Direct Analysis of hCGβcf Glycosylation in Normal and Aberrant Pregnancy by Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry
Abstract
:1. Introduction
2. Results and Discussion
2.1. Mass Spectral Profiles
2.2. Determination of Glycostructures
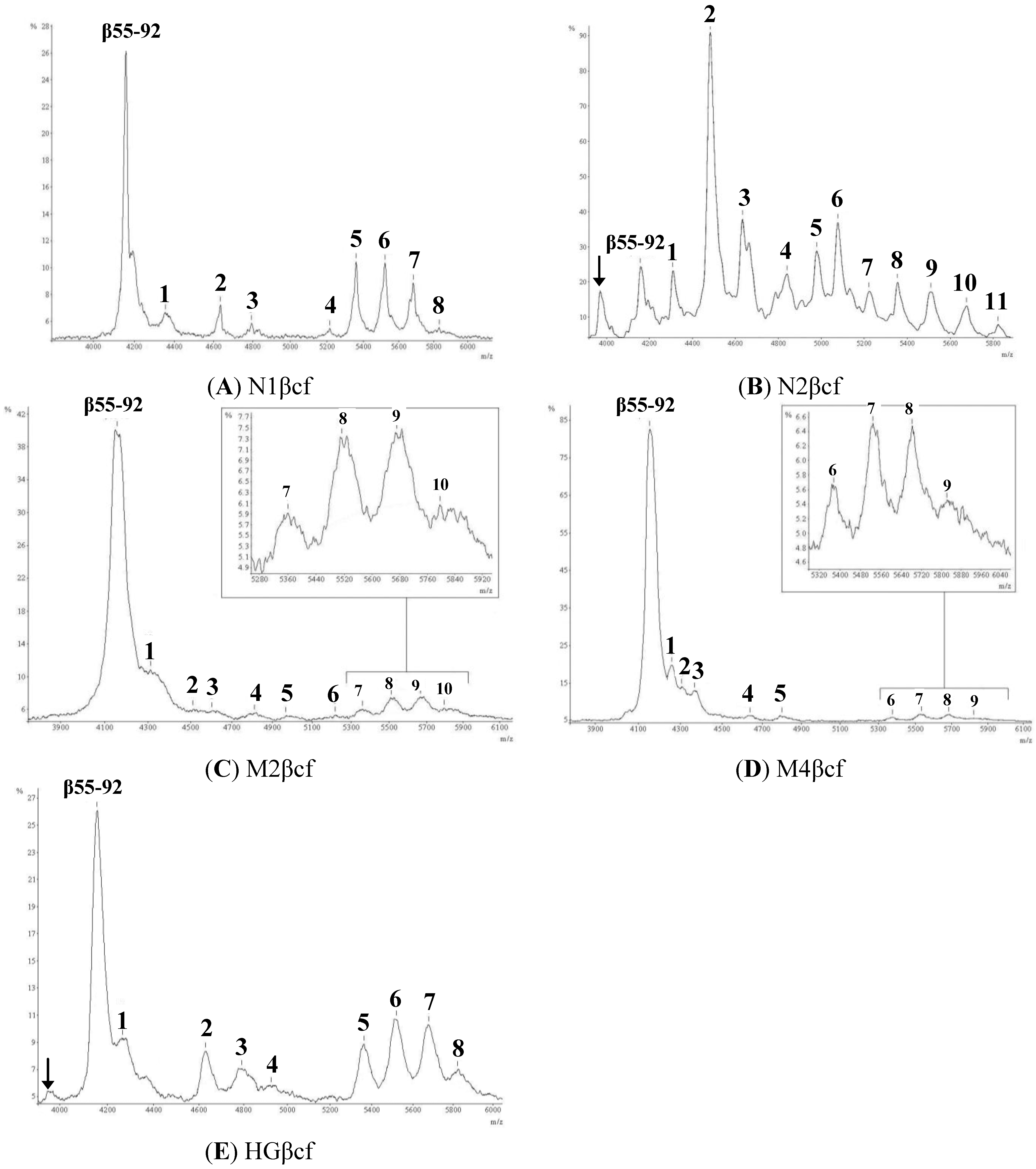
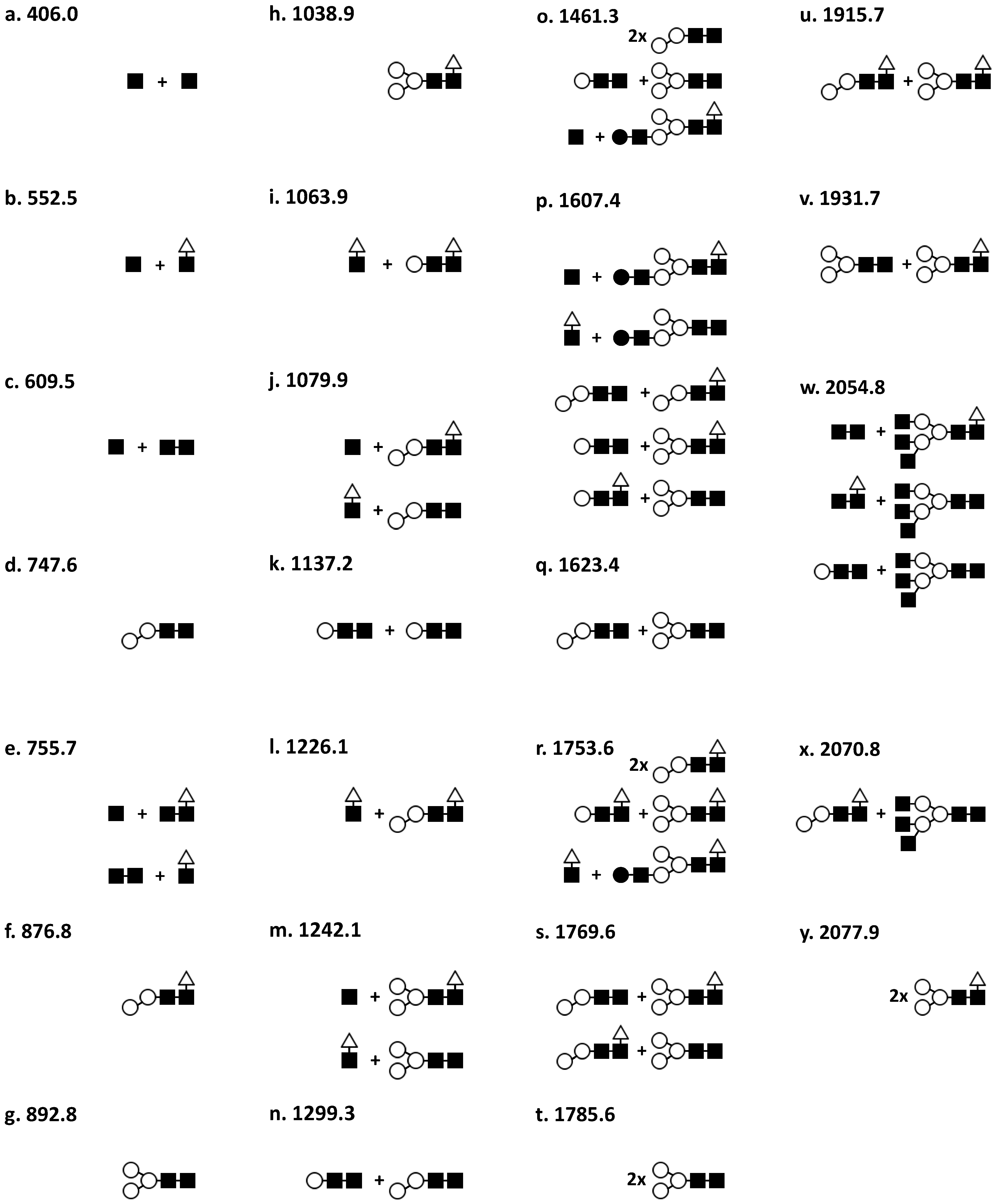
| Peak | Observed Mass (m/z) [M + H]+ | Predicted Carbohydrate Structure (Figure 2) | Predicted Mass (Da) of Glycopeptide | % Mass Match | % Abundance |
|---|---|---|---|---|---|
| N1bcf | |||||
| 1 | 4353.8 | c | 4361.9 | 0.9982 | 16.2 |
| 2 | 4634.7 | f | 4629.2 | 1.0012 | 5.1 |
| 3 | 4798.8 | h | 4790.8 | 1.0017 | 4.4 |
| 4 | 5220.8 | o | 5213.7 | 1.0014 | 3.2 |
| 5 | 5366.7 | p | 5359.8 | 1.0013 | 18.1 |
| 6 | 5529.1 | s | 5522.0 | 1.0013 | 22.8 |
| 7 | 5689.8 | v | 5684.1 | 1.0010 | 17.8 |
| 8 | 5840.6 | y | 5830.3 | 1.0018 | 12.4 |
| N2bcf | |||||
| 1 | 4307.7 | b | 4277.9 | 1.007 | 4.7 |
| 2 | 4477.6 | d | 4483.0 | 1.0012 | 35.2 |
| 3 | 4630.9 | g | 4630.9 | 1.0030 | 17.3 |
| 4 | 4837.4 | j | 4832.3 | 0.9989 | 6.6 |
| 5 | 4976.6 | l | 4978.5 | 1.0004 | 5.3 |
| 6 | 5074.3 | n | 5051.4 | 0.9955 | 7.5 |
| 7 | 5219.7 | o | 5213.7 | 0.9899 | 3.3 |
| 8 | 5352.6 | p | 5359.8 | 1.0013 | 5.9 |
| 9 | 5504.9 | r | 5506.0 | 1.0002 | 7.3 |
| 10 | 5672.8 | u | 5568.1 | 0.9992 | 5.3 |
| 11 | 5820.9 | x | 5823.2 | 1.0004 | 1.6 |
| M2bcf | |||||
| 1 | 4310 | b | 4304.9 | 0.9988 | 24 |
| 2 | 4515.2 | e | 4508.1 | 0.9984 | 0.3 |
| 3 | 4601.7 | f | 4628.4 | 1.0058 | 0.6 |
| 4 | 4805.8 | i | 4816.3 | 1.0022 | 13.1 |
| 5 | 4966.4 | m | 4994.5 | 1.0056 | 9.8 |
| 6 | 5219.1 | o | 5213.7 | 0.9970 | 1.7 |
| 7 | 5359.5 | p | 5359.8 | 1.0001 | 9.8 |
| 8 | 5518.9 | s | 5522.0 | 1.0006 | 13.4 |
| 9 | 5673 | u | 5668.1 | 0.9991 | 15.3 |
| 10 | 5796.1 | w | 5807.2 | 1.0019 | 12 |
| M4bcf | |||||
| 1 | 4256.3 | a | 4158.4 | 0.9770 | 25.6 |
| 2 | 4305.2 | b | 4304.9 | 0.9999 | 16.2 |
| 3 | 4369 | c | 4361.9 | 0.9984 | 24.5 |
| 4 | 4639.1 | f | 4629.2 | 0.9979 | 6.1 |
| 5 | 4801.5 | h | 4791.3 | 0.9979 | 8.1 |
| 6 | 5378.9 | q | 5375.8 | 0.9994 | 4.1 |
| 7 | 5535.7 | t | 5538.0 | 1.0004 | 7.3 |
| 8 | 5686.9 | v | 5684.1 | 0.9995 | 5.7 |
| 9 | 5824.9 | x | 5823.2 | 0.9997 | 2.4 |
| HGbcf | |||||
| 1 | 4265.4 | b | 4304.4 | 1.0091 | 12.2 |
| 2 | 4628.3 | f | 4629.2 | 1.0002 | 9.2 |
| 3 | 4790.2 | h | 4791.3 | 1.0002 | 8.9 |
| 4 | 4929.4 | k | 4889.6 | 0.9919 | 3.1 |
| 5 | 5361.6 | p | 5359.8 | 0.9997 | 13.5 |
| 6 | 5516.8 | s | 5522.0 | 1.0079 | 21.7 |
| 7 | 5675.7 | u | 5568.1 | 0.9987 | 14.9 |
| 8 | 5820.2 | x | 5823.2 | 1.0005 | 16.5 |
2.3. Relative Abundance of Glycoforms
2.4. Discussion
3. Materials and Methods
3.1. Biological Samples
3.2. Sample Treatments
3.3. hCGβcf Enzyme-Linked Immunosorbent Assay (ELISA)
3.4. Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry (MALDI TOF MS)
3.5. Whole Molecule hCGβcf Analysis (Non-Reduced)
3.6. Dithiothreitol (DTT)-Treated hCGβcf Analysis (Reduced)
3.7. Treatment of Spectra
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Smith, L.M.; Kelleher, N.L. Proteomics consortium down. Proteoform: A single term describing protein complexity. Nat. Methods 2013, 10, 186–187. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, N.; Wan, D.; Cui, M.; Liu, Z.; Liu, S. Mass spectrometry-based analysis of glycoproteins and its clinical applications in cancer biomarker discovery. Clin. Proteomics 2014, 11, 14. [Google Scholar]
- Kessler, M.J.; Reddy, M.S.; Shah, R.H.; Bahl, O.P. Structures of N-glycosidic carbohydrate units of human chorionic gonadotrophin. J. Biol. Chem. 1979, 254, 7901–7908. [Google Scholar]
- Boime, I.; Ben-Menahem, D.; Olijve, W. Studies of recombinant gonadotropins: Intersection of basic science and therapeutics. In Molecular Biology in Reproductive Medicine; Fauser, B.C.J.M., Rutherford, A.J., Strauss, J.F., van Steirteghem, A., Eds.; The Parthenon Publishing Group: London, UK, 1999; pp. 148–164. [Google Scholar]
- Kobata, A.; Takeuchi, M. Structure, pathology and function of the N-linked sugar chains of human chorionic gonadotropin. Biochim. Biophys. Acta 1999, 1455, 315–326. [Google Scholar]
- Kessler, M.J.; Mise, T.; Ghai, R.D.; Bahl, O.P. Structure and location of the O-glycosidic carbohydrate units of human chorionic gonadotrophin. J. Biol. Chem. 1979, 254, 7909–7914. [Google Scholar]
- Cole, L.A.; Birken, S.; Perini, F. The structures of the serine-linked sugar chains on human chorionic gonadotropin. Biochem. Biophys. Res. Commun. 1985, 126, 333–339. [Google Scholar] [CrossRef]
- Cole, L.A. Distribution of O-linked sugar units on hCG and its free α-subunit. Mol. Cell. Endocrinol. 1987, 50, 45–57. [Google Scholar] [CrossRef]
- Amano, J.; Nishimura, R.; Mochizuki, M.; Kobata, A. Comparative study of the mucin-type sugar chains of human chorionic gonadotropin present in the urine of patients with trophoblastic diseases and healthy pregnant women. J. Biol. Chem. 1988, 263, 1157–1165. [Google Scholar]
- Yoshimoto, Y.; Wolfsen, A.; Odell, W.D. Glycosylation, a variable in the production of hCG by cancers. Am. J. Med. 1979, 67, 414–420. [Google Scholar] [CrossRef]
- Mizuochi, T.; Nishimura, R.; Derappe, C.; Taniguchi, T.; Hamamoto, T.; Mochizuki, M.; Kobata, A. Structures of the asparagine-linked sugar chains of human chorionic gonadotropin produced in choriocarcinoma. J. Biol. Chem. 1983, 258, 14126–14129. [Google Scholar]
- Damm, J.B.L.; Voshol, H.; Hard, K.; Kamerling, J.P.; van Dedem, G.W.K.; Vliegenthart, J.F.G. The β-subunit of human chorionic gonadotropin contains N-glycosidictrisialo tri-antennary and tri-antennary carbohydrate chains. Glycoconj. J. 1988, 5, 221–233. [Google Scholar]
- Elliot, M.M.; Kardana, A.; Lustbader, J.W.; Cole, L.A. Carbohydrate and peptide structure of the α- and β-subunits of human chorionic gonadotropin from normal and aberrant pregnancy and choriocarcinoma. Endocrine 1997, 7, 15–32. [Google Scholar]
- Cole, L.A.; Cermik, D.; Bahado-Singh, R. Oligosaccharide variants of hCG-related molecules: Potential screening markers for Down syndrome. Prenat. Diagn. 1997, 17, 1187–1190. [Google Scholar] [CrossRef]
- Birken, S.; Armstrong, E.G.; Kolks, M.A.; Cole, L.A.; Agosto, G.M.; Krichevsky, A.; Vaitukaitis, J.L.; Canfield, R.E. Structure of the human chorionic gonadotrophin β-subunit fragment from pregnancy urine. Endocrinology 1988, 123, 572–583. [Google Scholar]
- Cole, L.A.; Tanaka, A.; Kim, G.S.; Park, S.Y.; Koh, M.W.; Schwartz, P.E.; Chambers, J.T.; Nam, J.H. β-Core fragment (β-core, UGP or UGF), a tumor marker: A 7-year report. Gynecol. Oncol. 1996, 60, 264–270. [Google Scholar] [CrossRef]
- Nisula, B.C.; Blithe, D.L.; Akar, A.; Lefort, G.; Wehmann, R.E. Metabolic fate of human choriogonadotropin. J. Steroid Biochem. 1989, 33, 733–737. [Google Scholar]
- Okamoto, T.; Matsuo, K.; Niu, R.; Osawa, M.; Suzuki, H. Human chorionic gonadotropin (hCG) β-core fragment is produced by degradation of hCG or free hCGβ in gestational trophoblastic tumors: A possible marker for early detection of persistent post molar gestational trophoblastic disease. J. Endocrinol. 2001, 171, 435–443. [Google Scholar] [CrossRef]
- Karas, M.; Hillenkamp, F. Laser desorption ionisation of proteins with molecular masses exceeding 10,000 daltons. Anal. Chem. 1988, 60, 2299–2301. [Google Scholar] [CrossRef]
- Dell, A.; Morris, H.R. Glycoprotein structure determination by mass spectrometry. Science 2001, 291, 2351–2356. [Google Scholar] [CrossRef]
- Sutton, C.W.; O’Neil, J.A.; Cottrell, J.S. Site-specific characterisation of glycoprotein carbohydrates by exoglycosidase digestion and laser desorption mass spectrometry. Anal. Biochem. 1994, 218, 34–46. [Google Scholar]
- Yang, Y.; Orlando, R. Identifying the glycosylation sites and site-specific carbohydrate heterogeneity of glycoproteins by matrix-assisted laser desorption/ionization mass spectrometry. Rapid Commun. Mass Spectrom. 1996, 10, 932–936. [Google Scholar] [CrossRef]
- Iwase, H.; Tanaka, A.; Hiki, Y.; Kokubo, T.; Ishii-Karakasa, I.; Nishikido, J.; Kobayashi, Y.; Hotta, K. Application of matrix-assisted laser desorption ionization time-of-flight mass spectrometry to the analysis of glycopeptide-containing multiple O-linked oligosaccharides. J. Chromatogr. B 1998, 709, 145–149. [Google Scholar] [CrossRef]
- Blithe, D.L.; Akar, A.H.; Wehmann, R.E.; Nisula, B.C. Purification of β-core fragment from pregnancy urine and demonstration that its carbohydrate moieties differ from those of native human chorionic gonadotropin-β. Endocrinology 1988, 122, 173–180. [Google Scholar]
- Blithe, D.L.; Wehmann, R.E.; Nisula, B.C. Carbohydrate composition of β-core. Endocrinology 1989, 125, 2267–2272. [Google Scholar] [CrossRef]
- Endo, T.; Nishimura, R.; Saito, S.; Kanazawa, K.; Nomura, K.; Katsuno, M.; Shii, K.; Mukhopadhyay, K.; Baba, S.; Kobata, A. Carbohydrate structures of β-core fragment of human chorionic gonadotropin isolated from a pregnant individual. Endocrinology 1992, 130, 2052–2058. [Google Scholar]
- De Medeiros, S.F.; Amato, F.; Matthews, C.D.; Norman, R.J. Molecular heterogeneity of the β-core fragment of human chorionic gonadotrophin. J. Endocrinol. 1993, 139, 519–532. [Google Scholar] [CrossRef]
- Jacoby, E.S.; Kicman, A.T.; Laidler, P.; Iles, R.K. Determination of the glycoforms of human chorionic gonadotrophin β-core fragment by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Clin. Chem. 2000, 46, 1796–1803. [Google Scholar]
- Waechter, C.J.; Lennarz, W.J. The role of polyprenol-linked sugars in glycoprotein synthesis. Annu. Rev. Biochem. 1976, 45, 95–112. [Google Scholar] [CrossRef]
- Kornfeld, S.; Li, E.; Tabas, I. The synthesis of complex-type oligosaccharides. II. Characterization of the processing intermediates in the synthesis of the complex oligosaccharide units of the vesicular stomatitis virus G protein. J. Biol. Chem. 1978, 253, 7771–7778. [Google Scholar]
- Hirschberg, C.B.; Snider, M.D. Topography of glycosylation in the rough endoplasmic reticulum and Golgi apparatus. Annu. Rev. Biochem. 1987, 56, 63–87. [Google Scholar] [CrossRef]
- Butler, S.A.; Khanlian, S.A.; Cole, L.A. Detection of early pregnancy forms of human chorionic gonadotropin by home pregnancy test devices. Clin. Chem. 2001, 47, 2131–2136. [Google Scholar]
- Cole, L.A.; Khanlian, S.A.; Sutton, J.M.; Davies, S.; Stephens, N.D. Hyperglycosylated hCG (invasive trophoblast antigen, ITA) a key antigen for early pregnancy detection. Clin. Biochem. 2003, 36, 647–655. [Google Scholar] [CrossRef]
- Birken, S.; Berger, P.; Bidart, J.M.; Weber, M.; Bristow, A.; Norman, R.; Sturgeon, C.; Stenman, U.H. Preparation and characterization of new WHO reference reagents for human chorionic gonadotropin and metabolites. Clin. Chem. 2003, 49, 144–154. [Google Scholar] [CrossRef]
- Berger, P.; Sturgeon, C.; Bidart, J.M.; Paus, E.; Gerth, R.; Niang, M.; Bristow, A.; Birken, S.; Stenman, U.H. The ISOBM TD-7 Workshop on hCG and related molecules. Towards user-oriented standardization of pregnancy and tumor diagnosis: Assignment of epitopes to the three-dimensional structure of diagnostically and commercially relevant monoclonal antibodies directed against human chorionic gonadotropin and derivatives. Tumor Biol. 2002, 23, 1–38. [Google Scholar]
- Canfield, R.E.; Ross, G.T. A new reference preparation of human chorionic gonadotrophin and its subunits. Bull. World Health Organ. 1976, 54, 463–472. [Google Scholar]
- Berger, P.; Bidart, J.M.; Delves, P.S.; Dirnhofer, S.; Hoermann, R.; Isaacs, N.; Jackson, A.; Klonisch, T.; Lapthorn, A.; Lund, T.; et al. Immunochemical mapping of gonadotropins. Mol. Cell. Endocrinol. 1996, 125, 33–43. [Google Scholar] [CrossRef]
- Lee, C.L.; Iles, R.K.; Shepherd, J.H.; Hudson, C.N.; Chard, T. The purification and development of a radioimmunoassay for β-core fragment of human chorionic gonadotrophin in urine: Application as a marker of gynaecological cancer in premenopausal and postmenopausal women. J. Endocrinol. 1991, 130, 481–489. [Google Scholar] [CrossRef]
- Jacoby, E.S.; Kicman, A.T.; Iles, R.K. Identification of post-translational modifications resulting from LHβ polymorphisms by matrix-assisted laser desorption time-of-flight mass spectrometric analysis of pituitary LHβ core fragment. J. Mol. Endocrinol. 2003, 30, 239–252. [Google Scholar] [CrossRef]
- Neubert, H.; Jacoby, E.S.; Bansal, S.S.; Iles, R.K.; Cowan, D.A.; Kicman, A.T. Enhanced affinity capture MALDI TOF MS: Orientation of an immunoglobulin G using recombinant protein G. Anal. Chem. 2002, 74, 3677–3683. [Google Scholar] [CrossRef]
- Wuhrer, M. Glycomics using mass spectrometry. Glycoconj. J. 2013, 30, 11–22. [Google Scholar] [CrossRef]
- Baycin Hizal, D.; Wolozny, D.; Colao, J.; Jacobson, E.; Tian, Y.; Krag, S.S.; Betenbaugh, M.J.; Zhang, H. Glycoproteomic and glycomic databases. Clin. Proteomics 2014, 11, 15. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Iles, R.K.; Cole, L.A.; Butler, S.A. Direct Analysis of hCGβcf Glycosylation in Normal and Aberrant Pregnancy by Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry. Int. J. Mol. Sci. 2014, 15, 10067-10082. https://doi.org/10.3390/ijms150610067
Iles RK, Cole LA, Butler SA. Direct Analysis of hCGβcf Glycosylation in Normal and Aberrant Pregnancy by Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry. International Journal of Molecular Sciences. 2014; 15(6):10067-10082. https://doi.org/10.3390/ijms150610067
Chicago/Turabian StyleIles, Ray K., Laurence A. Cole, and Stephen A. Butler. 2014. "Direct Analysis of hCGβcf Glycosylation in Normal and Aberrant Pregnancy by Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry" International Journal of Molecular Sciences 15, no. 6: 10067-10082. https://doi.org/10.3390/ijms150610067





