1. Introduction
B cell-activating factor (BAFF) also known as B lymphocyte stimulator (BLyS) is a member of the tumor necrosis factor (TNF) ligand super-family of cytokines [
1], and is expressed and secreted by monocytes, DCs, neutrophils, basophils, stromal cells, activated T cells, epithelial cells, and malignant B cells [
2]. BAFF binds B cells via three receptors: BAFF-R, TACI (transmembrane activator and calcium modulator ligand interactor) and BCMA (B cell maturation antigen). Binding stimulates B cell proliferation and promotes their survival [
3,
4] through activation of the alternative NF-κB pathway [
5,
6]. Over-expression of BAFF results in peripheral B cell numbers increasing significantly and may be involved in autoimmune disease and B cell malignancies [
7].
Clinical studies have demonstrated that BAFF plays crucial roles in B-cell lymphoma [
8]. First, BAFF levels both in Hodgkin’s lymphoma (HL) and non-Hodgkin’s lymphoma (NHL) patients are significantly elevated compared with those of healthy controls [
9,
10]. Serum BAFF levels of follicular non-Hodgkin’s lymphatic tumors (follicular lymphoma, FL) are 3–4 times higher than normal levels. Furthermore, BAFF and its receptors are highly expressed in reactive lymphoid tissues and B-cell lymphomas, including diffuse large B-cell lymphoma (DLBCL), primary central nervous system lymphoma (PCNSL), mantle cell lymphoma (MCL), marginal zone B-cell lymphoma (MZBCL) and plasma cell myeloma (PCM). Second, the expression of BAFF is correlated with Ann Arbor stage [
11]. BAFF mRNA levels in early B-NHL patients are significantly lower than those in patients with advanced B-NHL. Lastly, BAFF levels correlate with disease severity, response to therapy, and prediction of clinical outcome. Serum BAFF levels are correlated with responses to initial therapy, progression-free survival (PFS), and overall survival (OS). A high level of BAFF is associated with several poor prognostic characteristics. Patients with a high serum BAFF level at the time of diagnosis had a significantly worse median OS compared to those with a low serum BAFF level, and patients with high serum BAFF also had a lower CR rate than those with low BAFF levels and a shorter median PFS.
Currently, some strategies are designed to eliminate B cells through the use of anti-BAFF antibodies [
12] when elevated levels of BAFF are found in patients with autoimmune disease or B cell malignancies. However, the majority of approved antibodies use full-length IgG, which comes with certain limitations, including the immunogenicity of murine molecules [
13], inability to reach the cryptic site desired for efficacy [
14], distribution to normal organs, poor penetration of solid tumors, and difficulty of expression and purification. Along with progress in genetic engineering techniques, there have been major efforts to obtain smaller antibodies to cope with these shortcomings. Here, we describe a single variable domain of heavy-chain antibody only IgG from camels (HCAbs) [
15].
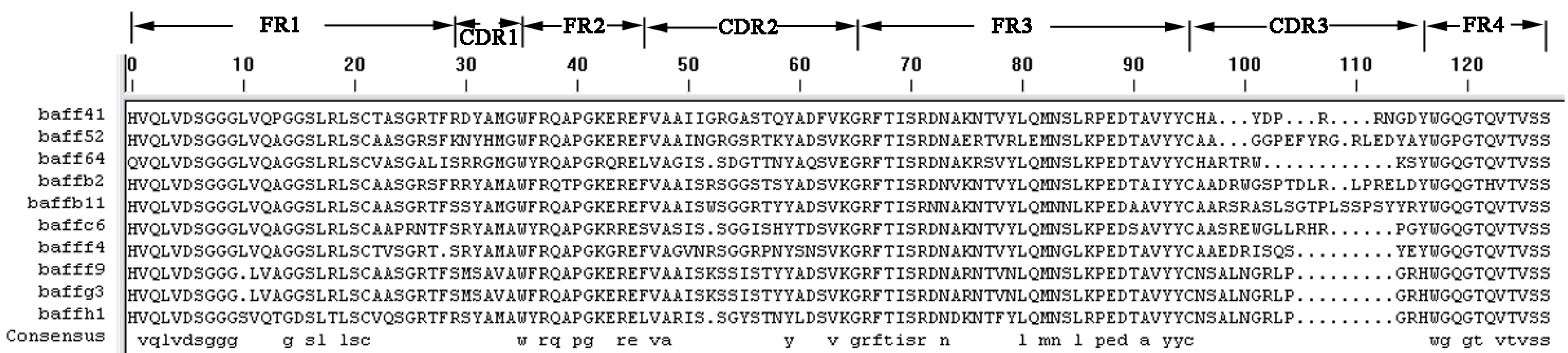
Heavy chain antibodies from camelid species lacking light chains and CH1 domain formed the basis for generating functional single-domain antibody fragments (sdAb) from their variable domains (VHH). The molecular weight of VHH is 15 kDa, much smaller than other recombinant antibody formats (60 kDa), hence it has also been called a nanobody. The smaller size results in lower immunologic response and better pharmacokinetics. VHH not only has a smaller size but also superior properties. There are only three complementarity-determining regions (CDRs) of VHH [
16,
17], and they are somewhat unique: the CDR1 and CDR2 loops are canonical in structure, and an unusually long surface CDR3 fold enables VHH to recognize alternative antigen epitopes and enhances solubility [
18,
19]. VHH is highly stable in extreme pH conditions, can bind to their target at high concentrations of chaotropic agents and is easily produced in large quantities [
20]. Because of those favorable properties, VHHs have been given much attention by researchers and have been used in settings ranging from scientific research to clinical detection and treatment. In an early study, researchers derived VHH agents from non-immune libraries, but immune libraries were subsequently found to lead more directly to VHHs with higher affinities [
21].
The aim of the current study was to obtain single-domain anti-BAFF camel antibodies from an immune library to improve clinical efficacy of B cell malignancies. Here, we detail the construction of an immune library as well as panning, expression and characterization of functional sdAbs with high specificities and sensitivities to BAFF.
3. Discussion
The purpose of this study was to generate anti-BAFF sdAbs and to determine their biological effects. To the best of our knowledge, this is the first report of the effects of anti-BAFF sdAbs with B-cell lymphoma cells. In the present study, we have successfully constructed the immunized alpaca sdAb library and isolated three anti-BAFF sdAbs (anti-BAFF64, anti-BAFF52 and anti-BAFFG3) from the phage library. Tests of their biological effects showed that the three sdAbs were high affinity and inhibited tumor cell proliferation in vitro, providing a promising opportunity for the development of new B-cell lymphoma therapeutics.
First, we constructed the immunized alpaca VHH library. The choice of phage display strategy depends on the required affinity and the target application of the desired antibody. In order to isolate sdAbs with high affinities for BAFF, with possible therapeutic applications in B-cell lymphoma, we chose to construct an immunized alpaca VHH library to produce anti-BAFF sdAbs. An immune phage antibody library has many advantages. First, the immune phage antibody library contained a large number of antigen-specific antibodies, some of which will also have been affinity matured by the immune system [
23,
24]. High-affinity antigen binding domains can be isolated by screening relatively few clones from immune libraries; Second, the immune response removes irrelevant antibodies, thus improving library quality before cloning starts. Animals can be made tolerant for certain irrelevant antigens, after which a mixture of the relevant and irrelevant antigens is used for immunization [
25]; Third, VHH libraries from immunized alpaca retain full functional diversity. The young alpaca was immunized with BAFF four times over more than two months, and the titer was tested immediately after the serum was obtained. The antibody titer of the immune serum against BAFF was a minimum of 1.992 after the fourth injection, as opposed to the normal serum’s titer of 0.1. Alpaca possesses two types of antibodies, conventional antibodies and a heavy chain IgGs (HCAbs), which account for 50% of alpaca sera. The heavy chain IgGs include two types, IgG2 and IgG3. IgG2, a homodimer of 46 kDa chains with an extended hinge could substitute for the CH1 domain, only binds to protein A. In contrast, IgG3, which consists of two chains of 43 kDa, binds to protein A and G after reduction. The antigen-specific heavy chain antibodies can therefore be determined by the presence of proteins A and G.
VHH genes were cloned from alpaca lymphocytes by nested PCR. VHH cloning was repeated several times to increase the number of
VHH genes in the library. Obviously, a larger the number of different VHH clones that bind BAFF antigen will increase the likelihood of finding a VHH with the specific properties that are most important to us. More than 11 pairs of different primers were selected to amplify
VHH genes in the first round of PCR, so that the different degenerate primers might amplify different antibody heavy chain genes, further increasing the diversity of the VHH library. Amplified
VHH genes were purified, digested, and ligated into phage vector pHEN-6. The recombinant plasmids were introduced and electroporated into
Escherichia coli TG1. Phage was precipitated with polyethylene glycol (PEG)/NaCl (20% PEG6000–2.5 M NaCl), and the phage titer was calculated. The resultant immunized alpaca VHH library contained 3 × 10
9 members.
Secondly, the single-chain antibody recognizing BAFF was selected from the immune phage library by panning. Equally important to immune phage library construction is the design of an appropriate selection strategy. Effective panning and gradual decreasing of the concentration of BAFF antigen resulted in the isolation of the most specific VHHs during each panning. Streptavidin-coated microbeads were used to capture the biotinylated antigen. Due to the high binding affinity of streptavidin to biotin, the interaction between the phage particle and antigen takes place in the solution, which permits precise control of the antigen concentration and exposure time to the immune phage library [
26,
27]. BAFF antigen was gradually reduced from 50 to 25 µg/mL to allow preferential selection of higher affinity mutants. Input/output ratio and affinity assays verified that the bio-panning and the enrichment of phages binding to BAFF were effective. To prepare crude phage to examine by monoclonal phage ELISA, 188 clones from either second or third round were chosen at random. More than half of the clones selected from the second and third round displayed positive results. Of these, 21 positive clones were collected randomly for DNA sequencing, which showed that the clones fell into different families according to sequence. Three different clones (clones 64, 52 and G3) were selected for expression of soluble periplasmic VHH and were transformed into
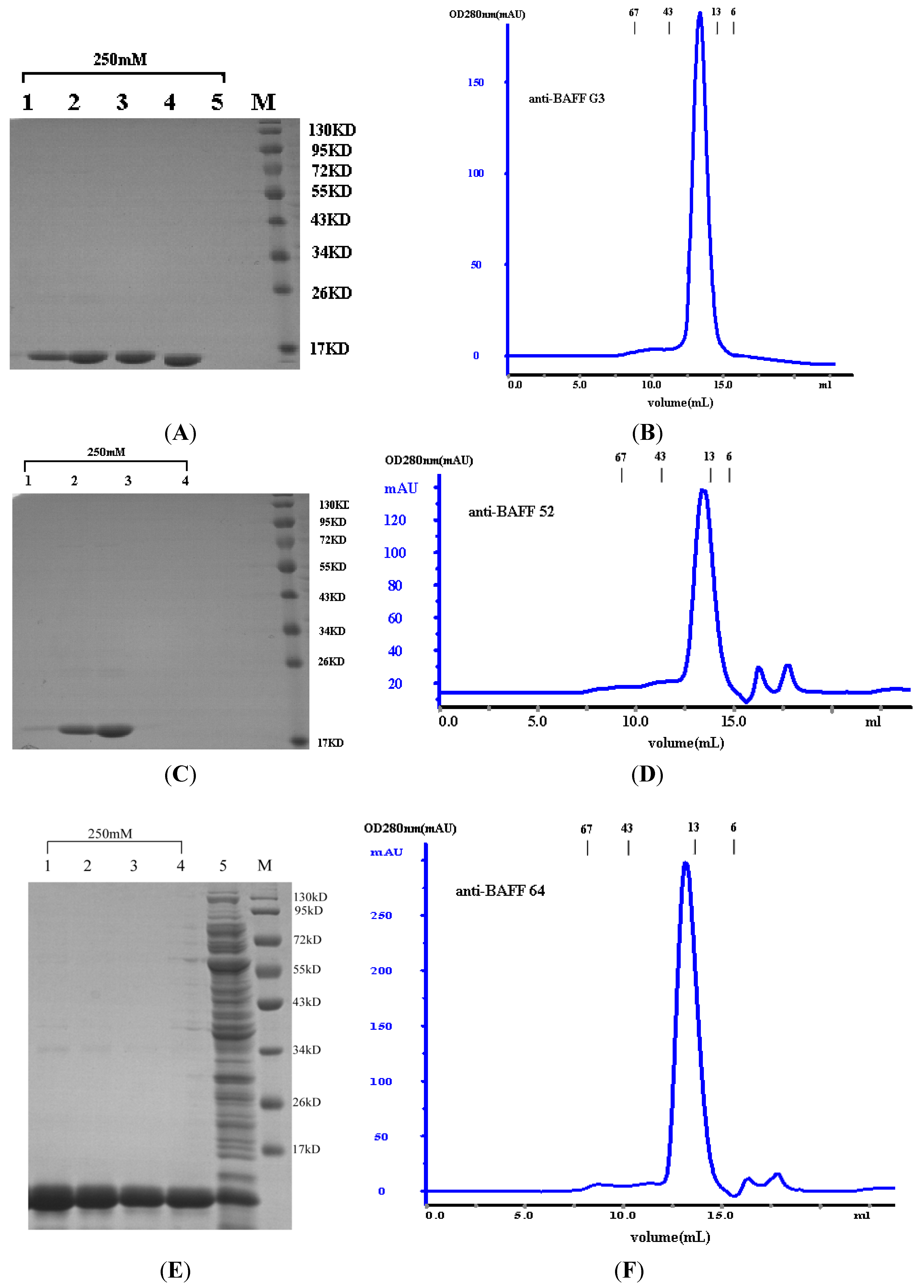
Escherichia coli TG1. The products of sdAbs (anti-BAFF64, anti-BAFF52 and anti-BAFFG3) were shown to be highly pure and homogeneous by SDS-PAGE (more than 95% pure, as estimated by absorbance scanning), where the purified sdAbs appeared as a single band of 15 kDa.
The three selected sdAbs, anti-BAFF64, anti-BAFF52 and anti-BAFFG3, possess unique characteristics in terms of their different CDR structures. The three sdAbs show more than 80% sequence identity with human VH domain; the differences in amino-acid sequence between the three alpaca VHH and human VH are localized in the CDR regions. As we all know, CDR provides complementarity of paratope and epitope surfaces, and this determines the antigen-recognizing capacity of apical regions of the variable domains, defining the antibody–antigen interaction. SdAbs consist of three distinct hyper-variable regions and CDRs. A long CDR3 results in new antigen binding modes, such as binding the active site of enzymes, and also covers the hydrophobic interface that would be formed with the variable light chain domain [
28,
29]. These appear to enhance both the stability and relative affinity of sdAbs. The three sdAbs recognize close but distinct epitopes on BAFF and bind with high affinity. The relative affinity constant for the three sdAbs against BAFF were calculated using Bio-Layer Interferometry. The range of mean Kaff values for the three sdAbs was 10
−6 to 10
−7 M; anti-BAFF64 has a higher affinity than the other two sdAbs.
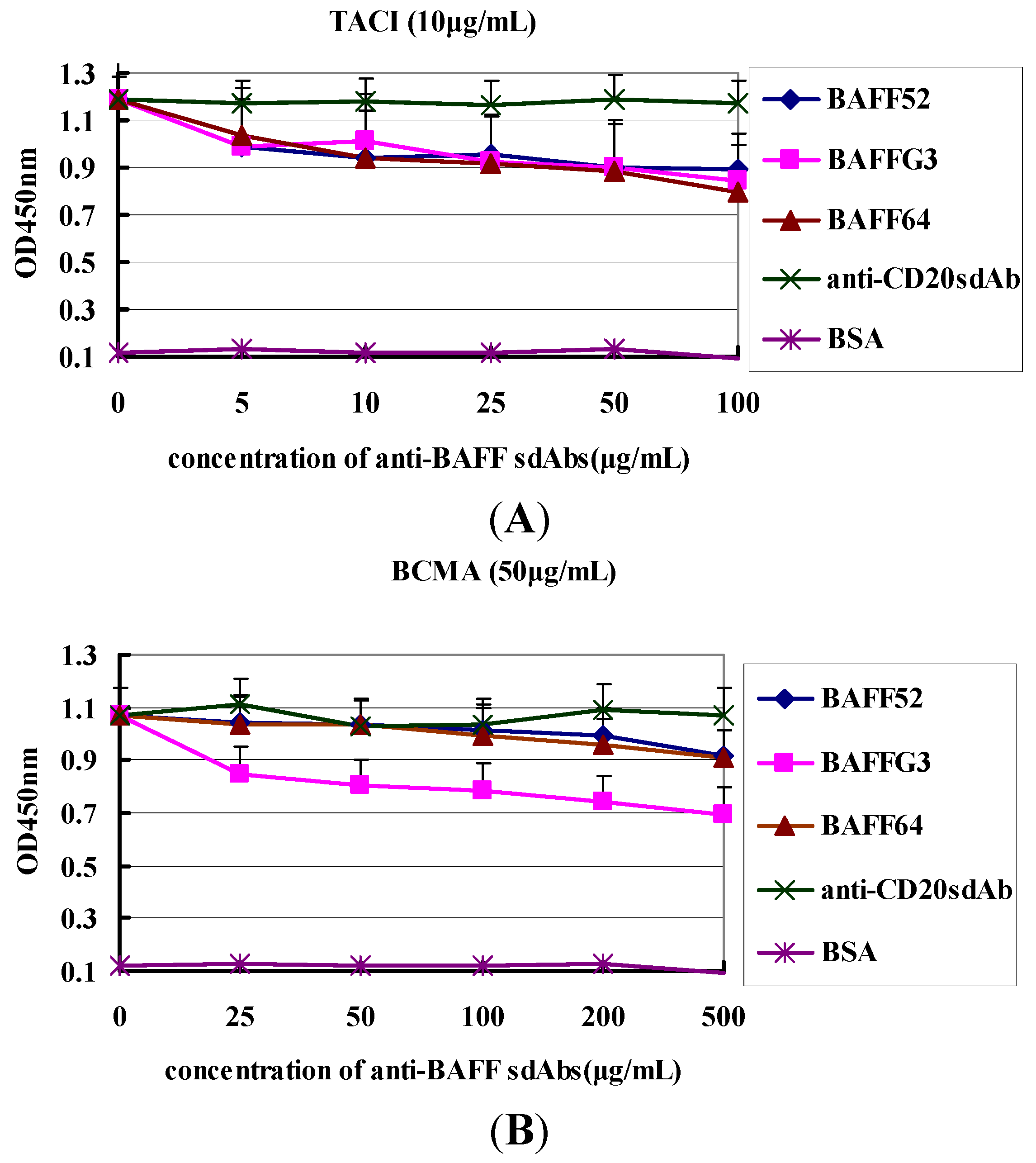
Importantly, the functionality of VHH was demonstrated. They can recognize BAFF molecules and were able to disturb binding between BAFF and its receptors. Anti-BAFF64 inhibited the binding of TACI to BAFF, while anti-BAFFG3 could block BMCA binding. The ratios of inhibition were 31% and 33%, respectively. TACI and BCMA are expressed by B cells at various times during their ontogeny. It has been reported that most autoantibody-producing plasma cells derive from extrafollicular foci and that their survival is largely dependent on TACI and BCMA [
30]. Anti-BAFF64 could significantly prevent the combination of BAFF and TACI, which makes it a good candidate for use in bi-specific formats for therapeutic use.
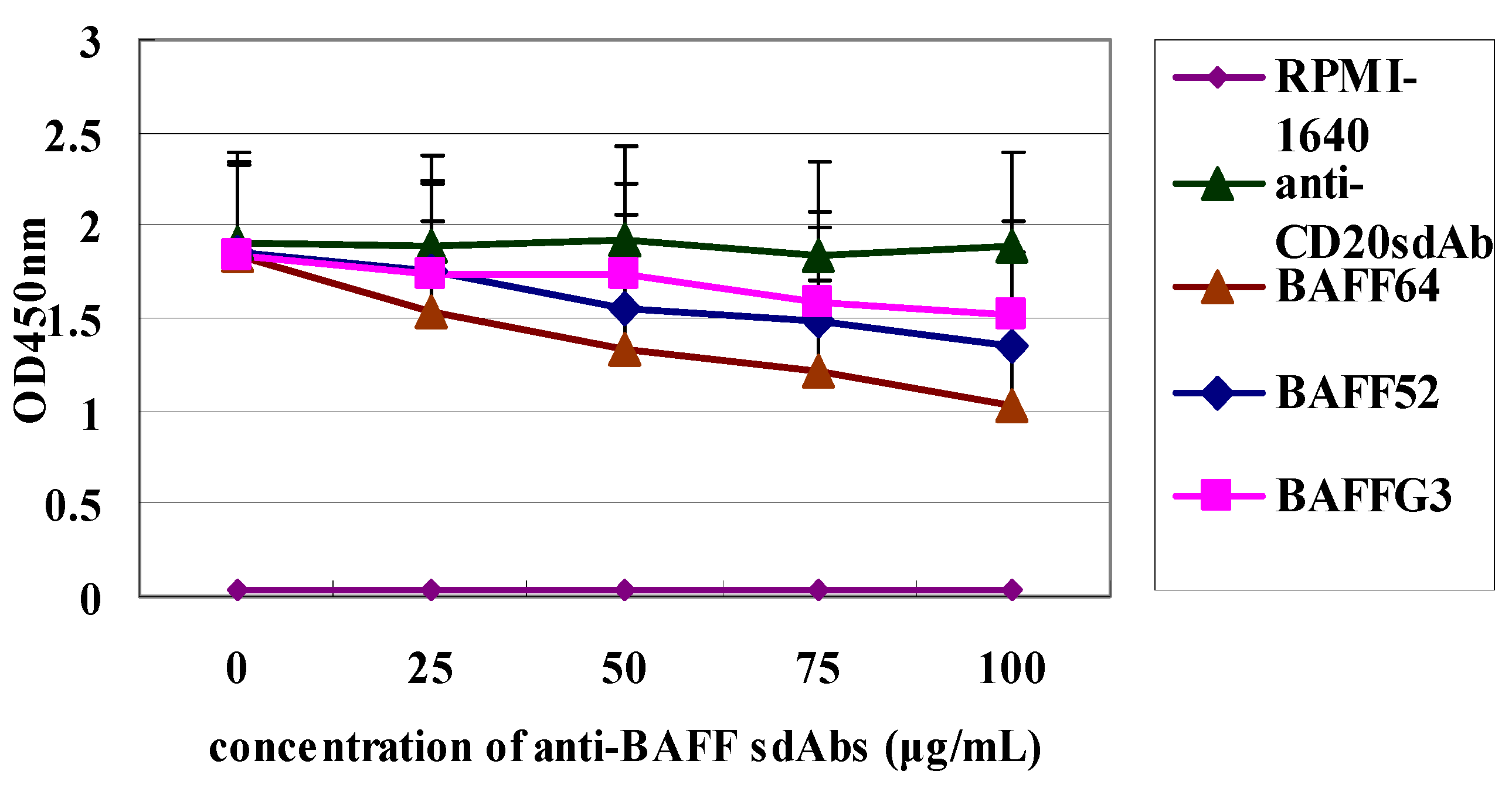
Finally, differences in the degree of inhibition by the three sdAbs may be due to the differences in their affinity for BAFF. Our functional assay showed that the three sdAbs could, in a dose dependent manner, inhibit BAFF-induced B-cell lymphoma cell proliferation and anti-BAFF64 sdAb, which had a higher affinity and was the most potent in inhibiting tumor cell proliferation. Anti-BAFF64 presents a potential therapeutic approach for growth inhibition of tumor cells and must bind at a site critical to BAFF function, as it had the greatest inhibitory effect in vitro. In fact, because the exact nature of the three sdAbs is not fully understood, determination of their in vivo activity is of interest.
4. Experimental Section
4.1. Alpaca Immunization
A young female alpaca was intramuscularly inoculated with BAFF (0.2 mg/0.2 mL), and booster treatments were administered 3, 6 and 9 weeks later. For the first injection, the antigen was mixed with an equal volume of complete Freund’s adjuvant (Sigma-Aldrich, St. Louis, MO, USA), and all subsequent boosts were with incomplete Freund’s adjuvant (Sigma-Aldrich). Pre-immune sera were obtained from the jugular vein before boosts and immune sera were taken after boosts one to two weeks to test the titer. One week after the last injection, 50 mL of immune sera was collected for total RNA purification.
4.2. Measurement of Antibody Titers
The titer of anti-BAFF antibody was measured by ELISA. First, 96-well plates were coated with BAFF (10 µg/mL) and incubated overnight at 4 °C. Wells were then blocked with 2% skimmed milk powder in phosphate-buffered saline (MPBS), and incubated at 37 °C for 2 h. Sera were diluted with 2% MPBS and added 100 μL/well, and incubated at 37 °C for 1 h. Then the wells were added horseradish peroxidase (HRP)-labeled goat anti-alpaca antibodies after three washes with PBS containing 0.05% Tween-20 (PBST, pH 7.4), and incubated at 37 °C for 1 h. The wells were added substrate 3,3',5,5'-Tetramethylbenzidine (TMB) after three washes with PBST, and incubated at room temperature for 10 min. The absorbance of each sample was read at 450 nm with an ELISA reader.
4.3. Antigen Biotinylation
Antigen was biotinylated with EZ-Link Sulfo–NHS–Biotin (sulfosuccinimidobiotin, Thermo Fischer Scientific, Waltham, MA, USA) dissolved in dimethylsulfoxide (1.0 g/L). The antigen was reacted with a 10:1 molar excess of NHS–LC–Biotin buffer (Succinimidyl 6-(biotinamido)-hexanoate, Thermo Fischer Scientific, Waltham, MA, USA). After 30 min at room temperature, the biotinylated antigen was dialyzed against PBS overnight.
4.4. Construction of Immunized Alpaca sdAb Library
White blood cells were isolated from alpaca blood with anticoagulant using the recommended protocol and total RNA was extracted from leukocytes using the QIAamp RNA Blood Mini™ Kit (QIAGEN, Hilden, Germany). The concentration of RNA was calculated from A260 measurements. cDNA was synthesized from 1.0 µg of total RNA using a First-Strand cDNA Synthesis Kit (Invitrogen, Carlsbad, CA, USA). The amplification of VHH was performed using nested PCR. In the first PCR, the amplified region ranged from the VHH domain to the CH2 domain. Sequences of the first-PCR primers are as follows: YTCh-1 5'-CGCCATCAAGGTACCAGTTGA-3'; YT1 BN 5'-GCCCAGCCGGCCATGGCCSMKGTRCAGCTGGTGGAKTCTGGGGGAG-3'. Amplified heavy domain antibodies (around 500 bp) were purified from agarose gels using the QIAquick Gel Extraction™ kit (QIAGEN), which was then subjected to the second round of PCR. The specific degenerate
VHH primers were used to amplify
VHH genes (5'-CATGTGTAGATTCCTGGCCGGCCTGGCCTGAGGAGACGGTGACCT GG-3' and 3'-CATGTGCATGGCCTAGACTCGCGGCCCAGCCGGCCATGGCC-5'). The amplified product incorporated a SfiI restriction site at the ends of
VHH genes.
VHH genes were purified with the QIAquick PCR Purification kit (QIAGEN) and were digested and ligated into phage vector pHEN-6 [
31], using a ratio of 800 ng of vector to 70 ng of VHH (which should provide the optimal vector-to-VHH ratio). The recombinant plasmids were introduced into
Escherichia coli strain TG1 by electroporation. A small amount of electroporated cells was diluted to determine the recombination rate and the remaining cells were incubated with 2YT/Amp at 37 °C. M13KO7 helper phage was added at a 20:1 ratio, allow for infection by incubation at 37 °C without shaking for 30 min. Phage was precipitated with PEG/NaCl and the phage titer was calculated.
4.5. Panning the Immunized Alpaca sdAb Library
Affinity selection for BAFF-binding recombinant phages was performed [
32]. First, 100 μL of M-280 streptavidin beads (Invitrogen) and 1 mL of 2% MPBS were added to a 1.5 mL microcentrifuge tube. This tube was then incubated with end-over-end rotation for 1 h at room temperature to block non-specific binding. The beads were drawn to one side of the tube with a magnetic tube holder (Dynal Biotech, Oslo, Norway) and the supernatant was discarded. Biotinylated BAFF antigen (500 nM) was mixed with the magnetic streptavidin beads and incubated with end-over-end rotation for a further 30 min at room temperature. Then, 5 × 10
11 t.u. phage were incubated with antigen-coated magnetic streptavidin beads for 2 h at room temperature. Meanwhile, 1–2 clones from a fresh
Escherichia coli plate were inoculated into 10 mL of 2YT, which was incubated at 37 °C and shaken at 250 rpm for 2~4 h until OD600 = 0.5. Unbound phage was discarded and the magnetic streptavidin beads were rinsed 5–10 times with 350 μL of PBST and then 5–10 times with 350 μL of PBS. Bound phage was eluted with 200 μL of freshly diluted triethylamine (TEA) (14 μL triethylamine + 1 mL distilled water), and then, the TEA was neutralized with 400 μL of 1 M Tris–HCl. TG1 culture (2 mL, OD600 = 0.5) was infected with 600 μL of eluted phage while standing at 37 °C for 30 min. Serial dilutions of infected cells were used to titer phages and the remainder was used to amplify the phage for further selection or analysis. Phages were purified with PEG (20% PEG6000–2.5 M NaCl). Three rounds of selection were carried out and populations from the second and third rounds were tested for specificity to BAFF by phage ELISA.
4.6. Phage Enzyme-Linked Immunosorbent Assay (ELISA)
After the second and third rounds of screening of anti-BAFF sdAb clones by phage ELISA, 188 clones were chosen at random. Bacteria from the second and third rounds of selection were diluted and plated on an LB/ampicillin plate to produce an isolated colony. The single colony was chose and cultured in 96-well plates at 37 °C for 4 to 5 h with shaking at 100 rpm. M13K07 helper phage was added and incubated at 37 °C for 30 min without shaking. Next, 5 μL ampicillin (100 mg/mL) and 15 μL kanamycin (50 mg/mL) was mixed with 50 mL 2× yeast extract-tryptone broth (2YT; composition per liter of distilled water: Tryptone 16;Yeast extract; NaCl 5 g) and added to each well before incubating overnight at 37 °C. Supernatant was collected and added to an ELISA plate, and the standard ELISA process was followed using an HRP-anti-M13 anti-body (GE Healthcare, Munich, Germany). The plate was analyzed at 450 nm with an ELISA reader. Positive phage clones were analyzed by DNA sequencing.
4.7. Expression and Purification of Anti-BAFF VHHs
VHHs were digested and ligated into the phage expression vector pSJF2. The recombinant plasmids were introduced into TG1 by electroporation. Individual clones were inoculated into 10 mL 2YT (supplemented with ampicillin (100 mg/mL) and grown overnight at 37 °C while shaking at 250 rpm. Soluble proteins were purified by nickel affinity using Ni-NTA (QIAGEN) as recommended by the manufacturer).
The sdAbs were assessed by size exclusion chromatography using a Superdex
75 (GE Healthcare Bio-Sciences AB) column on an AKTA purifier 2000 system according to the manufacturer’s instructions. Size exclusion chromatography was carried out in 0.1 mol/L phosphate buffer at a flow rate of 0.8 mL/min. Molecular weight was determined using the Gel Filtration Calibration Kit HMW (high molecular weight, GE healthcare). Additionally, marker retention volumes were incorporated into the top of all chromatographs (
Figure 2B,D,F). Purified proteins were also assessed by SDS-PAGE, and the protein concentrations were determined by BCA (Pierce, Rockford, IL, USA).
4.8. Affinity Measurements
The relative affinity constant of each sdAb against BAFF was calculated by BLI. The BAFF antigen was biotinylated using NHS–LC–Biotin buffer at a 3:1 molar ratio of biotin to protein for 30 min at room temperature, followed by dialysis against PBS. Binding assays were performed by BLI using an Octet RED system (Pall Forte Bio Europe, Portsmouth, UK) and Fortebio Acquisition Software in 96-well microtiter plates at room temperature with orbital sensor agitation of 1000 rpm. Biotinylated BAFF (20 μg/mL) was load onto Streptavidin (SA) biosensors, a baseline was established in PBST buffer (PBS pH 7.4, 0.1% BSA, albumin fraction and 0.02% Tween-20, both Merck KGaA, Darmstadt, Germany) prior to association at varying analyte concentrations. Serial dilutions were made to produce 200 μL of 20, 10, 5, 2.5 and 1.25 µM sdAbs in appropriate wells of the assay plate. Antigen-coated sensors were placed in five different concentrations of sdAbs for 300 s, during which antigen was bound by antibodies on the sensor. Sensors were rinsed in kinetics buffer for 1500 s, which also served as the background buffer. Octet Analysis Software version 6.4 was used for automatic data processing. Biosensor data were fit using a 1:1 binding model.
4.9. ELISA Competition Assays
First, two 96-well plates were coated with 100 µL BAFF (10 µg/mL in 50 mM NaCO3 buffer, pH 9.5) and incubated overnight at 4 °C, and unspecific binding was blocked by incubation with 380 µL of 3% BSA per well at 37 °C for 2 h. Then each of the three individual sdAbs (anti-BAFF64, anti-BAFF52 and anti-BAFFG3) fused to 50 µL BCMA (50 µg/mL), at various dilutions (0, 25, 50, 100, 200, 500 µg/mL) with 3% BSA, were added in the wells of one plate for 2 h at 37 °C. And in the other plate, each of the sdAbs against BAFF (64, 52 and G3) with different concentration (0, 5, 10, 20, 50, 100 µg/mL) was incubate with 50 µL TACI (10 µg/mL) for 2 h at 37 °C, and the total volume of each solution was 100 µL. Anti-CD20 sdAb was the negative control. Wells were washed three times with PBST, after which 100 µL of the HRP-labeled goat anti-human IgG (Sigma-Aldrich) were added and incubated for 1 h at room temperature. The wells were added substrate TMB after three washes with PBST, and incubated at room temperature for 10 min. Optical density was read at 450 nm.
4.10. Raji Cells Proliferation Inhibition Assay
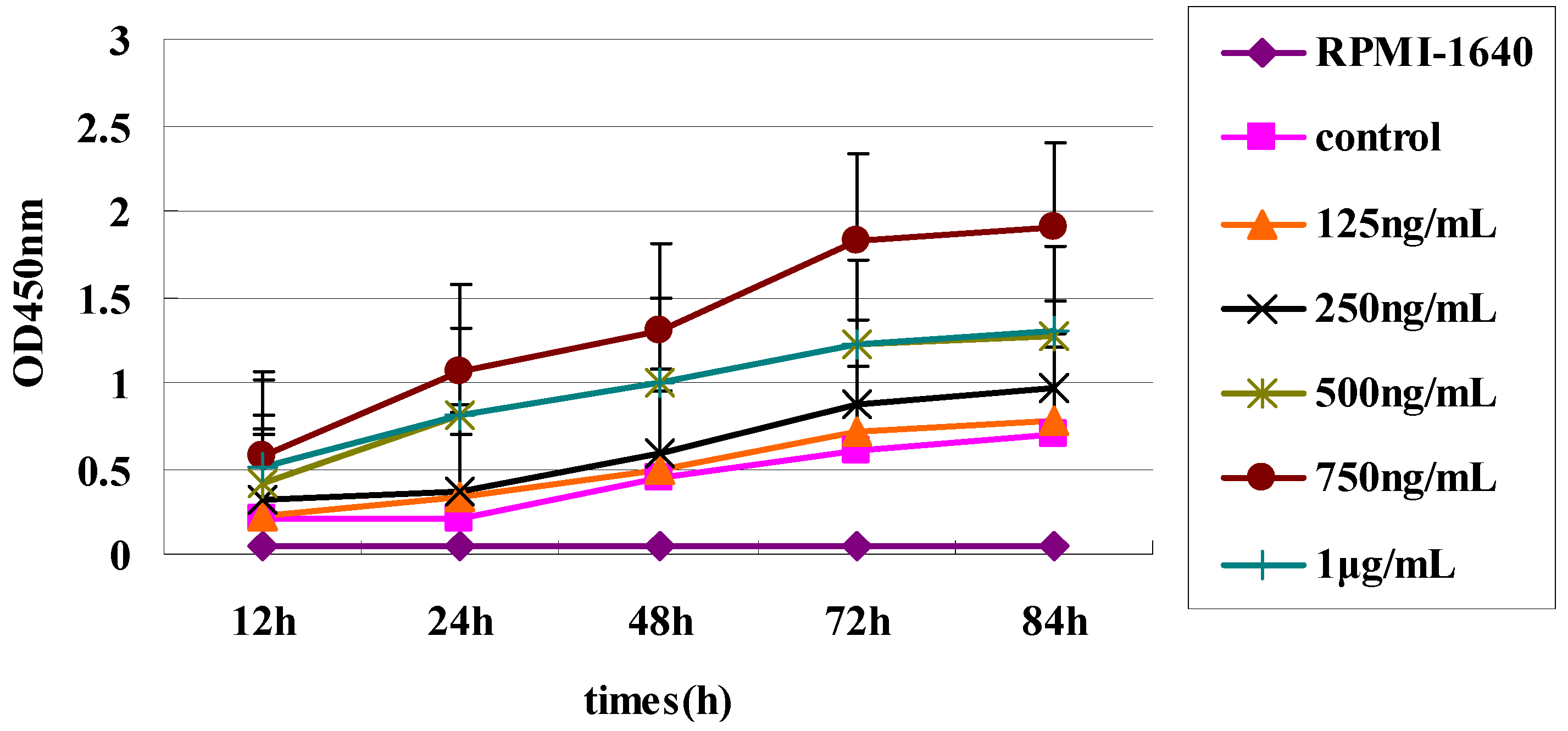
Raji Cells (American Type Culture Collection, Manassas, VA, USA) were adjusted to 106 cells/mL and cultured in 96-well flat-bottom plates with 100 µL RPMI-1640 (GibcoBRL, Gaithersburg, MD, USA) per well supplemented with 10% FBS (Sigma-Aldrich). BAFF (750 ng/mL final concentration) was added to each well, and the cells were incubated at 37 °C in a humidified atmosphere with 5% CO2. After 48 h, the three sdAbs were added to wells to achieve different concentrations (0, 25, 50, 75 and 100 µg/mL). Raji Cells were incubated for a total of 72 h and all assays were carried out in triplicate. Cell proliferation was measured with a Cell Counting Kit-8 (Dojindo, Kumamoto, Japan).