Identification of Proteins of Altered Abundance in Oil Palm Infected with Ganoderma boninense
Abstract
:1. Introduction
2. Results
2.1. Analysis of Oil Palm Root Proteins by 2-DE
2.2. Image Analysis of 2-DE Oil Palm Root Protein Profiles
2.3. Analysis by Tandem Mass Spectrometry
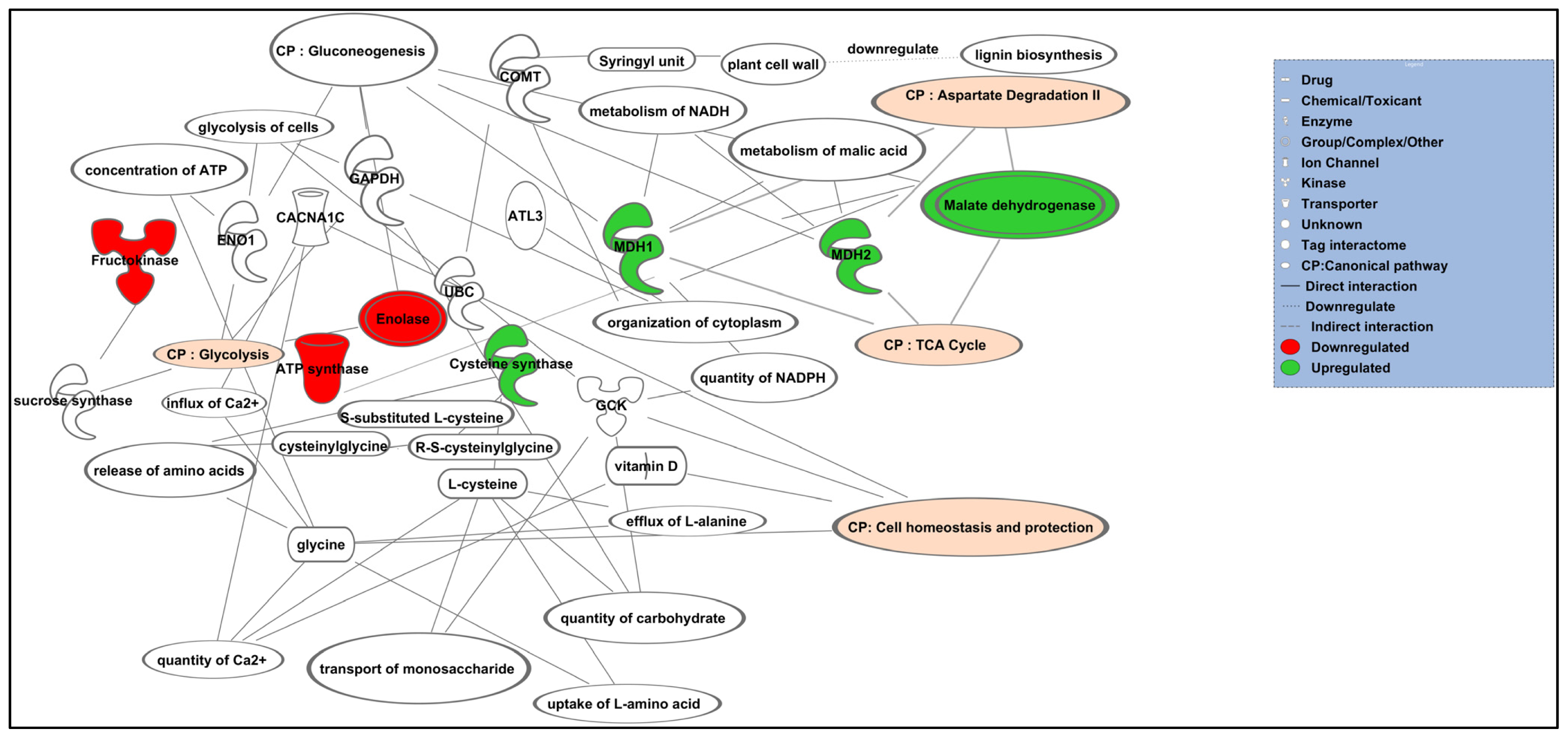
2.4. Pathway Interactions and Biological Process Analysis
3. Discussion
4. Experimental Section
4.1. Plant Materials
4.2. Ganoderma Culture
4.3. Preparation of Wood Blocks for Artificial Inoculation
4.4. Plant Challenge Experiments
4.5. Protein Extraction
4.6. Estimation of Proteins Content
4.7. 2-DE
4.8. Gel Scanning and Image Analysis of Protein Spots
4.9. Mass Spectrometry and Database Query
4.10. Functional Analysis
5. Conclusions
Acknowledgments
Conflicts of Interest
- Author ContributionsJ.R.A., Y.M.-Y., R.Y.O. and O.H.H. designed the experiments. C.-C.T. and N.M.-N. performed the plant challenge experiments. J.R.A. performed the 2-DE analysis. J.R.A. and J.J.J. analysed the MS/MS data and performed database search. N.R. and S.M.J. performed the IPA analysis. J.R.A., R.Y.O. and O.H.H. wrote the manuscript.
References
- Pilotti, C.A. Stem rots of oil palm caused by Ganoderma boninense: Pathogen biology and epidemiology. Mycopathologia 2005, 159, 129–137. [Google Scholar]
- Singh, R.; Ong-Abdullah, M.; Low, E.T.; Manaf, M.A.; Rosli, R.; Nookiah, R.; Ooi, L.C.; Ooi, S.E.; Chan, K.L.; Halim, M.A.; et al. Oil palm genome sequence reveals divergence of interfertile species in old and new worlds. Nature 2013, 500, 335–339. [Google Scholar]
- Santoso, H.; Gunawan, T.; Jatmiko, R.; Darmosarkoro, W.; Minasny, B. Mapping and identifying basal stem rot disease in oil palms in North Sumatra with QuickBird imagery. Precis. Agric. 2011, 12, 233–248. [Google Scholar]
- Susanto, A.; Sudharto, P.S.; Purba, R.Y. Enhancing biological control of basal stem rot disease (Ganoderma boninense) in oil palm plantations. Mycopathologia 2005, 159, 153–157. [Google Scholar]
- Kim, S.T.; Cho, K.S.; Yu, S.; Kim, S.G.; Hong, J.C.; Han, C.D.; Bae, D.W.; Nam, M.H.; Kang, K.Y. Proteomic analysis of differentially expressed proteins induced by rice blast fungus and elicitor in suspension-cultured rice cells. Proteomics 2003, 3, 2368–2378. [Google Scholar]
- Colditz, F.; Nyamsuren, O.; Niehaus, K.; Eubel, H.; Braun, H.P.; Krajinski, F. Proteomic approach: Identification of Medicago truncatula proteins induced in roots after infection with the pathogenic oomycete Aphanomyces euteiches. Plant Mol. Biol. 2004, 55, 109–120. [Google Scholar]
- Zhou, W.; Eudes, F.; Laroche, A. Identification of differentially regulated proteins in response to a compatible interaction between the pathogen Fusarium graminearum and its host Triticum aestivum. Proteomics 2006, 6, 4599–4609. [Google Scholar]
- Gómez-Vidal, S.; Salinas, J.; Tena, M.; Lopez-Llorca, L.V. Proteomic analysis of date palm (Phoenix dactylifera L) responses to endophytic colonization by entomopathogenic fungi. Electrophoresis 2009, 30, 2996–3005. [Google Scholar]
- Bari, R.; Jones, J.D. Role of plant hormones in plant defence responses. Plant Mol. Biol. 2009, 69, 473–488. [Google Scholar]
- Al-Obaidi, J.R.; Mohd-Yusuf, Y.; Chin-Chong, T.; Mhd-Noh, N.; Othman, R.Y. Identification of a partial oil palm polygalacturonase-inhibiting protein (EgPGIP) gene and its expression during basal stem rot infection caused by Ganoderma boninense. Afr. J. Biotechnol. 2010, 9, 7788–7797. [Google Scholar]
- De Franqueville, H.; Asmady, H.; Jacquemard, J.C.; Hayun, Z.; Durand-Gasselin, T. Indications on Sources of Oil Palm (Elæis guineensis Jacq) Genetic Resistance and Susceptibility to Ganoderma sp., the Cause of Basal Stem Rot. In Cutting-Edge Technologies for Sustained Competitiveness; Malaysian Palm Oil Board (MPOB): Kuala Lumpur, Malaysia, 2001; pp. 420–431. [Google Scholar]
- Bridge, P.; Panchal, G.; Sanderson, F.; Pilotti, C. Environmental Sampling for Ganoderma in Oil Palm: A Molecular Tool for Elucidating Epidemiology. Proceedings of International Palm Oil Congress; Canadian Association of Business Incubation, Bangi, Selangor, Malaysia, 20–22 August 2001; pp. 649–652.
- Grenville-Briggs, L.J.; Avrova, A.O.; Bruce, C.R.; Williams, A.; Whisson, S.C.; Birch, P.R.; van West, P. Elevated amino acid biosynthesis in Phytophthora infestans during appressorium formation and potato infection. Fungal Genet. Biol. 2005, 42, 244–256. [Google Scholar]
- Rampitsch, C.; Bykova, N.V.; McCallum, B.; Beimcik, E.; Ens, W. Analysis of the wheat and Puccinia triticina (leaf rust) proteomes during a susceptible host-pathogen interaction. Proteomics 2006, 6, 1897–1907. [Google Scholar]
- Mehta, A.; Brasileiro, A.C.; Souza, D.S.; Romano, E.; Campos, M.A.; Grossi-de-Sa, M.F.; Silva, M.S.; Franco, O.L.; Fragoso, R.R.; Bevitori, R.; et al. Plant–pathogen interactions: What is proteomics telling us? FEBS J. 2008, 275, 3731–3746. [Google Scholar]
- Acero, F.J.; Carbu, M.; El-Akhal, M.R.; Garrido, C.; Gonzalez-Rodriguez, V.E.; Cantoral, J.M. Development of proteomics-based fungicides: New strategies for environmentally friendly control of fungal plant diseases. Int. J. Mol. Sci. 2011, 12, 795–816. [Google Scholar]
- Gonzalez-Fernandez, R.; Prats, E.; Jorrin-Novo, J.V. Proteomics of plant pathogenic fungi. J. Biomed. Biotech. 2010, 2010, 932527. [Google Scholar]
- Shevchenko, A.; Jensen, O.N.; Podtelejnikov, A.V.; Sagliocco, F.; Wilm, M.; Vorm, O.; Mortensen, P.; Shevchenko, A.; Boucherie, H.; Mann, M. Linking genome and proteome by mass spectrometry: Large-scale identification of yeast proteins from two dimensional gels. Proc. Natl. Acad. Sci. USA 1996, 93, 14440–14445. [Google Scholar]
- Syahanim, S.; Abrizah, O.; Mohamad Arif, A.M.; Idris, A.S.; Mohd Din, A. Identification of differentially expressed proteins in oil palm seedlings artificially infected with ganoderma: A proteomics approach. J. Oil Palm Res. 2013, 25, 298–304. [Google Scholar]
- Castillejo, M.A.; Curto, M.; Fondevilla, S.; Rubiales, D.; Jorrin, J.V. Two-dimensional electrophoresis based proteomic analysis of the pea (Pisum sativum) in response to Mycosphaerella pinodes. J. Agric. Food Chem. 2010, 58, 12822–12832. [Google Scholar]
- Geddes, J.; Eudes, F.; Laroche, A.; Selinger, L.B. Differential expression of proteins in response to the interaction between the pathogen Fusarium graminearum and its host Hordeum vulgare. Proteomics 2008, 8, 545–554. [Google Scholar]
- Collins, R.M.; Afzal, M.; Ward, D.A.; Prescott, M.C.; Sait, S.M.; Rees, H.H.; Tomsett, A.B. Differential proteomic analysis of Arabidopsis thaliana genotypes rxhibiting tesistance or susceptibility to the insect herbivore Plutella xylostella. PLoS One 2010, 5, e10103. [Google Scholar]
- Lance, C.; Rustin, P. The central role of malate in plant-metabolism. Physiol. Veg. 1984, 22, 625–641. [Google Scholar]
- Mooney, B.P.; Miernyk, J.A.; Michael Greenlief, C.; Thelen, J.J. Using quantitative proteomics of Arabidopsis roots and leaves to predict metabolic activity. Physiol. Plant 2006, 128, 237–250. [Google Scholar]
- Capitani, G.; Hohenester, E.; Feng, L.; Storici, P.; Kirsch, J.F.; Jansonius, J.N. Structure of 1-aminocyclopropane-1-carboxylate synthase a key enzyme in the biosynthesis of the plant hormone ethylene. J. Mol. Biol. 1999, 294, 745–756. [Google Scholar]
- Tsang, E.T.; Yang, J.; Chang, Q.; Nowak, G.; Kolenovsky, A.; McGregor, D.I.; Keller, W. Chlorophyll reduction in the seed of Brassica napus with a glutamate 1-semialdehyde aminotransferase antisense gene. Plant Mol. Biol. 2003, 51, 191–201. [Google Scholar]
- Dahal, D.; Pich, A.; Braun, H.P.; Wydra, K. Analysis of cell wall proteins regulated in stem of susceptible and resistant tomato species after inoculation with Ralstonia solanacearum: A proteomic approach. Plant Mol. Biol. 2010, 73, 643–658. [Google Scholar]
- Miles, L.A.; Dahlberg, C.M.; Plescia, J.; Felez, J.; Kato, K.; Plow, E.F. Role of cell-surface lysines in plasminogen binding to cells: Identification of α-enolase as a candidate plasminogen receptor. Biochemistry 1991, 30, 1682–1691. [Google Scholar]
- Li, C.; Bonnema, G.; Che, D.; Dong, L.; Lindhout, P.; Visser, R.; Bai, Y. Biochemical and molecular mechanisms involved in monogenic resistance responses to tomato powdery mildew. Mol. Plant 2007, 20, 1161–1172. [Google Scholar]
- Rode, C.; Gallien, S.; Heintz, D.; van Dorsselaer, A.; Braun, H.P.; Winkelmann, T. Enolases: Storage compounds in seeds? Evidence from a proteomic comparison of zygotic and somatic embryos of Cyclamen persicum Mill. Plant Mol. Biol. 2011, 75, 305–319. [Google Scholar]
- Ostergaard, O.; Finnie, C.; Laugesen, S.; Roepstorff, P.; Svennson, B. Proteome analysis of barley seeds: Identification of major proteins from two-dimensional gels (pI 4–7). Proteomics 2004, 4, 2437–2447. [Google Scholar]
- Kortstee, A.J.; Appeldoorn, N.J.; Oortwijn, M.E.; Visser, R.G. Differences in regulation of carbohydrate metabolism during early fruit development between domesticated tomato and two wild relatives. Planta 2007, 226, 929–939. [Google Scholar]
- Curto, M.; Camafeita, E.; Lopez, J.A.; Maldonado, A.M.; Rubiales, D.; Jorrin, J.V. A proteomic approach to study pea (Pisum sativum) responses to powdery mildew (Erysiphe pisi). Proteomics 2006, 6, S163–S174. [Google Scholar]
- Sharma, R.; Tan, F.; Jung, K.H.; Sharma, M.K.; Peng, Z.; Ronald, P.C. Transcriptional dynamics during cell wall removal and regeneration reveals key genes involved in cell wall development in rice. Plant Mol. Biol. 2011, 77, 391–406. [Google Scholar]
- Ryu, H.S.; Han, M.; Lee, S.K.; Cho, J.I.; Ryoo, N.; Heu, S.; Lee, Y.H.; Bhoo, S.H.; Wang, G.L.; Hahn, T.R.; et al. A comprehensive expression analysis of the WRKY gene superfamily in rice plants during defense response. Plant Cell Rep. 2006, 25, 836–847. [Google Scholar]
- McCarty, R.E.; Evron, Y.; Johnson, E.A. The chloroplast atp synthase: A rotary enzyme? Ann. Rev. Plant Physiol. Plant Mol. Biol. 2000, 51, 83–109. [Google Scholar]
- Tsunezuka, H.; Fujiwara, M.; Kawasaki, T.; Shimamoto, K. Proteome analysis of programmed cell death and defense signaling using the rice lesion mimic mutant cdr2. Mol. Plant Microb. Interact. 2005, 18, 52–59. [Google Scholar]
- Wongpia, A.; Lomthaisong, K. Changes in the 2DE protein profiles of chilli pepper (Capsicum annuum) leaves in response to Fusarium oxysporum infection. Scienceasia 2010, 36, 259–270. [Google Scholar]
- Kim, J.Y.; Park, S.C.; Hwang, I.; Cheong, H.; Nah, J.W.; Hahm, K.S.; Park, Y. Protease inhibitors from plants with antimicrobial activity. Int. J. Mol. Sci. 2009, 10, 2860–2872. [Google Scholar]
- Zhang, J.; Erickson, L.R. Harvest-inducibility of the promoter of alfalfa S-adenosyl-L-methionine: trans-Caffeoyl-CoA3-O-methyltransferase gene. Mol. Biol. Rep. 2012, 39, 2489–2495. [Google Scholar]
- Chen, C.; Meyermans, H.; Burggraeve, B.; de Rycke, R.M.; Inoue, K.; de Vleesschauwer, V.; Steenackers, M.; van Montagu, M.C.; Engler, G.J.; Boerjan, W.A. Cell-specific and conditional expression of caffeoyl-coenzyme A-3-O-methyltransferase in poplar. Plant Physiol. 2000, 123, 853–867. [Google Scholar]
- Paterson, R.R.M.; Meon, S.; Abidin, M.A.Z.; Lima, N. Prospects for inhibition of lignin degrading enzymes to control Ganoderma white rot of oil palm. Curr. Enzym. Inhibit. 2008, 4, 172–179. [Google Scholar]
- Ye, Z.H.; Zhong, R.; Morrison, W.H., 3rd; Himmelsbach, D.S. Caffeoyl coenzyme A O-methyltransferase and lignin biosynthesis. Phytochemistry 2001, 57, 1177–1185. [Google Scholar]
- Paterson, R.R.M. Ganoderma disease of oil palm—A white rot perspective necessary for integrated control. Crop Protect. 2007, 26, 1369–1376. [Google Scholar] [Green Version]
- Paterson, R.R.M.; Moen, S.; Lima, N. The feasibility of producing oil palm with altered lignin content to control Ganoderma disease. J. Phytopathol. 2009, 157, 649–656. [Google Scholar]
- Baucher, M.; Halpin, C.; Petit-Conil, M.; Boerjan, W. Lignin: Genetic engineering and impact on pulping. Crit. Rev. Biochem. Mol. Biol. 2003, 38, 305–350. [Google Scholar]
- Vanholme, R.; Ralph, J.; Akiyama, T.; Lu, F.; Pazo, J.R.; Kim, H.; Christensen, J.H.; van Reusel, B.; Storme, V.; de Rycke, R.; et al. Engineering traditional monolignols out of lignin by concomitant up-regulation of F5H1 and down-regulation of COMT in Arabidopsis. Plant J. 2010, 64, 885–897. [Google Scholar]
- Pincon, G.; Chabannes, M.; Lapierre, C.; Pollet, B.; Ruel, K.; Joseleau, J.P.; Boudet, A.M.; Legrand, M. Simultaneous down-regulation of caffeic/5-hydroxy ferulic acid-O-methyltransferase I and cinnamoyl-coenzyme A reductase in the progeny from a cross between tobacco lines homozygous for each transgene Consequences for plant development and lignin synthesis. Plant Physiol. 2001, 126, 145–155. [Google Scholar]
- Piquemal, J.; Lapierre, C.; Myton, K.; O’connell, A.; Schuch, W.; Grima-pettenati, J.; Boudet, A.-M. Down-regulation of Cinnamoyl-CoA reductase induces significant changes of lignin profiles in transgenic tobacco plants. Plant J. 1998, 13, 71–83. [Google Scholar]
- Bhadauria, V.; Banniza, S.; Wei, Y.; Peng, Y.L. Reverse genetics for functional genomics of phytopathogenic fungi and oomycetes. Comp. Funct. Genomics 2009, 2009, 380719. [Google Scholar]
- Shimizu, M.; Yuda, N.; Nakamura, T.; Tanaka, H.; Wariishi, H. Metabolic regulation at the tricarboxylic acid and glyoxylate cycles of the lignin-degrading basidiomycete Phanerochaete chrysosporium against exogenous addition of vanillin. Proteomics 2005, 5, 3919–3931. [Google Scholar]
- Ketudat Cairns, J.R.; Esen, A. β-Glucosidases. Cell. Mol. Life Sci. 2010, 67, 3389–3405. [Google Scholar]
- Moldenhauer, J.; Moerschbacher, B.M.; van der Westhuizen, A.J. Histological investigation of stripe rust (Puccinia striiformis F sp tritici) development in resistant and susceptible wheat cultivars. Plant Pathol. 2006, 55, 469–474. [Google Scholar]
- Nair, K.S.; Jaleel, A.; Asmann, Y.W.; Short, K.R.; Raghavakaimal, S. Proteomic research: Potential opportunities for clinical and physiological investigators. Am. J. Physiol. 2004, 286, E863–E874. [Google Scholar]
- Ariffin, D.; Idris, S. The Ganoderma Selective Media (GSM); PORIM Information Series; Palm Oil Research Institute: Bangi, Malaysia, 1992. [Google Scholar]
- Idris, S.; Arifin, D.; Swinburne, R.; Watt, A.A. The Identity of Ganoderma Species Responsible for BSR Disease of Oil Palm in Malaysia Morphological Characteristics; MPOB Information Series; Palm Oil Research Institute: Bangi, Malaysia, 2000. [Google Scholar]
- Idris, S.; Khusair, I.D.; Ariffin, D.; Basri, W. Technique for Inoculation of Oil Palm Germinated Seeds with Ganoderma; MPOB Information Series; Palm Oil Research Institute: Bangi, Malaysia, 2006; pp. 1–4. [Google Scholar]
- NurAinIzzati, M.Z.; Abdullah, F. Disease suppression in Ganoderma-infected oil palm seedlings treated with Trichoderma harzianum. Plant Protect. Sci. 2008, 3, 101–107. [Google Scholar]
- Carpentier, S.C.; Witters, E.; Laukens, K.; Deckers, P.; Swennen, R.; Panis, B. Preparation of protein extracts from recalcitrant plant tissues: An evaluation of different methods for two-dimensional gel electrophoresis analysis. Proteomics 2005, 5, 2497–2507. [Google Scholar]
- Xiao, X.; Yang, F.; Zhang, S.; Korpelainen, H.; Li, C. Physiological and proteomic responses of two contrasting Populus cathayana populations to drought stress. Physiol. Plant 2009, 136, 150–168. [Google Scholar]
- Bringans, S.; Eriksen, S.; Kendrick, T.; Gopalakrishnakone, P.; Livk, A.; Lock, R.; Lipscombe, R. Proteomic analysis of the venom of Heterometrus longimanus (Asian black scorpion). Proteomics 2008, 8, 1081–1096. [Google Scholar]


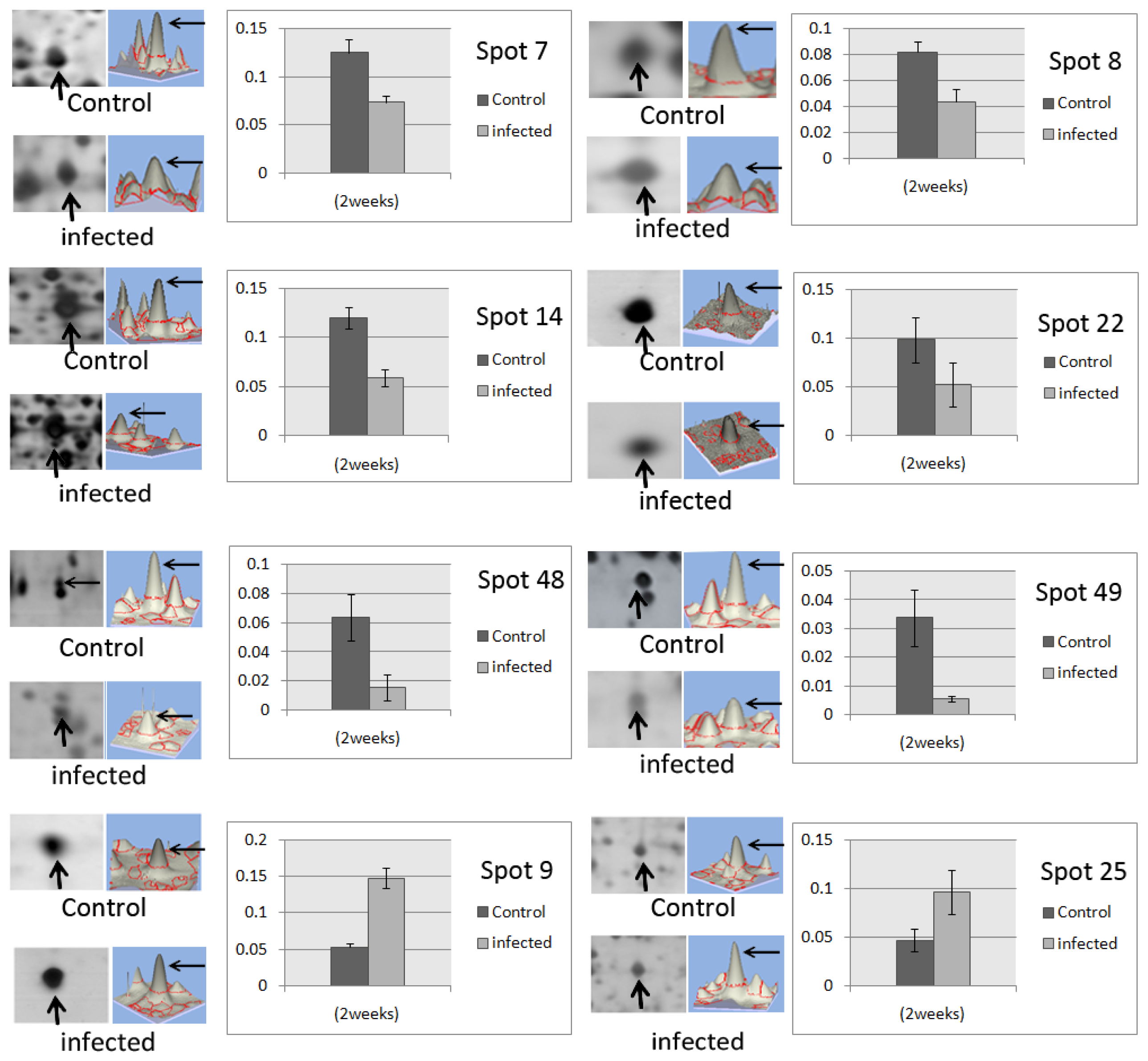
| Spot ID | Accession a | Protein | FC b | p-value | MW c | pI | Score | MP d | Cov (%) e | Organism | Biological process f | Molecular function f |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 7 | G9JLN6 | ATP synthase subunit alpha | −1.64 | 0.0043 | 55,602 | 5.84 | 387 | 29 | 33 | Oryza brachyantha | ATP hydrolysis coupled proton transport; ATP synthesis coupled proton transport | ATP binding |
| 8 | Q9SYR8 | Catechol O-methyltransferase | −1.80 | 0.002 | 40,192 | 5.57 | 88 | 2 | 6 | Thalictrum tuberosum | O-methyltransferase activity | |
| 9 | I1R814 | Cysteine synthase | +3.17 | 0.0056 | 43,774 | 8.76 | 63 | 2 | 4 | Oryza glaberrima | cysteine biosynthetic process from serine | cysteine synthase activity; transferase activity |
| 14 | B9R9N6 | Enolase | −2.01 | 0.0078 | 48,149 | 5.71 | 199 | 16 | 28 | Ricinus communis | glycolysis | magnesium ion binding; phosphopyruvate hydratase activity |
| 22 | B3TLU4 | Enolase | −1.88 | 0.00016 | 48,127 | 5.98 | 80 | 7 | 11 | Elaeis guineensis | glycolysis | magnesium ion binding; phosphopyruvate hydratase activity |
| 25 | F6HZK0 | Malate dehydrogenase | +2.06 | 0.0040 | 39,405 | 6.67 | 121 | 5 | 18 | Vitis vinifera | cellular carbohydrate metabolic process; malate metabolic process; tricarboxylic acid cycle | L-malate dehydrogenase activity |
| 28 | I1HVU4 | Uncharacterized protein | +10.43 | 0.005 | 38,996 | 6.09 | 66 | 2 | 4 | Brachypodiu m distachyon | nutrient reservoir activity | |
| 36 | M0YUL2 | Uncharacterized protein | −2.60 | 0.00011 | 38,971 | 8.47 | 105 | 4 | 13 | Hordeum vulgare | ||
| 48 | Q0J8G4 | Fructokinase-2 | −4.15 | 0.0072 | 35,893 | 5.02 | 86 | 3 | 9 | Oryza sativa | starch biosynthestic process | ATP binding; fructokinase activity |
| 49 | G1FCF5 | Fructokinase | −6.04 | 0.013 | 36,460 | 5.61 | 72 | 2 | 7 | Dimocarpus longan | D-ribose metabolic process | ribokinase activity |
| Top canonical pathway | p value | Ratio | List of protein |
|---|---|---|---|
| Gluconeogenesis | 4.36 × 10−8 | 2/47 (1.043) | Enolase, Malate dehydrogenase |
| Cell homeostasis | 1.53 × 10−6 | 1/14 (0.071) | Catechol O-methyltransferase |
| TCA cycle | 2.47 × 10−5 | 2/41 (0.049) | Malate dehydrogenase, ATP synthase alpha subunit |
| Glycolysis | 8.28 × 10−3 | 3/40 (0.075) | Fructokinase, enolase, ATP synthase alpha subunit |
| Aspartate degradation II | 8.35 × 10−3 | 1/14 (0.071) | Malate dehydrogenase |
| ID | Associated Network Function | Score a | |
| 1 | Cellular Function and Maintenance, Energy Production and Lipid Metabolism | 20 | |
| 2 | Amino Acid Metabolism, Small Molecule Biochemistry, Molecular Transport | 2 | |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Al-Obaidi, J.R.; Mohd-Yusuf, Y.; Razali, N.; Jayapalan, J.J.; Tey, C.-C.; Md-Noh, N.; Junit, S.M.; Othman, R.Y.; Hashim, O.H. Identification of Proteins of Altered Abundance in Oil Palm Infected with Ganoderma boninense. Int. J. Mol. Sci. 2014, 15, 5175-5192. https://doi.org/10.3390/ijms15035175
Al-Obaidi JR, Mohd-Yusuf Y, Razali N, Jayapalan JJ, Tey C-C, Md-Noh N, Junit SM, Othman RY, Hashim OH. Identification of Proteins of Altered Abundance in Oil Palm Infected with Ganoderma boninense. International Journal of Molecular Sciences. 2014; 15(3):5175-5192. https://doi.org/10.3390/ijms15035175
Chicago/Turabian StyleAl-Obaidi, Jameel R., Yusmin Mohd-Yusuf, Nurhanani Razali, Jaime Jacqueline Jayapalan, Chin-Chong Tey, Normahnani Md-Noh, Sarni Mat Junit, Rofina Yasmin Othman, and Onn Haji Hashim. 2014. "Identification of Proteins of Altered Abundance in Oil Palm Infected with Ganoderma boninense" International Journal of Molecular Sciences 15, no. 3: 5175-5192. https://doi.org/10.3390/ijms15035175






