Arginine Enhances Osteoblastogenesis and Inhibits Adipogenesis through the Regulation of Wnt and NFATc Signaling in Human Mesenchymal Stem Cells
Abstract
:1. Introduction
2. Results and Discussion
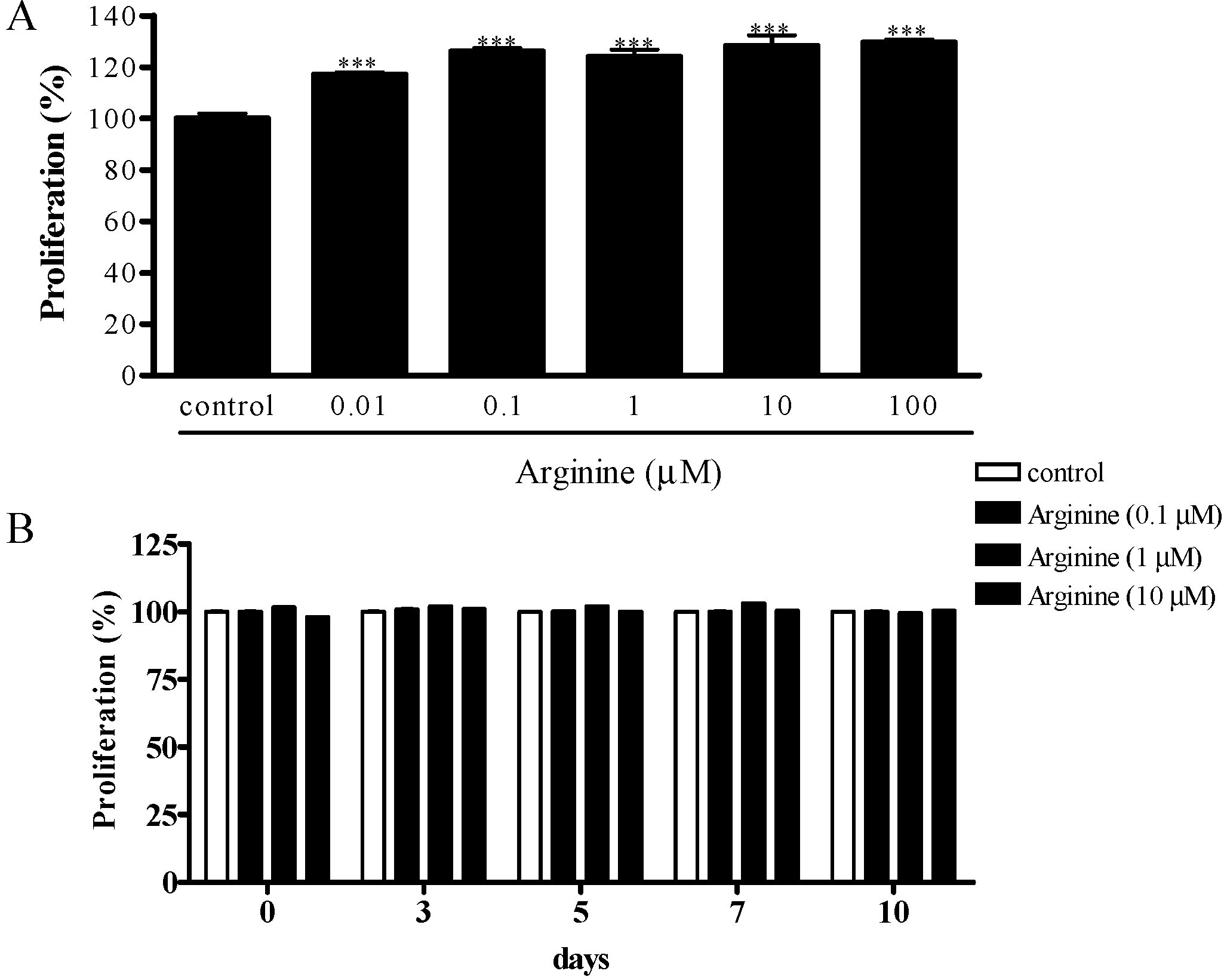
2.1. Effect of Arginine on the Proliferation of MSCs
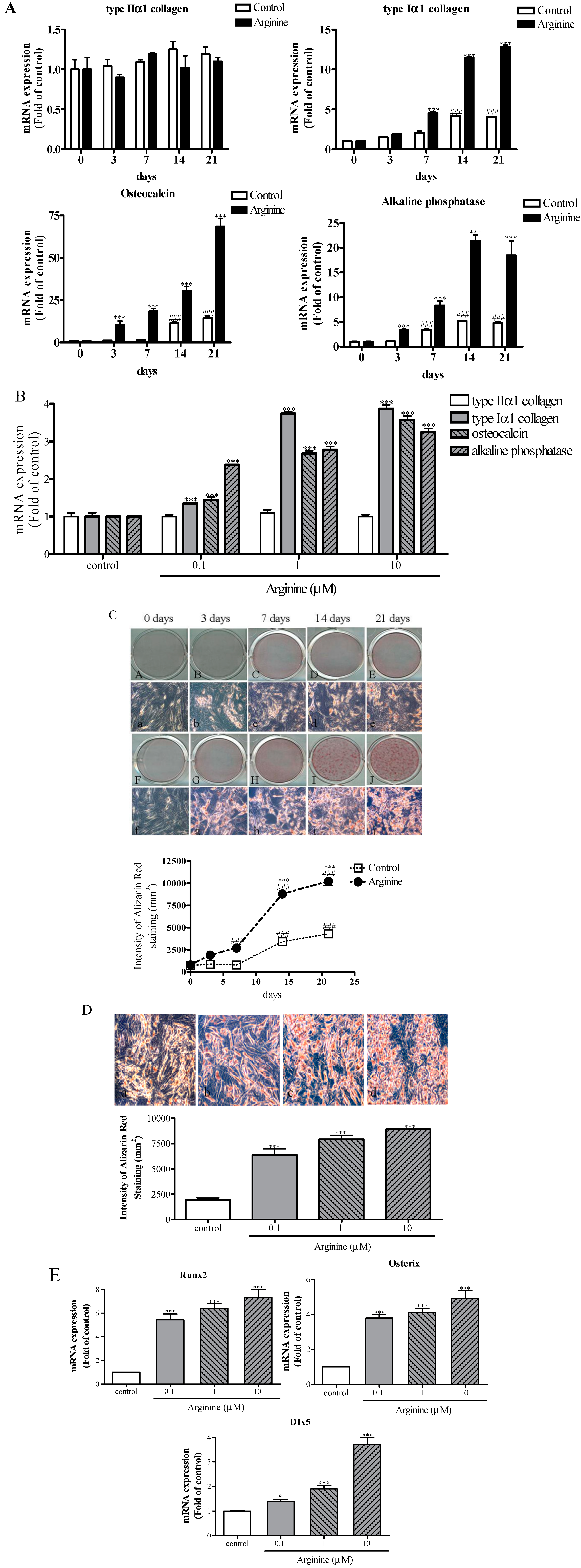
2.2. Effect of Arginine on Osteogenic Differentiation of MSCs
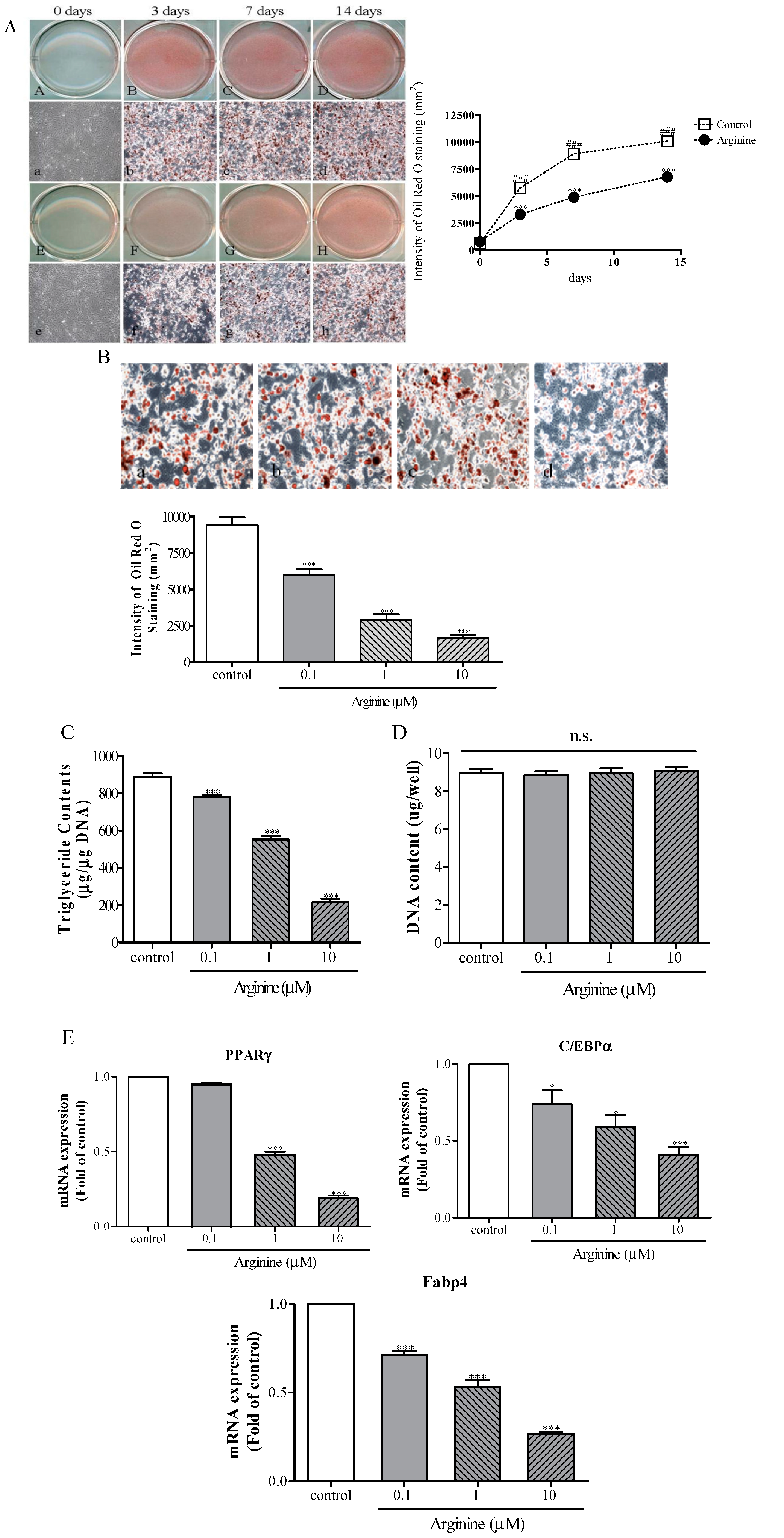
2.3. Effect of Arginine on Adipogenesis in MSCs
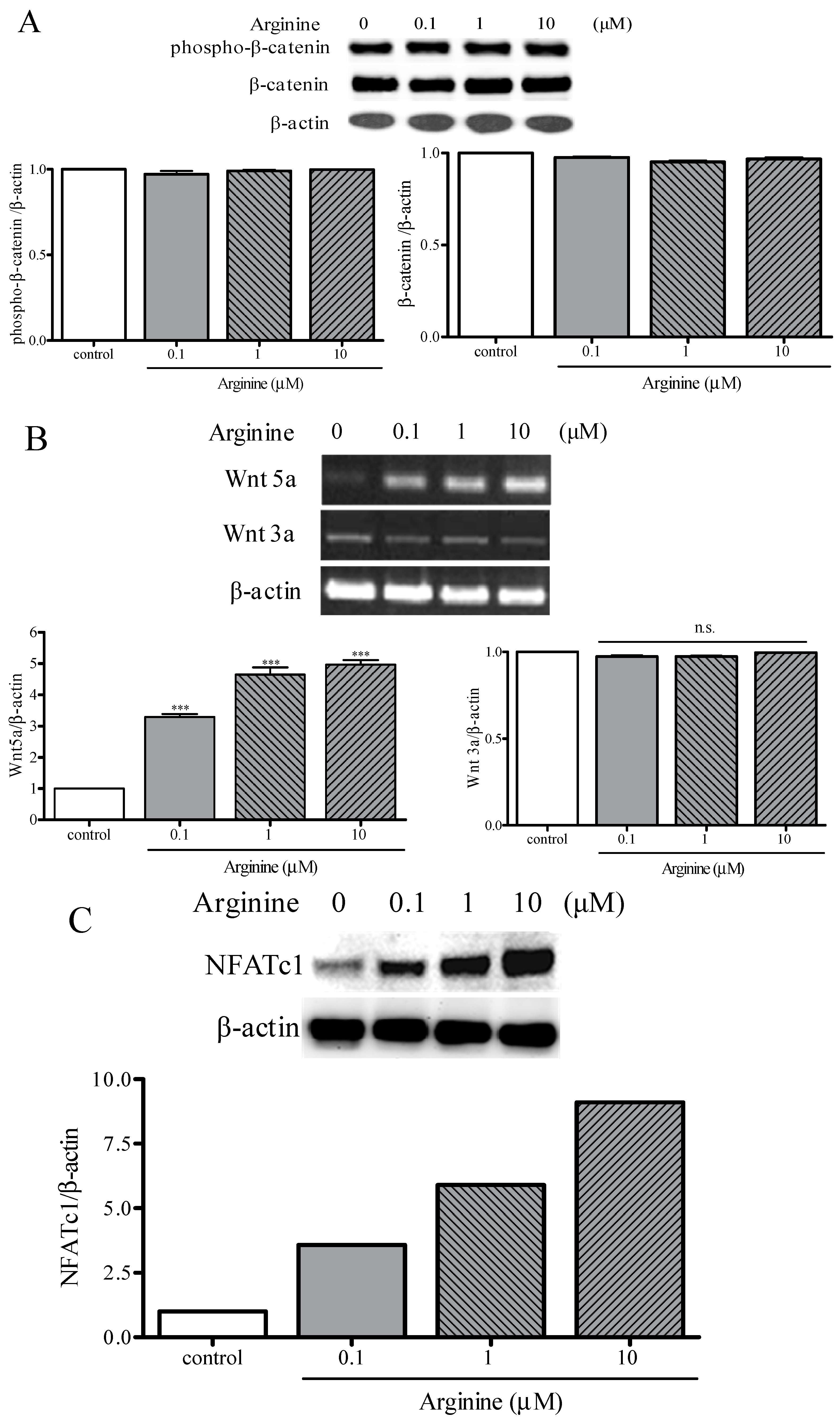
2.4. Osteogenic and Adipogenic Differentiation Is Regulated through the NFATc and Wnt Signaling Pathways

3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Cell Culture and Treatments
3.3. Cell Proliferation Assay
3.4. Assessment of Osteogenic Differentiation
3.5. Alizarin Red S Staining
3.6. Assessment of Adipogenic Differentiation
3.7. Oil Red O Staining
3.8. TG Assay and DNA-Content Measurement
3.9. Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR) and Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR).
| mRNA | Primers | Annealing Tm (Cycle) | |
|---|---|---|---|
| Type II α1 collagen | Fw: 5'-AACACTGCAACGTCCAGAT-3' | 58 °C (32) | |
| Rv: 5'-CTGCAGCACGGTATAGGTGA-3' | |||
| Type I α1 collagen | Fw: 5'-TGACCTCAAGATGTGCCACT-3' | 58 °C (32) | |
| Rv: 5'-GGGAGTTTCCATGAAGCCAC-3' | |||
| Osteocalcin | Fw: 5'-CATGAGAGCCCTCACA-3' | 55 °C (32) | |
| Rv: 5'-AGAGCGACACCCTAGAC-3' | |||
| Alkaline Phosphatase | Fw: 5'-TCAGAAGCTCAACACCAACG-3' | 58 °C (32) | |
| Rv: 5'-GTCAGGGACCTGGGCATT-3' | |||
| Runx2 | Fw: 5'-ACAACCACAGAACCACAAG-3' | 58 °C (30) | |
| Rv: 5'-TCTCGGTGGCTGGTAGTGA-3' | |||
| DIx5 | Fw: 5'-GACAGGATCCCTATGACAGGAGTG-3' | 58 °C (30) | |
| Rv: 5'-GGACTCGAGATCTAATAAAGCGTC-3' | |||
| Osterix | Fw: 5'-TGAGGAAGAAGCCCATTCAC-3' | 58 °C (30) | |
| Rv: 5'-ACTTCTTCTCCCGGGTGTG-3' | |||
| Proliferators-activated receptor γ | Fw: 5'-AGAACACCTCTGAAAGTAAG-3' | 58 °C (30) | |
| Rv: 5'-ACTGTGATGTACTGCTGAAC-3' | |||
| CCAAT/enhancer binding protein α | Fw: 5'-GAGTGACAAGCCTGTAGCC-3' | 58 °C (30) | |
| Rv: 5'-GGTTGACTTTCTCCTGGTAT-3' | |||
| Fatty acid binding protein 4 | Fw: 5'-TCAGTTCGTCCTCACTCCAG-3' | 58 °C (30) | |
| Rv: 5'-TTGGTCCACCTGTCATCTTC-3' | |||
| β-actin | Fw: 5'-GCTCTCCAGAACATCACTCCTGCC-3' | 58 °C (30) | |
| Rv: 5'-CGTTGTCATACCAGGAAATGAGCTT-3' | |||
| mRNA | Primers | Annealing Tm (Cycle) |
|---|---|---|
| Wnt5a | Fw: 5'-ACGCTA AGGGTTCCTATGAG-3' | 58 °C (32) |
| Rv: 5'-CATAGCAGCACCAGTGAAAC-3' | ||
| Wnt3a | Fw: 5'-GTTCTGCAGCGAAGTGGTG-3' | 58 °C (32) |
| Rv: 5'-CTGCAGCACGGTATAGGTGA-3' | ||
| β-actin | Fw: 5'-GCTCTCCAGAACATCACTCCTGCC-3' | 58 °C (30) |
| Rv: 5'-CGTTGTCATACCAGGAAATGAGCTT-3' |
3.10. Western Blotting
3.11. Statistical Analysis
4. Conclusions
 Indicates activation or induction;
Indicates activation or induction;  indicates inhibition or blockade.
indicates inhibition or blockade.
 Indicates activation or induction;
Indicates activation or induction;  indicates inhibition or blockade.
indicates inhibition or blockade.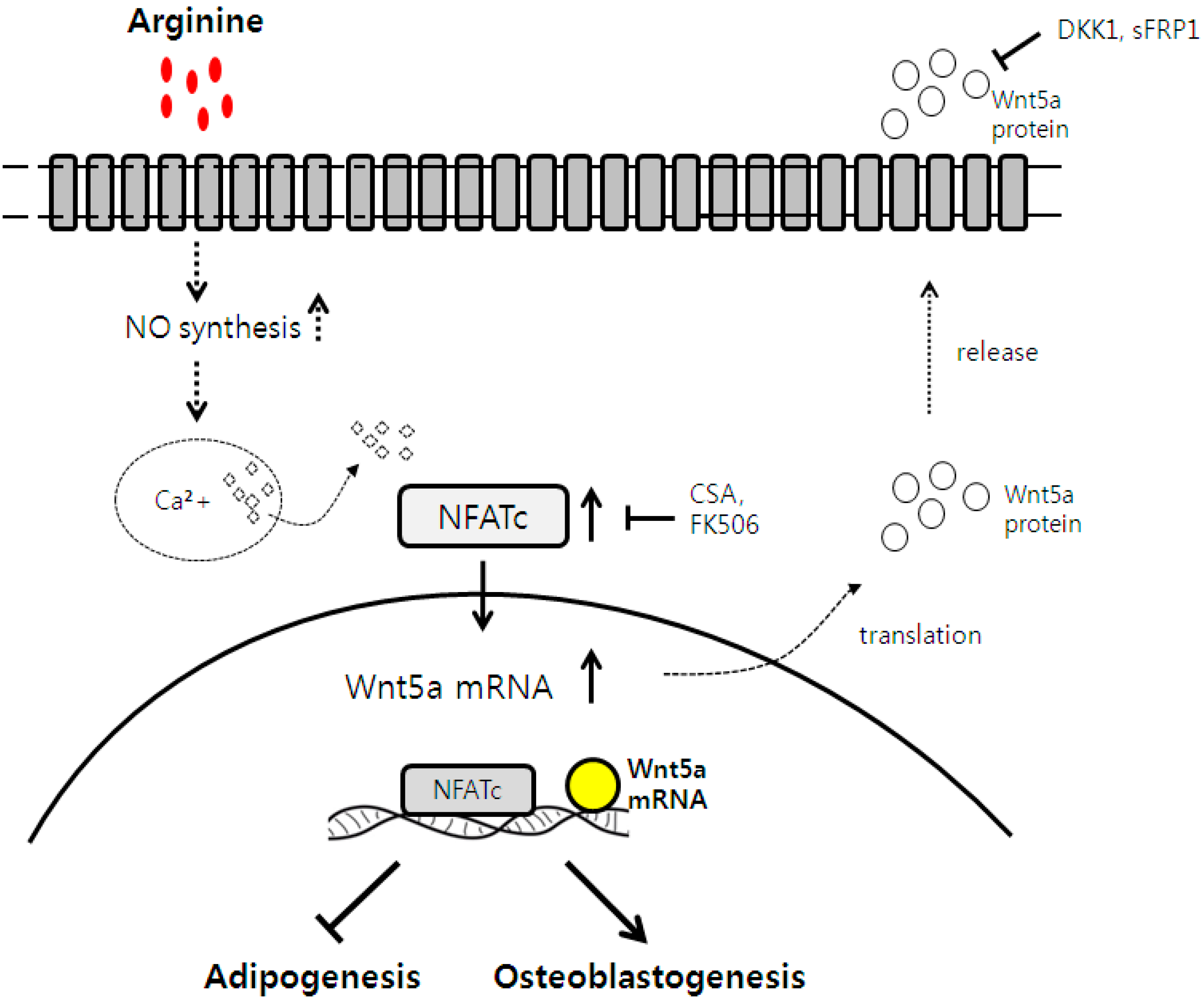
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Webster, R.A.; Blaber, S.P.; Herbert, B.R.; Wilkins, M.R.; Vesey, G. The role of mesenchymal stem cells in veterinary therapeutics—A review. N. Z. Vet. J. 2012, 60, 265–272. [Google Scholar] [CrossRef]
- Raisz, L.G. Pathogenesis of osteoporosis: Concepts, conflicts, and prospects. J. Clin. Investig. 2005, 115, 3318–3325. [Google Scholar] [CrossRef]
- Duque, G. Bone and fat connection in aging bone. Curr. Opin. Rheumatol. 2008, 20, 429–434. [Google Scholar] [CrossRef]
- Nelson-Dooley, C.; della-Fera, M.A.; Hamrick, M.; Baile, C.A. Novel treatments for obesity and osteoporosis: Targeting apoptotic pathways in adipocytes. Curr. Med. Chem. 2005, 12, 2215–2225. [Google Scholar] [CrossRef]
- Komi, J.; Lankinen, K.S.; DeGregorio, M.; Heikkinen, J.; Saarikoski, S.; Tuppurainen, M.; Halonen, K.; Lammintausta, R.; Väänänen, K.; Ylikorkala, O.; et al. Effects of ospemifene and raloxifene on biochemical markers of bone turnover in postmenopausal women. J. Bone Miner. Metab. 2006, 24, 314–318. [Google Scholar] [CrossRef]
- Seeman, E. Reduced bone formation and increased bone resorption: Rational targets for the treatment of osteoporosis. Osteoporos. Int. 2003, 3, S2–S8. [Google Scholar]
- Abdallah, B.M.; Kassem, M. New factors controlling the balance between osteoblastogenesis and adipogenesis. Bone 2012, 50, 540–545. [Google Scholar] [CrossRef]
- Franceschi, R.T.; Xiao, G. Regulation of the osteoblast-specific transcription factor, Runx2: Responsiveness to multiple signal transduction pathways. J. Cell. Biochem. 2003, 88, 446–454. [Google Scholar] [CrossRef]
- Nakashima, K.; Zhou, X.; Kunkel, G.; Zhang, Z.; Deng, J.M.; Behringer, R.R.; de, Crombrugghe, B. The novel zinc finger-containing transcription factor osterix is required for osteoblast differentiation and bone formation. Cell 2002, 108, 17–29. [Google Scholar] [CrossRef]
- Takada, I.; Kouzmenko, A.P.; Kato, S. Wnt and PPARγ signaling in osteoblastogenesis and adipogenesis. Nat. Rev. Rheumatol. 2009, 5, 442–447. [Google Scholar] [CrossRef]
- Takada, I.; Kouzmenko, A.P.; Kato, S. Molecular switching of osteoblastogenesis versus adipogenesis: implications for targeted therapies. Expert Opin. Ther. Targets 2009, 13, 593–603. [Google Scholar] [CrossRef]
- Bäckesjö, C.M.; Li, Y.; Lindgren, U.; Haldosén, L.A. Activation of Sirt1 decreases adipocyte formation during osteoblast differentiation of mesenchymal stem cells. J. Bone Miner. Res. 2006, 21, 993–1002. [Google Scholar] [CrossRef]
- Taipaleenmäki, H.; Abdallah, B.M.; AlDahmash, A.; Säämänen, A.M.; Kassem, M. Wnt signalling mediates the cross-talk between bone marrow derived pre-adipocytic and pre-osteoblastic cell populations. Exp. Cell Res. 2011, 317, 745–756. [Google Scholar] [CrossRef]
- Gao, B.; Huang, Q.; Lin, Y.S.; Wei, B.Y.; Guo, Y.S.; Sun, Z.; Wang, L.; Fan, J.; Zhang, H.Y.; Han, Y.H.; et al. Dose-dependent effect of estrogen suppresses the osteo-adipogenic transdifferentiation of osteoblasts via canonical Wnt signaling pathway. PLoS One 2014, 9, e99137. [Google Scholar]
- Franceschi, R.T.; Ge, C.; Xiao, G.; Roca, H.; Jiang, D. Transcriptional regulation of osteoblasts. Cells Tissues Organs 2009, 189, 144–152. [Google Scholar] [CrossRef]
- Takada, I.; Suzawa, M.; Matsumoto, K.; Kato, S. Suppression of PPAR transactivation switches cell fate of bone marrow stem cells from adipocytes into osteoblasts. Ann. N. Y. Acad. Sci. 2007, 1116, 182–195. [Google Scholar] [CrossRef]
- Yang, F.; Yang, D.; Tu, J.; Zheng, Q.; Cai, L.; Wang, L. Strontium enhances osteogenic differentiation of mesenchymal stem cells and in vivo bone formation by activating Wnt/catenin signaling. Stem Cells 2011, 29, 981–991. [Google Scholar] [CrossRef]
- Sen, B.; Styner, M.; Xie, Z.; Case, N.; Rubin, C.T.; Rubin, J. Mechanical loading regulates NFATc1 and beta-catenin signaling through a GSK3beta control node. J. Biol. Chem. 2009, 284, 34607–34617. [Google Scholar] [CrossRef]
- Hedgepeh, C.M.; Conrad, L.J.; Zhang, J.; Huang, H.C.; Lee, V.M.; Klein, P.S. Activation of the Wnt signaling pathway: A molecular mechanism for lithium action. Dev. Biol. 1997, 185, 82–91. [Google Scholar] [CrossRef]
- Kong, X.; Wu, G.; Yin, Y. Roles of phytochemicals in amino acid nutrition. Front. Biosci. 2011, 3, 372–384. [Google Scholar] [CrossRef]
- Wu, G. Amino acids: Metabolism, functions, and nutrition. Amino Acids 2009, 37, 1–17. [Google Scholar] [CrossRef]
- McKnight, J.R.; Satterfield, M.C.; Jobgen, W.S.; Smith, S.B.; Spencer, T.E.; Meininger, C.J.; McNeal, C.J.; Wu, G. Beneficial effects of l-arginine on reducing obesity: potential mechanisms and important implications for human health. Amino Acids 2010, 39, 349–357. [Google Scholar] [CrossRef]
- Ghigo, E.; Arvat, E.; Rizzi, G.; Bellone, J.; Nicolosi, M.; Boffano, G.M.; Mucci, M.; Boghen, M.F.; Camanni, F. Arginine enhances the growth hormone-releasing activity of a synthetic hexapeptide (GHRP-6) in elderly but not in young subjects after oral administration. J. Endocrinol. Investig. 1994, 17, 157–162. [Google Scholar] [CrossRef]
- Chevalley, T.; Rizzoli, R.; Manen, D.; Caverzasio, J.; Bonjour, J.P. Arginine increases insulin-like growth factor-I production and collagen synthesis in osteoblast-like cells. Bone 1998, 23, 103–109. [Google Scholar] [CrossRef]
- Tan, B.; Yin, Y.; Liu, Z.; Li, X.; Xu, H.; Kong, X.; Huang, R.; Tang, W.; Shinzato, I.; Smith, S.B.; et al. Dietary L-arginine supplementation increases muscle gain and reduces body fat mass in growing-finishing pigs. Amino Acids 2009, 37, 169–175. [Google Scholar] [CrossRef]
- Zhang, L.; Su, P.; Xu, C.; Chen, C.; Liang, A.; Du, K.; Peng, Y.; Huang, D. Melatonin inhibits adipogenesis and enhances osteogenesis of human mesenchymal stem cells by suppressing PPARγ expression and enhancing Runx2 expression. J. Pineal Res. 2010, 49, 364–372. [Google Scholar] [CrossRef]
- Lee, J.Y.; Lee, Y.M.; Kim, M.J.; Choi, J.Y.; Park, E.K.; Kim, S.Y.; Lee, S.P.; Yang, J.S.; Kim, D.S. Methylation of the mouse DIx5 and Osx gene promoters regulates cell type-specific gene expression. Mol. Cells 2006, 22, 182–188. [Google Scholar]
- Khosla, S.; Riggs, B.L. Pathophysiology of age-related bone loss and osteoporosis. Metab. Clin. N. Am. 2005, 34, 1015–1030. [Google Scholar] [CrossRef]
- Coskun, B.I.; Guzel, R. Oral bisphosphonate related osteonecrosis of the jaw: A challenging adverse effect. ISRN. Rheumatol. 2013, 16, 215034. [Google Scholar]
- Watts, N.B.; Diab, D.L. Long-term use of bisphosphonates in osteoporosis. J. Clin. Endocrinol. Metab. 2010, 95, 1555–1565. [Google Scholar] [CrossRef]
- Jones, J.R.; Barrick, C.; Kim, K.A.; Lindner, J.; Blondeau, B.; Fujimoto, Y.; Shiota, M.; Kesterson, R.A.; Kahn, B.B.; Magnuson, M.A. Deletion of PPARγ in adipose tissues of mice protects against high fat diet-induced obesity and insulin resistance. Proc. Natl. Acad. Sci. USA. 2005, 102, 6207–6212. [Google Scholar] [CrossRef]
- Farmer, S.R. Regulation of PPARγ activity during adipogenesis. Int. J. Obes. 2005, 29, S13–S16. [Google Scholar] [CrossRef]
- Bernlohr, D.A.; Simpson, M.A.; Hertzel, A.V.; Banaszak, L.J. Intracellular lipid-binding proteins and their genes. Annu. Rev. Nutr. 1997, 17, 277–303. [Google Scholar] [CrossRef]
- Nuttall, M.E.; Gimble, J.M. Controlling the balance between osteoblastogenesis and adipogenesis and the consequent therapeutic implications. Curr. Opin. Pharmacol. 2004, 4, 290–294. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, R.; Wen, S.; McCafferty, D.M.; Beck, P.L.; MacNaughton, W.K. Nitric oxide increases Wnt-induced secreted protein-1 (WISP-1/CCN4) expression and function in colitis. J. Mol. Med. 2009, 87, 435–445. [Google Scholar] [CrossRef]
- Saidak, Z.; Haÿ, E.; Marty, C.; Barbara, A.; Marie, P.J. Strontium ranelate rebalances bone marrow adipogenesis and osteoblastogenesis in senescent osteopenic mice through NFATc/Maf and Wnt signaling. Aging Cell 2012, 11, 467–474. [Google Scholar] [CrossRef]
- Zhu, D.; Cardenas, M.E.; Heitman, J. Calcineurin mutants render T lymphocytes resistant to cyclosporin A. Mol. Pharmacol. 1996, 50, 506–511. [Google Scholar]
- Fromigué, O.; Haÿ, E.; Barbara, A.; Marie, P.J. Essential role of nuclear factor of activated T cells (NFAT)-mediated Wnt signaling in osteoblast differentiation induced by strontium ranelate. J. Biol. Chem. 2010, 285, 25251–25258. [Google Scholar] [CrossRef]
- Winslow, M.M.; Pan, M.; Starbuck, M.; Gallo, E.M.; Deng, L.; Karsenty, G.; Crabtree, G.R. Calcineurin/NFAT signaling in osteoblasts regulates bone mass. Dev. Cell 2006, 10, 771–782. [Google Scholar] [CrossRef]
- Muñoz, F.J.; Godoy, J.A.; Cerpa, W.; Poblete, I.M.; Huidobro-Toro, J.P.; Inestrosa, N.C. Wnt-5a increases NO and modulates NMDA receptor in rat hippocampal neurons. Biochem. Biophys. Res. Commun. 2014, 444, 189–194. [Google Scholar] [CrossRef]
- Li, Y.; Li, B.; Wan, X.; Zhang, W.; Zhong, L.; Tang, S.J. NMDA receptor activation stimulates transcription-independent rapid wnt5a protein synthesis via the MAPK signaling pathway. Mol. Brain 2012, 5. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huh, J.-E.; Choi, J.-Y.; Shin, Y.-O.; Park, D.-S.; Kang, J.W.; Nam, D.; Choi, D.-Y.; Lee, J.-D. Arginine Enhances Osteoblastogenesis and Inhibits Adipogenesis through the Regulation of Wnt and NFATc Signaling in Human Mesenchymal Stem Cells. Int. J. Mol. Sci. 2014, 15, 13010-13029. https://doi.org/10.3390/ijms150713010
Huh J-E, Choi J-Y, Shin Y-O, Park D-S, Kang JW, Nam D, Choi D-Y, Lee J-D. Arginine Enhances Osteoblastogenesis and Inhibits Adipogenesis through the Regulation of Wnt and NFATc Signaling in Human Mesenchymal Stem Cells. International Journal of Molecular Sciences. 2014; 15(7):13010-13029. https://doi.org/10.3390/ijms150713010
Chicago/Turabian StyleHuh, Jeong-Eun, Jun-Young Choi, Ye-Ok Shin, Dong-Suk Park, Jung Won Kang, Dongwoo Nam, Do-Young Choi, and Jae-Dong Lee. 2014. "Arginine Enhances Osteoblastogenesis and Inhibits Adipogenesis through the Regulation of Wnt and NFATc Signaling in Human Mesenchymal Stem Cells" International Journal of Molecular Sciences 15, no. 7: 13010-13029. https://doi.org/10.3390/ijms150713010




