1. Introduction
Organotin (Ot) compounds, used for many years as antifouling biocides on ships, marine structures and fishing nets, became a problem, because of their toxicity and environmental accumulation [
1].
The movement towards global regulation began in October 2001, when the International Maritime Organization (IMO) adopted the International Convention on the Control of Harmful Antifouling Systems (AFS Convention) [
2]. The regulation took effect on the 17 September 2008, prohibiting the use of Ots as active ingredients in antifouling agents on marine vessels. Therefore, instead of the prohibited Ot compounds, paint manufacturers had to develop many alternative products to be used as antifouling coatings.
At first, it was thought that Ot alternative antifoulants would not accumulate as much in sea water and sediment as organotin compounds, because the resolution speed of Ot alternative antifoulants was faster [
3]. However, Ot alternative antifoulants have subsequently been detected at higher levels than initially expected in sea water and sediment, which has caused concern about the possible effects.
The presence of Ot alternative antifoulants in surface sediment had previously been researched in areas, such as Hong Kong [
4], India [
5], Malaysia [
6], Thailand [
7], Vietnam [
8], Greece [
9], Denmark [
10],
etc. However, little was known about the accumulation of these compounds in the sediment core. The detection of biocides in the sediment core in earlier studies in the United Kingdom [
11], Brazil [
12], Korea [
13], Japan [
14],
etc., were only of organotin compounds. In Korea, Brazil and Japan, the highest concentration of tributyltin (TBT) was detected at 46,100, 6000 and 962 µg/kg, respectively.
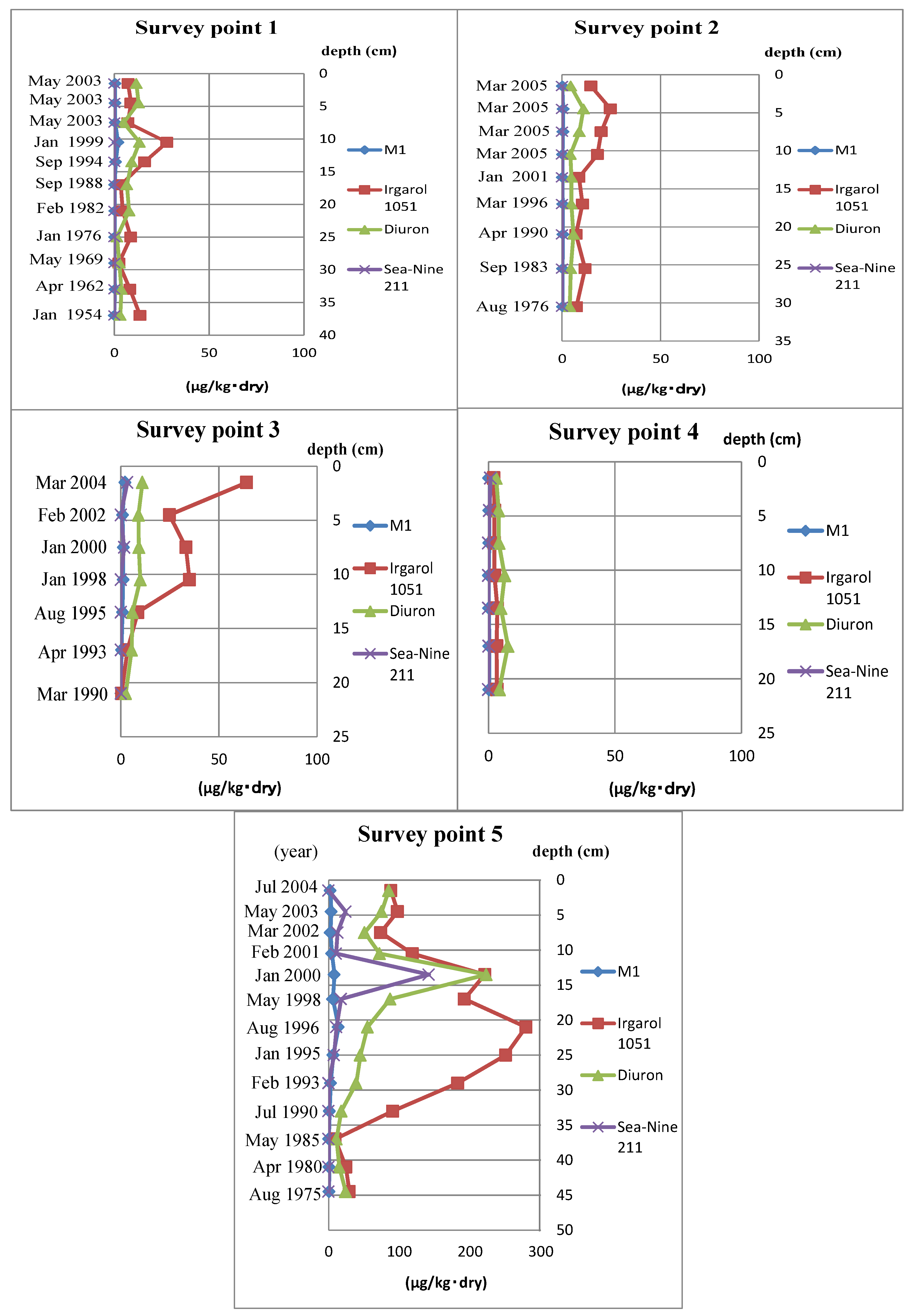
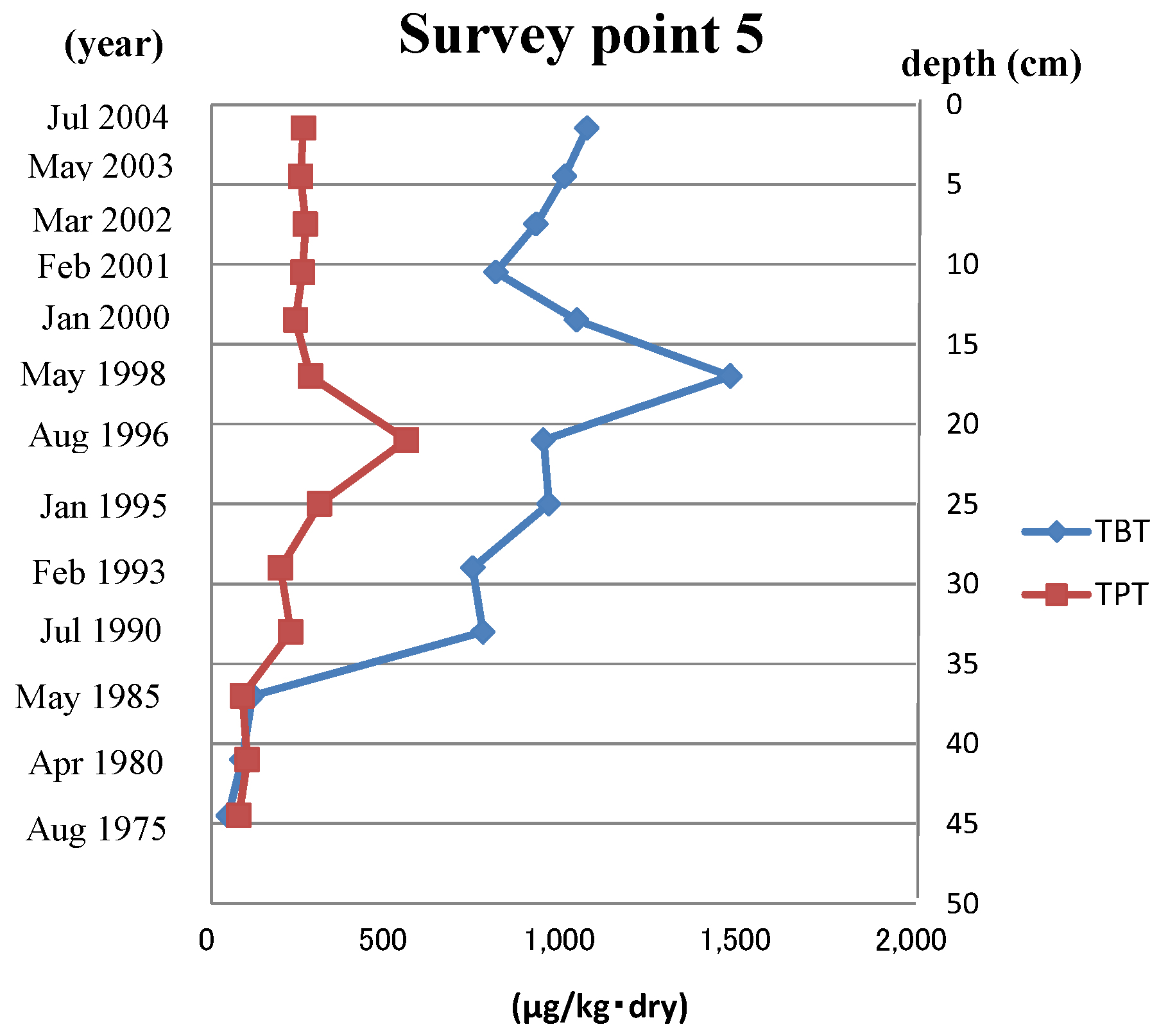
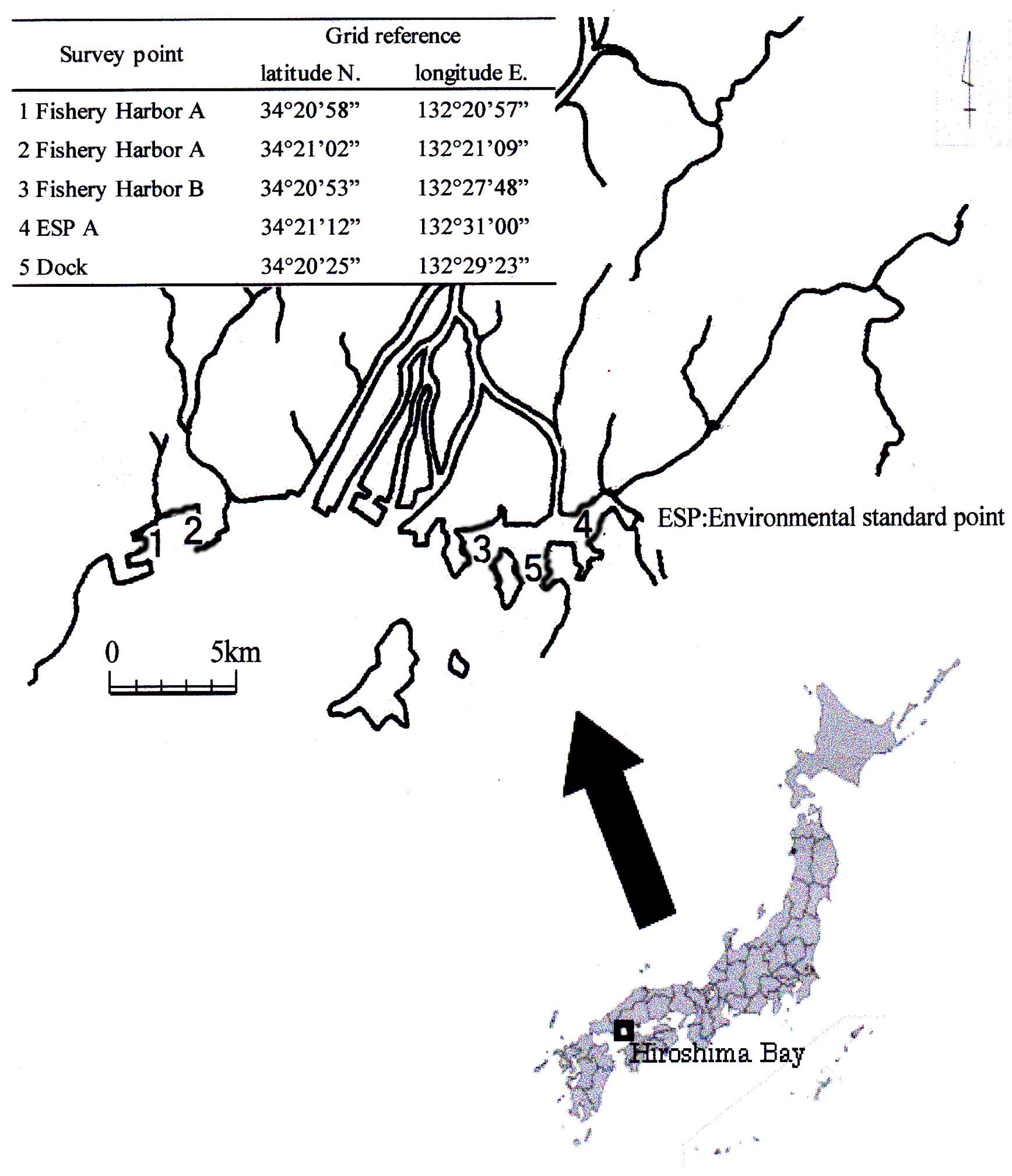
In this report, the Ot alternative antifoulants, Diuron, Sea-nine211 and Irgarol 1051, and the latter’s degradation product, M1, were investigated in five sediment core samples taken from the northern part of Hiroshima Bay. Ot compounds were also investigated for comparison with alternative antifoulants. In addition, metal (Pb, Cu, Zn, Fe and Mn) levels and chronology were measured to better understand what occurs after metals accumulate in the sediment core.
3. Discussion
In general, chemical compounds are often attached to the smaller particles. The grain distribution of the sediment core’s surface is shown in
Table 4.
Survey Points 1 and 2 were located in the same fishery harbor. Point 2 is nearer than Point 1 with respect to its entrance, so the specific surface area, below 67 µm (%), of Point 2 is lower than Point 1. However, their concentration level was almost the same. It was thought that many leisure boats anchored in the fishery harbor long term.
Though the specific surface area, below 67 µm (%), of Point 4 was relatively high, the concentration of Ot alternative antifoulants was much lower, due to the boats only passing through the survey point.
Survey Point 5 was mostly high in specific surface area and below 67 µm (%) in all samples. The Ot alternative antifoulants were thought to have come from the nearby dock, so it is believed that the accumulation speed was high.
Table 4.
Grain distribution of the sediment core’s surface.
Table 4.
Grain distribution of the sediment core’s surface.
| Survey Point | Specific Surface Area (cm2/cm3) | Median Particle Size (µm) | Arithmetic Mean Particle Size (µm) | Below 67 µm (%) |
|---|
| 1 | 33,926 | 10.1930 | 15.0119 | 97.5 |
| 2 | 24,890 | 14.4307 | 33.6879 | 86.0 |
| 3 | 33,374 | 10.1875 | 19.3025 | 93.4 |
| 4 | 37,229 | 8.9690 | 15.4780 | 95.7 |
| 5 | 40,214 | 7.6898 | 12.1456 | 98.1 |
The concentration of antifouling biocides in sediment core samples from Hiroshima Bay was much higher than the corresponding sea floor sample concentrations [
25]. In particular, the survey Point 5 sample had the highest observed concentration of the alternative biocides.
As shown in
Figure 1 and
Figure 2, the concentration of Ot alternative antifoulants and Ot compounds at survey Point 5 began to increase from almost the same point. In the case of Ot alternative antifoulants, it was thought that the rise in the concentration was caused by the increase of the consumption after the regulation of Ot compounds in 1990. On the other hand, in the case of Ot compounds, the cause was mainly due to the way the dock replaced the Ot compound paints with the Ot alternative antifoulants. When the regulation became effective in Japan, the Ot compounds were stripped from ships before the Ot alternative antifoulants were painted on to the ships. In the case of this dock, the stripped pieces of Ot compound paint were not cleared from the dry dock, so when the ships were relaunched, the stripped paint chips on the ground were washed into the sea water. Obviously, this would have had an impact on the Ot compound concentration levels shown in
Figure 2.
The sediment core’s layers were thought to indicate the chronology of the biocide’s accumulation. At first, it was thought that the concentration of biocides would be higher in the layers closest to the surface, with a gradual decrease in the deeper layers. In fact, the results indicated differently, as the highest concentration levels were detected at deeper points. Metals in the sediment, especially Mn, Fe and Cu, are known to go through a process of dissolution as the sediment conditions cycle through oxidation and reduction, as new layers of sediment form [
27].
Metal concentrations in every core section were measured to elucidate the distribution in every layer and their relationship to the concentrations of biocides analyzed. In this study, only the concentrations of Cu, which were obtained at survey Points 3 and 5 showed signs of accumulation. However, it was thought that this accumulation was not as a result of oxidation and reduction, due to accumulation of Mn or Fe not being detected. Both Mn and Fe generally react more to oxidation and reduction than Cu, making it unlikely that oxidation and reduction were the cause of accumulation. Furthermore, the increase of Zn in the same layer with a similar pattern to Cu support this theory. The more likely cause of Cu accumulation is that Cu was present in the paint fragments of the antifouling biocides. Therefore, from this research, it is inconclusive whether or not alternative antifouling biocides are affected by the change in conditions due to oxidation and reduction.
According to Jacobson
et al. [
3], the half-life of Sea-Nine 211 and TBT in sea water is one hour and six to nine months, respectively. It was thought that Sea-Nine 211 did not accumulate in sediment. However, high concentrations were measured in sediment cores. Therefore, it is clear that the binding to sediment protects molecules from breakdown.
Furthermore, according to Tsunemasa
et al. [
28,
29], the toxicity (LC
50) of Sea-Nine 211 and TBT on oyster embryos is 28 and 16 µg/L, respectively. The highest value of Sea-Nine 211 and TBT in the survey Point 5 sediment core is five and 87 times more compared with the toxicity values on oyster embryos, respectively. If these compounds dissolve in sea water, it is clear that they have an effect on the marine organisms (for example, oyster and sea urchin embryos).
As the consumption of these biocides increases, it will have a negative impact through the retardation of the development of eggs and embryos, leading to an increased mortality rate. Therefore, it is thought that the antifouling biocide resolution speed in sea water and sediment should be improved.
5. Conclusions
1. Irgarol 1051 clearly had the highest concentration measured in four of the five survey points, with survey Point 4 being the exception.
2. Higher concentrations of antifouling biocides in the sediment core samples than the surface sediment samples indicated that this is where accumulation occurs.
3. Oxidation and reduction were not the causes of the high concentration levels of Cu in survey Points 3 and 5, due to the accumulation of Mn or Fe not being detected.
4. The 1990 regulation led to higher concentrations of Ot alternative antifoulants and Ot compounds in survey Point 5. The former was due to increased consumption; the latter due to paint chips containing Ot compounds that were stripped being washed into the sea.
5. After metals have accumulated in the sediment core, they tend not to shift.