Combined Elevation of microRNA-196a and microRNA-196b in Sera Predicts Unfavorable Prognosis in Patients with Osteosarcomas
Abstract
:Aim
Methods
Results
Conclusion
1. Introduction
2. Results and Discussion
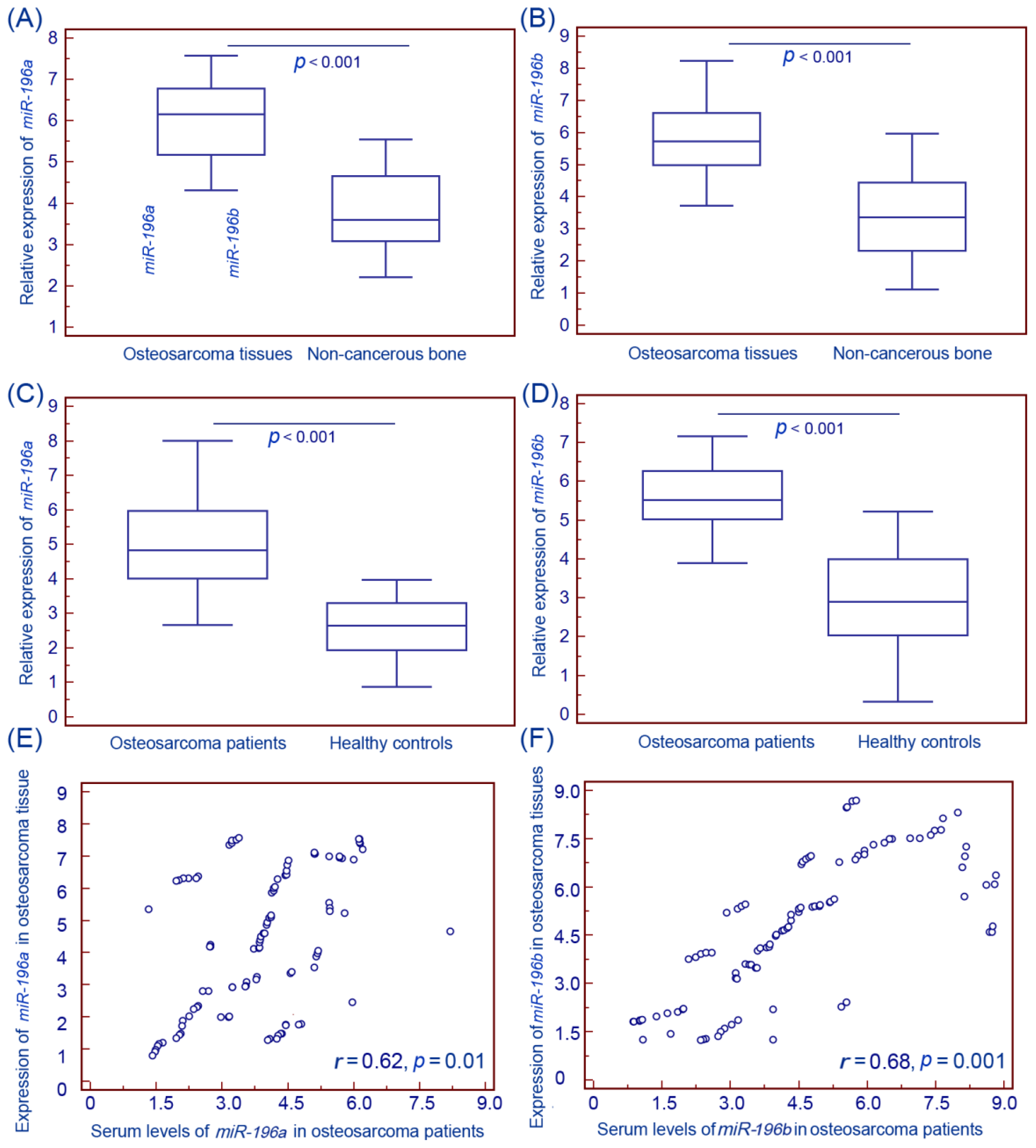
2.1. Upregulation of miR (microRNA)-196a and miR-196b in Human Osteosarcoma Tissues and Patients’ Sera
2.2. Serum Levels of miR-196a and miR-196b Associate with Clinicopathological Features of Human Osteosarcoma
2.3. Serum Levels of miR-196a and miR-196b Predicts Prognosis in Patients with Osteosarcoma
2.4. Discussion
3. Experimental Section
3.1. Ethics Statement
3.2. Patients and Tissue Samples
3.3. RNA Extraction
3.4. QRT-PCR for miRNA
3.5. Statistical Analysis
4. Conclusions
Conflicts of Interest
- Author ContributionsX.H., C.Z., and C.Y. designed this study. C.Z. and C.Y. performed the experiments. H.L. and G.W. performed the statistical analysis.
References
- Fletcher, C.D.M.; Unni, K.K.; Mertens, F. World Health Organization Classification of Tumours. Pathology and Genetics of Tumours of Soft Tissue and Bone; IARC Press: Lyon, France, 2002. [Google Scholar]
- Kager, L.; Zoubek, A.; Pötschger, U.; Kastner, U.; Flege, S.; Kempf-Bielack, B.; Branscheid, D.; Kotz, R.; Salzer-Kuntschik, M.; Winkelmann, W.; et al. Primary metastatic osteosarcoma: Presentation and outcome of patients treated on neoadjuvant cooperative osteosarcoma study group protocols. J. Clin. Oncol 2003, 21, 2011–2018. [Google Scholar]
- Longhi, A.; Fabbri, N.; Donati, D.; Capanna, R.; Briccoli, A.; Biagini, R.; Bernini, G.; Ferrari, S.; Versari, M.; Bacci, G. Neoadjuvant chemotherapy for patients with synchronous multifocal osteosarcoma: Results in eleven cases. J. Chemother 2001, 13, 324–330. [Google Scholar]
- Bacci, G.; Briccoli, A.; Rocca, M.; Ferrari, S.; Donati, D.; Longhi, A.; Bertoni, F.; Bacchini, P.; Giacomini, S.; Forni, C.; et al. Neoadjuvant chemotherapy for osteosarcoma of the extremities with metastases at presentation: Recent experience at the Rizzoli Institute in 57 patients treated with cisplatin, doxorubicin, and a high dose of methotrexate and ifosfamide. Ann. Oncol 2003, 14, 1126–1134. [Google Scholar]
- Bentwich, I.; Avniel, A.; Karov, Y.; Aharonov, R.; Gilad, S.; Barad, O. Identification of hundreds of conserved and nonconserved human microRNAs. Nat. Genet 2005, 37, 766–770. [Google Scholar]
- Sassen, S.; Miska, E.A.; Caldas, C. MicroRNA: Implications for cancer. Virchows Arch 2008, 452, 1–10. [Google Scholar]
- Maire, G.; Martin, J.W.; Yoshimoto, M.; Chilton-MacNeill, S.; Zielenska, M. Analysis of miRNA-gene expression-genomic profiles reveals complex mechanisms of microRNA deregulation in osteosarcoma. Cancer Genet 2011, 204, 138–146. [Google Scholar]
- Bouyssou, J.M.; Manier, S.; Huynh, D.; Issa, S.; Roccaro, A.M.; Ghobrial, I.M. Regulation of microRNAs in Cancer Metastasis. Biochim. Biophys. Acta 2014. [Google Scholar]
- Mollaie, H.R.; Monavari, S.H.; Arabzadeh, S.A.; Shamsi-Shahrabadi, M.; Fazlalipour, M.; Afshar, R.M. RNAi and miRNA in viral infections and cancers. Asian Pac. J. Cancer Prev 2013, 14, 7045–7056. [Google Scholar]
- Nugent, M. MicroRNA function and dysregulation in bone tumors: The evidence to date. Cancer Manag. Res 2014, 6, 15–25. [Google Scholar]
- Gomez, G.G.; Wykosky, J.; Zanca, C.; Furnari, F.B.; Cavenee, W.K. Therapeutic resistance in cancer: microRNA regulation of EGFR signaling networks. Cancer Biol. Med 2013, 10, 192–205. [Google Scholar]
- Suzuki, H.; Maruyama, R.; Yamamoto, E.; Kai, M. Epigenetic alteration and microRNA dysregulation in cancer. Front. Genet 2013. [Google Scholar] [CrossRef]
- Yu, H.; Lindsay, J.; Feng, Z.P.; Frankenberg, S.; Hu, Y.; Carone, D.; Shaw, G.; Pask, A.J.; O’Neill, R.; Papenfuss, A.T.; et al. Evolution of coding and non-coding genes in HOX clusters of a marsupial. BMC Genomics 2012. [Google Scholar] [CrossRef]
- Karsy, M.; Arslan, E.; Moy, F. Current progress on understanding microRNAs in glioblastoma multiforme. Genes Cancer 2012, 3, 3–15. [Google Scholar]
- Liu, C.J.; Tsai, M.M.; Tu, H.F.; Lui, M.T.; Cheng, H.W.; Lin, S.C. miR-196a overexpression and miR-196a2 gene polymorphism are prognostic predictors of oral carcinomas. Ann. Surg. Oncol 2013, 20, S406–S414. [Google Scholar]
- Namløs, H.M.; Meza-Zepeda, L.A.; Barøy, T.; Østensen, I.H.; Kresse, S.H.; Kuijjer, M.L.; Serra, M.; Bürger, H.; Cleton-Jansen, A.M.; Myklebost, O. Modulation of the osteosarcoma expression phenotype by microRNAs. PLoS One 2012, 7, e48086. [Google Scholar]
- Hauptman, N.; Glavac, D. MicroRNAs and long non-coding RNAs: Prospects in diagnostics and therapy of cancer. Radiol. Oncol 2013, 47, 311–318. [Google Scholar]
- McGinnis, W.; Krumlauf, R. Homeobox genes and axial patterning. Cell 1992, 68, 283–302. [Google Scholar]
- Tanzer, A.; Amemiya, C.T.; Kim, C.B. Evolution of microRNAs located within HOX gene clusters. J. Exp. Zool. B Mol. Dev. Evol 2005, 304, 75–85. [Google Scholar]
- Guan, Y.; Mizoguchi, M.; Yoshimoto, K.; Hata, N.; Shono, T.; Suzuki, S.O.; Araki, Y.; Kuga, D.; Nakamizo, A.; Amano, T.; et al. miRNA-196 is upregulated in glioblastoma but not in anaplastic astrocytoma and has prognostic significance. Clin. Cancer Res 2010, 16, 4289–4297. [Google Scholar]
- Schultz, N.A.; Werner, J.; Willenbrock, H.; Roslind, A.; Giese, N.; Horn, T.; Wøjdemann, M.; Johansen, J.S. MicroRNA expression profiles associated with pancreatic adenocarcinoma and ampullary adenocarcinoma. Mod. Pathol 2012, 25, 1609–1622. [Google Scholar]
- Dou, T.; Wu, Q.; Chen, X.; Ribas, J.; Ni, X.; Tang, C.; Huang, F.; Zhou, L.; Lu, D. A polymorphism of microRNA196a genome region was associated with decreased risk of glioma in Chinese population. J. Cancer Res. Clin. Oncol 2010, 136, 1853–1859. [Google Scholar]
- Peng, S.; Kuang, Z.; Sheng, C.; Zhang, Y.; Xu, H.; Cheng, Q. Association of microRNA-196a-2 gene polymorphism with gastric cancer risk in a Chinese population. Dig. Dis. Sci 2010, 55, 2288–2293. [Google Scholar]
- Hoffman, A.E.; Zheng, T.; Yi, C.; Leaderer, D.; Weidhaas, J.; Slack, F.; Zhang, Y.; Paranjape, T.; Zhu, Y. microRNA miR-196a-2 and breast cancer: A genetic and epigenetic association study and functional analysis. Cancer Res 2009, 69, 5970–5977. [Google Scholar]
- Mueller, D.W.; Bosserhoff, A.K. MicroRNA miR-196a controls melanoma-associated genes by regulating HOX-C8 expression. Int. J. Cancer 2011, 129, 1064–1074. [Google Scholar]
- Bhatia, S.; Kaul, D.; Varma, N. Potential tumor suppressive function of miR-196b in B-cell lineage acute lymphoblastic leukemia. Mol. Cell. Biochem 2010, 340, 97–106. [Google Scholar]
- Tsai, K.W.; Hu, L.Y.; Wu, C.W.; Li, S.C.; Lai, C.H.; Kao, H.W.; Fang, W.L.; Lin, W.C. Epigenetic regulation of miR-196b expression in gastric cancer. Genes Chromosomes Cancer 2010, 49, 969–980. [Google Scholar]
- Ma, R.; Yan, W.; Zhang, G.; Lv, H.; Liu, Z.; Fang, F.; Zhang, W.; Zhang, J.; Tao, T.; You, Y.; et al. Upregulation of miR-196b confers a poor prognosis in glioblastoma patients via inducing a proliferative phenotype. PLoS One 2012, 7, e38096. [Google Scholar]
- Li, Y.; Zhang, M.; Chen, H.; Dong, Z.; Ganapathy, V.; Thangaraju, M.; Huang, S. Ratio of miR-196s to HOXC8 messenger RNA correlates with breast cancer cell migration and metastasis. Cancer Res 2010, 70, 7894–7904. [Google Scholar]
- Hamamoto, J.; Soejima, K.; Yoda, S.; Naoki, K.; Nakayama, S.; Satomi, R.; Terai, H.; Ikemura, S.; Sato, T.; Yasuda, H.; et al. Identification of microRNAs differentially expressed between lung squamous cell carcinoma and lung adenocarcinoma. Mol. Med. Rep 2013, 8, 456–462. [Google Scholar]
- How, C.; Hui, A.B.; Alajez, N.M.; Shi, W.; Boutros, P.C.; Clarke, B.A.; Yan, R.; Pintilie, M.; Fyles, A.; Hedley, D.W.; et al. MicroRNA-196b regulates the homeobox B7-vascular endothelial growth factor axis in cervical cancer. PLoS One 2013, 8, e67846. [Google Scholar]
- Bacci, G.; Bertoni, F.; Longhi, A.; Ferrari, S.; Forni, C.; Biagini, R.; Bacchini, P.; Donati, D.; Manfrini, M.; Bernini, G.; et al. Neoadjuvant chemotherapy for high-grade central osteosarcoma of the extremity. Histologic response to preoperative chemotherapy correlates with histologic subtype of the tumor. Cancer 2003, 97, 3068–3075. [Google Scholar]
- Kroh, E.M.; Parkin, R.K.; Mitchell, P.S.; Tewari, M. Analysis of circulating microRNA biomarkers in plasma and serum using quantitative reverse transcription-PCR (qRT-PCR). Methods 2010, 50, 298–301. [Google Scholar]

| Clinicopathological Features | No. of Cases | miR-196a-High (n, %) | p | miR-196b-High (n, %) | p | miR-196a-High/miR-196b-High (n, %) | p |
|---|---|---|---|---|---|---|---|
| Age | |||||||
| <18 | 32 (32.00) | 20 (62.51) | NS | 14 (43.75) | NS | 12 (37.50) | NS |
| ≥18 | 68 (68.00) | 37 (54.41) | 38 (55.88) | 28 (41.18) | |||
| Sex | |||||||
| Male | 70 (70.00) | 40 (57.14) | NS | 36 (51.73) | NS | 28 (40.00) | NS |
| Female | 30 (30.00) | 17 (56.67) | 16 (53.33) | 12 (40.00) | |||
| Tumor Site | |||||||
| Femur | 58 (58.00) | 35 (60.34) | NS | 30 (51.72) | NS | 22 (37.93) | NS |
| Tibia | 20 (20.00) | 10 (50.00) | 11 (55.00) | 9 (45.00) | |||
| Humeral bone | 15 (15.00) | 8 (53.33) | 8 (53.33) | 6 (40.00) | |||
| Other | 7 (7.00) | 4 (57.14) | 3 (42.86) | 3 (42.86) | |||
| Histologic Type | |||||||
| Osteoblastic | 52 (52.00) | 30 (57.69) | NS | 28 (53.85) | NS | 20 (38.46) | NS |
| Chondroblastic | 18 (18.00) | 10 (55.56) | 9 (50.00) | 8 (44.44) | |||
| Fibroblastic | 20 (20.00) | 11 (55.00) | 10 (50.00) | 8 (40.00) | |||
| Telangiectatic | 10 (10.00) | 6 (60.00) | 5 (50.00) | 4 (40.00) | |||
| Tumor Grade | |||||||
| Low | 38 (38.00) | 15 (39.47) | 0.008 | 12 (31.58) | 0.01 | 8 (23.53) | <0.001 |
| High | 62 (62.00) | 42 (67.74) | 40 (64.52) | 32 (51.61) | |||
| Metastasis | |||||||
| Absent | 60 (60.00) | 30 (50.00) | 0.001 | 26 (43.33) | 0.006 | 12 (20.00) | <0.001 |
| Present | 40 (40.00) | 27 (67.50) | 26 (65.00) | 28 (70.00) | |||
| Recurrence | |||||||
| Absent | 65 (65.00) | 27 (41.54) | 0.001 | 26 (40.00) | 0.006 | 14 (21.54) | <0.001 |
| Present | 35 (35.00) | 30 (85.71) | 26 (74.29) | 26 (74.29) | |||
| Response to Pre-Operative Chemotherapy | |||||||
| Good | 60 (60.00) | 31 (51.67) | NS | 32 (53.33) | NS | 22 (36.67) | NS |
| Poor | 40 (40.00) | 26 (65.00) | 20 (50.00) | 18 (45.00) | |||
| Variables | OS | DFS | ||||
|---|---|---|---|---|---|---|
| RR | 95% CI | p | RR | 95% CI | p | |
| Tumor grade | 6.86 | 1.69–15.02 | 0.01 | 7.37 | 1.72–16.31 | 0.008 |
| Response to pre-operative chemotherapy | 3.58 | 0.80–7.21 | 0.04 | 4.02 | 1.01–8.38 | 0.03 |
| Metastasis status | 3.19 | 0.76–6.82 | NS | 3.56 | 0.80–7.19 | 0.04 |
| Recurrence status | 3.48 | 0.79–7.03 | NS | 4.00 | 1.00–8.06 | 0.03 |
| Serum miR-196a level | 6.28 | 1.62–13.39 | 0.01 | 6.95 | 1.63–14.61 | 0.01 |
| Serum miR-196b level | 6.33 | 1.61–13.48 | 0.01 | 6.98 | 1.65–14.82 | 0.01 |
| miR-196a/miR-196b expression | 9.89 | 2.66–20.98 | 0.001 | 10.09 | 2.82–21.99 | 0.001 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, C.; Yao, C.; Li, H.; Wang, G.; He, X. Combined Elevation of microRNA-196a and microRNA-196b in Sera Predicts Unfavorable Prognosis in Patients with Osteosarcomas. Int. J. Mol. Sci. 2014, 15, 6544-6555. https://doi.org/10.3390/ijms15046544
Zhang C, Yao C, Li H, Wang G, He X. Combined Elevation of microRNA-196a and microRNA-196b in Sera Predicts Unfavorable Prognosis in Patients with Osteosarcomas. International Journal of Molecular Sciences. 2014; 15(4):6544-6555. https://doi.org/10.3390/ijms15046544
Chicago/Turabian StyleZhang, Chun, Cong Yao, Haopeng Li, Guoyu Wang, and Xijng He. 2014. "Combined Elevation of microRNA-196a and microRNA-196b in Sera Predicts Unfavorable Prognosis in Patients with Osteosarcomas" International Journal of Molecular Sciences 15, no. 4: 6544-6555. https://doi.org/10.3390/ijms15046544




