Estrogen Rapidly Enhances Incisional Pain of Ovariectomized Rats Primarily through the G Protein-Coupled Estrogen Receptor
Abstract
:1. Introduction
2. Results and Discussion
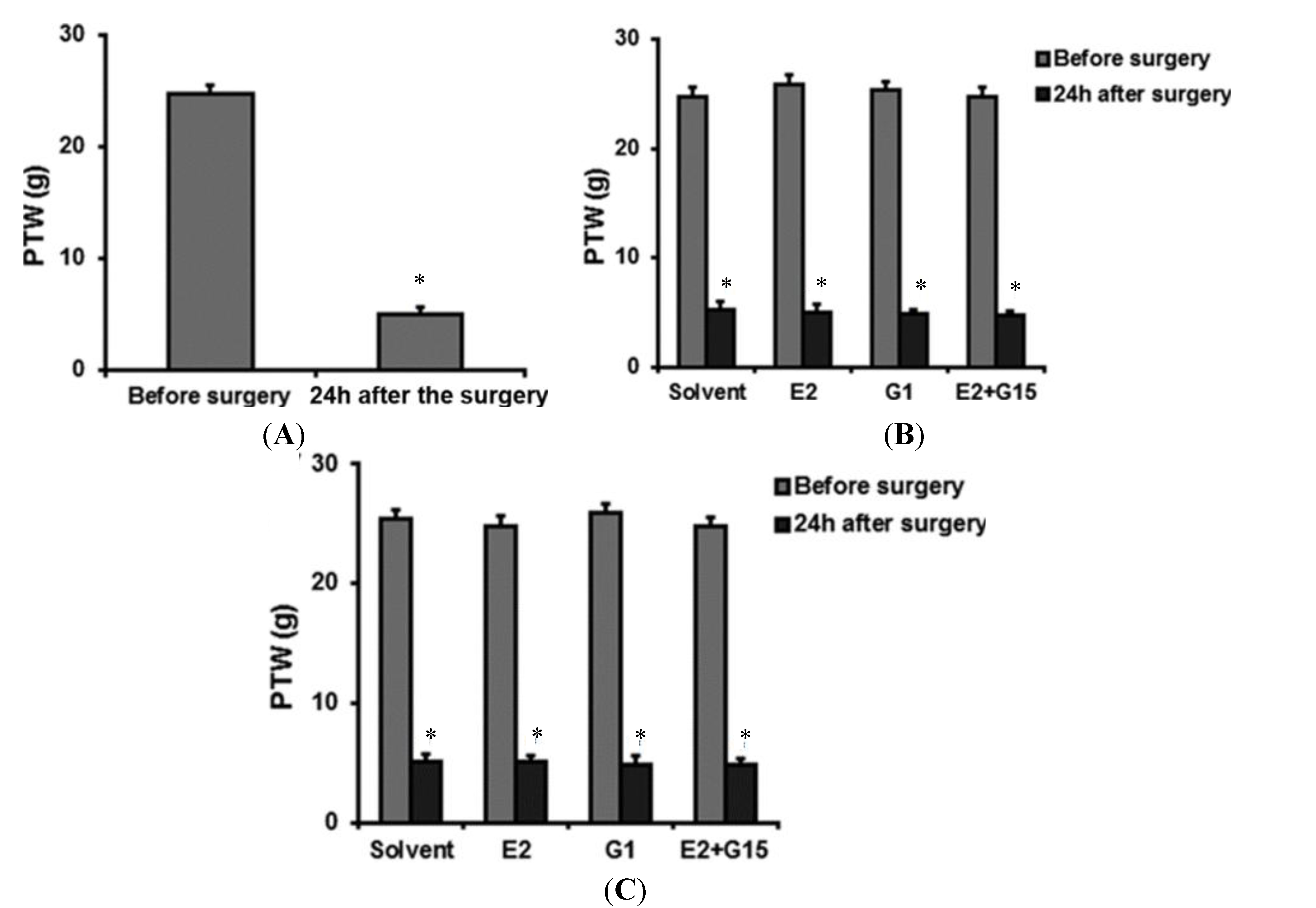
2.1. Hind-Paw Withdrawal Threshold (PWT)
| Administration | Group | Before Surgery | Before Drugs | After Drugs |
|---|---|---|---|---|
| IV | Solvent | 24.8 ± 0.78 g | 5.23 ± 0.72 g | 5.00 ± 0.71 g |
| E2 | 25.9 ± 0.88 g | 5.03 ± 0.74 g | 2.65 ± 0.66 g | |
| G1 | 25.4 ± 0.78 g | 4.87 ± 0.40 g | 2.50 ± 0.58 g | |
| E2 + G15 | 24.8 ± 0.78 g | 4.67 ± 0.40 g | 4.60 ± 0.51 g | |
| IT | Solvent | 25.4 ± 0.78 g | 5.10 ± 0.63 g | 5.05 ± 0.61 g |
| E2 | 24.8 ± 0.88 g | 5.10 ± 0.56 g | 1.98 ± 0.36 g | |
| G1 | 25.9 ± 0.78 g | 4.80 ± 0.86 g | 2.56 ± 0.33 g | |
| E2 + G15 | 24.8 ± 0.78 g | 4.90 ± 0.41 g | 4.48 ± 0.55 g | |
| Total | 25.2 ± 0.49 g | 4.90 ± 0.20 g |
2.2. Intravenously (IV) Group
2.2.1. The Effect of 17-β-Estradiol (E2) Administration on PWT
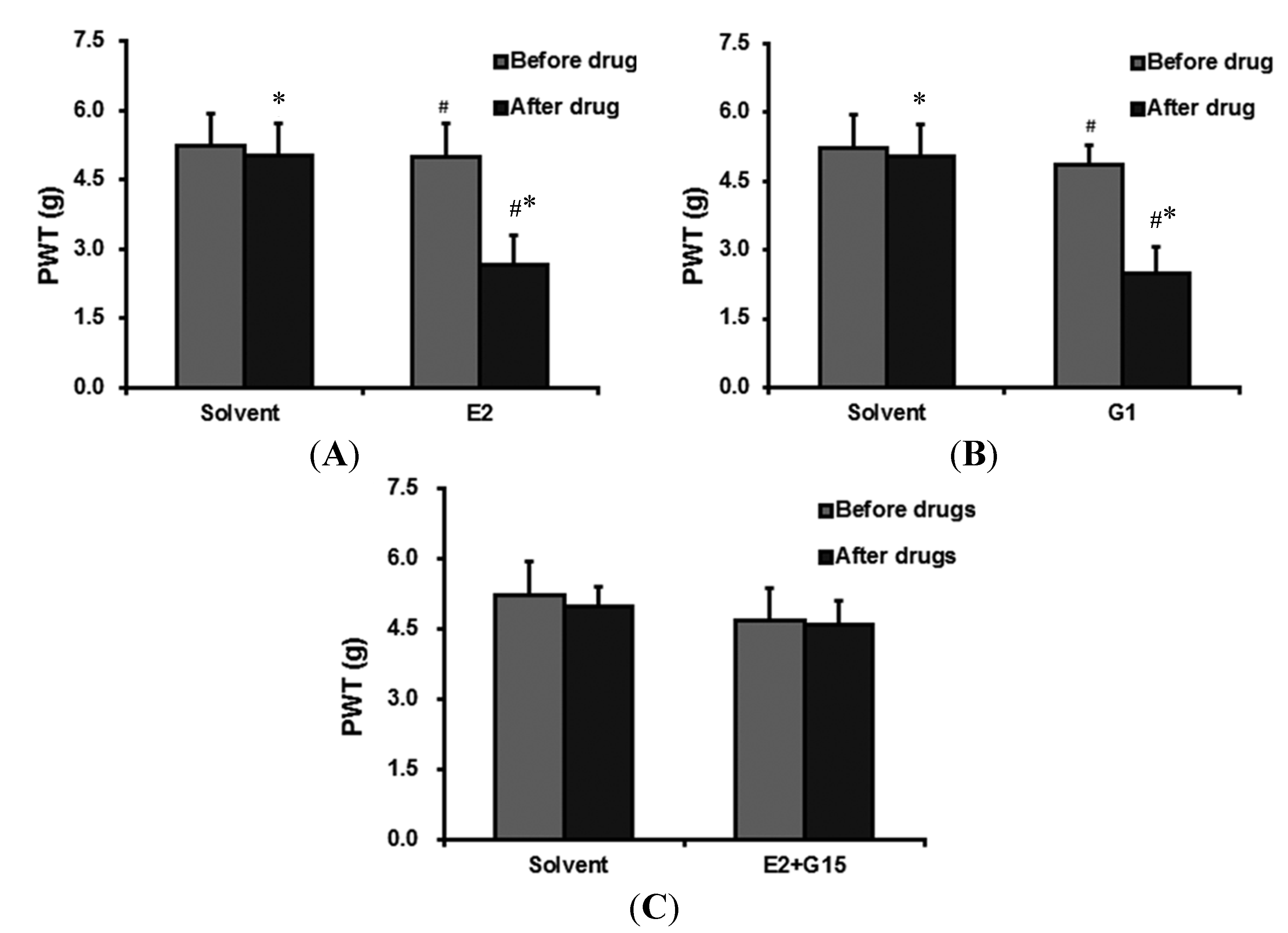
2.2.2. G Protein-Coupled Estrogen Receptor (GPER)-Selective Agonist (G1) Administration Ecreases PWT
2.2.3. The Effect of 17-β-Estradiol (E2) + GPER-Selective Antagonist (G15) Administration on PWT
2.3. Intrathecal (IT) Group
2.3.1. The Effect of E2 Administration on PWT

2.3.2. G1 Administration Decreases PWT
2.3.3. The Effect of E2 + G15 Administration on PWT
2.4. Discussion
3. Materials and Methods
3.1. Animals
3.2. Drugs
3.3. Equipment
3.4. Ovariectomized (OVX) Surgery
3.5. The Incisional Pain Model
3.6. Subarachnoid Catheter
3.7. Evaluation of the Incisional Pain Threshold
3.8. Administration of Drugs
3.9. Doses of the Drugs
3.10. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Craft, R.M. Modulation of pain by estrogens. Pain 2007, 132 (Suppl. 1), S3–S12. [Google Scholar] [CrossRef]
- Chesterton, L.S.; Barlas, P.; Foster, N.E.; Baxter, G.D.; Wright, C.C. Gender differences in pressure pain threshold in healthy humans. Pain 2003, 101, 259–266. [Google Scholar]
- Sarlani, E.; Farooq, N.; Greenspan, J.D. Gender and laterality differences in thermosensation throughout the perceptible range. Pain 2003, 106, 9–18. [Google Scholar] [CrossRef]
- Frot, M.; Feine, J.S.; Bushnell, M.C. Sex differences in pain perception and anxiety. A psychophysical study with topical capsaicin. Pain 2004, 108, 230–236. [Google Scholar]
- Unruh, A.M. Gender variations in clinical pain experience. Pain 1996, 65, 123–167. [Google Scholar] [CrossRef]
- Khasar, S.G.; Dina, O.A.; Green, P.G.; Levine, J.D. Estrogen regulates adrenal medullary function producing sexual dimorphism in nociceptive threshold and beta-adrenergic receptor-mediated hyperalgesia in the rat. Eur. J. Neurosci. 2005, 21, 3379–3386. [Google Scholar] [CrossRef]
- Hucho, T.; Levine, J.D. Signaling pathways in sensitization: Toward a nociceptor cell biology. Neuron 2007, 55, 365–376. [Google Scholar] [CrossRef]
- Woolf, C.J.; Ma, Q. Nociceptors—Noxious stimulus detectors. Neuron 2007, 55, 353–364. [Google Scholar] [CrossRef]
- Hucho, T.B.; Dina, O.A.; Kuhn, J.; Levine, J.D. Estrogen controls PKCε-dependent mechanical hyperalgesia through direct action on nociceptive neurons. Eur. J. Neurosci. 2006, 24, 527–534. [Google Scholar] [CrossRef]
- Evrard, H.C.; Balthazart, J. Rapid regulation of pain by estrogens synthesized in spinal dorsal horn neurons. J. Neurosci. 2004, 24, 7225–7229. [Google Scholar] [CrossRef]
- Mannino, C.A.; South, S.M.; Quinones-Jenab, V.; Inturrisi, C.E. Estradiol replacement in ovariectomized rats is antihyperalgesic in the formalin test. J. Pain 2007, 8, 334–342. [Google Scholar] [CrossRef]
- Gaumond, I.; Arsenault, P.; Marchand, S. Specificity of female and male sex hormones on excitatory and inhibitory phases of formalin-induced nociceptive responses. Brain Res. 2005, 1052, 105–111. [Google Scholar]
- Ceccarelli, I.; Scaramuzzino, A.; Massafra, C.; Aloisi, A.M. The behavioral and neuronal effects induced by repetitive nociceptive stimulation are affected by gonadal hormones in male rats. Pain 2003, 104, 35–47. [Google Scholar] [CrossRef]
- Sanoja, R.; Cervero, F. Estrogen modulation of ovariectomy-induced hyperalgesia in adult mice. Eur. J. Pain 2008, 12, 573–581. [Google Scholar] [CrossRef]
- Revankar, C.M.; Cimino, D.F.; Sklar, L.A.; Arterburn, J.B.; Prossnitz, E.R. A transmembrane intracellular estrogen receptor mediates rapid cell signaling. Science 2005, 307, 1625–1630. [Google Scholar] [CrossRef]
- Bjornstrom, L.; Sjoberg, M. Mechanisms of estrogen receptor signaling: Convergence of genomic and nongenomic actions on target genes. Mol. Endocrinol. 2005, 19, 833–842. [Google Scholar] [CrossRef]
- Dennis, M.K.; Burai, R.; Ramesh, C.; Petrie, W.K.; Alcon, S.N.; Nayak, T.K.; Bologa, C.G.; Leitao, A.; Brailoiu, E.; Deliu, E.; et al. In vivo effects of a GPR30 antagonist. Nat. Chem. Biol. 2009, 5, 421–427. [Google Scholar] [CrossRef]
- Khasar, S.G.; Lin, Y.H.; Martin, A.; Dadgar, J.; McMahon, T.; Wang, D.; Hundle, B.; Aley, K.O.; Isenberg, W.; McCarter, G.; et al. A novel nociceptor signaling pathway revealed in protein kinase C epsilon mutant mice. Neuron 1999, 24, 253–260. [Google Scholar] [CrossRef]
- Kuhn, J.; Dina, O.A.; Goswami, C.; Suckow, V.; Levine, J.D.; Hucho, T. GPR30 estrogen receptor agonists induce mechanical hyperalgesia in the rat. Eur. J. Neurosci. 2008, 27, 1700–1709. [Google Scholar]
- Tang, B.; Ji, Y.; Traub, R.J. Estrogen alters spinal NMDA receptor activity via a PKA signaling pathway in a visceral pain model in the rat. Pain 2008, 137, 540–549. [Google Scholar] [CrossRef]
- Dai, Y.; Fukuoka, T.; Wang, H.; Yamanaka, H.; Obata, K.; Tokunaga, A.; Noguchi, K. Contribution of sensitized P2X receptors in inflamed tissue to the mechanical hypersensitivity revealed by phosphorylated ERK in DRG neurons. Pain 2004, 108, 258–266. [Google Scholar] [CrossRef]
- Ma, B.; Yu, L.H.; Fan, J.; Cong, B.; He, P.; Ni, X.; Burnstock, G. Estrogen modulation of peripheral pain signal transduction: Involvement of P2X3 receptors. Purinergic Signal. 2011, 7, 73–83. [Google Scholar] [CrossRef]
- Fan, J.; Yu, L.H.; Zhang, Y.; Ni, X.; Ma, B.; Burnstock, G. Estrogen altered visceromotor reflex and P2X3 mRNA expression in a rat model of colitis. Steroids 2009, 74, 956–962. [Google Scholar] [CrossRef]
- Evrard, H.C. Estrogen synthesis in the spinal dorsal horn: A new central mechanism for the hormonal regulation of pain. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006, 291, R291–R299. [Google Scholar] [CrossRef]
- Carmeci, C.; Thompson, D.A.; Ring, H.Z.; Francke, U.; Weigel, R.J. Identification of a gene (GPR30) with homology to the G-protein-coupled receptor superfamily associated with estrogen receptor expression in breast cancer. Genomics 1997, 45, 607–617. [Google Scholar] [CrossRef]
- Prossnitz, E.R.; Arterburn, J.B.; Smith, H.O.; Oprea, T.I.; Sklar, L.A.; Hathaway, H.J. Estrogen signaling through the transmembrane G protein-coupled receptor GPR30. Annu. Rev. Physiol. 2008, 70, 165–190. [Google Scholar] [CrossRef]
- Strader, C.D.; Fong, T.M.; Tota, M.R.; Underwood, D.; Dixon, R.A. Structure and function of G protein-coupled receptors. Annu. Rev. Biochem. 1994, 63, 101–132. [Google Scholar] [CrossRef]
- Bologa, C.G.; Revankar, C.M.; Young, S.M.; Edwards, B.S.; Arterburn, J.B.; Kiselyov, A.S.; Parker, M.A.; Tkachenko, S.E.; Savchuck, N.P.; Sklar, L.A.; et al. Virtual and biomolecular screening converge on a selective agonist for GPR30. Nat. Chem. Biol. 2006, 2, 207–212. [Google Scholar]
- Julius, D.; Basbaum, A.I. Molecular mechanisms of nociception. Nature 2001, 413, 203–210. [Google Scholar] [CrossRef]
- Dun, S.L.; Brailoiu, G.C.; Gao, X.; Brailoiu, E.; Arterburn, J.B.; Prossnitz, E.R.; Oprea, T.I.; Dun, N.J. Expression of estrogen receptor GPR30 in the rat spinal cord and in autonomic and sensory ganglia. J. Neurosci. Res. 2009, 87, 1610–1619. [Google Scholar]
- Takanami, K.; Sakamoto, H.; Matsuda, K.; Hosokawa, K.; Nishi, M.; Prossnitz, E.R.; Kawata, M. Expression of G protein-coupled receptor 30 in the spinal somatosensory system. Brain Res. 2010, 1310, 17–28. [Google Scholar] [CrossRef]
- Brennan, T.J.; Vandermeulen, E.P.; Gebhart, G.F. Characterization of a rat model of incisional pain. Pain 1996, 64, 493–501. [Google Scholar] [CrossRef]
- Chaplan, S.R.; Bach, F.W.; Pogrel, J.W.; Chung, J.M.; Yaksh, T.L. Quantitative assessment of tactile allodynia in the rat paw. J. Neurosci. Methods 1994, 53, 55–63. [Google Scholar] [CrossRef]
- Valenzano, K.J.; Tafesse, L.; Lee, G.; Harrison, J.E.; Boulet, J.M.; Gottshall, S.L.; Mark, L.; Pearson, M.S.; Miller, W.; Shan, S.; et al. harmacological and pharmacokinetic characterization of the cannabinoid receptor 2 agonist, GW405833, utilizing rodent models of acute and chronic pain, anxiety, ataxia and catalepsy. Neuropharmacology 2005, 48, 658–672. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
An, G.; Li, W.; Yan, T.; Li, S. Estrogen Rapidly Enhances Incisional Pain of Ovariectomized Rats Primarily through the G Protein-Coupled Estrogen Receptor. Int. J. Mol. Sci. 2014, 15, 10479-10491. https://doi.org/10.3390/ijms150610479
An G, Li W, Yan T, Li S. Estrogen Rapidly Enhances Incisional Pain of Ovariectomized Rats Primarily through the G Protein-Coupled Estrogen Receptor. International Journal of Molecular Sciences. 2014; 15(6):10479-10491. https://doi.org/10.3390/ijms150610479
Chicago/Turabian StyleAn, Guanghui, Wenhui Li, Tao Yan, and Shitong Li. 2014. "Estrogen Rapidly Enhances Incisional Pain of Ovariectomized Rats Primarily through the G Protein-Coupled Estrogen Receptor" International Journal of Molecular Sciences 15, no. 6: 10479-10491. https://doi.org/10.3390/ijms150610479




