Mangiferin Facilitates Islet Regeneration and β-Cell Proliferation through Upregulation of Cell Cycle and β-Cell Regeneration Regulators
Abstract
:1. Introduction
2. Results
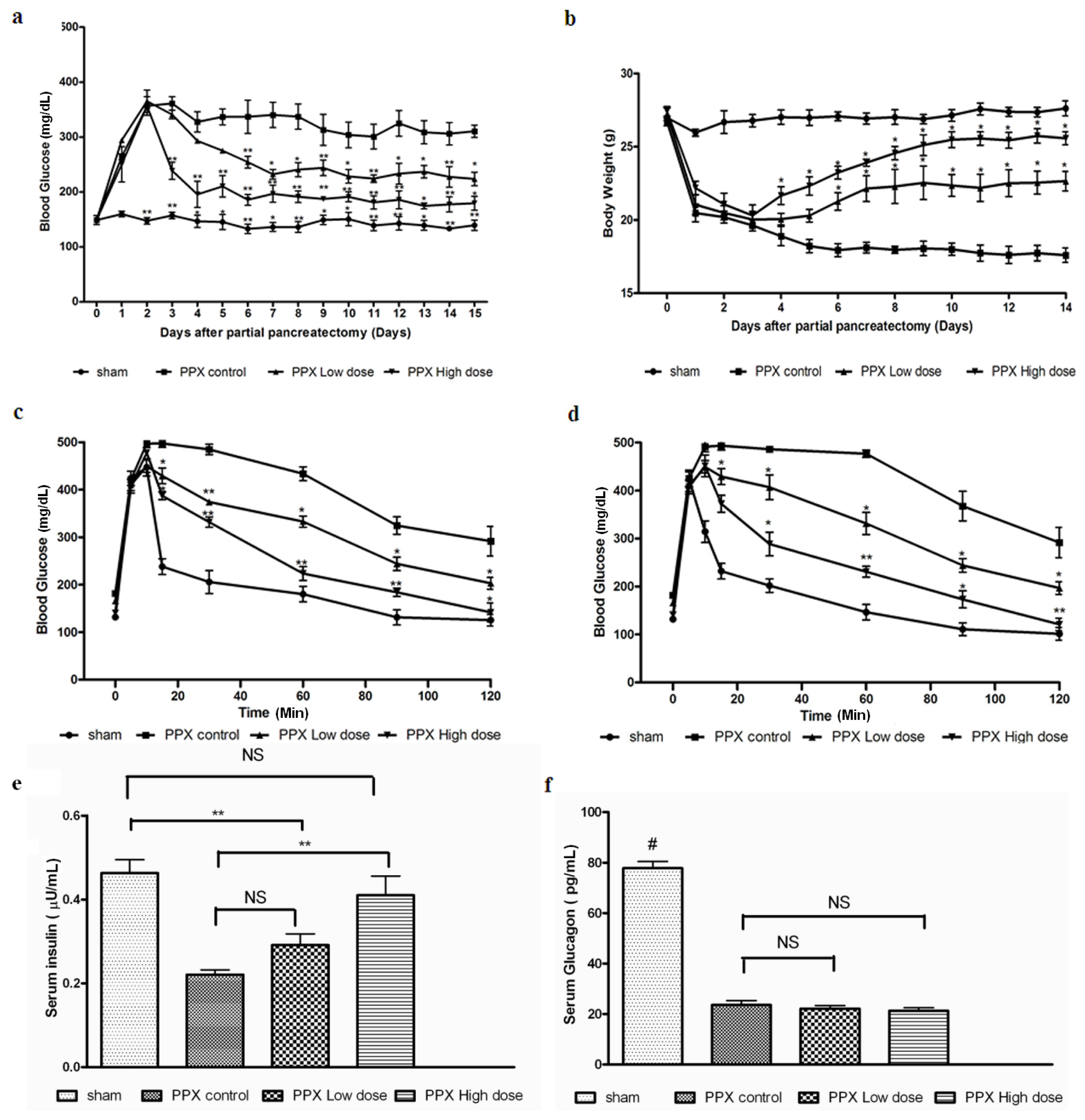
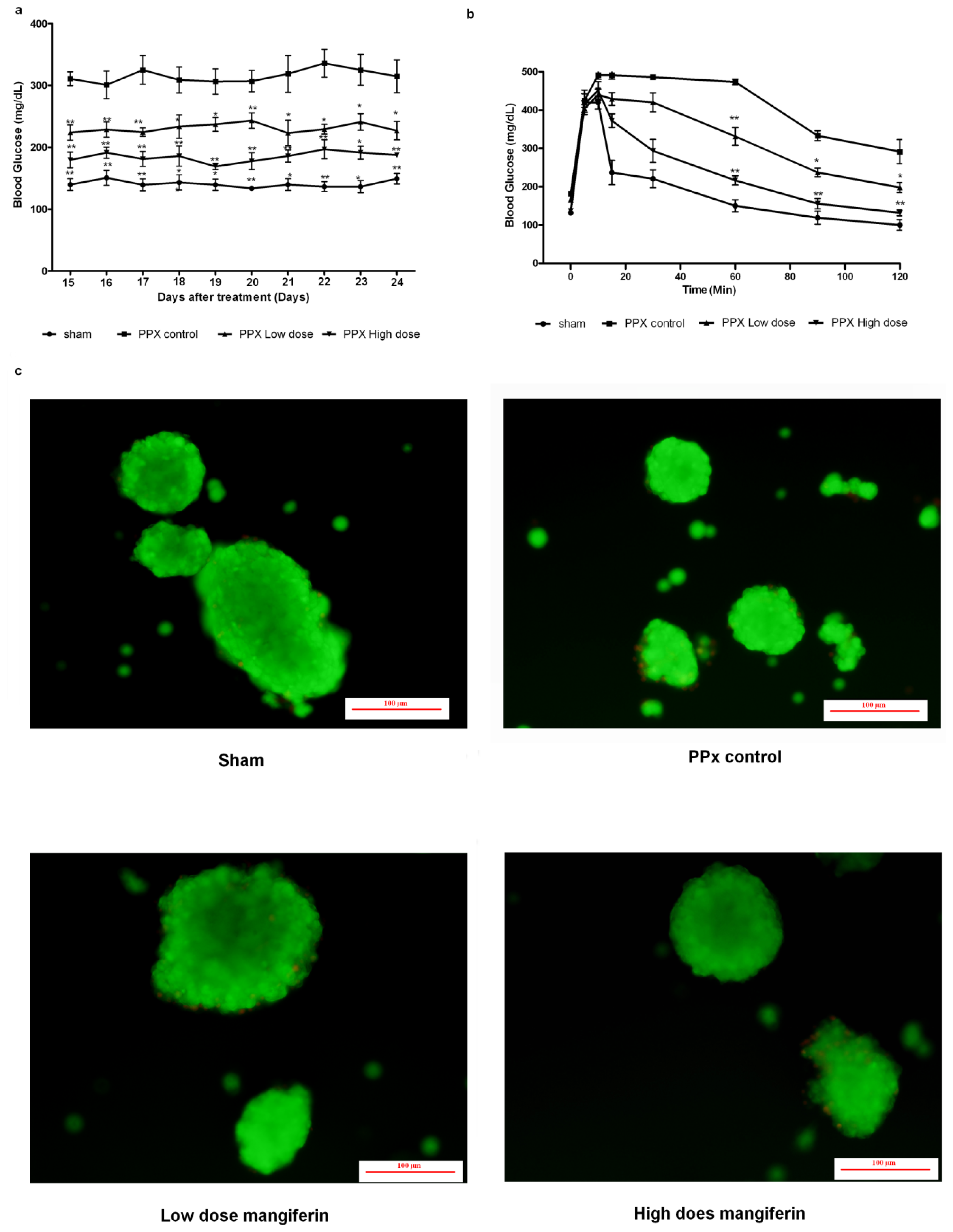
2.1. Mangiferin Induces an Increased Insulin Release to Promote Glucose Homeostasis
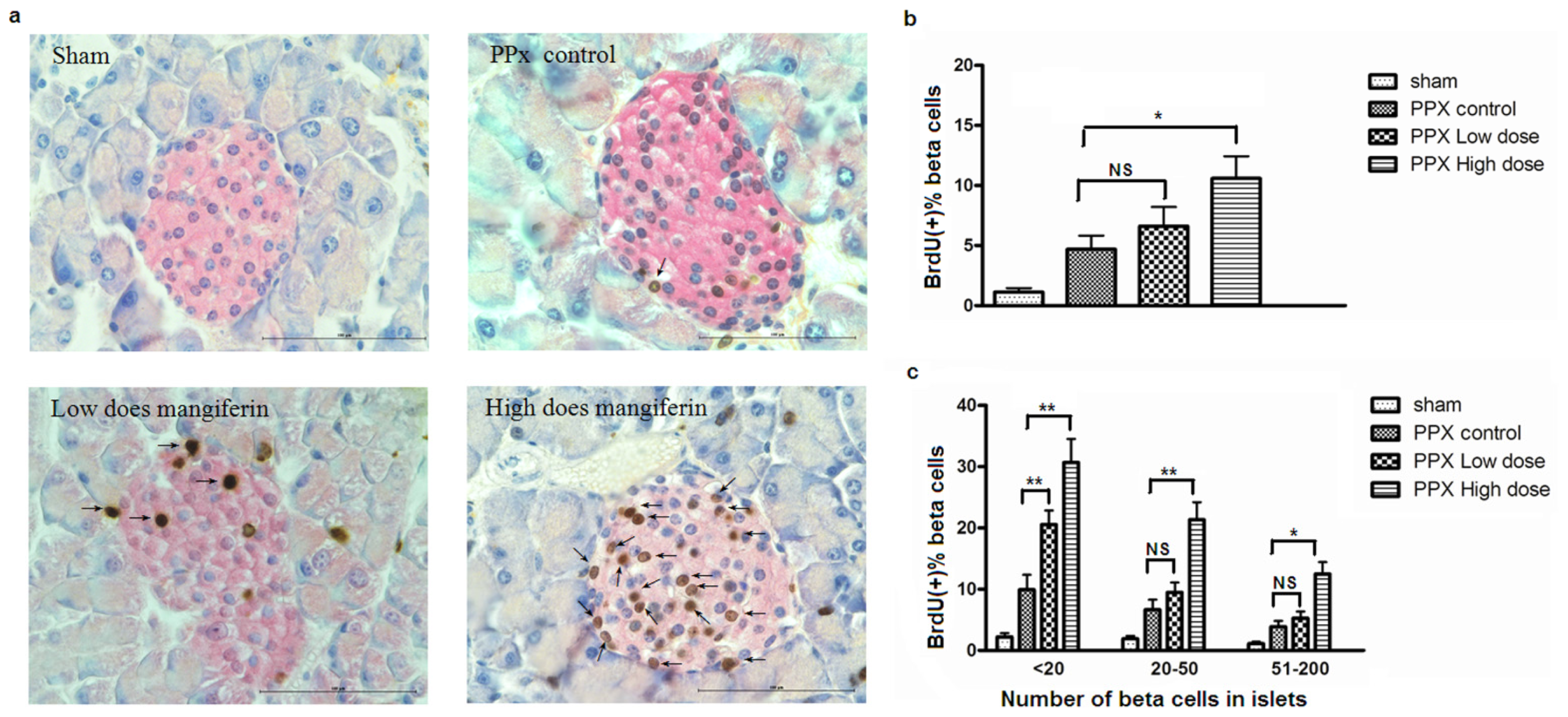
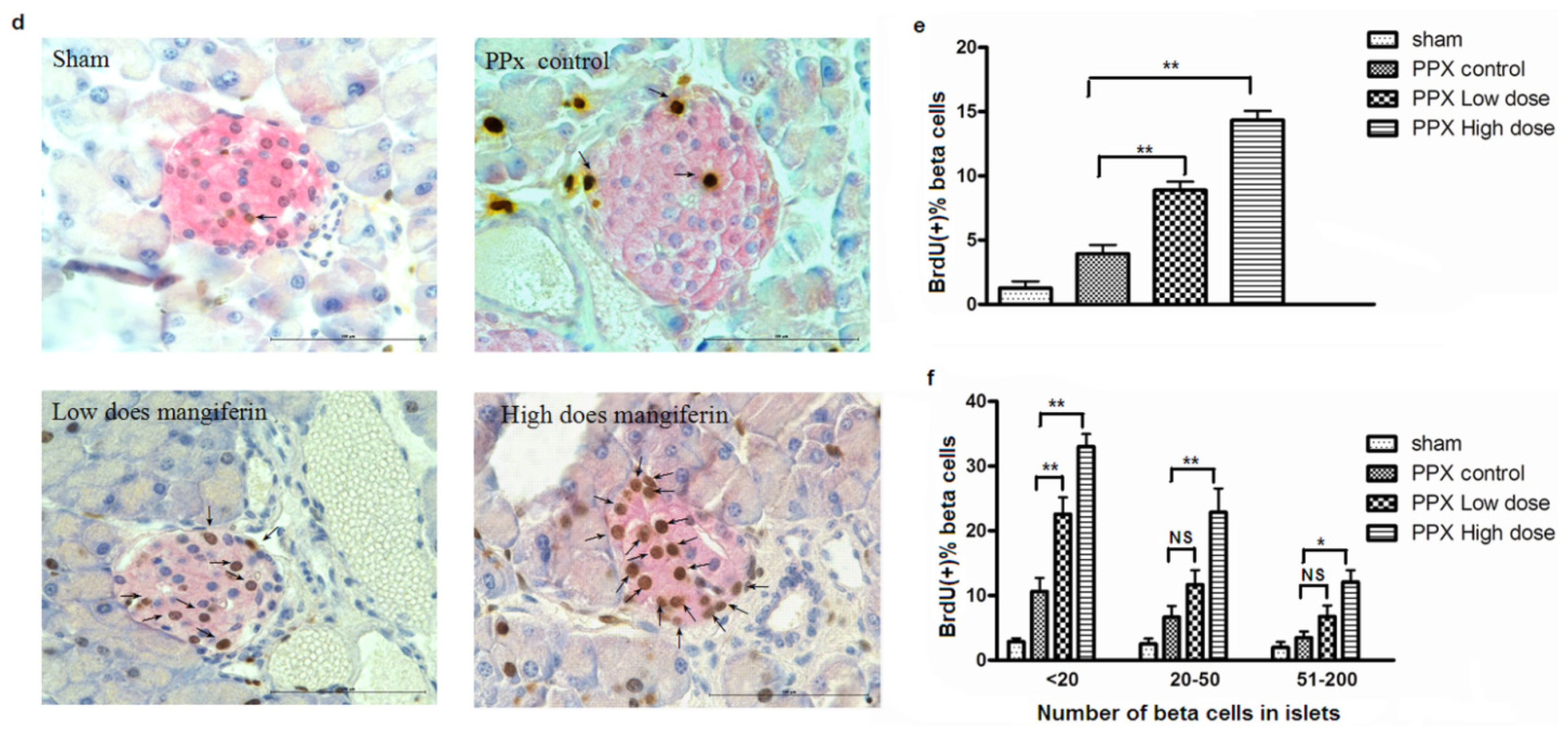
2.2. Mangiferin Treatment Enhances Proliferation of β-Cells and Duct Cells
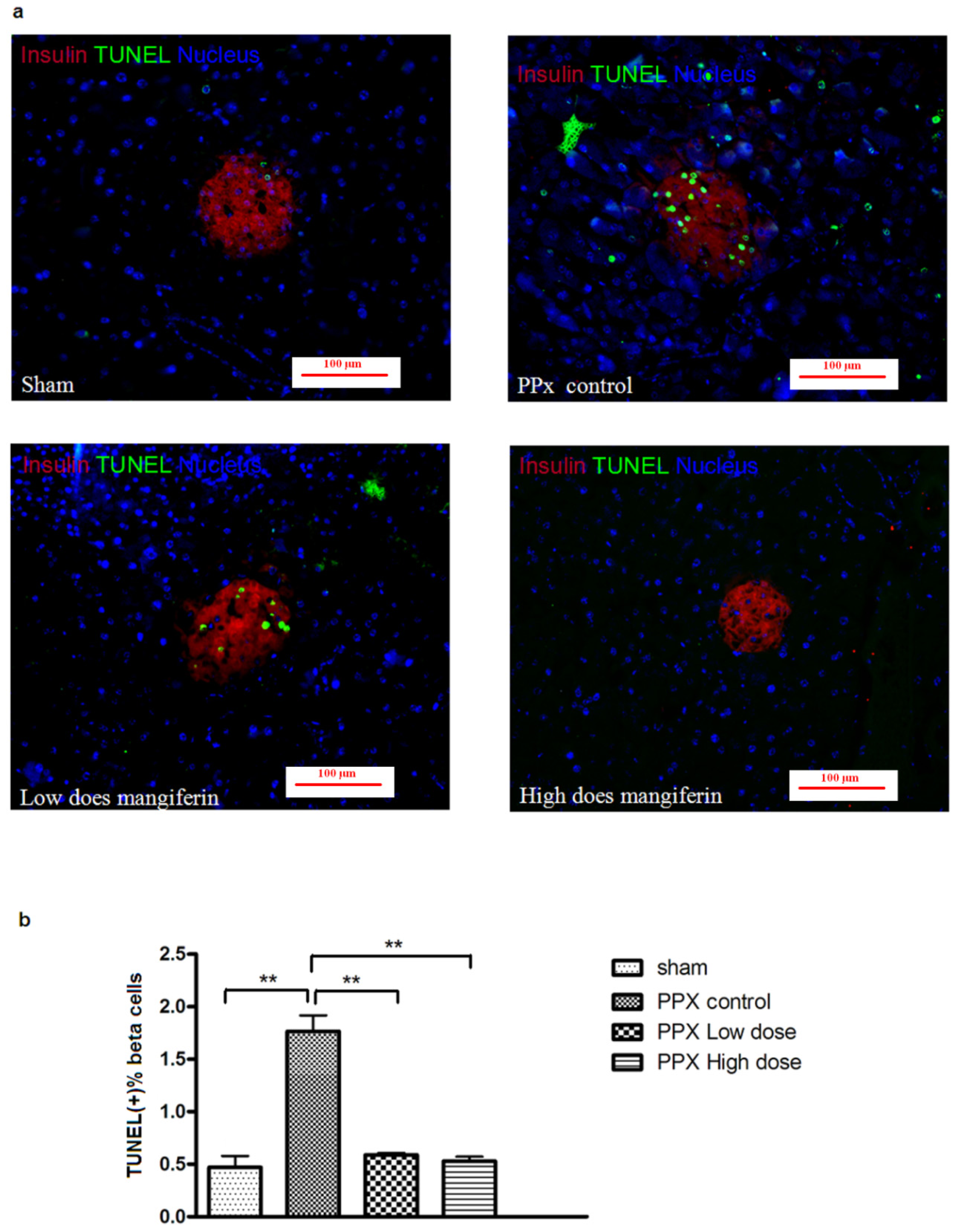
2.3. β-Cell Apoptosis Is Inhibited by Mangiferin
2.4. Mangiferin Increases β-Cell Mass
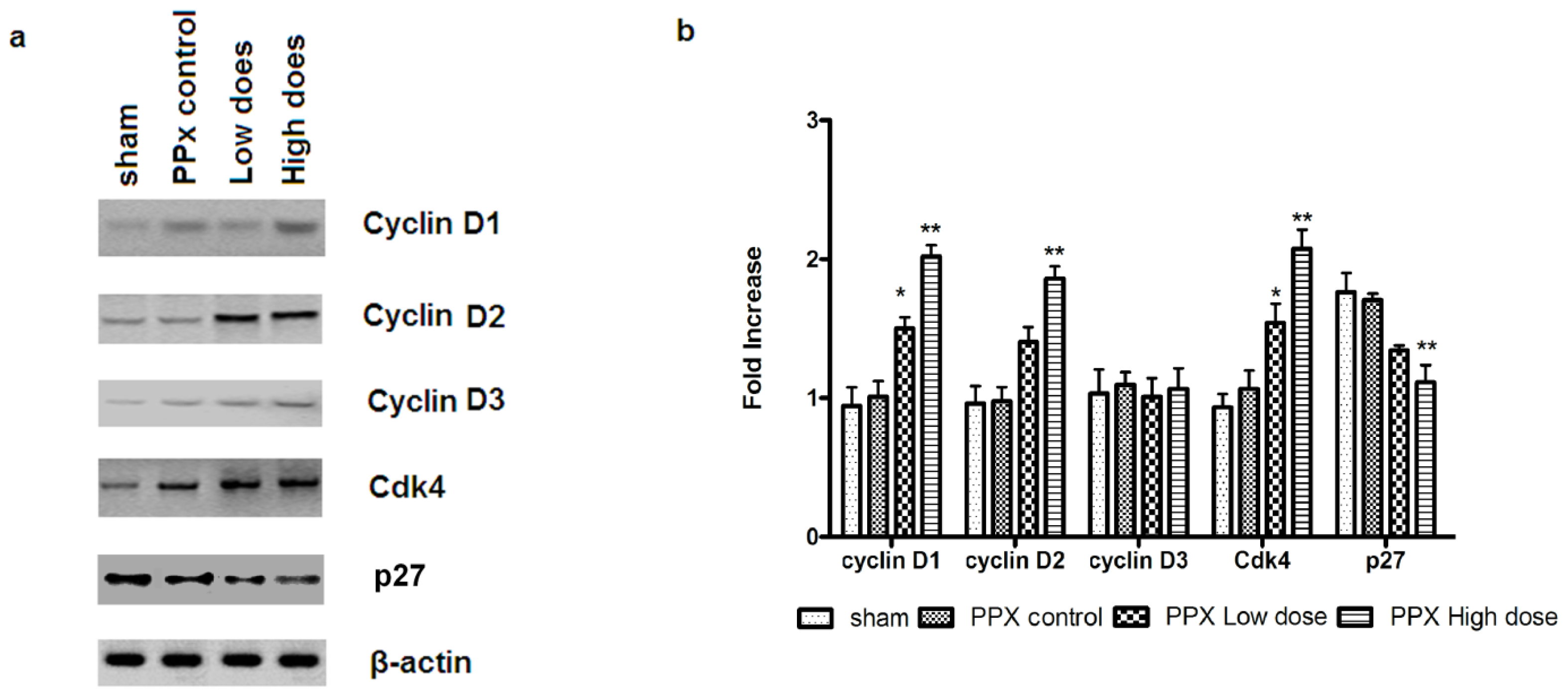
2.5. Mangiferin Up-Regulates both Expression and Activity of Cell Cycle Regulators
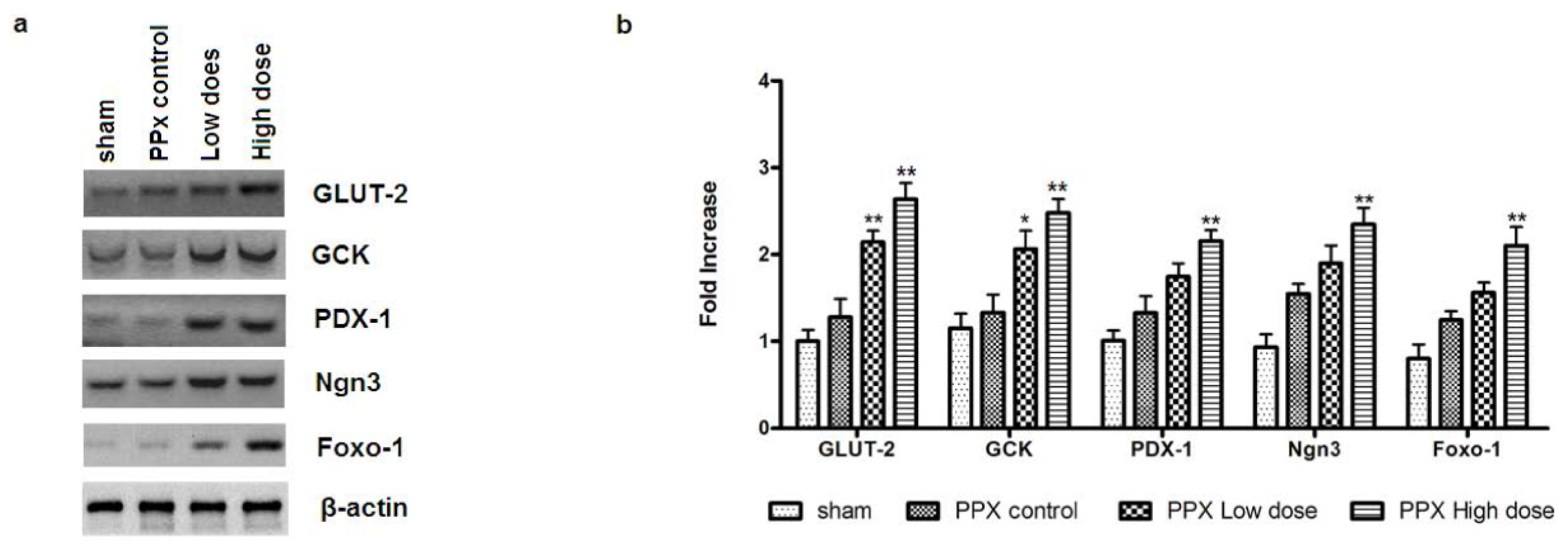
2.6. Mangiferin Modulates the Expression of β-Cell-Specific Genes
2.7. Islet Viability Analysis
3. Discussion
4. Materials and Methods
4.1. Study Design
4.2. Animal Surgery
4.3. Biochemical Measurements
4.4. Immunoblotting
4.5. Quantitative Reverse Transcription Polymerase Chain Reaction Analysis
4.6. Cdk4 Kinase Activity
4.7. Histology Staining
4.8. Islet Viability Detection
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Conflicts of Interest
Abbreviations
| PPx | partial pancreatectomy |
| STZ | streptozotocin |
| CPK | creatine phosphokinase |
| DMSO | dimethyl sulfoxide |
| BrdU | bromodeoxyuridine |
| IVGTTs | Intravenous glucose tolerance tests |
| ELISA | enzyme-linked immunosorbent assay |
| real-time PCR | real-time polymerase chain reaction |
| SD-PAGE | sodium dodecylsulfate polyacrylamide gel electrophoresis |
| TUNEL | transferase-mediated dUTP nick-end labeling |
| Cdk4 | cyclin-dependent kinase 4 |
| GLUT-2 | glucose transporter 2 |
| GCK | glucokinase |
| PDX-1 | pancreatic and duodenal homeobox 1 |
| Ngn3 | neurogenin 3 |
| Foxo-1 | Forkhead box protein O1 |
| DN | diabetic nephropathy |
| T1DM | type 1 diabetes mellitus |
| T2DM | type 2 diabetes mellitus |
| TCM | traditional Chinese medicine |
- Author ContributionsHai-lian Wang, Jin-ku Bao and Yi Wang designed the research; Chun-yang Li and Bin Zhang performed the experiments; Zheng shi, Na An, Liang-Kai Zhao, Yuan-de Liu and Jing-jing Zhang contributed to the analysis of the data; Hai-lian Wang, Chun-yang Li, Jin-ku Bao and Yi Wang wrote the paper.
References
- Dean, P.G.; Kudva, Y.C.; Stegall, M.D. Long-term benefits of pancreas transplantation. Curr. Opin. Organ Tranplant 2008, 13, 85–90. [Google Scholar]
- Shapiro, A.J.; Ricordi, C.; Hering, B.J.; Auchincloss, H.; Lindblad, R.; Robertson, R.P.; Secchi, A.; Brendel, M.D.; Berney, T.; Brennan, D.C. International trial of the Edmonton protocol for islet transplantation. N. Engl. J. Med 2006, 355, 1318–1330. [Google Scholar]
- Bonner-Weir, S.; Sharma, A. Are there pancreatic progenitor cells from which new islets form after birth? Nat. Clin. Pract 2006, 2, 240–224. [Google Scholar]
- Palmer, J.P.; McCulloch, D.K. Prediction and prevention of IDDM—1991. Diabetes 1991, 40, 943–947. [Google Scholar]
- Mauricio, D.; Mandrup-Poulsen, T. Apoptosis and the pathogenesis of IDDM: A question of life and death. Diabetes 1998, 47, 1537–1543. [Google Scholar]
- Bergada, I.; Suissa, S.; Dufresne, J.; Schiffrin, A. Severe hypoglycemia in IDDM children. Diabetes Care 1989, 12, 239–244. [Google Scholar]
- Xu, X.; D’Hoker, J.; Stangé, G.; Bonné, S.; de Leu, N.; Xiao, X.; van de Casteele, M.; Mellitzer, G.; Ling, Z.; Pipeleers, D.; et al. β-Cells can be generated from endogenous progenitors in injured adult mouse pancreas. Cell 2008, 132, 197–207. [Google Scholar]
- Dar, A.; Faizi, S.; Naqvi, S.; Roome, T.; Zikr-ur-Rehman, S.; Ali, M.; Firdous, S.; Moin, S.T. Analgesic and antioxidant activity of mangiferin and its derivatives: The structure activity relationship. Biol. Pharm. Bull 2005, 28, 596–600. [Google Scholar]
- Guha, S.; Ghosal, S.; Chattopadhyay, U. Antitumor, immunomodulatory and anti-HIV effect of mangiferin, a naturally occurring glucosylxanthone. Chemotherapy 1996, 42, 443–451. [Google Scholar]
- Duang, X.Y.; Wang, Q.; Zhou, X.D.; Huang, D.M. Mangiferin: A possible strategy for periodontal disease to therapy. Med. Hypotheses 2011, 76, 486–488. [Google Scholar]
- Muruganandan, S.; Srinivasan, K.; Gupta, S.; Gupta, P.; Lal, J. Effect of mangiferin on hyperglycemia and atherogenicity in streptozotocin diabetic rats. J. Ethnopharmacol 2005, 97, 497–501. [Google Scholar]
- Muruganandan, S.; Gupta, S.; Kataria, M.; Lal, J.; Gupta, P. Mangiferin protects the streptozotocin-induced oxidative damage to cardiac and renal tissues in rats. Toxicology 2002, 176, 165–173. [Google Scholar]
- Bwititi, P.; Musabayane, C.; Nhachi, C. Effects of Opuntia megacantha on blood glucose and kidney function in streptozotocin diabetic rats. J. Ethnopharmacol 2000, 69, 247–252. [Google Scholar]
- Jouad, H.; Eddouks, M.; Lacaille-Dubois, M.A.; Lyoussi, B. Hypoglycaemic effect of Spergularia purpurea in normal and streptozotocin-induced diabetic rats. J. Ethnopharmacol 2000, 71, 169–177. [Google Scholar]
- Miura, T.; Ichiki, H.; Iwanmoto, N.; Kato, M.; Kubo, M.; Sasaki, H.; Okada, M.; Ishida, T.; Seino, Y.; Tanigawa, K. Antidiabetic activity of the rhizoma of Anemarrhena asphodeloides and active components, mangiferin and its glucoside. Biol. Pharm. Bull 2001, 24, 1009–1011. [Google Scholar]
- Miura, T.; Iwamoto, N.; Kato, M.; Ichiki, H.; Kubo, M.; Komatsu, Y.; Ishida, T.; Okada, M.; Tanigawa, K. The suppressive effect of mangiferin with exercise on blood lipids in type 2 diabetes. Biol. Pharm. Bull 2001, 24, 1091–1092. [Google Scholar]
- Wang, Y.; Liu, Y.; Wang, H.; Li, C.; Qi, P.; Bao, J. Agaricus bisporus lectins mediates islet β-cell proliferation through regulation of cell cycle proteins. Exp. Biol. Med 2012, 237, 287–296. [Google Scholar]
- Fatrai, S.; Elghazi, L.; Balcazar, N.; Cras-Méneur, C.; Krits, I.; Kiyokawa, H.; Bernal-Mizrachi, E. Akt induces β-cell proliferation by regulating cyclin D1, cyclin D2, and p21 levels and cyclin-dependent kinase-4 activity. Diabetes 2006, 55, 318–325. [Google Scholar]
- Matsushime, H.; Quelle, D.; Shurtleff, S.; Shibuya, M.; Sherr, C.; Kato, J. D-type cyclin-dependent kinase activity in mammalian cells. Mol. Cell. Biol 1994, 14, 2066–2076. [Google Scholar]
- Kim, S.Y.; Rane, S.G. The Cdk4-E2F1 pathway regulates early pancreas development by targeting Pdx1+ progenitors and Ngn3+ endocrine precursors. Development 2011, 138, 1903–1912. [Google Scholar]
- Kim, Y.C.; Kim, S.Y.; Mellado-Gil, J.M.; Yadav, H.; Neidermyer, W.; Kamaraju, A.K.; Rane, S.G. RB regulates pancreas development by stabilizing Pdx1. EMBO J 2011, 30, 1563–1576. [Google Scholar]
- Chappell, J.H.; Wang, X.D.; Loeken, M.R. Diabetes and apoptosis: Neural crest cells and neural tube. Apoptosis 2009, 14, 1472–1483. [Google Scholar]
- Freinkel, N. Banting Lecture 1980: Of pregnancy and progeny. Diabetes 1980, 29, 1023–1035. [Google Scholar]
- Rossini, A.A.; Mordes, J.P.; Like, A.A. Immunology of insulin-dependent diabetes mellitus. Annu. Rev. Immunol 1985, 3, 289–320. [Google Scholar]
- Kahn, S.E. The importance of β-cell failure in the development and progression of type 2 diabetes. J. Clin. Endocrinol. Metab 2001, 86, 4047–4058. [Google Scholar]
- Kahn, B.B.; Flier, J.S. Obesity and insulin resistance. J. Clin. Investig 2000, 106, 473–481. [Google Scholar]
- Brownlee, M. Biochemistry and molecular cell biology of diabetic complications. Nature 2001, 414, 813–820. [Google Scholar]
- Nishikawa, T.; Edelstein, D.; Du, X.L.; Yamagishi, S.I.; Matsumura, T.; Kaneda, Y.; Yorek, M.A.; Beebe, D.; Oates, P.J.; Hammes, H.P. Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature 2000, 404, 787–790. [Google Scholar]
- Mehta, J.L.; Rasouli, N.; Sinha, A.K.; Molavi, B. Oxidative stress in diabetes: A mechanistic overview of its effects on atherogenesis and myocardial dysfunction. Int. J. Biochem. Cell Biol 2006, 38, 794–803. [Google Scholar]
- Scott, L.J.; Mohlke, K.L.; Bonnycastle, L.L.; Willer, C.J.; Li, Y.; Duren, W.L.; Erdos, M.R.; Stringham, H.M.; Chines, P.S.; Jackson, A.U. A genome-wide association study of type 2 diabetes in Finns detects multiple susceptibility variants. Science 2007, 316, 1341–1345. [Google Scholar]
- Frayling, T.M.; Timpson, N.J.; Weedon, M.N.; Zeggini, E.; Freathy, R.M.; Lindgren, C.M.; Perry, J.R.; Elliott, K.S.; Lango, H.; Rayner, N.W. A common variant in the FTO gene is associated with body mass index and predisposes to childhood and adult obesity. Science 2007, 316, 889–894. [Google Scholar]
- Kushner, J.A.; Ciemerych, M.A.; Sicinska, E.; Wartschow, L.M.; Teta, M.; Long, S.Y.; Sicinski, P.; White, M.F. Cyclins D2 and D1 are essential for postnatal pancreatic β-cell growth. Mol. Cell. Biol 2005, 25, 3752–3762. [Google Scholar]
- Maruo, S.; Wu, Y.; Ishikawa, S.; Kanda, T.; Iwakiri, D.; Takada, K. Epstein–Barr virus nuclear protein EBNA3C is required for cell cycle progression and growth maintenance of lymphoblastoid cell. Proc. Natl. Acad. Sci. USA 2006, 103, 19500–19505. [Google Scholar]
- Krishnamurthy, J.; Ramsey, M.R.; Ligon, K.L.; Torrice, C.; Koh, A.; Bonner-Weir, S.; Sharpless, N.E. p16INK4a induces an age-dependent decline in islet regenerative potential. Nature 2006, 443, 453–457. [Google Scholar]
- Tsutsui, T.; Hesabi, B.; Moons, D.S.; Pandolfi, P.P.; Hansel, K.S.; Koff, A.; Kiyokawa, H. Targeted disruption of Cdk4 delays cell cycle entry with enhanced p27Kip1 activity. Mol. Cell. Biol 1999, 19, 7011–7019. [Google Scholar]
- Rane, S.G.; Dubus, P.; Mettus, R.V.; Galbreath, E.J.; Boden, G.; Reddy, E.P.; Barbacid, M. Loss of Cdk4 expression causes insulin-deficient diabetes and Cdk4 activation results in β-islet cell hyperplasia. Nat. Genet 1999, 22, 44–52. [Google Scholar]
- Kulkarni, R.N.; Jhala, U.S.; Winnay, J.N.; Krajewski, S.; Montminy, M.; Kahn, C.R. PDX-1 haploinsufficiency limits the compensatory islet hyperplasia that occurs in response to insulin resistance. J. Clin. Investig 2004, 114, 828–836. [Google Scholar]
- Kushner, J.A.; Ye, J.; Schubert, M.; Burks, D.J.; Dow, M.A.; Flint, C.L.; Dutta, S.; Wright, C.V.; Montminy, M.R.; White, M.F. PDX-1 restores β cell function in Irs2 knockout mice. J. Clin. Investig 2002, 109, 1193–1201. [Google Scholar]
- Faustman, D.; Hauptfeld, V.; Lacy, P.; Davie, J. Prolongation of murine islet allograft survival by pretreatment of islets with antibody directed to Ia determinants. Proc. Natl. Acad. Sci. USA 1981, 78, 5156–5159. [Google Scholar]
- Coulombe, M.G.; Warnock, G.L.; Rajotte, R.V. Prolongation of islet xenograft survival by cryopreservation. Diabetes 1987, 36, 1086–1088. [Google Scholar]
- Wang, Y.; Wang, H.; Liu, Y.; Li, C.; Qi, P.; Bao, J. Antihyperglycemic effect of ginsenoside Rh2 by inducing islet β-cell regeneration in mice. Horm. Metab. Res 2012, 44, 33–40. [Google Scholar]



© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, H.-L.; Li, C.-Y.; Zhang, B.; Liu, Y.-D.; Lu, B.-M.; Shi, Z.; An, N.; Zhao, L.-K.; Zhang, J.-J.; Bao, J.-K.; et al. Mangiferin Facilitates Islet Regeneration and β-Cell Proliferation through Upregulation of Cell Cycle and β-Cell Regeneration Regulators. Int. J. Mol. Sci. 2014, 15, 9016-9035. https://doi.org/10.3390/ijms15059016
Wang H-L, Li C-Y, Zhang B, Liu Y-D, Lu B-M, Shi Z, An N, Zhao L-K, Zhang J-J, Bao J-K, et al. Mangiferin Facilitates Islet Regeneration and β-Cell Proliferation through Upregulation of Cell Cycle and β-Cell Regeneration Regulators. International Journal of Molecular Sciences. 2014; 15(5):9016-9035. https://doi.org/10.3390/ijms15059016
Chicago/Turabian StyleWang, Hai-Lian, Chun-Yang Li, Bin Zhang, Yuan-De Liu, Bang-Min Lu, Zheng Shi, Na An, Liang-Kai Zhao, Jing-Jing Zhang, Jin-Ku Bao, and et al. 2014. "Mangiferin Facilitates Islet Regeneration and β-Cell Proliferation through Upregulation of Cell Cycle and β-Cell Regeneration Regulators" International Journal of Molecular Sciences 15, no. 5: 9016-9035. https://doi.org/10.3390/ijms15059016



