Mitigating the Effects of Xuebijing Injection on Hematopoietic Cell Injury Induced by Total Body Irradiation with γ rays by Decreasing Reactive Oxygen Species Levels
Abstract
:1. Introduction
2. Results and Discussion
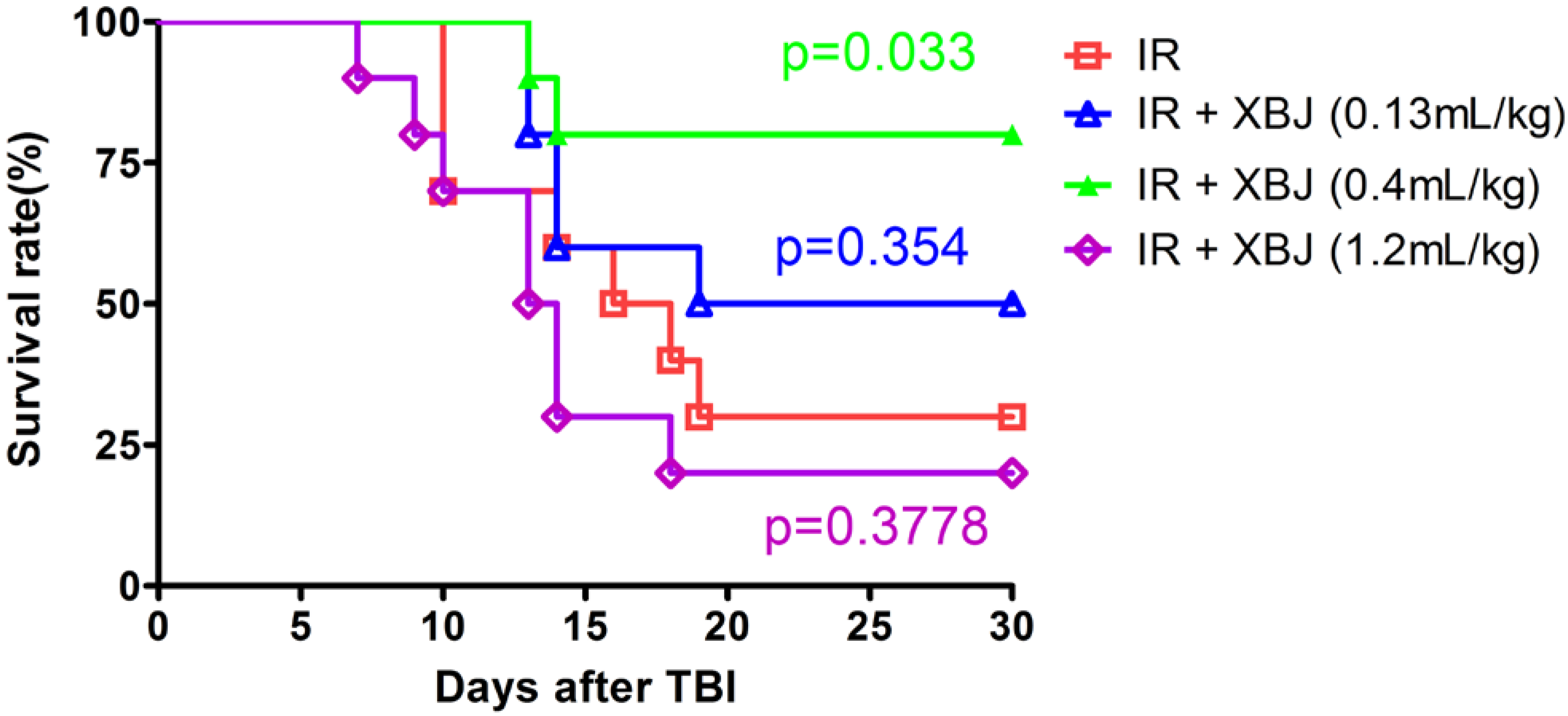
2.1. XBJ Increased the 30-Day Survival Rate of Mice Exposed to a Lethal Dose of TBI
2.2. XBJ Mitigated IR-Induced Hematopoietic Cell Number Decreases
| XBJ (μL/mL) | 0 Gy | 1 Gy | 4 Gy |
|---|---|---|---|
| 0 | 130 ± 10 | 100 ± 3 # | 74 ± 3 # |
| 1 | 139 ± 9 * | 107 ± 2 **,# | 92 ± 7 **,# |
| 5 | 143 ± 10 ** | 111 ± 2 *,# | 89 ± 4 **,## |
| 10 | 127 ± 7 | 109 ± 6 ## | 91 ± 4 **,## |
| 25 | 131 ± 8 | 99 ± 3 # | 89 ± 6 *,## |
| 50 | 121 ± 8 * | 93 ± 6 ## | 77 ± 4 *,## |
| 100 | 81 ± 5 ** | 73 ± 2 **,# | 60 ± 3 *,## |
| 200 | 26 ± 2 ** | 23 ± 2 **,## | 19 ± 1 **,## |
| Mice | WBCs (109/L) | RBCs (1012/L) | HGB (g/L) | HCT (%) | PLT (109/L) |
|---|---|---|---|---|---|
| Ctr | 8.6 ± 1.5 | 9.0 ± 0.3 | 144.2 ± 2.8 | 35.0 ± 1.0 | 494.8 ± 51.1 |
| 0.4 mL/kg XBJ | 9.8 ± 0.7 | 8.7 ± 0.3 | 138.2 ± 6.8 | 33.4 ± 1.8 | 522.4 ± 117.5 |
| 2 Gy | 3.8 ± 0.4 ** | 7.6 ± 0.3 ** | 125.0 ± 6.5 ** | 30.2 ± 1.6 ** | 306.8 ± 41.8 ** |
| 2 Gy + 0.4 mL/kg XBJ | 6.5 ± 1.6 *,# | 8.2 ± 0.3 **,# | 135.2 ± 6.6 *,# | 32.6 ± 1.4 *,# | 349.0 ± 88.7 * |
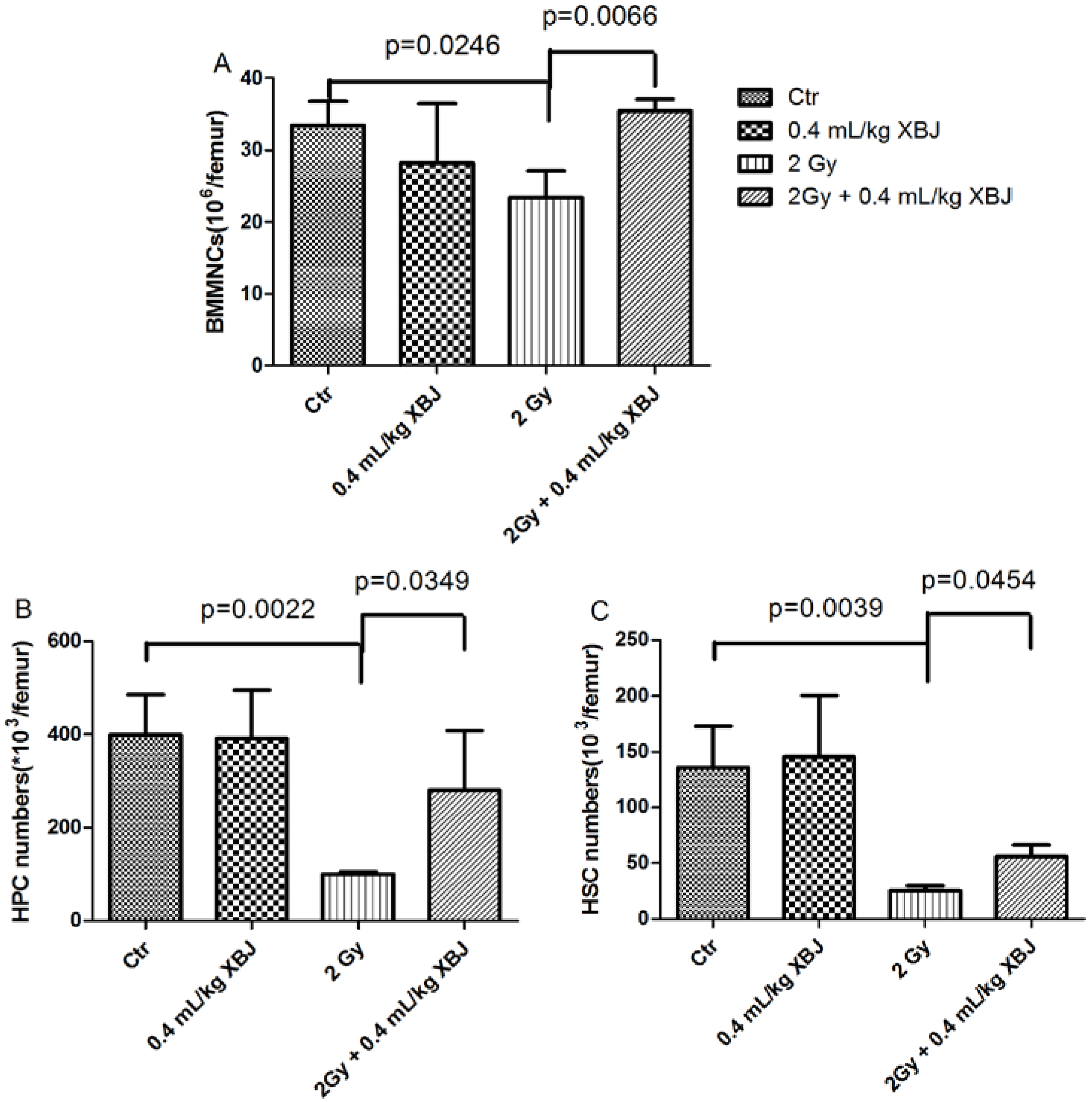
2.3. XBJ Attenuated IR-Induced CFU-GM Reduction

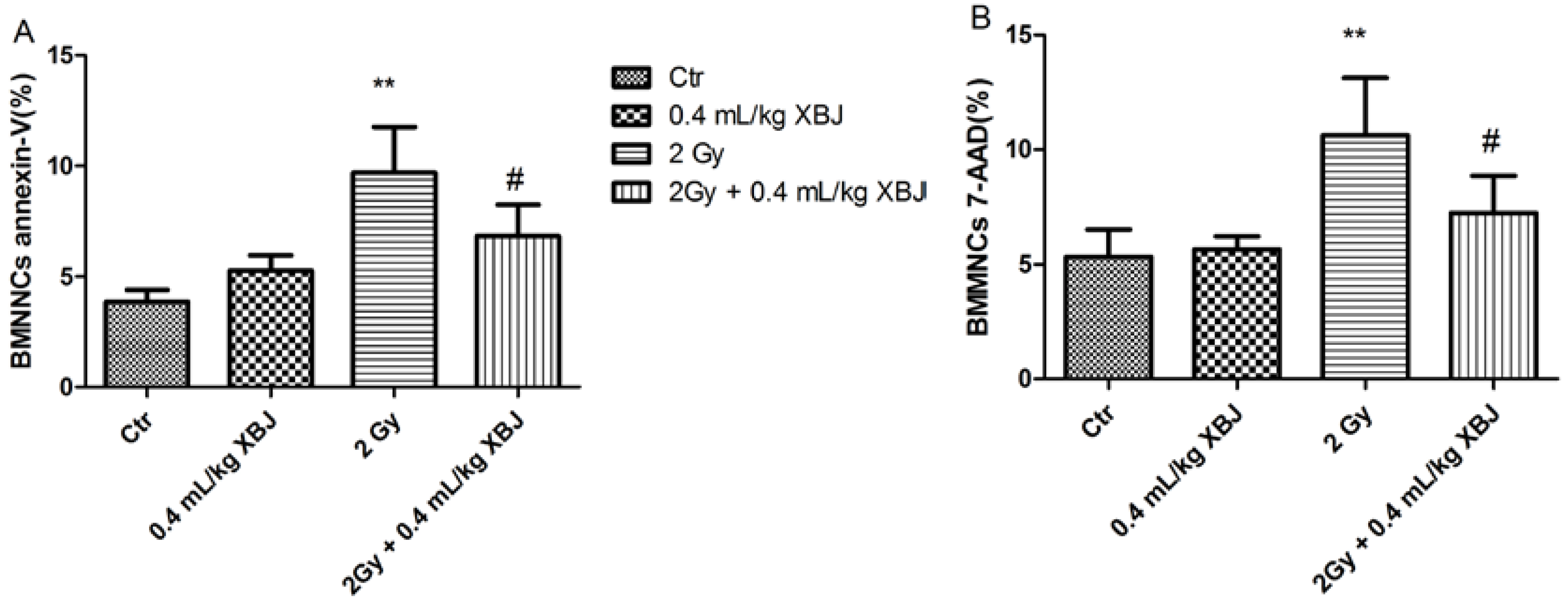
2.4. XBJ Attenuated IR-Induced BMMNC Apoptosis
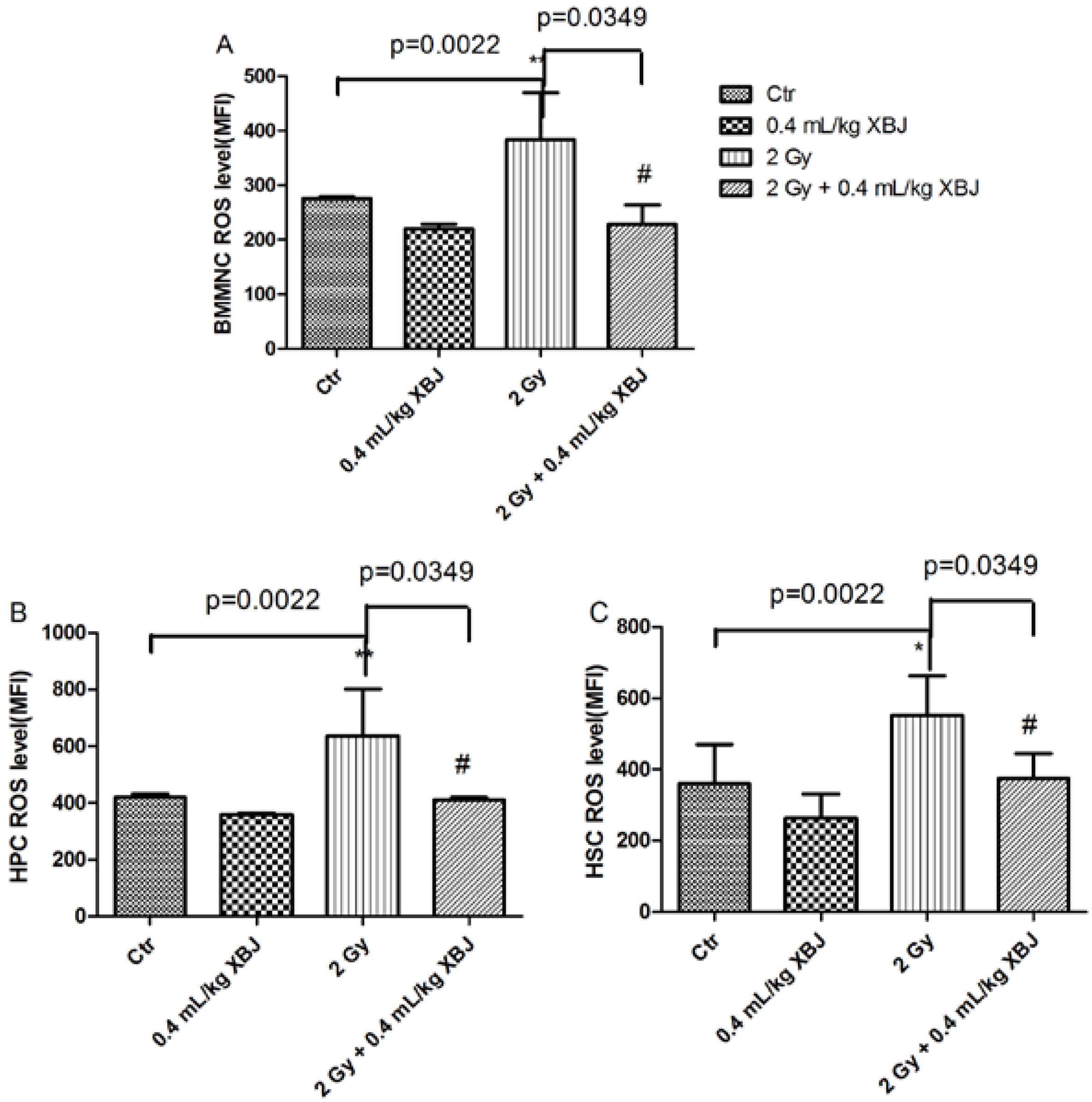
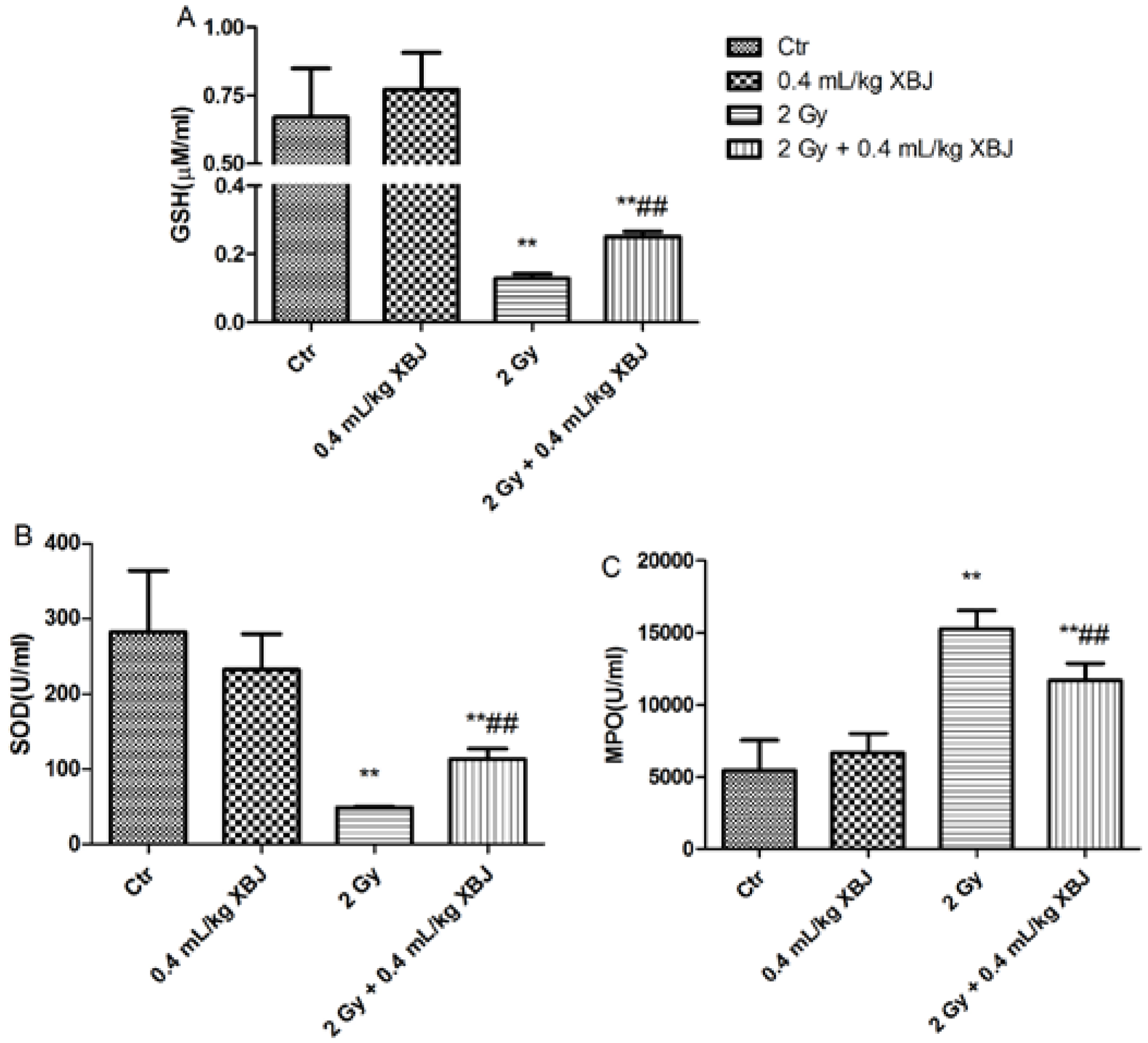
2.5. XBJ Inhibited Radiation-Induced ROS Production
3. Experimental
3.1. Animals and Reagents
3.2. Irradiation
3.3. Cell Viability Assays
3.4. XBJ Administration
3.5. Peripheral Blood Cell and BM Mononucleated Cells (BMMNCs) Counts
3.6. Detection of HPCs and HSCs
3.7. Colony-Forming Assays
3.8. Intracellular ROS Analysis
3.9. Assay of SOD, GSH and MPO Levels in Serum
3.10. Statistical Analysis
4. Conclusions
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Coleman, C.N.; Blakely, W.F.; Fike, J.R.; MacVittie, T.J.; Metting, N.F.; Mitchell, J.B.; Moulder, J.E.; Preston, R.J.; Seed, T.M.; Stone, H.B.; et al. Molecular and cellular biology of moderate-dose (1–10 gy) radiation and potential mechanisms of radiation protection: Report of a workshop at Bethesda, Maryland, December 17–18, 2001. Radiat. Res. 2003, 159, 812–834. [Google Scholar] [CrossRef]
- Xiao, M.; Whitnall, M.H. Pharmacological countermeasures for the acute radiation syndrome. Curr. Mol. Pharmacol. 2009, 2, 122–133. [Google Scholar] [CrossRef]
- Coleman, C.N.; Stone, H.B.; Moulder, J.E.; Pellmar, T.C. Medicine. Modulation of radiation injury. Science 2004, 304, 693–694. [Google Scholar] [CrossRef]
- Weiss, J.F.; Landauer, M.R. History and development of radiation-protective agents. Int. J. Radiat. Biol. 2009, 85, 539–573. [Google Scholar] [CrossRef]
- Ganesh, C.J. Radioprotective potential of plants and herbs against the effects of ionizing radiation. J. Clin. Biochem. Nutr. 2007, 40, 74–81. [Google Scholar] [CrossRef]
- Hosseinimehr, S.J. Trends in the development of radioprotective agents. Drug Discov. Today 2007, 12, 794–805. [Google Scholar] [CrossRef]
- Hosseinimehr, S.J.; Tavakoli, H.; Pourheidari, G.; Sobhani, A.; Shafiee, A. Radioprotective effects of citrus extract against gamma-irradiation in mouse bone marrow cells. J. Radiat. Res. 2003, 44, 237–241. [Google Scholar] [CrossRef]
- Hosseinimehr, S.J.; Azadbakht, M.; Mousavi, S.M.; Mahmoudzadeh, A.; Akhlaghpoor, S. Radioprotective effects of hawthorn fruit extract against gamma irradiation in mouse bone marrow cells. J. Radiat. Res. 2007, 48, 63–68. [Google Scholar] [CrossRef]
- Huang, H.; Ji, L.; Song, S.; Wang, J.; Wei, N.; Jiang, M.; Bai, G.; Luo, G. Identification of the major constituents in xuebijing injection by hplc-esi-ms. Phytochem. Anal. 2011, 22, 330–338. [Google Scholar] [CrossRef]
- Qi, F.; Liang, Z.X.; She, D.Y.; Yan, G.T.; Chen, L.A. A clinical study on the effects and mechanism of xuebijing injection in severe pneumonia patients. J. Tradit. Chin. Med. 2011, 31, 46–49. [Google Scholar] [CrossRef]
- Chen, Y.; Tong, H.; Zhang, X.; Tang, L.; Pan, Z.; Liu, Z.; Duan, P.; Su, L. Xuebijing injection alleviates liver injury by inhibiting secretory function of kupffer cells in heat stroke rats. J. Tradit. Chin. Med. 2013, 33, 243–249. [Google Scholar] [CrossRef]
- Fang, K.; Wang, X.L. Treatment of multiple organ dysfunction syndrome by xuebijing injection: A clinical research. Zhongguo Zhong Xi Yi Jie He Za Zhi 2013, 33, 205–207. [Google Scholar]
- Shao, M.; Liu, B.; Wang, J.Q.; Tao, X.G.; Zhou, S.S.; Jin, K.; Zhang, C.P. Effect of xuebijing injection on t helper 17 and cd4+ cd25+ regulatory t cells in patients with sepsis. Zhongguo Wei Zhong Bing Ji Jiu Yi 2011, 23, 430–434. [Google Scholar]
- Jiang, M.; Zhou, M.; Han, Y.; Xing, L.; Zhao, H.; Dong, L.; Bai, G.; Luo, G. Identification of nf-kappab inhibitors in xuebijing injection for sepsis treatment based on bioactivity-integrated uplc-q/tof. J. Ethnopharmacol. 2013, 147, 426–433. [Google Scholar] [CrossRef]
- Li, D.; Wang, Y.; Wu, H.; Lu, L.; Wang, X.; Zhang, J.; Zhang, H.; Fan, S.; Fan, F.; Zhou, D.; et al. The effects of p38 mapk inhibition combined with g-csf administration on the hematoimmune system in mice with irradiation injury. PLoS One 2013, 8, e62921. [Google Scholar]
- Li, D.; Wang, Y.; Wu, H.; Lu, L.; Zhang, H.; Chang, J.; Zhai, Z.; Zhang, J.; Zhou, D.; Meng, A. Mitigation of ionizing radiation-induced bone marrow suppression by p38 inhibition and g-csf administration. J. Radiat. Res. 2011, 52, 712–716. [Google Scholar] [CrossRef]
- Tak, J.K.; Park, J.W. The use of ebselen for radioprotection in cultured cells and mice. Free Radic. Biol. Med. 2009, 46, 1177–1185. [Google Scholar]
- Singh, V.K.; Brown, D.S.; Kao, T.C. Tocopherol succinate: A promising radiation countermeasure. Int. Immunopharmacol. 2009, 9, 1423–1430. [Google Scholar] [CrossRef]
- Singh, V.K.; Grace, M.B.; Parekh, V.I.; Whitnall, M.H.; Landauer, M.R. Effects of genistein administration on cytokine induction in whole-body gamma irradiated mice. Int. Immunopharmacol. 2009, 9, 1401–1410. [Google Scholar] [CrossRef]
- Wang, K.; Liu, C.; Di, C.J.; Ma, C.; Han, C.G.; Yuan, M.R.; Li, P.F.; Li, L.; Liu, Y.X. Kojic acid protects c57bl/6 mice from gamma-irradiation induced damage. Asian Pac. J. Cancer Prev. 2014, 15, 291–297. [Google Scholar]
- Hoggatt, J.; Singh, P.; Stilger, K.N.; Plett, P.A.; Sampson, C.H.; Chua, H.L.; Orschell, C.M.; Pelus, L.M. Recovery from hematopoietic injury by modulating prostaglandin e (2) signaling post-irradiation. Blood Cells Mol. Dis. 2013, 50, 147–153. [Google Scholar] [CrossRef]
- Wang, Y.; Schulte, B.A.; Zhou, D. Hematopoietic stem cell senescence and long-term bone marrow injury. Cell Cycle 2006, 5, 35–38. [Google Scholar] [CrossRef]
- Wang, Y.; Schulte, B.A.; LaRue, A.C.; Ogawa, M.; Zhou, D. Total body irradiation selectively induces murine hematopoietic stem cell senescence. Blood 2006, 107, 358–366. [Google Scholar] [CrossRef]
- Porter, R.L.; Georger, M.A.; Bromberg, O.; McGrath, K.E.; Frisch, B.J.; Becker, M.W.; Calvi, L.M. Prostaglandin e2 increases hematopoietic stem cell survival and accelerates hematopoietic recovery after radiation injury. Stem Cells 2013, 31, 372–383. [Google Scholar] [CrossRef]
- Yamamori, T.; Yasui, H.; Yamazumi, M.; Wada, Y.; Nakamura, Y.; Nakamura, H.; Inanami, O. Ionizing radiation induces mitochondrial reactive oxygen species production accompanied by upregulation of mitochondrial electron transport chain function and mitochondrial content under control of the cell cycle checkpoint. Free Radic. Biol. Med. 2012, 53, 260–270. [Google Scholar] [CrossRef]
- Shao, L.; Li, H.; Pazhanisamy, S.K.; Meng, A.; Wang, Y.; Zhou, D. Reactive oxygen species and hematopoietic stem cell senescence. Int. J. Hematol. 2011, 94, 24–32. [Google Scholar] [CrossRef]
- Kalyanaraman, B. Teaching the basics of redox biology to medical and graduate students: Oxidants, antioxidants and disease mechanisms. Redox Biol. 2013, 1, 244–257. [Google Scholar] [CrossRef]
- Bast, A.; Haenen, G.R. Ten misconceptions about antioxidants. Trends Pharmacol. Sci. 2013, 34, 430–436. [Google Scholar] [CrossRef]
- Liu, Y.K.; Huang, L.J.; Zhao, S.; Lin, W.; He, J.B.; Ying, L.; You, X.; Wang, W.T. [effect of xuebijing injection on tlr4--nf-kappab--il-1beta pathway of myocardial hypoxia/reoxygenation in rats]. Zhongguo Ying Yong Sheng Li Xue Za Zhi 2014, 30, 55–59. [Google Scholar]
- Zhang, H.; Zhai, Z.; Wang, Y.; Zhang, J.; Wu, H.; Li, C.; Li, D.; Lu, L.; Wang, X.; Chang, J.; et al. Resveratrol ameliorates ionizing irradiation-induced long-term hematopoietic stem cell injury in mice. Free Radic. Biol. Med. 2013, 54, 40–50. [Google Scholar] [CrossRef]
- Li, H.; Wang, Y.; Pazhanisamy, S.K.; Shao, L.; Batinic-Haberle, I.; Meng, A.; Zhou, D. Mn(iii) meso-tetrakis-(n-ethylpyridinium-2-yl) porphyrin mitigates total body irradiation-induced long-term bone marrow suppression. Free Radic. Biol. Med. 2011, 51, 30–37. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, D.; Lu, L.; Zhang, J.; Wang, X.; Xing, Y.; Wu, H.; Yang, X.; Shi, Z.; Zhao, M.; Fan, S.; et al. Mitigating the Effects of Xuebijing Injection on Hematopoietic Cell Injury Induced by Total Body Irradiation with γ rays by Decreasing Reactive Oxygen Species Levels. Int. J. Mol. Sci. 2014, 15, 10541-10553. https://doi.org/10.3390/ijms150610541
Li D, Lu L, Zhang J, Wang X, Xing Y, Wu H, Yang X, Shi Z, Zhao M, Fan S, et al. Mitigating the Effects of Xuebijing Injection on Hematopoietic Cell Injury Induced by Total Body Irradiation with γ rays by Decreasing Reactive Oxygen Species Levels. International Journal of Molecular Sciences. 2014; 15(6):10541-10553. https://doi.org/10.3390/ijms150610541
Chicago/Turabian StyleLi, Deguan, Lu Lu, Junling Zhang, Xiaochun Wang, Yonghua Xing, Hongying Wu, Xiangdong Yang, Zhexin Shi, Mingfeng Zhao, Saijun Fan, and et al. 2014. "Mitigating the Effects of Xuebijing Injection on Hematopoietic Cell Injury Induced by Total Body Irradiation with γ rays by Decreasing Reactive Oxygen Species Levels" International Journal of Molecular Sciences 15, no. 6: 10541-10553. https://doi.org/10.3390/ijms150610541




