1. Introduction
Cerium oxide nanoparticles (nano-ceria) are engineered nanomaterials (ENMs) that possess unique catalytic, optical and anti-oxidant properties [
1]. The widespread use of nano-ceria in industry has raised occupational and environmental health concerns over the potential risks of nano-ceria exposure. Therefore, nano-ceria are identified by the Organisation for Economic Co-operation and Development as one of the 13 priority listed representative ENMs for immediate testing [
2].
Nano-ceria are used increasingly as an additive to improve the burning efficiency of fuels, reducing fuel consumption, greenhouse gases and particle numbers in vehicle exhaust [
3]. Nanoscale ceria could be found in the soot matrix and, thereby, be released into the environment [
4]. By using a modeling method, Park
et al. predicted that receptors in the car passenger area of a street canyon would be exposed to ceria at a concentration up to 80 ng/m
3 in the worst case [
5]. The combusted nano-ceria would interact with the carbonaceous aggregates or other co-pollutants in the exhaust, thereby exhibited a toxicological profile different from nano-ceria alone. Therefore, risk-assessment studies should be conducted based on the combusted particulates emitted from engines using nano-ceria-based fuel additives [
6]. Furthermore, the toxicity of the combusted nano-ceria should be correlated to their physicochemical properties, so that researchers are able to predict potential risks and design ceria-based additives with minimum toxicity.
However, the physicochemical properties of nano-ceria would be changed during combustion, and the details of the changes remain unknown. For example, the reported crystallite size of nano-ceria in diesel exhaust were quite controversial: ranging from 1–3 nm [
6], 5–7 nm [
7], up to about 43 nm [
4]. There is very limited knowledge about the other physicochemical properties of the combusted ceria (e.g., surface chemistry). The gap makes it difficult to understand the mechanisms underlying the toxic impact from the combusted nano-ceria when considering the hazard posed by the use of nano-ceria as a fuel additive. Alternatively, toxicological assessments of a series of nano-ceria with diverse, but well-defined, nano-properties were conducted in previous studies [
5,
8,
9] to relate the physicochemical properties of nano-ceria to their toxicity.
Recent literature has shown that nano-ceria exposure is associated with pulmonary injury, leading to oxidative stress, inflammation and cytotoxicity [
9–
11]. In our previous study, mice were exposed to 0.04, 0.4, 4 and 40 μg of nano-ceria with a mean diameter of 6.6 nm (6.6-Ceria) to explore the pulmonary toxicity of nano-ceria after an acute intratracheal instillation [
8]. It was indicated that only the exposure to 40 μg of 6.6-Ceria caused changes in the differential bronchoalveolar lavage (BAL) fluid (BALF) cell counts and BALF lactate dehydrogenase (LDH) activity as an acute inflammatory reaction; the cell counts returned to normal at seven days post-instillation (PI) and did not cause oxidative damage to the lungs; the particle overload in pulmonary macrophages led to multifocal acute alveolitis and pulmonary granulomas at seven days PI and recovered at 28 days PI.
The size of ENMs plays a key role in determining the toxicity of ENMs [
12]. Compared with their larger counterparts, smaller ENMs are of a larger number concentration at the same mass concentration and have a higher surface energy and reactivity, greater mobility and greater resistance to systemic clearance. In the present work, we tried to test whether exposure to nano-ceria sized 3–5 nm could cause more severe pulmonary toxicity than 6.6-Ceria did. Mice were intratracheally exposed to two kinds of nano-ceria were are of a similar size, but have different redox activity, and the pulmonary toxicities in terms of oxidative damage, inflammation and histopathological changes were assessed.
3. Discussion
Nano-ceria have recently gained a wide range of applications, which might pose unwanted risks to both the environment and human health. The greatest potential for the environmental discharge of nano-ceria appears to be in their use as a diesel fuel additive. According to the prediction by Park
et al., the commercial use of nano-ceria will increase the air-borne nano-ceria, to a level of 80 ng/m
3 in the worst case [
5]. In the present work, mice were exposed to 40 μg of nano-ceria via a single intratracheal instillation, corresponding to 10,000 times the maximum daily exposure dose for mice [
8].
Although the toxic mechanisms for inhaled ENMs were not very clear, the results of older toxicological and epidemiological studies with airborne fine or ultrafine particles can be viewed as the basis for the expanding field of nanotoxicology [
13]. Due to their small size, inhaled ENMs could be transported into the deep lung and could not be removed efficiently, leading to particle-overload in lung. In the present work, particle-laden macrophages could be found in the lungs instilled with either Ceria-p or Ceria-h during the whole experimental period. Our results implied that the pulmonary toxicity of nano-ceria was associated with lung particle-overload.
Studies in rodents imply that the excessive burden of insoluble particles of low cytotoxicity would lead to pulmonary inflammation [
14]. Oyabu
et al. reviewed the reports focusing on particle-overload in lung and concluded that a dose that can induce inflammation is between one and 5 mg/kg [
15]. Our previous work also showed that the intratracheal instillation of nano-ceria (with a mean size of 6.6 nm) at a dose of ~1.5 mg/kg (40 μg per mouse) induced moderate and transient inflammation in mouse lung, which recovered by four weeks, whereas 0.15 mg/kg did not cause any inflammation. In the present work, neutrophil inflammation was induced by the exposure to either Ceria-p or Ceria-h at a dose of 1.5 mg/kg (40 μg per mouse). The acute intratracheal instillation of Ceria-p and Ceria-h also influenced the total protein content and LDH leakage in the BALF and induced oxidative stress and inflammatory response in the lung homogenates.
However, there were some differences between the toxicological profiles of Ceria-p and Ceria-h. In general, intratracheal instillation of Ceria-p induced transient inflammatory responses in both BAL cells and lung tissue at the early stage of exposure (one and/or three days PI), which recovered at 7 or 28 days PI. Ceria-h induced initially minor, but more persistent inflammation in BAL cells and delayed lipid peroxidation and pro-inflammation in lung tissue at 28 days PI.
The differences in the initial inflammatory responses may be due to the different agglomeration state of Ceria-p and Ceria-h. The size of agglomerates would determine their deposition in the lung, their ability to cross biological barriers and their cellular internalization [
12]. Noël
et al. found that the pulmonary toxicity of ENMs might depend not solely on the dimension of the ENMs, but also on the dimension of the ENMs agglomerates [
16]. However, in their study, large nano-TiO
2 agglomerates (>100 nm) caused acute inflammatory responses; small ones (<100 nm) exerted both cytotoxicity and oxidative stress without apparent neutrophil influx into the airways, because agglomerates smaller than 100 nm could easily escape the pulmonary clearance via phagocytosis [
12,
17]. In the present work, Ceria-p agglomerates and Ceria-h agglomerates were both larger than 100 nm (313 ± 30 and 1731 ± 165 nm, respectively). Therefore, macrophage phagocytosis was activated in both treatments. Due to the smaller size of Ceria-p agglomerates, there was a larger number concentration of agglomerates in the Ceria-p exposure. Moreover, smaller agglomerates had a higher deposition rate in the deep lung; particle-laden macrophages and granulomas could even be found at the peripheral area of the lungs instilled with Ceria-p. Therefore, Ceria-p caused a more potent particle-overload inflammation at the early stage of exposure.
In the lungs instilled with Ceria-h, oxidative damage was found at seven and 28 days PI, and a pro-inflammatory effect was found at 28 days PI. These delayed responses seemed not to be attributed exclusively to the excessive ENMs burdens in the lung, as particle-laden macrophages were less observed in the Ceria-h treated mice than in the Ceria-p group. ROS generation is identified as one of the major mechanisms by which inhaled ENMs exert adverse biological effects [
18,
19]. We speculated that the retention of Ceria-h in lung and their capability to generate ROS might also be involved in the pulmonary toxicity of Ceria-h. Although Ceria-h tended to agglomerate, the agglomeration would be gradually reduced in the airway due to the interactions with surfactant proteins or vascular proteins (in the case of a breakdown of the integrity of the alveolar-capillary barrier caused by ENMs exposure) [
1,
20]. Deagglomerated Ceria-h might be deposited in the BAL area or translocated into the lung interstitium, where it catalyzes the generation of ROS and increases the risk of oxidative damage to lung tissue. Therefore, Ceria-h exposure caused two waves of lung injury: BAL inflammation and cytotoxicity induced by particle overload in the early stage, and pulmonary lipid peroxidation and pro-inflammation in the latter stage. Since the intratracheal instillation of Ceria-h caused a more persistent lung injury compared to Ceria-p, the long-term pulmonary and systemic toxicity of Ceria-h needs further study.
Our results implied that the agglomeration, as well as the surface chemistry plays an important role in the pulmonary toxicity of nano-ceria. Due to agglomeration, Ceria-p did not exert more severe pulmonary toxicity when compared to nano-ceria sized 6.6 nm. Therefore, the characterization of the size distribution of the ceria-contained agglomerates in the exhaust is essential to assess the potential risks of using nano-ceria as a fuel additive. Meanwhile, more attention should be paid to the surface chemistry of nano-ceria after combustion. The ability to reversibly switch between Ce
3+ and Ce
4+ makes nano-ceria attractive as a fuel additive to reduce the release of greenhouse gases and particles. Meanwhile, nano-ceria are being proven promising for their SOD mimetic activity [
21]. It was reported that nano-ceria with a high Ce
3+/Ce
4+ ratio on the surface could exhibit SOD mimetic activity, whereas nano-ceria with a lower Ce
3+/Ce
4+ ratio show no SOD mimetic activity [
22]. In the present work, Ceria-p and Ceria-h both have no detectable fraction of Ce
3+ (
Figure 1B), thereby exhibiting negligible SOD mimetic activity (data not shown). The redox activity may also endow nano-ceria with the ability to catalyze the generation of ROS. Our previous work suggested that the oxidative stress caused by nano-ceria exposure at the environmentally relevant concentration would shorten worms’ lifespans [
23]. Therefore, surface chemistry is also an important parameter to determine the pulmonary toxicity of the combusted nano-ceria. Unfortunately, so far, there was only very limited and controversial information on the properties of the combusted nano-ceria in the exhaust.
4. Subjects and Methods
4.1. Synthesis and Characterization of Nano-Ceria
The primary particle diameters of the two types of nano-ceria used in the present work were both 3–5 nm. One of the nano-ceria (Ceria-p) was synthesized by a precipitation method [
24]. In brief, 1.736 g Ce(NO
3)
3 was added to NaOH solution (0.4 g NaOH dissolved in 128 mL water) followed by 48 h of magnetic stirring. The resulting white precipitate was collected and washed several times in ultrapure water.
Another type of nano-ceria (Ceria-h) was synthesized by a surfactant-assisted hydrothermal approach [
25]. Briefly, 15 mL of 116.7 mmol/L Ce(NO
3)
3 solution was mixed with 15 mL toluene; then, 22.5 mL of 233.3 mmol/L sodium oleate aqueous was dropped into the above mixture solution with magnetic stirring. The upper layer toluene with cerium precursor was transferred to a 50-mL Teflon-lined stainless-steel autoclave with 15 mL deionized water and 0.35 mL
tert-butylamine. The sealed autoclave was transferred to a 180 °C oven, held there for 12 h and then cooled to room temperature. Then, the brown supernatant solution with synthesized nano-ceria was precipitated with an adequate volume of ethanol. The precipitate was obtained by centrifugation for 10 min and re-dispersed in 15 mL ultrapure water containing 400 μL of 3-mercaptopropionic acid (3-MPA). After centrifuging and washing 3 times, a Ceria-p suspension was obtained.
The physicochemical properties of nano-ceria were characterized by transmission electron microscopy (TEM, JEM-2010, JEOL, Tokyo, Japan), dynamic light scattering (DLS, Zetasizer nano-ZS 90, Malvern Instrument, Worcestershire, UK), X-ray absorption near-edge structure (XANES, 1W1B beamline at Beijing Synchrotron Radiation Facility, Beijing, China), and X-ray diffraction (XRD, X’pert PRO MPD, PANalytical, Almelo, The Netherlands). The ROS generation catalyzed by nano-ceria was determined by the method described by the literature of Heckert [
26], using 2,2’-azinobis-(3-ethylbenzthiazoline-6-sulfonic acid) (ABTS) as the free radical capture agent. After catching ROS, the ABTS can form a stable blue-green product, cation radicals ABTS
+#, which was measured at 405 nm by using a UV-vis spectrophotometer (TU-1901, PGENENAL, Beijing, China). Reactions with nano-ceria were buffered by Tris (100 μM, pH 7.0). The solution containing 100 μM nano-CeO
2, 88 mM H
2O
2 and 100 μM ABTS was added into a 1-cm path length quartz cuvette, and the absorbance at 405 nm was recorded once every 3 s for 10 min.
4.2. Animals
Male CD-1 (ICR) mice (6 weeks old) were purchased from Beijing Vital River Laboratories. A commercial pellet diet and deionized water were available ad libitum. After one week of acclimation, mice were randomly divided into 12 groups, with 8 mice per group and housed individually in stainless steel cages under standard laboratory conditions (temperature 23 ± 1 °C, humidity 50% ± 3% and a 12-h light/dark cycle). All the animal experiments were performed with the approval of the Ethics Committee of Animal Care and Experimentation of the National Institute for Environmental Studies, China.
4.3. Intratracheal Instillation of Nano-Ceria
The Ceria-p and Ceria-h were dispersed in ultrapure water at a concentration of 0.8 mg/mL, respectively, and sonicated for 15 min before use. By using a non-surgical intratracheal instillation method [
8], mice were intratracheally instilled with 40 μg of either nano-ceria (50 μL of nano-ceria suspension) followed by 800 μL of air in 2 s. The control group was intratracheally instilled with ultrapure water instead of nano-ceria.
4.4. Cytological and Biochemical Analysis of Bronchoalveolar Lavage Fluid
The animals were sacrificed at 1, 3, 7 and 28 days post-instillation (PI). Bronchoalveolar lavage was performed 3 times with sodium phosphate buffer (PBS, pH 7.4, 1 mL for each lavage), and the cells in BALF were collected by centrifugation at 400× g and 4 °C for 10 min. The supernatant was used for biochemical analyses, while the recovered cells from the pellet were resuspended for the macrophage, neutrophil and lymphocyte cell counting with Wright’s staining. The activities of lactate dehydrogenase (LDH) and acid phosphatase (ACP) in the BALF were analyzed using colorimetric assay kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). The concentration of total protein (TP) was determined by the bicinchoninic acid (BCA) method using a BCA protein assay kit (Thermo Fisher Scientific Incorporated, Rockford, IL, USA) with bovine serum albumin as a standard.
4.5. Biochemical Assay of Lung Homogenates
The lung tissues were separated and homogenized by a glass homogenizer in pre-cooled 0.01 M PBS (pH 7.4, v/v = 1/4). The homogenates were centrifuged at 10,000 rpm for 10 min at 4 °C, and the supernatants were aliquoted and stored at −80 °C for the biochemical analysis. The concentrations of malondialdehyde (MDA) and GSH, as well as the activities of SOD, GSH-Px and CAT in the lung homogenates were measured using the reagent kits purchased from Jiancheng Bioengineering Co., Ltd., Nanjing, China, according to the manufacturer’s protocols. Protein levels in the lung homogenates were determined by the bicinchoninic acid (BCA) protein assay kit (Thermo Fisher Scientific Incorporated, Rockford, IL, USA). The levels of IL-6 in the lung homogenates were determined using commercially available ELISA kits (Wuhan Boster Biological Technology Co., Ltd., Wuhan, China).
4.6. Histopathological Examination
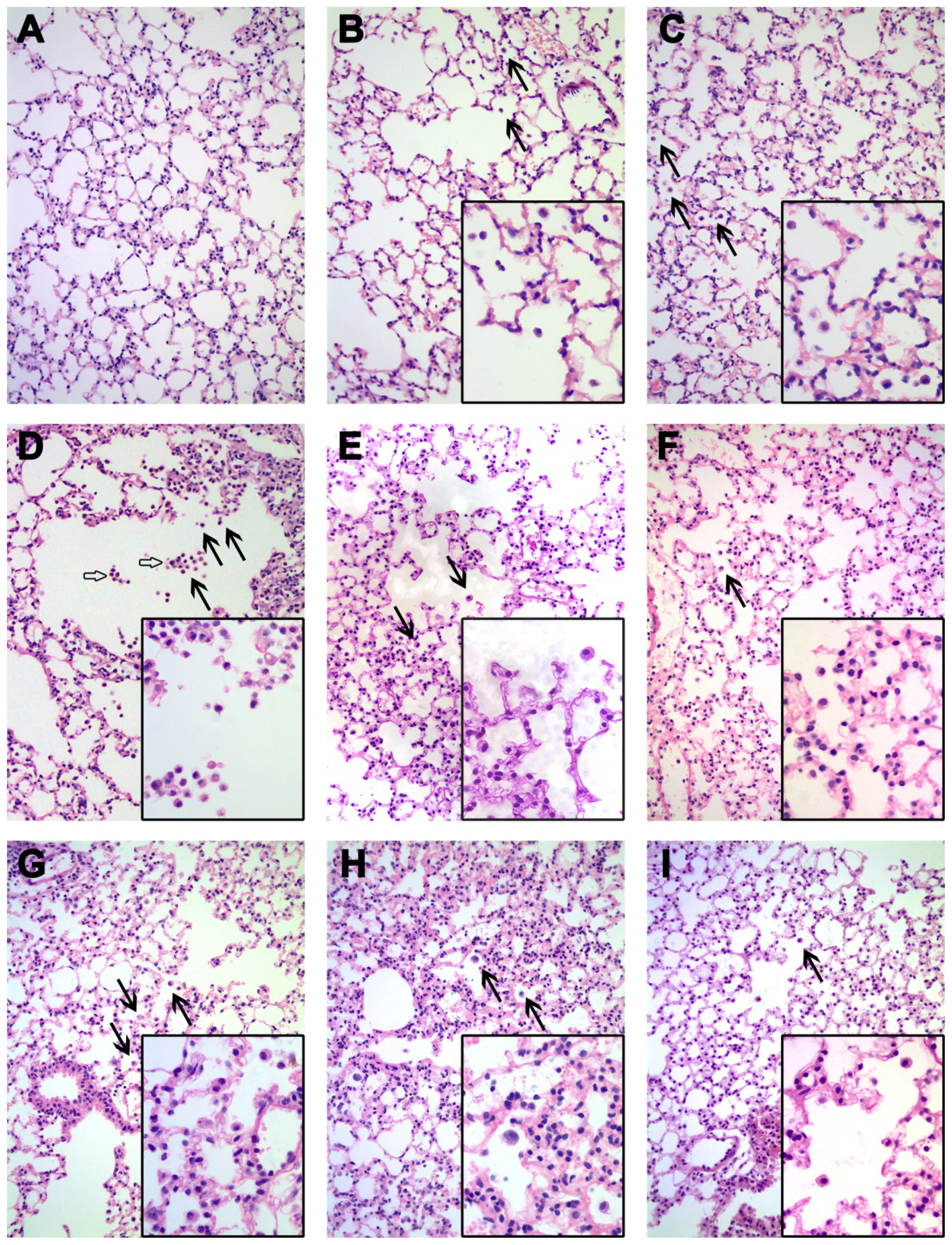
Three pieces of lungs were selected randomly in each group at each time point. The lung tissues were immersed in 10% formaldehyde solution and subsequently embedded into paraffin wax, then sectioned into 5 μm-thick slices and mounted on a glass microscope slide. Then, the slices were stained with HE dye and observed using an optical microscope (Leica DM4000M, Wetzlar, Germany).
4.7. Statistical Analyses
All data were expressed as the mean ± standard deviation (SD). Statistical analyses were conducted with SPSS 16.0 (SPSS Inc., Chicago, IL, USA) statistical software for Windows. A one-way analysis of variance (ANOVA) and Tukey’s test were carried out to illustrate the significant difference between the treatment, and a difference of * p < 0.05 was considered statistically significant.