2.2. Inhibitory Effect of Genistein on the Osteoclastic Differentiation of RAW 264.7 Cells
Osteoclast cells are responsible for bone resorption in the bone remodeling process and are coupled tightly with osteoblasts in order to form a new bone matrix. Excessive bone resorption by osteoclasts can result in bone loss, causing diseases, such as osteoporosis and rheumatoid arthritis [
13]. In the present study, the inhibitory effects of genistein on osteoclast differentiation were investigated to establish their antiosteoporotic activity through suppressing excessive bone resorption by osteoclasts. Osteoclast differentiation from murine macrophage RAW 264.7 cells was induced by RANKL, which is essential for the terminal differentiation of monocytes/macrophages into osteoclasts [
15]. Genistein at 1–20 μm was not toxic to RAW 264.7 macrophage cells during a period of five days of differentiation (data not shown). TRAP-positive multinucleated osteoclasts were visualized by light microphotography (
Figure 2A). RANKL treatment induced osteoclast formation from RAW 264.7 pre-osteoclast cells and dramatically enhanced TRAP activity up to 297.9%, as shown in
Figure 2A,C. Treatment with genistein reduced the number of multinucleated TRAP-positive cells and TRAP activity in a dose-dependent manner (
Figure 2B,C). The inhibitory effects of natural flavonoids, such as luteolin [
24], baicalein [
25], resveratrol [
26], epigallocatechin-3-gallate [
27], scopoletin [
28], curcumin [
29] and fisetin [
30], as potent antioxidants on osteoclast differentiation from osteoclastic precursors through controlling ROS generation, have also been reported. The suppressive effect on osteoclastogenesis via attenuating ROS production in RANKL-treated RAW 264.7 cells was observed in luteolin, which showed potent cellular antioxidant activity in rat C6 astroglioma cells [
22,
24]. Scopoletin at 1–10 µM also alleviated general ROS and superoxide anions produced in RANKL-treated RAW 264.7 cells to inhibit osteoclastic differentiation [
28]. In addition, fisetin inhibited osteoclastogenesis through the prevention of RANKL-induced ROS production by nuclear transcription factor-erythroid 2-related factor 2 (Nrf2)-mediated upregulation of phase II antioxidant enzymes [
30]. Thus, this study’s data support the hypothesis that the suppressive effect of genistein on RANKL-induced osteoclast differentiation may be closely related with its intracellular antioxidant activity.
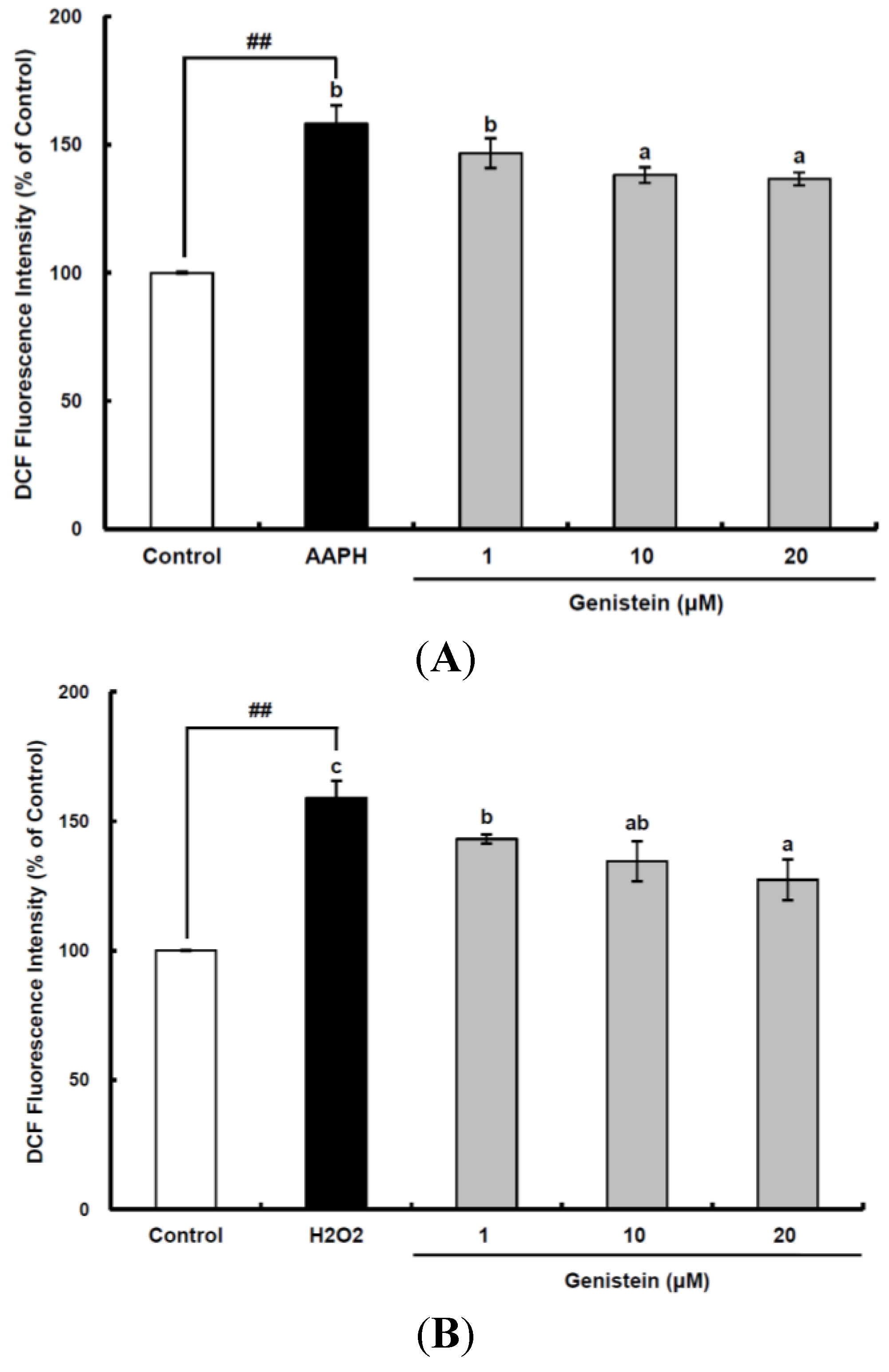
Figure 1.
Cellular antioxidant capacity of genistein against oxidative stress induced by 2,2'-azobis(2-amidinopropane) dihydrochloride (AAPH) (A) and H2O2 (B) in HepG2 cells. HepG2 cells were first cultured in 96-well plates (5 × 105/mL) with Dulbecco’s modified Eagle’s medium (DMEM) for 24 h. After the cells were incubated with different concentrations of sample dissolved in dimethylsulfoxide (DMSO) for 30 min, Hank’s balanced salt solution (HBSS), which is fluorescently stable, was then added to each well. After the cells were treated with 60 μM AAPH or 1 mM H2O2 for 30 min, 2',7'-dichlorodihydrofluorescein-diacetate (DCFH-DA) was added to the culture plates at a final concentration of 40 µM, and the cells were incubated for 30 min at 37 °C in the dark. Then, the cells were washed with HBSS and 2',7'-dichlorofluorescein (DCF) fluorescence intensity was measured at an excitation wavelength of 485 nm and an emission wavelength of 535 nm using a Tecan GENios fluorometric plate reader. Data are expressed as percentages of the value of untreated cells (mean ± standard deviation, n = 3). Different corresponding letters indicate significant differences at p < 0.05 by Duncan’s test. ## p < 0.01 vs. control.
Figure 1.
Cellular antioxidant capacity of genistein against oxidative stress induced by 2,2'-azobis(2-amidinopropane) dihydrochloride (AAPH) (A) and H2O2 (B) in HepG2 cells. HepG2 cells were first cultured in 96-well plates (5 × 105/mL) with Dulbecco’s modified Eagle’s medium (DMEM) for 24 h. After the cells were incubated with different concentrations of sample dissolved in dimethylsulfoxide (DMSO) for 30 min, Hank’s balanced salt solution (HBSS), which is fluorescently stable, was then added to each well. After the cells were treated with 60 μM AAPH or 1 mM H2O2 for 30 min, 2',7'-dichlorodihydrofluorescein-diacetate (DCFH-DA) was added to the culture plates at a final concentration of 40 µM, and the cells were incubated for 30 min at 37 °C in the dark. Then, the cells were washed with HBSS and 2',7'-dichlorofluorescein (DCF) fluorescence intensity was measured at an excitation wavelength of 485 nm and an emission wavelength of 535 nm using a Tecan GENios fluorometric plate reader. Data are expressed as percentages of the value of untreated cells (mean ± standard deviation, n = 3). Different corresponding letters indicate significant differences at p < 0.05 by Duncan’s test. ## p < 0.01 vs. control.
![Ijms 15 10605 g001]()
Figure 2.
Inhibitory effects of genistein on differentiation (A,B) and tartrate-resistant acid phosphatase (TRAP) activity (C) of osteoclastic RAW 264.7 cells. RAW 264.7 cells were exposed to the receptor activator of nuclear factor kappa-B ligand (RANKL) (50 ng/mL) for five days in the absence and the presence of genistein. After five days in culture, the cells were fixed and stained using a leukocyte acid phosphatase kit. TRAP-positive multinucleated osteoclasts were visualized under light microphotography (A); TRAP-positive multi-nucleated osteoclasts were counted (B). Data are expressed as the percentages of the value of cells treated with RANKL (means ± standard deviations (SD), n = 3); TRAP activity was measured at λ = 405 nm (C). Data are expressed as percentages of the values of untreated cells (means ± standard deviations, n = 3). Different corresponding letters indicate significant differences at p < 0.05 by Duncan’s test. ### p < 0.001 vs. C (C: control, which was not treated; TC: treated control, which was treated with RANKL).
Figure 2.
Inhibitory effects of genistein on differentiation (A,B) and tartrate-resistant acid phosphatase (TRAP) activity (C) of osteoclastic RAW 264.7 cells. RAW 264.7 cells were exposed to the receptor activator of nuclear factor kappa-B ligand (RANKL) (50 ng/mL) for five days in the absence and the presence of genistein. After five days in culture, the cells were fixed and stained using a leukocyte acid phosphatase kit. TRAP-positive multinucleated osteoclasts were visualized under light microphotography (A); TRAP-positive multi-nucleated osteoclasts were counted (B). Data are expressed as the percentages of the value of cells treated with RANKL (means ± standard deviations (SD), n = 3); TRAP activity was measured at λ = 405 nm (C). Data are expressed as percentages of the values of untreated cells (means ± standard deviations, n = 3). Different corresponding letters indicate significant differences at p < 0.05 by Duncan’s test. ### p < 0.001 vs. C (C: control, which was not treated; TC: treated control, which was treated with RANKL).
![Ijms 15 10605 g002]()
2.3. The Suppressive Effect of ROS Level by Genistein during the Osteoclastic Differentiation of RAW 264.7 Cells
Accumulating evidence indicates that ROS are not only dangerous byproducts of cellular metabolism, but also important factors of signaling pathways in various cellular functions, including regulation, apoptosis and differentiation [
31]. Previous studies have reported that the intracellular ROS level increases during RANKL-induced osteoclast differentiation, and this ROS generation can be attenuated by several antioxidants, including curcumin and fisetin [
29,
30]. Our previous study also showed that the intracellular ROS level in RANKL-stimulated Raw 264.7 cells increased to its largest value within two days and then declined [
28]. Thus, to assess the suppressive effects of genistein on the production of different ROS during two days of incubation, the cell-permeable, fluorescent probes, DCFH-DA and dihydroethidium (DHE), were used. After DCFH-DA is permeated into the cell membrane, it is going to be hydrolyzed by esterase to release DCFH. DCFH then is easily oxidized by different kinds of ROS, including the peroxyl radical, hydrogen peroxide and peroxynitrite, except superoxide anions, to become fluorescent DCF. Accordingly, the fluorescence due to DCF formation can provide information on the intracellular oxidative stress level caused by different ROS, but cannot differentiate the species of ROS. On the other hand, DHE specifically reacting with superoxide anions can demonstrate the amount of superoxide anions intracellularly generated. The general ROS level determined by DCFH-DA was significantly (
p < 0.01) increased by RANKL treatment to 130.6%, as compared to that of the control, and declined by genistein at 20 μm to 117.3% (
Figure 3A). The superoxide anion level was also significantly (
p < 0.001) augmented by 174.1% when compared to that in the control following RANKL stimulation and suppressed by genistein at 1–10 µM to 128.9% in a dose-dependent manner (
Figure 3B). It is known that glutathione (GSH) exists in a high concentration (mm) depending upon the types of cell and tissue in comparison with other antioxidants and is involved in the front line of cellular antioxidant defense system. Therefore, GSH as a redox status biomarker has a vital role in the antioxidant defense system in cells. The GSH level was analyzed using monochlorobimane (mBCL), a fluorescent probe that conjugates with GSH. As expected, RANKL treatment significantly (
p < 0.05) induced depletion in GSH level to 72.1%, as compared to that of the control (
Figure 3C). However, genistein at 1–20 µM significantly (
p < 0.05) enhanced the cellular GSH level depleted by RANKL treatment in a dose-dependent manner. These results imply that the suppressive effect of genistein on general ROS and superoxide anions level is likely to be potent, which is similar to its inhibitory effects on the RANKL-induced differentiation of RAW 264.7 cells into osteoclasts, indicating that genistein may suppress osteoclast differentiation from osteoclastic precursors through controlling ROS level.
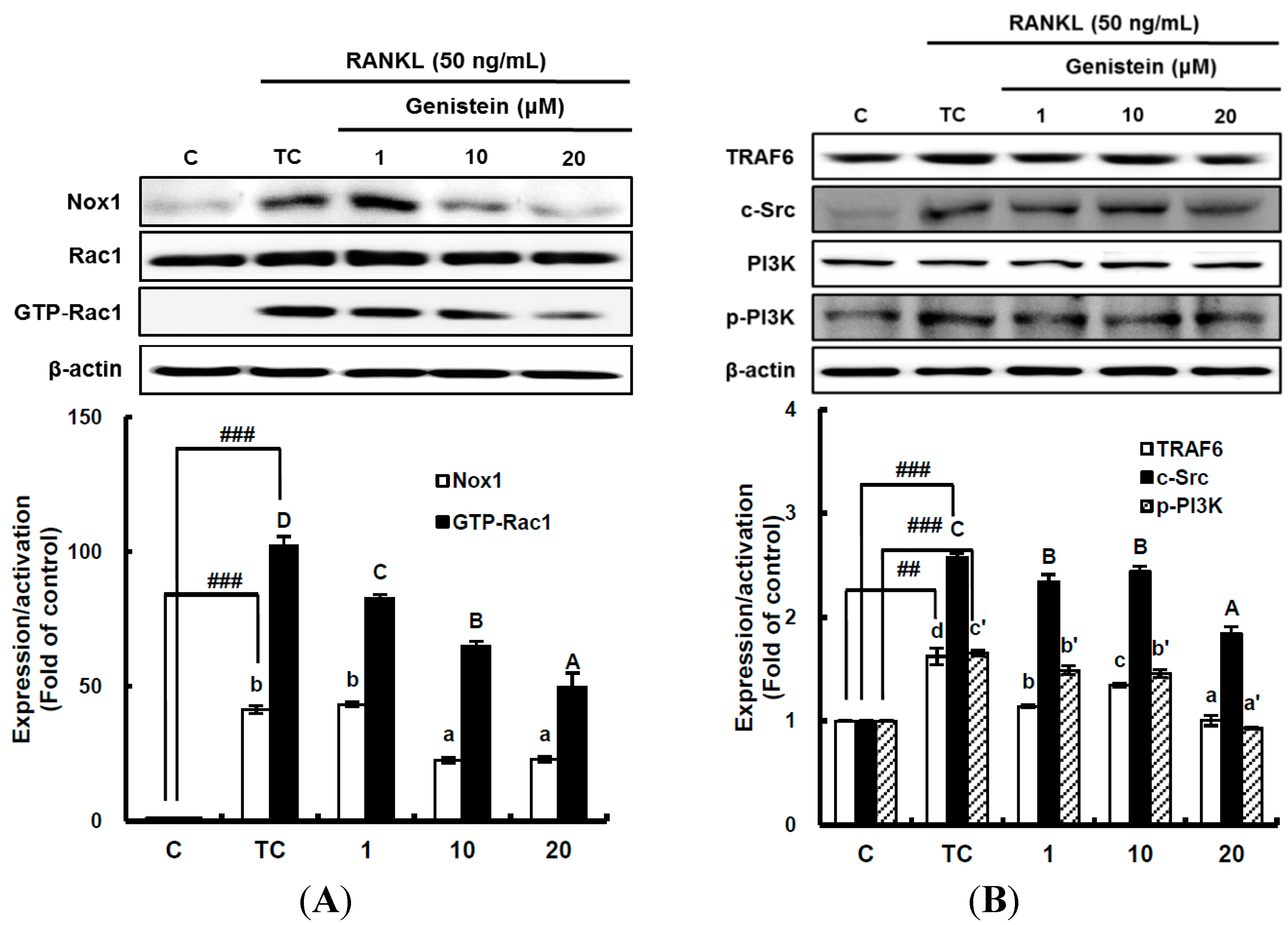
2.4. The Inhibitory Effect of Genistein on the Translation and Activation of Nox1 and the Disruption of the Mitochondrial Electron Transport Chain System
It has been suggested that ROS-mediated RANKL-induced differentiation of RAW 264.7 cells into osteoclasts occurs through the activation of Nox homologues and the electron transport chain in mitochondria [
4,
8]. This means that superoxide anions are first produced by Nox and the mitochondrial electron transport chain system as ROS during osteoclast differentiation. Thus, to determine whether genistein suppresses superoxide anion production by Nox1, the translation and activation level of Nox1 was analyzed by western blotting. The translation level of Nox1, known as a major contributing factor in superoxide anion generation, was significantly (
p < 0.01) stimulated by 50 ng/mL RANKL treatment, but it was significantly (
p < 0.05) attenuated by genistein at 1–10 μm in a dose-dependent manner (
Figure 4A). The small guanosine triphosphatase (GTPase) Rac1 existing as a cytosolic component of Nox1 in macrophages is essential for the activation of Nox1. Therefore, to examine the effect of genistein on Nox1 activation, the guanosine triphosphate (GTP) bound Rac1 (GTP-Rac1) and Rac1 levels were determined [
6]. As seen in
Figure 4A, Rac1 was not changed, but GTP-Rac1 significantly (
p < 0.001) augmented by the RANKL treatment was significantly (
p < 0.05) reduced by genistein at 1–20 μm in a dose-dependent manner, suggesting that genistein ameliorates Nox1 activation via controlling the GTP-Rac1 level. In addition, upstream signals, such as tumor necrosis factor receptor-associated factor (TRAF 6), tyrosine kinase c-Src, and phosphatidylinositol 3-kinase (PI3K), which are upregulated by RANKL and required for Nox1 activation, were analyzed. As shown in
Figure 4B, TRAF6 expression significantly (
p < 0.01) induced by RANKL treatment was ameliorated by genistein at 10–20 μm. c-Src expression significantly (
p < 0.001) augmented by RANKL treatment was also markedly attenuated by genistein at 10–20 μm in a dose-dependent manner when compared to the treated control, and PI3k activation significantly (
p < 0.001) induced by RANKL treatment was dose-dependently downregulated by genistein at 10–20 μm. These data indicate that genistein controls ROS generation by attenuating Nox1 translation, as well as Nox1 activation via the TRAF6/cSrc/PI3k signaling pathway in RANKL-mediated osteoclast differentiation from RAW 264.7 cells.
Figure 3.
The suppressive effects of genistein on general ROS (A), superoxide anions (B) and GSH levels (C) during the receptor activator of nuclear factor kappa-B ligand (RANKL)-induced osteoclast differentiation. RAW 264.7 cells were seeded in 12-well plates (3 × 104 cells/well) containing DMEM medium plus 10% FBS and incubated for 24 h. The medium was then replaced with a differentiation medium containing 50 ng/mL RANKL, and genistein at 1–10 µM was tested. After two days of incubation, the wells were gently washed twice with PBS. HBSS, which is fluorescently stable, was then added to each well instead of normal medium. DCFH-DA, dihydroethidium (DHE) or monochlorobimane (mBCl) was added to the culture plates at a final concentration of 40, 50 or 50 μM, respectively, and incubated for 30 min at 37 °C in the dark. After the cells were washed twice with HBSS, the fluorescence intensity of 2',7'-dichlorofluorescein (DCF), DHE and mBCl were measured using a Tecan GENios fluorometric plate reader. Data are expressed as the percentages of the value of untreated cells (means ± standard deviations, n = 3). Different corresponding letters indicate significant differences. Different corresponding letters indicate significant differences at p < 0.05 by Duncan’s test. # p < 0.05, ## p < 0.01, ### p < 0.001 vs. C (C: control, which was not treated; TC: treated control, which was treated with RANKL).
Figure 3.
The suppressive effects of genistein on general ROS (A), superoxide anions (B) and GSH levels (C) during the receptor activator of nuclear factor kappa-B ligand (RANKL)-induced osteoclast differentiation. RAW 264.7 cells were seeded in 12-well plates (3 × 104 cells/well) containing DMEM medium plus 10% FBS and incubated for 24 h. The medium was then replaced with a differentiation medium containing 50 ng/mL RANKL, and genistein at 1–10 µM was tested. After two days of incubation, the wells were gently washed twice with PBS. HBSS, which is fluorescently stable, was then added to each well instead of normal medium. DCFH-DA, dihydroethidium (DHE) or monochlorobimane (mBCl) was added to the culture plates at a final concentration of 40, 50 or 50 μM, respectively, and incubated for 30 min at 37 °C in the dark. After the cells were washed twice with HBSS, the fluorescence intensity of 2',7'-dichlorofluorescein (DCF), DHE and mBCl were measured using a Tecan GENios fluorometric plate reader. Data are expressed as the percentages of the value of untreated cells (means ± standard deviations, n = 3). Different corresponding letters indicate significant differences. Different corresponding letters indicate significant differences at p < 0.05 by Duncan’s test. # p < 0.05, ## p < 0.01, ### p < 0.001 vs. C (C: control, which was not treated; TC: treated control, which was treated with RANKL).
![Ijms 15 10605 g003]()
Figure 4.
The effect of genistein on the translation and activation of NADPH oxidase (Nox1) (A) and its upstream signals (B). RAW 264.7 cells were exposed to the receptor activator of nuclear factor kappa-B ligand (RANKL) (50 ng/mL) for two days in the absence and presence of genistein. GTP-Rac1 was recovered by centrifugation from proteins complexed to the beads, which have a glutathione S-transferase-p21-binding domain: human Pak1 as 67–150 and binds to GTP-bound Rac1 of the lysates. Western blotting analysis was performed on the lysates of cells that had been incubated with 1–20 μm genistein. Different corresponding letters indicate significant differences at p < 0.05 by Duncan’s test. ## p < 0.01, ### p < 0.001 vs. C (C: control, which was not treated; TC: treated control, which was treated with RANKL).
Figure 4.
The effect of genistein on the translation and activation of NADPH oxidase (Nox1) (A) and its upstream signals (B). RAW 264.7 cells were exposed to the receptor activator of nuclear factor kappa-B ligand (RANKL) (50 ng/mL) for two days in the absence and presence of genistein. GTP-Rac1 was recovered by centrifugation from proteins complexed to the beads, which have a glutathione S-transferase-p21-binding domain: human Pak1 as 67–150 and binds to GTP-bound Rac1 of the lysates. Western blotting analysis was performed on the lysates of cells that had been incubated with 1–20 μm genistein. Different corresponding letters indicate significant differences at p < 0.05 by Duncan’s test. ## p < 0.01, ### p < 0.001 vs. C (C: control, which was not treated; TC: treated control, which was treated with RANKL).
Figure 5.
The effect of genistein on mitochondrial membrane potential in RANKL-treated Raw264.7 cells. RAW 264.7 cells were exposed to the receptor activator of nuclear factor kappa-B ligand (RANKL) (50 ng/mL) for two days in the absence and the presence of genistein. After two days of incubation, the wells were gently washed twice with PBS. HBSS, which is fluorescently stable, was then added to each well. Rhodamine 123, a fluorescent probe detecting mitochondrial membrane potential, was added to the culture plates at a final concentration of 5 µM and incubated for 30 min at 37 °C in the dark. After the cells were washed with HBSS, the fluorescence intensity of rhodamine 123 was measured with an excitation wavelength of 485 nm and an emission wavelength of 535 nm using a Tecan GENios fluorometric plate reader. Data are expressed as percentages of the values of untreated cells (means ± standard deviations, n = 3). Different corresponding letters indicate significant differences at p < 0.05 by Duncan’s test. ### p < 0.001 vs. C (C: control, which was not treated; TC: treated control, which was treated with RANKL).
Figure 5.
The effect of genistein on mitochondrial membrane potential in RANKL-treated Raw264.7 cells. RAW 264.7 cells were exposed to the receptor activator of nuclear factor kappa-B ligand (RANKL) (50 ng/mL) for two days in the absence and the presence of genistein. After two days of incubation, the wells were gently washed twice with PBS. HBSS, which is fluorescently stable, was then added to each well. Rhodamine 123, a fluorescent probe detecting mitochondrial membrane potential, was added to the culture plates at a final concentration of 5 µM and incubated for 30 min at 37 °C in the dark. After the cells were washed with HBSS, the fluorescence intensity of rhodamine 123 was measured with an excitation wavelength of 485 nm and an emission wavelength of 535 nm using a Tecan GENios fluorometric plate reader. Data are expressed as percentages of the values of untreated cells (means ± standard deviations, n = 3). Different corresponding letters indicate significant differences at p < 0.05 by Duncan’s test. ### p < 0.001 vs. C (C: control, which was not treated; TC: treated control, which was treated with RANKL).
![Ijms 15 10605 g005]()
Superoxide anions are also produced as a normal metabolic byproduct of the electron transport chain in mitochondria. Previous studies have shown that intra-mitochondrial ROS generation can disrupt the mitochondrial membrane potential to induce a retrograde signaling pathway resulting in osteoclastogenesis in addition to mitochondrial dysfunction [
8,
32]. Therefore, the mitochondrial membrane potential was measured using a stable fluorescence probe, rhodamine 123 [
33]. In the assay system, the fluorescence value measured is inversely proportional to the mitochondrial membrane potential, because most of the rhodamine 123 added to RAW 264.7 cells without RANKL treatment is concentrated in the mitochondrial matrix and quenched, resulting in a low fluorescence value. The mitochondrial membrane potential of preosteoclastic precursor Raw264.7 cells was significantly (
p < 0.001) decreased by RANKL treatment, but it was significantly (
p < 0.05) recovered by the 20-μm genistein treatment (
Figure 5), indicating that mitochondrial ROS generation contributing to osteoclast differentiation may be attenuated by genistein. However, a further study is required for a clear understanding of the inhibitory effect of genistein on the disruption of the mitochondrial electron transport chain system in the osteoclast differentiation of RAW 264.7 cells by RANKL treatment. Overall, in view of controlling ROS generation in RANKL-treated RAW 264.7 cells, the above results provide solid evidence for the modulation of genistein on the superoxide anion level through suppressing both the translation and activation of Nox1 and protecting the disruption of the mitochondrial electron transport chain system.
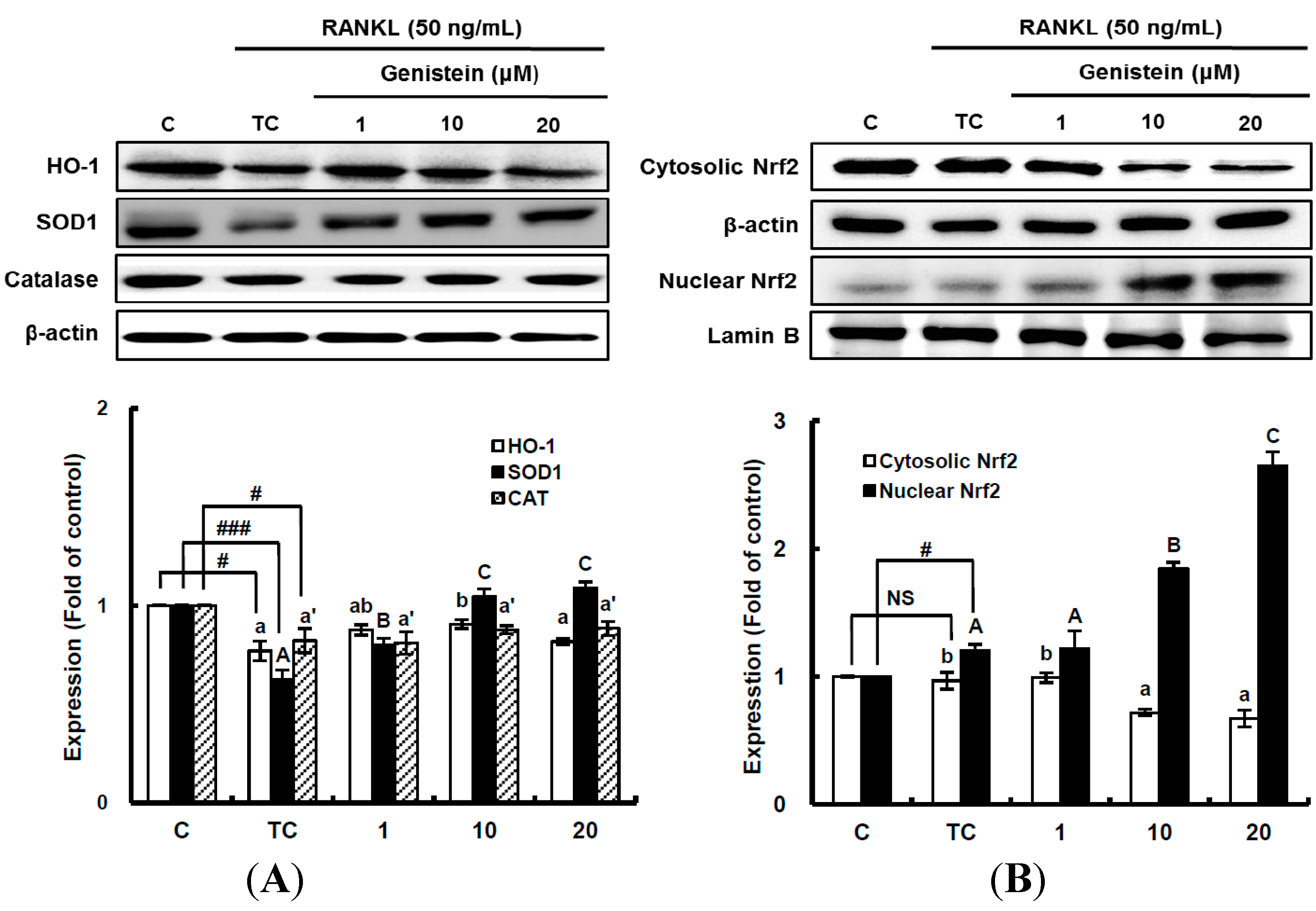
Figure 6.
The effects of genistein on the expression of detoxifying and antioxidant enzymes (A) and the activation of Nrf2 (B) in RANKL-treated RAW 264.7 cells. RAW 264.7 cells were exposed to the receptor activator of nuclear factor kappa-B ligand (RANKL) (50 ng/mL) for two days in the absence and presence of genistein. RAW 264.7 cells were lysed in radioimmunoprecipitation assay (RIPA) buffer (50 mM Tris-HCl (pH 8.0), 1% nonyl phenoxypolyethoxylethanol (NP)-40, 0.5% sodium deoxycholate, 150 mM NaCl and 1 mM phenylmethylsulfonyl fluoride (PMSF)) that contained a phosphatase inhibitor cocktail. The nuclear proteins in the RAW 264.7 cells were extracted by using Buffer A (10 mM hydroxyethyl piperazineethanesulfonic acid (HEPES), pH 7.9), 10 mM KCl, 2 mM MgCl2, 1 mM dithiothreitol (DTT), 0.5 mM ethylenediamine tetra-acetic acid (EDTA) (pH 7.9), 0.1 μM PMSF and 1× protease inhibitor cocktail) and Buffer B (50 mM HEPES (pH 7.9), 50 mM KCl, 0.3 mM NaCl, 1 mm DTT, 1 mM EDTA (pH 7.9), 0.1 μM PMSF and 10% glycerol). Western blotting analysis was performed on the lysates of cells that had been incubated with 1–20 μm genistein. Different corresponding letters indicate significant differences at p < 0.05 by Duncan’s test. # p < 0.05, ### p < 0.001 vs. C (C: control, which was not treated; TC: treated control, which was treated with RANKL; NS: not significant).
Figure 6.
The effects of genistein on the expression of detoxifying and antioxidant enzymes (A) and the activation of Nrf2 (B) in RANKL-treated RAW 264.7 cells. RAW 264.7 cells were exposed to the receptor activator of nuclear factor kappa-B ligand (RANKL) (50 ng/mL) for two days in the absence and presence of genistein. RAW 264.7 cells were lysed in radioimmunoprecipitation assay (RIPA) buffer (50 mM Tris-HCl (pH 8.0), 1% nonyl phenoxypolyethoxylethanol (NP)-40, 0.5% sodium deoxycholate, 150 mM NaCl and 1 mM phenylmethylsulfonyl fluoride (PMSF)) that contained a phosphatase inhibitor cocktail. The nuclear proteins in the RAW 264.7 cells were extracted by using Buffer A (10 mM hydroxyethyl piperazineethanesulfonic acid (HEPES), pH 7.9), 10 mM KCl, 2 mM MgCl2, 1 mM dithiothreitol (DTT), 0.5 mM ethylenediamine tetra-acetic acid (EDTA) (pH 7.9), 0.1 μM PMSF and 1× protease inhibitor cocktail) and Buffer B (50 mM HEPES (pH 7.9), 50 mM KCl, 0.3 mM NaCl, 1 mm DTT, 1 mM EDTA (pH 7.9), 0.1 μM PMSF and 10% glycerol). Western blotting analysis was performed on the lysates of cells that had been incubated with 1–20 μm genistein. Different corresponding letters indicate significant differences at p < 0.05 by Duncan’s test. # p < 0.05, ### p < 0.001 vs. C (C: control, which was not treated; TC: treated control, which was treated with RANKL; NS: not significant).
![Ijms 15 10605 g006]()