Autophagy-Related Direct Membrane Import from ER/Cytoplasm into the Vacuole or Apoplast: A Hidden Gateway also for Secondary Metabolites and Phytohormones?
Abstract
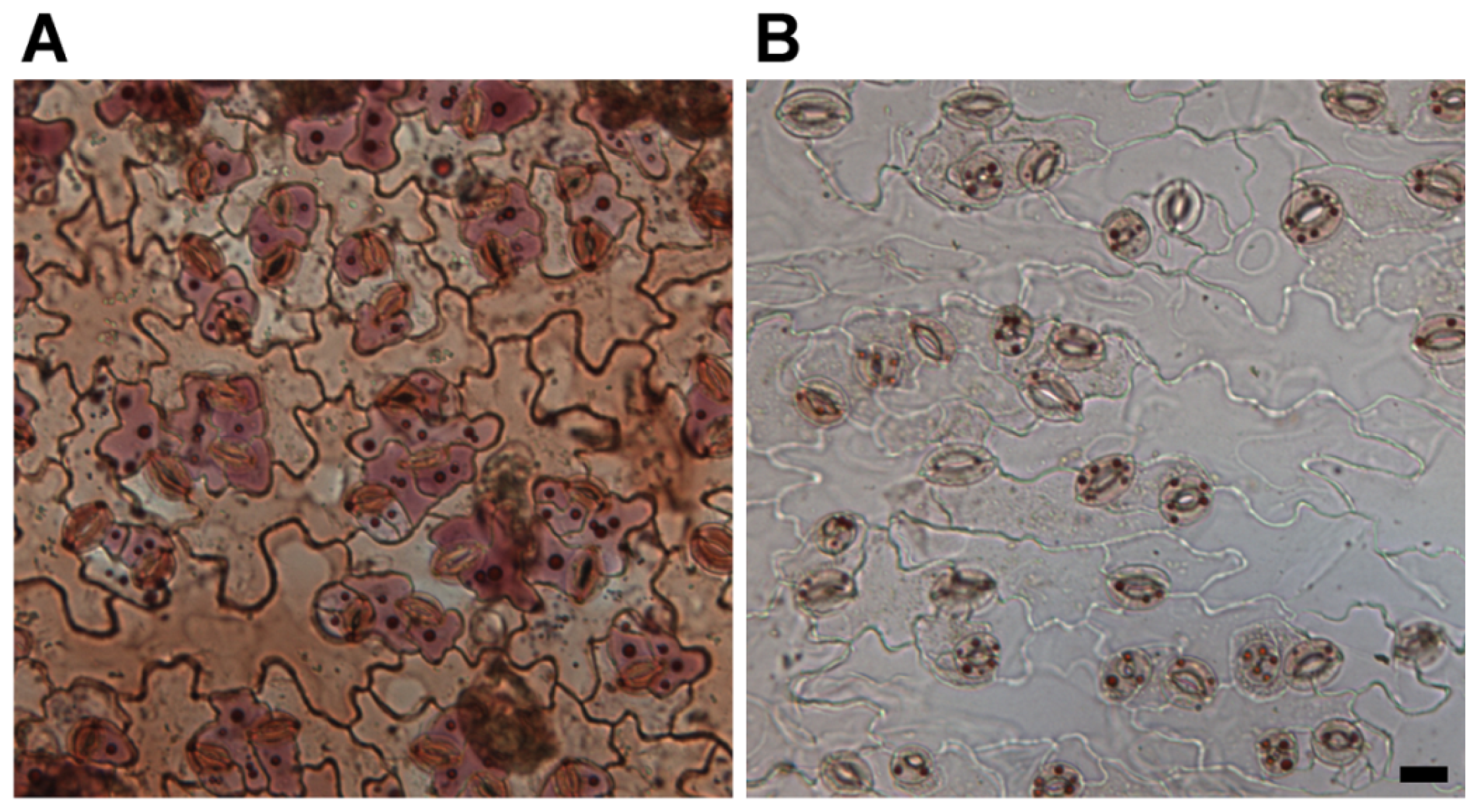
:1. Introduction: Intravacuolar Neutral Red Stained Bodies Have an Autophagic Origin
2. Anthocyanins and the Anthocyanin Vesicular Inclusions
3. Autophagic Tubes in Plants: Microautophagy with Macro Consequences?
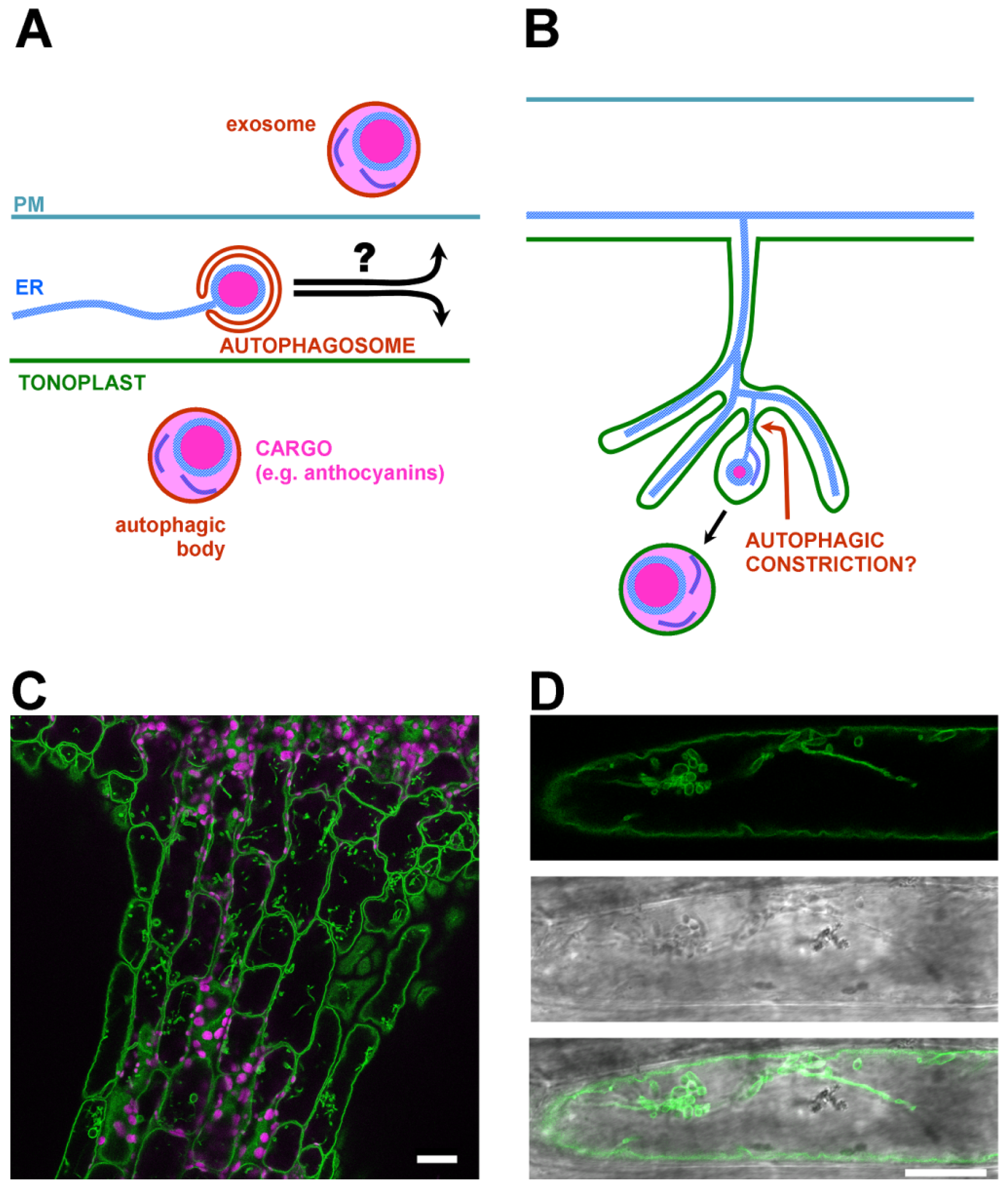
4. Endoplasmic Reticulum Initiated Autophagy as a General Pathway for the Delivery of Secondary Metabolites and Phytohormones into the Vacuole and Apoplast?
5. Putative Autophagy-Related Export of Secondary Metabolites to the Apoplast/Cell Wall
6. Conclusions
Acknowledgments
Conflicts of Interest
- Author ContributionsBoth authors Ivan Kulich and Viktor Žárský contributed equally to the published results critical discussion, new concepts and hypotheses development as well as writing.
References
- Robinson, D.G.; Jiang, L.; Schumacher, K. The endosomal system of plants: Charting new and familiar territories. Plant Physiol 2008, 147, 1482–1492. [Google Scholar]
- Frigerio, L.; Hinz, G.; Robinson, D.G. Multiple vacuoles in plant cells: Rule or exception? Traffic 2008, 9, 1564–1570. [Google Scholar]
- Foresti, O.; Denecke, J. Intermediate organelles of the plant secretory pathway: Identity and function. Traffic 2008, 9, 1599–1612. [Google Scholar]
- Zouhar, J.; Rojo, E. Plant vacuoles: Where did they come from and where are they heading? Curr. Opin. Plant Biol 2009, 12, 677–684. [Google Scholar]
- Bottanelli, F.; Foresti, O.; Hanton, S.; Denecke, J. Vacuolar transport in tobacco leaf epidermis cells involves a single route for soluble cargo and multiple routes for membrane cargo. Plant Cell 2011, 23, 3007–3025. [Google Scholar]
- Liu, Y.; Bassham, D.C. Autophagy: Pathways for self-eating in plant cells. Annu. Rev. Plant Biol 2012, 63, 215–237. [Google Scholar]
- Pourcel, L.; Irani, N.G.; Lu, Y.; Riedl, K.; Schwartz, S.; Grotewold, E. The formation of anthocyanic vacuolar inclusions in Arabidopsis thaliana and implications for the sequestration of anthocyanin pigments. Mol. Plant 2010, 3, 78–90. [Google Scholar]
- Inoue, Y.; Suzuki, T.; Hattori, M.; Yoshimoto, K.; Ohsumi, Y.; Moriyasu, Y. AtATG genes, homologs of yeast autophagy genes, are involved in constitutive autophagy in Arabidopsis root tip cells. Plant Cell Physiol 2006, 47, 1641–1652. [Google Scholar]
- Kulich, I.; Pečenková, T.; Sekereš, J.; Smetana, O.; Fendrych, M.; Foissner, I.; Höftberger, M.; Zárský, V. Arabidopsis exocyst subcomplex containing subunit Exo70B1 is involved in autophagy-related transport to the vacuole. Traffic 2013, 14, 1155–1165. [Google Scholar]
- Viotti, C.; Krüger, F.; Krebs, M.; Neubert, C.; Fink, F.; Lupanga, U.; Scheuring, D.; Boutté, Y.; Frescatada-Rosa, M.; Wolfenstetter, S.; et al. The endoplasmic reticulum is the main membrane source for biogenesis of the lytic vacuole in Arabidopsis. Plant Cell 2013, 25, 3434–3449. [Google Scholar]
- Stigliano, E.; Faraco, M.; Neuhaus, J.M.; Montefusco, A.; Dalessandro, G.; Piro, G.; di Sansebastiano, G.P. Two glycosylated vacuolar GFPs are new markers for ER-to-vacuole sorting. Plant Physiol. Biochem 2013, 73, 337–343. [Google Scholar]
- Saslowsky, D.E.; Warek, U.; Winkel, B.S.J. Nuclear localization of flavonoid enzymes in Arabidopsis. J. Biol. Chem 2005, 280, 23735–23740. [Google Scholar]
- Winkel-Shirley, B. Evidence for enzyme complexes in the phenylpropanoid and flavonoid pathways. Physiol. Plant 1999, 107, 142–149. [Google Scholar]
- Poustka, F.; Irani, N.G.; Feller, A.; Lu, Y.; Pourcel, L.; Frame, K.; Grotewold, E. A trafficking pathway for anthocyanins overlaps with the endoplasmic reticulum-to-vacuole protein-sorting route in Arabidopsis and contributes to the formation of vacuolar inclusions. Plant Physiol 2007, 145, 1323–1335. [Google Scholar]
- Sun, Y.; Li, H.; Huang, J.R. Arabidopsis TT19 functions as a carrier to transport anthocyanin from the cytosol to tonoplasts. Mol. Plant 2012, 5, 387–400. [Google Scholar]
- Conn, S.; Franco, C.; Zhang, W. Characterization of anthocyanic vacuolar inclusions in Vitis vinifera L. cell suspension cultures. Planta 2010, 231, 1343–1360. [Google Scholar]
- Gomez, C.; Conejero, G.; Torregrosa, L.; Cheynier, V.; Terrier, N.; Ageorges, A. In vivo grapevine anthocyanin transport involves vesicle-mediated trafficking and the contribution of anthoMATE transporters and GST. Plant J 2011, 67, 960–970. [Google Scholar]
- Irani, N.G.; Grotewold, E. Light-induced morphological alteration in anthocyanin-accumulating vacuoles of maize cells. BMC Plant Biol 2005, 5. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Wang, L.; Deroles, S.; Bennett, R.; Davies, K. New insight into the structures and formation of anthocyanic vacuolar inclusions in flower petals. BMC Plant Biol 2006, 6. [Google Scholar] [CrossRef]
- Markham, K.R.; Gould, K.S.; Winefield, C.S.; Mitchell, K.A.; Bloor, S.J.; Boase, M.R. Anthocyanic vacuolar inclusions—Their nature and significance in flower colouration. Phytochemistry 2000, 55, 327–336. [Google Scholar]
- Bae, R.N.; Kim, K.W.; Kim, T.C.; Lee, S.K. Anatomical observations of anthocyanin rich cells in apple skins. HortScience 2006, 41, 733–736. [Google Scholar]
- Müller, O.; Sattler, T.; Flötenmeyer, M.; Schwarz, H.; Plattner, H.; Mayer, A. Autophagic tubes vacuolar invaginations involved in lateral membrane sorting and inverse vesicle budding. J. Cell Biol 2000, 151, 519–528. [Google Scholar]
- Thompson, A.R.; Doelling, J.H.; Suttangkakul, A.; Vierstra, R.D. Autophagic nutrient recycling in Arabidopsis directed by the ATG8 and ATG12 conjugation pathways. Plant Physiol 2005, 138, 2097–2110. [Google Scholar]
- Yamamoto, Y.; Nishimura, M.; Hara-Nishimura, I.; Noguchi, T. Behavior of vacuoles during microspore and pollen development in Arabidopsis thaliana. Plant Cell Physiol 2003, 44, 1192–1201. [Google Scholar]
- Saito, C.; Ueda, T.; Abe, H.; Wada, Y.; Kuroiwa, T.; Hisada, A.; Furuya, M.; Nakano, A. A complex and mobile structure forms a distinct subregion within the continuous vacuolar membrane in young cotyledons of Arabidopsis. Plant J 2002, 29, 245–255. [Google Scholar]
- Sattler, T.; Mayer, A. Cell-free reconstitution of microautophagic vacuole invagination and vesicle formation. J. Cell Biol 2000, 151, 529–538. [Google Scholar]
- Kunz, J.B.; Schwarz, H.; Mayer, A. Determination of four sequential stages during Microautophagy in Vitro. J. Biol. Chem. 2004, 279, 9987–9996. [Google Scholar]
- Zhao, Y.; Liu, J.; Yang, C.; Capraro, B.R.; Baumgart, T.; Bradley, R.P.; Ramakrishnan, N.; Xu, X.; Radhakrishnan, R.; Svitkina, T.; et al. Exo70 generates membrane curvature for morphogenesis and cell migration. Dev. Cell 2013, 26, 266–278. [Google Scholar]
- Liu, Y.; Burgos, J.S.; Deng, Y.; Srivastava, R.; Howell, S.H.; Bassham, D.C. Degradation of the endoplasmic reticulum by autophagy during endoplasmic reticulum stress in Arabidopsis. Plant Cell 2012, 24, 4635–4651. [Google Scholar]
- Lin, Y.; Irani, N.G.; Grotewold, E. Sub-cellular trafficking of phytochemicals explored using auto-fluorescent compounds in maize cells. BMC Plant Biol 2003, 3. [Google Scholar] [CrossRef] [Green Version]
- Yoshimoto, K.; Jikumaru, Y.; Kamiya, Y.; Kusano, M.; Consonni, C.; Panstruga, R.; Ohsumi, Y.; Shirasu, K. Autophagy negatively regulates cell death by controlling NPR1-dependent salicylic acid signaling during senescence and the innate immune response in Arabidopsis. Plant Cell 2009, 21, 2914–2927. [Google Scholar]
- Métraux, J.P. Recent breakthroughs in the study of salicylic acid biosynthesis. Trends Plant Sci 2002, 7, 332–334. [Google Scholar]
- Brillouet, J.M.; Romieu, C.; Schoefs, B.; Solymosi, K.; Cheynier, V.; Fulcrand, H.; Verdeil, J.L.; Conéjéro, G. The tannosome is an organelle forming condensed tannins in the chlorophyllous organs of Tracheophyta. Ann. Bot 2013, 112, 1003–1014. [Google Scholar]
- Dixon, R.; Paiva, N. Stress-induced phenylpropanoid metabolism. Plant Cell 1995, 7, 1085–1097. [Google Scholar]
- Fritz, C.; Palacios-Rojas, N.; Feil, R.; Stitt, M. Regulation of secondary metabolism by the carbon-nitrogen status in tobacco: Nitrate inhibits large sectors of phenylpropanoid metabolism. Plant J. Cell Mol. Biol 2006, 46, 533–548. [Google Scholar]
- De Geyter, N.; Gholami, A.; Goormachtig, S.; Goossens, A. Transcriptional machineries in jasmonate-elicited plant secondary metabolism. Trends Plant Sci 2012, 17, 349–359. [Google Scholar]
- Alcantara, J.; Bird, D.A.; Franceschi, V.R.; Facchini, P.J. Sanguinarine biosynthesis is associated with the endoplasmic reticulum in cultured opium poppy cells after elicitor treatment. Plant Physiol 2005, 138, 173–183. [Google Scholar]
- Ralston, L.; Yu, O. Metabolons involving plant cytochrome P450s. Phytochem. Rev 2006, 5, 459–472. [Google Scholar]
- St-Pierre, B.; Luca, V.D. A cytochrome P-450 monooxygenase catalyzes the first step in the conversion of tabersonine to vindoline in Catharanthus roseus. Plant Physiol 1995, 109, 131–139. [Google Scholar]
- Wagner, G.J.; Hrazdina, G. Endoplasmic reticulum as a site of phenylpropanoid and flavonoid metabolism in hippeastrum. Plant Physiol 1984, 74, 901–906. [Google Scholar]
- Hrazdina, G.; Zobel, A.M.; Hoch, H.C. Biochemical, Immunological, And immunocytochemical evidence for the association of chalcone synthase with endoplasmic reticulum membranes. Proc. Natl. Acad. Sci. USA 1987, 84, 8966–8970. [Google Scholar]
- Jones, P.; Vogt, T. Glycosyltransferases in secondary plant metabolism: Tranquilizers and stimulant controllers. Planta 2001, 213, 164–174. [Google Scholar]
- Vlot, A.C.; Dempsey, D.A.; Klessig, D.F. Salicylic acid, a multifaceted hormone to combat disease. Annu. Rev. Phytopathol 2009, 47, 177–206. [Google Scholar]
- Burla, B.; Pfrunder, S.; Nagy, R.; Francisco, R.M.; Lee, Y.; Martinoia, E. Vacuolar transport of abscisic acid glucosyl ester is mediated by ATP-binding cassette and proton-antiport mechanisms in Arabidopsis. Plant Physiol 2013, 163, 1446–1458. [Google Scholar]
- Lee, K.H.; Piao, H.L.; Kim, H.Y.; Choi, S.M.; Jiang, F.; Hartung, W.; Hwang, I.; Kwak, J.M.; Lee, I.J.; Hwang, I. Activation of glucosidase via stress-induced polymerization rapidly increases active pools of abscisic acid. Cell 2006, 126, 1109–1120. [Google Scholar]
- Okita, T.W.; Li, X.; Roberts, M.W. Targeting of mRNAs to domains of the endoplasmic reticulum. Trends Cell Biol 1994, 4, 91–96. [Google Scholar]
- Sirikantaramas, S.; Yamazaki, M.; Saito, K. Mutations in topoisomerase I as a self-resistance mechanism coevolved with the production of the anticancer alkaloid camptothecin in plants. Proc. Natl. Acad. Sci. USA 2008, 105, 6782–6786. [Google Scholar]
- Mravec, J.; Skupa, P.; Bailly, A.; Hoyerová, K.; Krecek, P.; Bielach, A.; Petrásek, J.; Zhang, J.; Gaykova, V.; Stierhof, Y.D.; et al. Subcellular homeostasis of phytohormone auxin is mediated by the ER-localized PIN5 transporter. Nature 2009, 459, 1136–1140. [Google Scholar]
- Brooks, D.A.; Bader, C.; Ng, Y.S.; Brooks, R.D.; Borlace, G.N.; Shandal, T. At the intersection of the pathways for exocytosis and autophagy. In Crosstalk and Integration of Membrane Trafficking Pathways; Roberto, W., Ed.; InTech: Rijeka, Croatia, 2012; pp. 109–136. [Google Scholar]
- Pfeffer, S.R. Unconventional secretion by autophagosome exocytosis. J. Cell Biol 2010, 188, 451–452. [Google Scholar]
- Ding, Y.; Wang, J.; Wang, J.; Stierhof, Y.D.; Robinson, D.G.; Jiang, L. Unconventional protein secretion. Trends Plant Sci 2012, 17, 606–615. [Google Scholar]
- Markham, K.R.; Ryan, K.G.; Gould, K.S.; Rickards, G.K. Cell wall sited flavonoids in lisianthus flower petals. Phytochemistry 2000, 54, 681–687. [Google Scholar]
- Žárský, V.; Kulich, I.; Fendrych, M.; Pečenková, T. Exocyst complexes multiple functions in plant cells secretory pathways. Curr. Opin. Plant Biol 2013, 16, 726–733. [Google Scholar]
- Zhao, J.; Dixon, R.A. MATE transporters facilitate vacuolar uptake of epicatechin 3′-O-glucoside for proanthocyanidin biosynthesis in Medicago truncatula and Arabidopsis. Plant Cell 2009, 21, 2323–2340. [Google Scholar]
- Ting, H.M.; Wang, B.; Rydén, A.M.; Woittiez, L.; van Herpen, T.; Verstappen, F.W.A.; Ruyter-Spira, C.; Beekwilder, J.; Bouwmeester, H.J.; van der Krol, A. The metabolite chemotype of Nicotiana benthamiana transiently expressing artemisinin biosynthetic pathway genes is a function of CYP71AV1 type and relative gene dosage. New Phytol 2013, 199, 352–366. [Google Scholar]
- Tabata, M. The mechanism of shikonin biosynthesis in Lithospermum cell cultures. Plant Tissue Cult. Lett 1996, 13, 117–125. [Google Scholar]
- Yazaki, K. Transporters of secondary metabolites. Curr. Opin. Plant Biol 2005, 8, 301–307. [Google Scholar]
- Agati, G.; Azzarello, E.; Pollastri, S.; Tattini, M. Flavonoids as antioxidants in plants: Location and functional significance. Plant Sci 2012, 196, 67–76. [Google Scholar]
- Meyer, D.; Pajonk, S.; Micali, C.; O’Connell, R.; Schulze-Lefert, P. Extracellular transport and integration of plant secretory proteins into pathogen-induced cell wall compartments. Plant J 2009, 57, 986–999. [Google Scholar]
- Roepke, J.; Salim, V.; Wu, M.; Thamm, A.M.K.; Murata, J.; Ploss, K.; Boland, W.; Luca, V.D. Vinca drug components accumulate exclusively in leaf exudates of Madagascar periwinkle. Proc. Natl. Acad. Sci. USA 2010, 107, 15287–15292. [Google Scholar]
- Marty, F. Cytochemical studies on GERL, Provacuoles, and vacuoles in root meristematic cells of Euphorbia. Proc. Natl. Acad. Sci. USA 1978, 75, 852–856. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kulich, I.; Žárský, V. Autophagy-Related Direct Membrane Import from ER/Cytoplasm into the Vacuole or Apoplast: A Hidden Gateway also for Secondary Metabolites and Phytohormones? Int. J. Mol. Sci. 2014, 15, 7462-7474. https://doi.org/10.3390/ijms15057462
Kulich I, Žárský V. Autophagy-Related Direct Membrane Import from ER/Cytoplasm into the Vacuole or Apoplast: A Hidden Gateway also for Secondary Metabolites and Phytohormones? International Journal of Molecular Sciences. 2014; 15(5):7462-7474. https://doi.org/10.3390/ijms15057462
Chicago/Turabian StyleKulich, Ivan, and Viktor Žárský. 2014. "Autophagy-Related Direct Membrane Import from ER/Cytoplasm into the Vacuole or Apoplast: A Hidden Gateway also for Secondary Metabolites and Phytohormones?" International Journal of Molecular Sciences 15, no. 5: 7462-7474. https://doi.org/10.3390/ijms15057462



