-1 Act Synergistically in the Transcriptional Activation of the Porcine S100A12 Gene
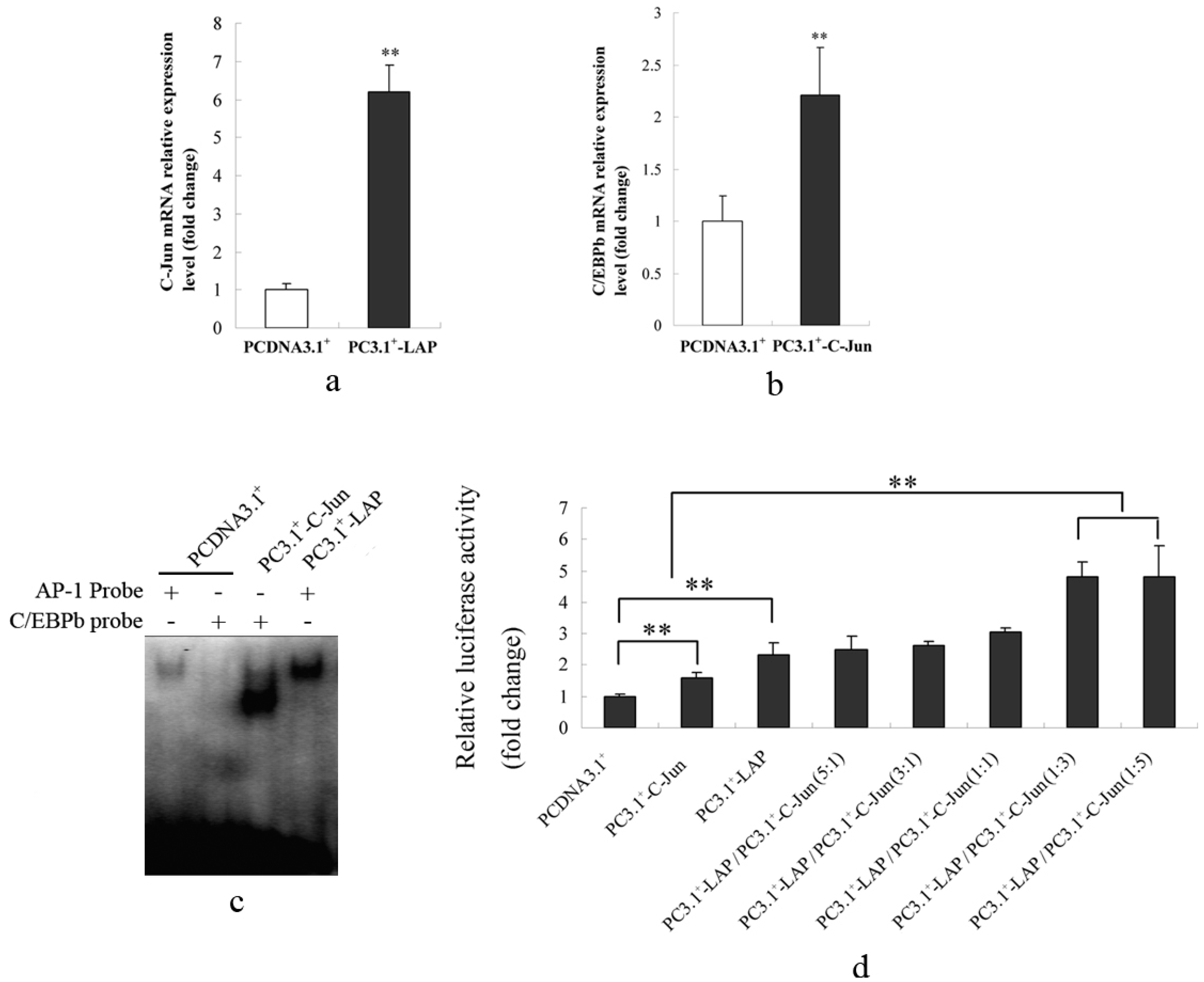
2.2. Discussion
3. Materials and Methods
3.1. Cells and Vectors
3.2. Quantitative Polymerase Chain Reaction
3.3. Luciferase Reporter Gene Analysis
3.4. Electromobility Shift Assay (EMSA) for Detection of DNA Binding Abilities of C/EBPβ and AP-1
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Santamaria-Kisiel, L.; Rintala-Dempsey, A.C.; Shaw, G.S. Calcium-dependent and -independent interactions of the S100 protein family. Biochem. J. 2006, 396, 201–214. [Google Scholar] [CrossRef]
- Ravasi, T.; Hsu, K.; Goyette, J.; Schroder, K.; Yang, Z.; Rahimi, F.; Miranda, L.P.; Alewood, P.F.; Hume, D.A.; Geczy, C. Probing the S100 protein family through genomic and functional analysis. Genomics 2004, 84, 10–22. [Google Scholar] [CrossRef]
- Foell, D.; Frosch, M.; Sorg, C.; Roth, J. Phagocyte-specific calcium-binding S100 proteins as clinical laboratory markers of inflammation. Clin. Chim. Acta 2004, 344, 37–51. [Google Scholar] [CrossRef]
- Foell, D.; Seeliger, S.; Vogl, T.; Koch, H.G.; Maschek, H.; Harms, E.; Sorg, C.; Roth, J. Expression of S100A12 (EN-RAGE) in cystic fibrosis. Thorax 2003, 58, 613–617. [Google Scholar] [CrossRef]
- Yang, Z.; Tao, T.; Raftery, M.J.; Youssef, P.; di Girolamo, N.; Geczy, C.L. Proinflammatory properties of the human S100 protein S100A12. J. Leukoc. Biol. 2001, 69, 986–994. [Google Scholar]
- Yang, Z.; Yan, W.X.; Cai, H.; Tedla, N.; Armishaw, C.; di Girolamo, N.; Wang, H.W.; Hampartzoumian, T.; Simpson, J.L.; Gibson, P.G.; et al. S100A12 provokes mast cell activation: A potential amplification pathway in asthma and innate immunity. J. Allergy Clin. Immunol. 2007, 119, 106–114. [Google Scholar]
- Hofmann, M.A.; Drury, S.; Fu, C.; Qu, W.; Taguchi, A.; Lu, Y.; Avila, C.; Kambham, N.; Bierhaus, A.; Nawroth, P.; et al. RAGE mediates a novel proinflammatory axis: A central cell surface receptor for S100/calgranulin polypeptides. Cell 1999, 97, 889–901. [Google Scholar] [CrossRef]
- Neeper, M.; Schmidt, A.M.; Brett, J.; Yan, S.D.; Wang, F.; Pan, Y.C.; Elliston, K.; Stern, D.; Shaw, A. Cloning and expression of a cell surface receptor for advanced glycosylation end products of proteins. J. Biol. Chem. 1992, 267, 14998–15004. [Google Scholar]
- Srikrishna, G.; Huttunen, H.J.; Johansson, L.; Weigle, B.; Yamaguchi, Y.; Rauvala, H.; Freeze, H.H. N-Glycans on the receptor for advanced glycation end products influence amphoterin binding and neurite outgrowth. J. Neurochem. 2002, 80, 998–1008. [Google Scholar] [CrossRef]
- Foell, D.; Wittkowski, H.; Kessel, C.; Luken, A.; Weinhage, T.; Varga, G.; Vogl, T.; Wirth, T.; Viemann, D.; Bjork, P.; et al. Proinflammatory S100A12 can activate human monocytes via Toll-like receptor 4. Am. J. Respir. Crit. Care Med. 2013, 187, 1324–1334. [Google Scholar]
- Hofmann, B.M.; Wilk, J.; Heydemann, A.; Kim, G.; Rehman, J.; Lodato, J.A.; Raman, J.; McNally, E.M. S100A12 mediates aortic wall remodeling and aortic aneurysm. Circ. Res. 2010, 106, 145–154. [Google Scholar] [CrossRef]
- Kosaki, A.; Hasegawa, T.; Kimura, T.; Iida, K.; Hitomi, J.; Matsubara, H.; Mori, Y.; Okigaki, M.; Toyoda, N.; Masaki, H.; et al. Increased plasma S100A12 (EN-RAGE) levels in patients with type 2 diabetes. J. Clin. Endocrinol. Metab. 2004, 89, 5423–5428. [Google Scholar] [CrossRef]
- Yao, D.; Brownlee, M. Hyperglycemia-induced reactive oxygen species increase expression of the receptor for advanced glycation end products (RAGE) and RAGE ligands. Diabetes 2010, 59, 249–255. [Google Scholar] [CrossRef]
- Hatakeyama, T.; Okada, M.; Shimamoto, S.; Kubota, Y.; Kobayashi, R. Identification of intracellular target proteins of the calcium-signaling protein S100A12. Eur. J. Biochem. 2004, 271, 3765–3775. [Google Scholar] [CrossRef]
- Dell’Angelica, E.C.; Schleicher, C.H.; Santome, J.A. Primary structure and binding properties of calgranulin C, a novel S100-like calcium-binding protein from pig granulocytes. J. Biol. Chem. 1994, 269, 28929–28936. [Google Scholar]
- Guignard, F.; Mauel, J.; Markert, M. Identification and characterization of a novel human neutrophil protein related to the S100 family. Biochem. J. 1995, 309, 395–401. [Google Scholar]
- Chen, H.; Li, C.; Fang, M.; Zhu, M.; Li, X.; Zhou, R.; Li, K.; Zhao, S. Understanding Haemophilus parasuis infection in porcine spleen through a transcriptomics approach. BMC Genomics 2009, 10, 64. [Google Scholar] [CrossRef]
- Chen, H.; Lunney, J.K.; Cheng, L.; Li, X.; Cao, J.; Zhu, M.; Zhao, S. Porcine S100A8 and S100A9: Molecular characterizations and crucial functions in response to Haemophilus parasuis infection. Dev. Comp. Immunol. 2011, 35, 490–500. [Google Scholar] [CrossRef]
- Chen, H.; Cheng, L.; Yang, S.; Liu, X.; Liu, Y.; Tang, J.; Li, X.; He, Q.; Zhao, S. Molecular characterization, induced expression, and transcriptional regulation of porcine S100A12 gene. Mol. Immunol. 2010, 47, 1601–1607. [Google Scholar] [CrossRef]
- Vogl, T.; Tenbrock, K.; Ludwig, S.; Leukert, N.; Ehrhardt, C.; van Zoelen, M.A.; Nacken, W.; Foell, D.; van der, Poll T.; Sorg, C.; et al. Mrp8 and Mrp14 are endogenous activators of Toll-like receptor 4, promoting lethal, endotoxin-induced shock. Nat. Med. 2007, 13, 1042–1049. [Google Scholar] [CrossRef]
- Wang, Y.; Qu, L.; Uthe, J.J.; Bearson, S.M.; Kuhar, D.; Lunney, J.K.; Couture, O.P.; Nettleton, D.; Dekkers, J.C.; Tuggle, C.K. Global transcriptional response of porcine mesenteric lymph nodes to Salmonella enterica serovar Typhimurium. Genomics 2007, 90, 72–84. [Google Scholar] [CrossRef]
- Sakitani, K.; Nishizawa, M.; Inoue, K.; Masu, Y.; Okumura, T.; Ito, S. Synergistic regulation of inducible nitric oxide synthase gene by CCAAT/enhancer-binding protein beta and nuclear factor-kappaB in hepatocytes. Genes Cells 1998, 3, 321–330. [Google Scholar] [CrossRef]
- Agrawal, A.; Cha-Molstad, H.; Samols, D.; Kushner, I. Transactivation of C-reactive protein by IL-6 requires synergistic interaction of CCAAT/enhancer binding protein beta (C/EBP beta) and Rel p50. J. Immunol. 2001, 166, 2378–2384. [Google Scholar] [CrossRef]
- Bretz, J.D.; Williams, S.C.; Baer, M.; Johnson, P.F.; Schwartz, R.C. C/EBP-related protein 2 confers lipopolysaccharide-inducible expression of interleukin 6 and monocyte chemoattractant protein 1 to a lymphoblastic cell line. Proc. Natl. Acad. Sci. USA 1994, 91, 7306–7310. [Google Scholar] [CrossRef]
- Matsumoto, M.; Sakao, Y.; Akira, S. Inducible expression of nuclear factor IL-6 increases endogenous gene expression of macrophage inflammatory protein-1 alpha, osteopontin and CD14 in a monocytic leukemia cell line. Int. Immunol. 1998, 10, 1825–1835. [Google Scholar] [CrossRef]
- Uematsu, S.; Kaisho, T.; Tanaka, T.; Matsumoto, M.; Yamakami, M.; Omori, H.; Yamamoto, M.; Yoshimori, T.; Akira, S. The C/EBP beta isoform 34-kDa LAP is responsible for NF-IL-6-mediated gene induction in activated macrophages, but is not essential for intracellular bacteria killing. J. Immunol. 2007, 179, 5378–5386. [Google Scholar] [CrossRef]
- Tanaka, T.; Akira, S.; Yoshida, K.; Umemoto, M.; Yoneda, Y.; Shirafuji, N.; Fujiwara, H.; Suematsu, S.; Yoshida, N.; Kishimoto, T. Targeted disruption of the NF-IL6 gene discloses its essential role in bacteria killing and tumor cytotoxicity by macrophages. Cell 1995, 80, 353–361. [Google Scholar] [CrossRef]
- Screpanti, I.; Romani, L.; Musiani, P.; Modesti, A.; Fattori, E.; Lazzaro, D.; Sellitto, C.; Scarpa, S.; Bellavia, D.; Lattanzio, G. Lymphoproliferative disorder and imbalanced T-helper response in C/EBP beta-deficient mice. EMBO J. 1995, 14, 1932–1941. [Google Scholar]
- Descombes, P.; Schibler, U. A liver-enriched transcriptional activator protein, LAP, and a transcriptional inhibitory protein, LIP, are translated from the same mRNA. Cell 1991, 67, 569–579. [Google Scholar] [CrossRef]
- Liu, W.; Ouyang, X.; Yang, J.; Liu, J.; Li, Q.; Gu, Y.; Fukata, M.; Lin, T.; He, J.C.; Abreu, M.; et al. AP-1 activated by toll-like receptors regulates expression of IL-23 p19. J. Biol. Chem. 2009, 284, 24006–24016. [Google Scholar] [CrossRef]
- Rohrbach, S.; Engelhardt, S.; Lohse, M.J.; Werdan, K.; Holtz, J.; Muller-Werdan, U. Activation of AP-1 contributes to the beta-adrenoceptor-mediated myocardial induction of interleukin-6. Mol. Med. 2007, 13, 605–614. [Google Scholar]
- Kang, S.S.; Woo, S.S.; Im, J.; Yang, J.S.; Yun, C.H.; Ju, H.R.; Son, C.G.; Moon, E.Y.; Han, S.H. Human placenta promotes IL-8 expression through activation of JNK/SAPK and transcription factors NF-kappaB and AP-1 in PMA-differentiated THP-1 cells. Int. Immunopharmacol. 2007, 7, 1488–1495. [Google Scholar] [CrossRef]
- Casals, C.; Barrachina, M.; Serra, M.; Lloberas, J.; Celada, A. Lipopolysaccharide up-regulates MHC class II expression on dendritic cells through an AP-1 enhancer without affecting the levels of CIITA. J. Immunol. 2007, 178, 6307–6315. [Google Scholar]
- Vanden Bush, T.J.; Bishop, G.A. TLR7 and CD40 cooperate in IL-6 production via enhanced JNK and AP-1 activation. Eur. J. Immunol. 2008, 38, 400–409. [Google Scholar] [CrossRef]
- Park, J.; Chung, S.W.; Kim, S.H.; Kim, T.S. Up-regulation of interleukin-4 production via NF-AT/AP-1 activation in T cells by biochanin A, a phytoestrogen and its metabolites. Toxicol. Appl. Pharmacol. 2006, 212, 188–199. [Google Scholar] [CrossRef]
- Shaulian, E.; Karin, M. AP-1 as a regulator of cell life and death. Nat. Cell Biol. 2002, 4, E131–E136. [Google Scholar]
- Hess, J.; Angel, P.; Schorpp-Kistner, M. AP-1 subunits: Quarrel and harmony among siblings. J. Cell Sci. 2004, 117, 5965–5973. [Google Scholar] [CrossRef]
- Angel, P.; Karin, M. The role of Jun, Fos and the AP-1 complex in cell-proliferation and transformation. Biochim. Biophys. Acta 1991, 1072, 129–157. [Google Scholar]
- Yuan, H.; Pan, Y.; Young, C.Y. Overexpression of c-Jun induced by quercetin and resverol inhibits the expression and function of the androgen receptor in human prostate cancer cells. Cancer Lett. 2004, 213, 155–163. [Google Scholar] [CrossRef]
- Duan, L.; Sterba, K.; Kolomeichuk, S.; Kim, H.; Brown, P.H.; Chambers, T.C. Inducible overexpression of C-Jun in MCF7 cells causes resistance to vinblastine via inhibition of drug-induced apoptosis and senescence at a step subsequent to mitotic arrest. Biochem. Pharmacol. 2007, 73, 481–490. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, X.; Tang, J.; Xu, J.; Zhu, M.; Cao, J.; Liu, Y.; Yu, M.; Zhao, S. The Inflammation-Related Gene S100A12 Is Positively Regulated by C/EBPβ and AP-1 in Pigs. Int. J. Mol. Sci. 2014, 15, 13802-13816. https://doi.org/10.3390/ijms150813802
Li X, Tang J, Xu J, Zhu M, Cao J, Liu Y, Yu M, Zhao S. The Inflammation-Related Gene S100A12 Is Positively Regulated by C/EBPβ and AP-1 in Pigs. International Journal of Molecular Sciences. 2014; 15(8):13802-13816. https://doi.org/10.3390/ijms150813802
Chicago/Turabian StyleLi, Xinyun, Juan Tang, Jing Xu, Mengjin Zhu, Jianhua Cao, Ying Liu, Mei Yu, and Shuhong Zhao. 2014. "The Inflammation-Related Gene S100A12 Is Positively Regulated by C/EBPβ and AP-1 in Pigs" International Journal of Molecular Sciences 15, no. 8: 13802-13816. https://doi.org/10.3390/ijms150813802




