Bovine Induced Pluripotent Stem Cells Are More Resistant to Apoptosis than Testicular Cells in Response to Mono-(2-ethylhexyl) Phthalate
Abstract
:1. Introduction
2. Results
2.1. Generation of iPSCs from Bovine Testis Cells
2.2. Pluripotency of 1F-iPSCs
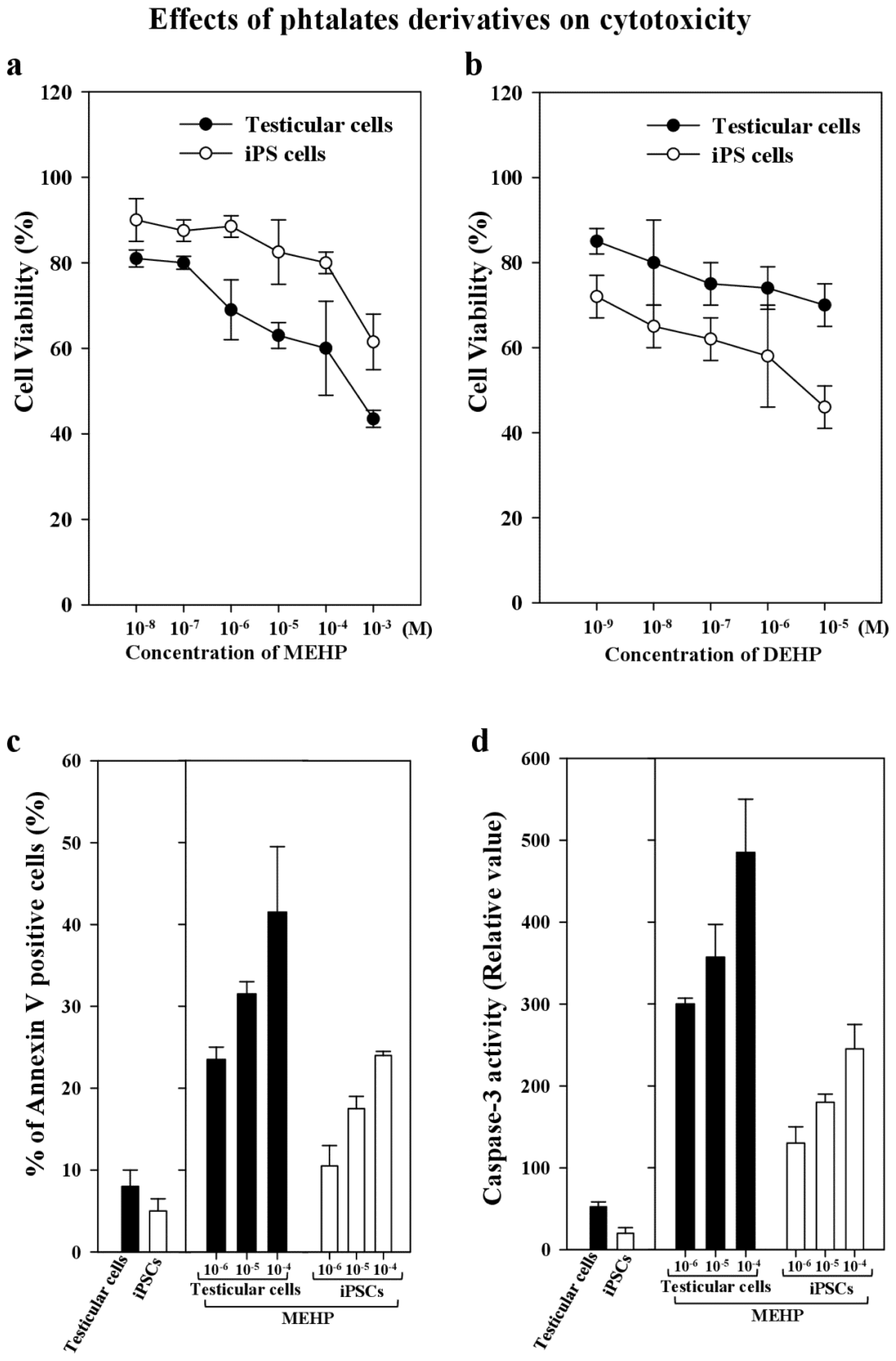
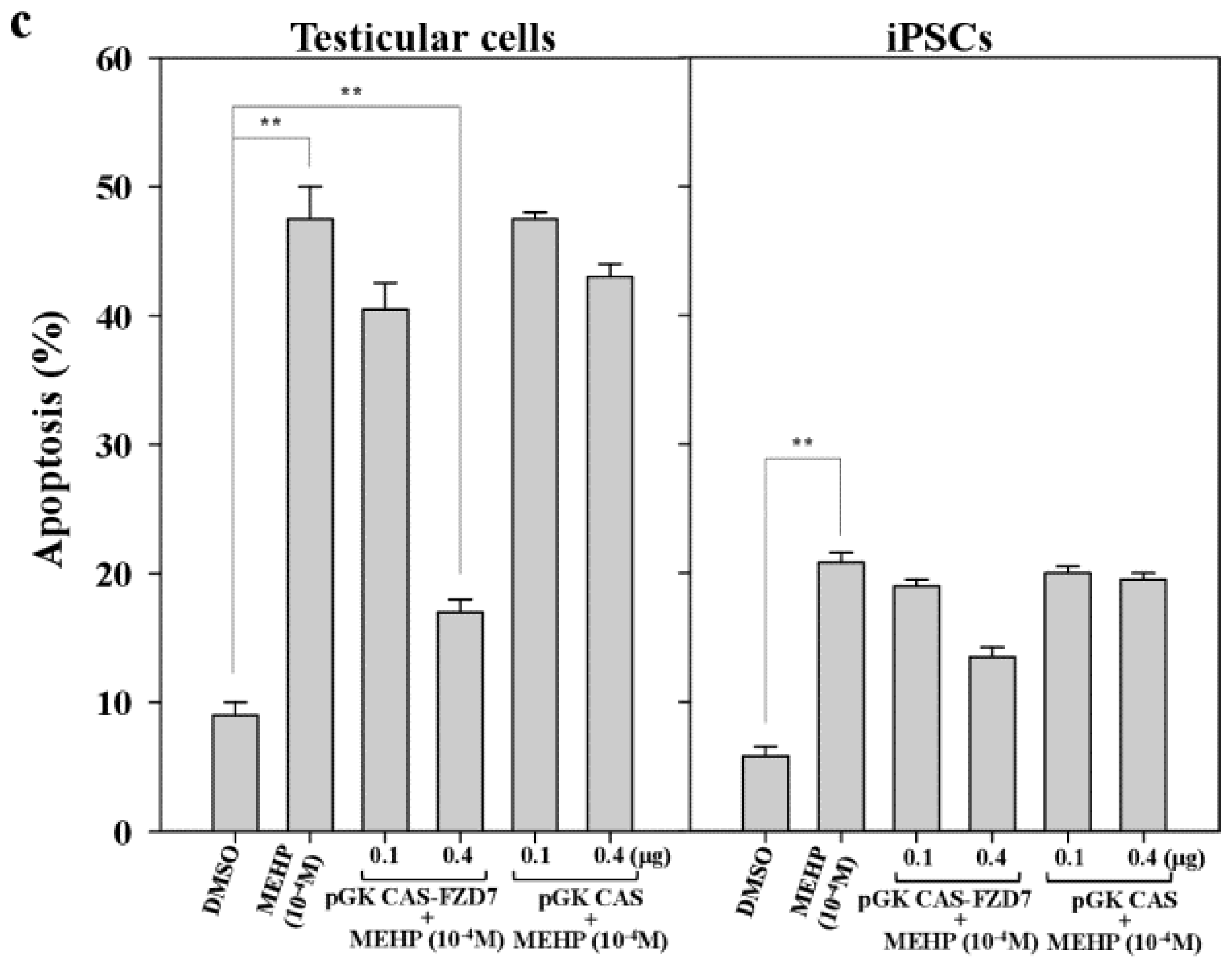
2.3. Effects of the Phthalate Ester MEHP on Apoptosis
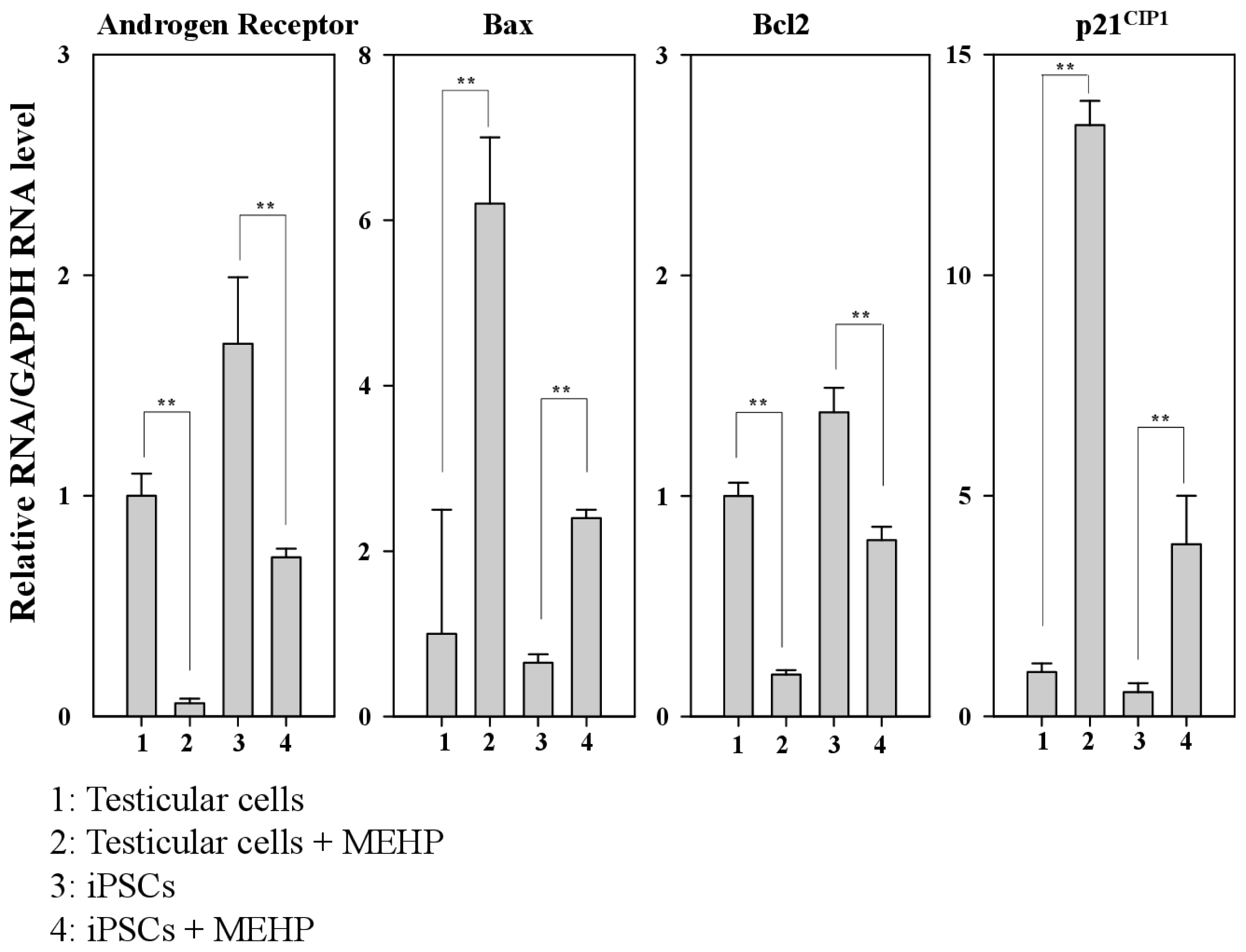
2.4. Regulation of AR, p21CIP1, and Apoptosis
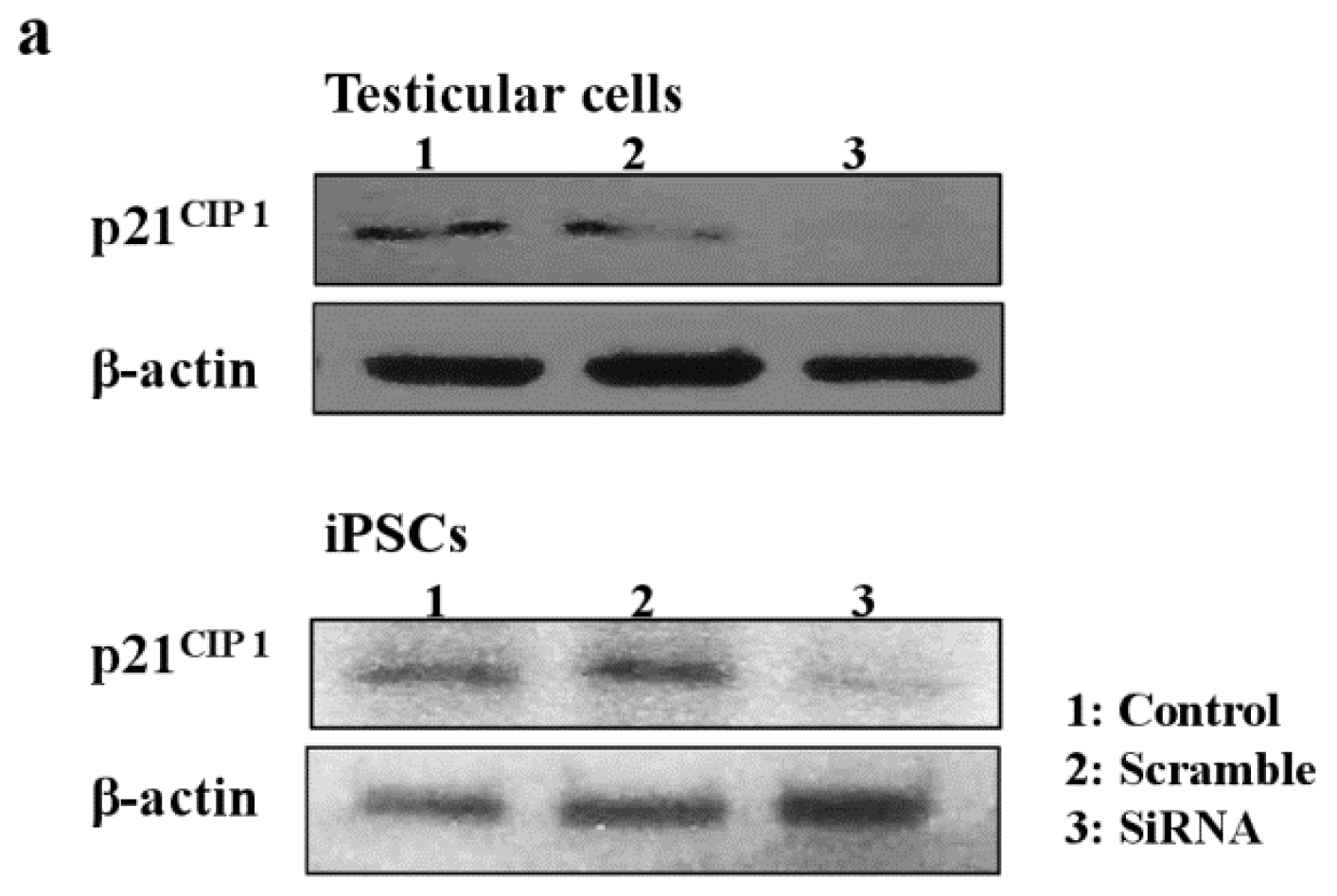
2.5. Role of p21CIP1 in Phthalate-Induced Apoptosis
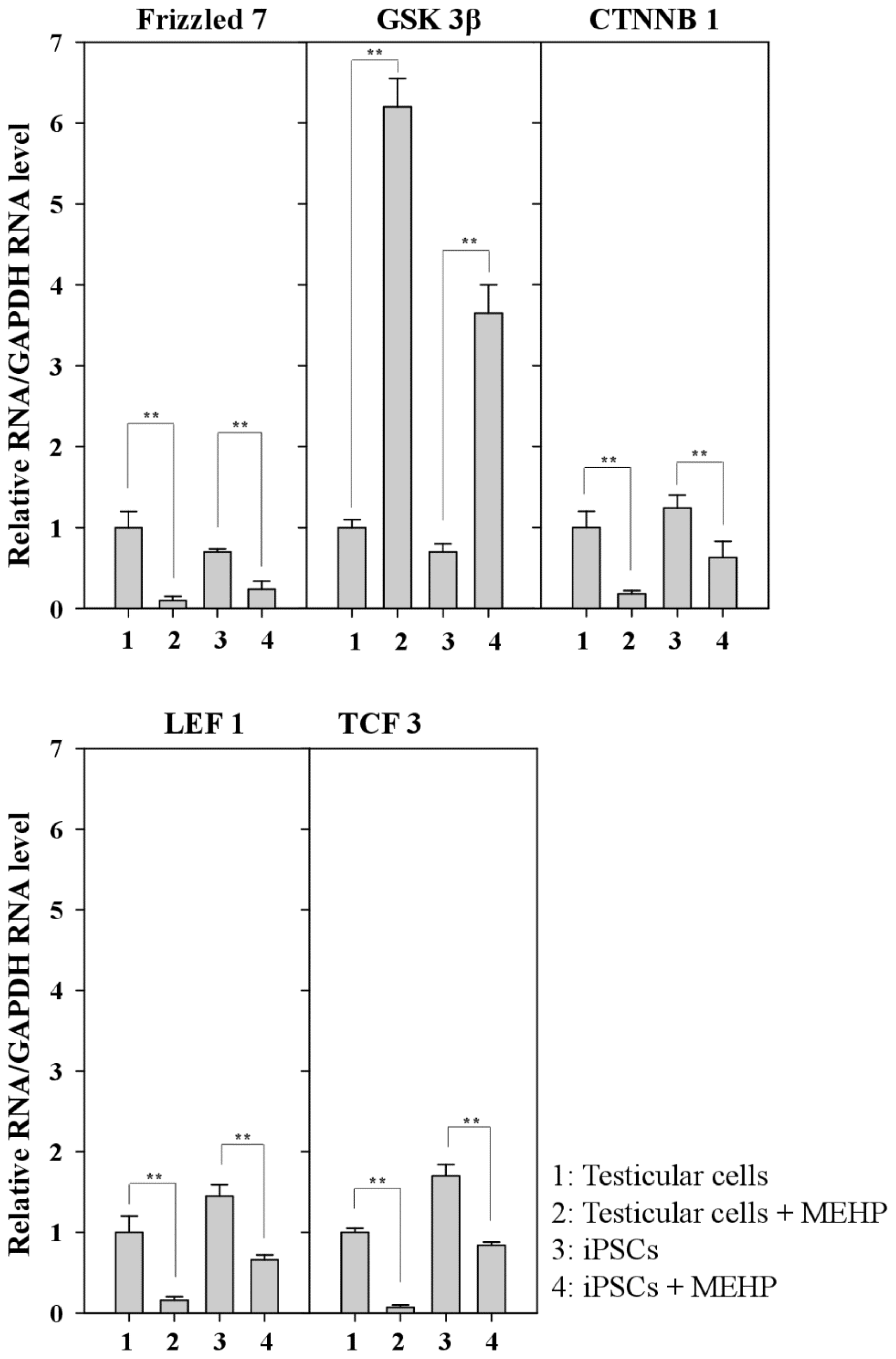
2.6. Repression of the Wnt Receptor Frizzled 7 by MEHP
2.7. Effect of MEHP on WNT/Frizzled Signaling and AR-Mediated Apoptosis
3. Discussion
4. Experimental Section
4.1. Reagents and Plasmids
4.2. Culture of Bovine Testicular Cells
4.3. Generation of iPSCs
4.4. Stemness Assay and Karyotyping
4.5. Cell Viability and Apoptosis
4.6. RNA Extraction and Quantitative Polymerase Chain Reaction (qPCR)
4.7. Transfection, Knock down and Luciferase Assay
4.8. Western Blot Analysis
4.9. Microwestern Arrays
4.10. Teratoma Formation Assay
4.11. Statistical Analysis
5. Conclusions
Supplementary Information
ijms-15-05011-s001.pdfAcknowledgments
Conflicts of Interest
Abbreviations
| AR | androgen receptor |
| DEHP | di-(2-ethylhexyl) phthalate |
| DMSO | dimethyl sulfoxide |
| EDCs | endocrine-disrupting chemicals |
| FZD7 | Frizzled receptor 7 |
| iPSCs | induced pluripotent stem cells |
| MEFs | mouse embryonic fibroblasts |
| MEHP | mono-(2-ethylhexyl) phthalate |
| OCT4 | octamer-binding transcription factor 4 |
| p21CIP1 | cycling-dependent kinase inhibitor 1 |
| qPCR | quantitative RT–PCR |
| RT–PCR | reverse transcription polymerase chain reaction |
- Author ContributionsYCL, SS and KKY designed and performed the experiments, analyzed the data, and wrote the manuscript; KW, SHL, CCK, YHY, CCW, and SWW performed the experiments, and analyzed the data; and KKK, SWW, DCW, CYC, CLL, CSL, MK, HM, YN, SH, KM, CJ, and SKH analyzed the data, consulted, and helped the manuscript preparation.
References
- Lyche, J.L.; Gutleb, A.C.; Bergman, A.; Eriksen, G.S.; Murk, A.J.; Ropstad, E.; Saunders, M.; Skaare, J.U. Reproductive and developmental toxicity of phthalates. J. Toxicol. Environ. Health Part B 2009, 12, 225–249. [Google Scholar]
- Jurewicz, J.; Hanke, W. Exposure to phthalates: Reproductive outcome and children health A review of epidemiological studies. Int. J. Occup. Med. Environ. Health 2011, 24, 115–141. [Google Scholar]
- Sjoberg, P.; Lindqvist, N.G.; Ploen, L. Age-dependent response of the rat testes to di(2-ethylhexyl) phthalate. Environ. Health Perspect. 1986, 65, 237–242. [Google Scholar]
- Awal, M.A.; Kurohmaru, M.; Ishii, M.; Andriana, B.B.; Kanai, Y.; Hayashi, Y. Mono-(2-ethylhexyl) phthalate (MEHP) induces spermatogenic cell apoptosis in guinea pig testes at prepubertal stage in vitro. Int. J. Toxicol. 2004, 23, 349–355. [Google Scholar]
- Lehraiki, A.; Racine, C.; Krust, A.; Habert, R.; Levacher, C. Phthalates impair germ cell number in the mouse fetal testis by an androgen- and estrogen-independent mechanism. Toxicol. Sci. 2009, 111, 372–382. [Google Scholar]
- Casals-Casas, C.; Desvergne, B. Endocrine disruptors: From endocrine to metabolic disruption. Annu. Rev. Physiol. 2011, 73, 135–162. [Google Scholar]
- Kwack, S.J.; Kim, K.B.; Kim, H.S.; Lee, B.M. Comparative toxicological evaluation of phthalate diesters and metabolites in Sprague-Dawley male rats for risk assessment. J. Toxicol. Environ. Health Part A 2009, 72, 1446–1454. [Google Scholar]
- Lambrot, R.; Muczynski, V.; Lecureuil, C.; Angenard, G.; Coffigny, H.; Pairault, C.; Moison, D.; Frydman, R.; Habert, R.; Rouiller-Fabre, V. Phthalates impair germ cell development in the human fetal testis in vitro without change in testosterone production. Environ. Health Perspect. 2009, 117, 32–37. [Google Scholar]
- Muczynski, V.; Cravedi, J.P.; Lehraiki, A.; Levacher, C.; Moison, D.; Lecureuil, C.; Messiaen, S.; Perdu, E.; Frydman, R.; Habert, R.; et al. Effect of mono-(2-ethylhexyl) phthalate on human and mouse fetal testis: In vitro and in vivo approaches. Toxicol. Appl. Pharmacol. 2012, 261, 97–104. [Google Scholar]
- Takahashi, K.; Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006, 126, 663–676. [Google Scholar]
- Ying, Q.L.; Wray, J.; Nichols, J.; Batlle-Morera, L.; Doble, B.; Woodgett, J.; Cohen, P.; Smith, A. The ground state of embryonic stem cell self-renewal. Nature 2008, 453, 519–523. [Google Scholar]
- Hanna, J.H.; Saha, K.; Jaenisch, R. Pluripotency and cellular reprogramming: Facts hypotheses unresolved issues. Cell 2010, 143, 508–525. [Google Scholar]
- Martello, G.; Sugimoto, T.; Diamanti, E.; Joshi, A.; Hannah, R.; Ohtsuka, S.; Gottgens, B.; Niwa, H.; Smith, A. Esrrb is a pivotal target of the Gsk3/Tcf3 axis regulating embryonic stem cell self-renewal. Cell Stem Cell 2012, 11, 491–504. [Google Scholar]
- MacDonald, B.T.; Tamai, K.; He, X. Wnt/beta-catenin signaling: Components mechanisms and diseases. Dev. Cell 2009, 17, 9–26. [Google Scholar]
- Fernandez, A.; Huggins, I.J.; Perna, L.; Brafman, D.; Lu, D.; Yao, S.; Gaasterland, T.; Carson, D.A.; Willert, K. The WNT receptor FZD7 is required for maintenance of the pluripotent state in human embryonic stem cells. Proc. Natl. Acad. Sci. USA 2014, 111, 1409–1414. [Google Scholar]
- Catena, R.; Tiveron, C.; Ronchi, A.; Porta, S.; Ferri, A.; Tatangelo, L.; Cavallaro, M.; Favaro, R.; Ottolenghi, S.; Reinbold, R.; et al. Conserved POU binding DNA sites in the Sox2 upstream enhancer regulate gene expression in embryonic and neural stem cells. J. Biol. Chem. 2004, 279, 41846–41857. [Google Scholar]
- Saito, S.; Sawai, K.; Ugai, H.; Moriyasu, S.; Minamihashi, A.; Yamamoto, Y.; Hirayama, H.; Kageyama, S.; Pan, J.; Murata, T.; et al. Generation of cloned calves and transgenic chimeric embryos from bovine embryonic stem-like cells. Biochem. Biophys. Res. Commun. 2003, 309, 104–113. [Google Scholar]
- Kim, J.B.; Sebastiano, V.; Wu, G.; Arauzo-Bravo, M.J.; Sasse, P.; Gentile, L.; Ko, K.; Ruau, D.; Ehrich, M.; van den Boom, D.; et al. Oct4-induced pluripotency in adult neural stem cells. Cell 2009, 136, 411–419. [Google Scholar]
- Wang, S.W.; Wang, S.S.; Wu, D.C.; Lin, Y.C.; Ku, C.C.; Wu, C.C.; Chai, C.Y.; Lee, J.N.; Tsai, E.M.; Lin, C.L.; et al. Androgen receptor-mediated apoptosis in bovine testicular induced pluripotent stem cells in response to phthalate esters. Cell Death Dis. 2013, 4, e907. [Google Scholar]
- Fisher, J.S. Environmental anti-androgens and male reproductive health: Focus on phthalates and testicular dysgenesis syndrome. Reproduction 2004, 127, 305–315. [Google Scholar]
- Lin, Y.; Kokontis, J.; Tang, F.; Godfrey, B.; Liao, S.; Lin, A.; Chen, Y.; Xiang, J. Androgen and its receptor promote Bax-mediated apoptosis. Mol. Cell. Biol. 2006, 26, 1908–1916. [Google Scholar]
- Balk, S.P.; Knudsen, K.E. AR the cell cycle and prostate cancer. Nuclear Recept. Signal. 2008, 6, e001. [Google Scholar]
- Lin, H.K.; Yeh, S.; Kang, H.Y.; Chang, C. Akt suppresses androgen-induced apoptosis by phosphorylating and inhibiting androgen receptor. Proc. Natl. Acad. Sci. USA 2001, 98, 7200–7205. [Google Scholar]
- Tang, F.; Kokontis, J.; Lin, Y.; Liao, S.; Lin, A.; Xiang, J. Androgen via p21 inhibits tumor necrosis factor alpha-induced JNK activation and apoptosis. J. Biol. Chem. 2009, 284, 32353–32358. [Google Scholar]
- Garay, J.P.; Karakas, B.; Abukhdeir, A.M.; Cosgrove, D.P.; Gustin, J.P.; Higgins, M.J.; Konishi, H.; Konishi, Y.; Lauring, J.; Mohseni, M.; et al. The growth response to androgen receptor signaling in ERalpha-negative human breast cells is dependent on p21 and mediated by MAPK activation. Breast Cancer Res. 2012, 14, R27. [Google Scholar]
- Terry, S.; Yang, X.; Chen, M.W.; Vacherot, F.; Buttyan, R. Multifaceted interaction between the androgen and Wnt signaling pathways and the implication for prostate cancer. J. Cell. Biochem. 2006, 99, 402–410. [Google Scholar]
- Wang, G.; Wang, J.; Sadar, M.D. Crosstalk between the androgen receptor and beta-catenin in castrate-resistant prostate cancer. Cancer Res. 2008, 68, 9918–9927. [Google Scholar]
- Rodda, D.J.; Chew, J.L.; Lim, L.H.; Loh, Y.H.; Wang, B.; Ng, H.H.; Robson, P. Transcriptional regulation of nanog by OCT4 and SOX2. J. Biol. Chem. 2005, 280, 24731–24737. [Google Scholar]
- Golestaneh, N.; Kokkinaki, M.; Pant, D.; Jiang, J.; DeStefano, D.; Fernandez-Bueno, C.; Rone, J.D.; Haddad, B.R.; Gallicano, G.I.; Dym, M. Pluripotent stem cells derived from adult human testes. Stem Cells Dev. 2009, 18, 1115–1126. [Google Scholar]
- Niwa, H.; Miyazaki, J.; Smith, A.G. Quantitative expression of Oct-3/4 defines differentiation dedifferentiation or self-renewal of ES cells. Nat. Genet. 2000, 24, 372–376. [Google Scholar]
- Montserrat, N.; Bahima, E.G.; Batlle, L.; Hafner, S.; Rodrigues, A.M.; Gonzalez, F.; Izpisua Belmonte, J.C. Generation of pig iPS cells: A model for cell therapy. J. Cardiovasc. Transl. Res. 2011, 4, 121–130. [Google Scholar]
- Han, X.; Han, J.; Ding, F.; Cao, S.; Lim, S.S.; Dai, Y.; Zhang, R.; Zhang, Y.; Lim, B.; Li, N. Generation of induced pluripotent stem cells from bovine embryonic fibroblast cells. Cell Res. 2011, 21, 1509–1512. [Google Scholar]
- Saito, S.; Lin, Y.C.; Murayama, Y.; Hashimoto, K.; Yokoyama, K.K. Human amnion-derived cells as a reliable source of stem cells. Curr. Mol. Med. 2012, 12, 1340–1349. [Google Scholar]
- Li, H.; Kim, K.H. Effects of mono-(2-ethylhexyl) phthalate on fetal and neonatal rat testis organ cultures. Biol. Reprod. 2003, 69, 1964–1972. [Google Scholar]
- Ugarte, F.; Forsberg, E.C. Haematopoitic stem cell and niches: New insights in spire new questions. EMBO J. 2013, 32, 2535–2547. [Google Scholar]
- Katoh, M.; Katoh, M. Comparative integromics on FZD7 orthologs: Conserved binding sites for PU1 SP1 CCAAT-box and TCF/LEF/SOX transcription factors within 5′-promoter region of mammalian FZD7 orthologs. Int. J. Mol. Med. 2007, 19, 529–533. [Google Scholar]
- Saito, S.; Sawai, K.; Minamihashi, A.; Ugai, H.; Murata, T.; Yokoyama, K.K. Derivation maintenance and induction of the differentiation in vitro of equine embryonic stem cells. Methods Mol. Biol. 2006, 329, 59–79. [Google Scholar]
- Ford, C.E.; Pollock, D.L.; Gustavsson, I. Proceedings of the first international conference for the standardisation of banded karyotypes of domestic animals University of Reading England 2nd-6th August 1976. Hereditas 1980, 92, 145–162. [Google Scholar]
- Tang, F.; Tang, G.; Xiang, J.; Dai, Q.; Rosner, M.R.; Lin, A. The absence of NF-kappaB-mediated inhibition of c-Jun N-terminal kinase activation contributes to tumor necrosis factor alpha-induced apoptosis. Mol. Cell. Biol. 2002, 22, 8571–8579. [Google Scholar]
- Nakade, K.; Pan, J.; Yoshiki, A.; Ugai, H.; Kimura, M.; Liu, B.; Li, H.; Obata, Y.; Iwama, M.; Itohara, S.; et al. JDP2 suppresses adipocyte differentiation by regulating histone acetylation. Cell Death Differ. 2007, 14, 1398–1405. [Google Scholar]

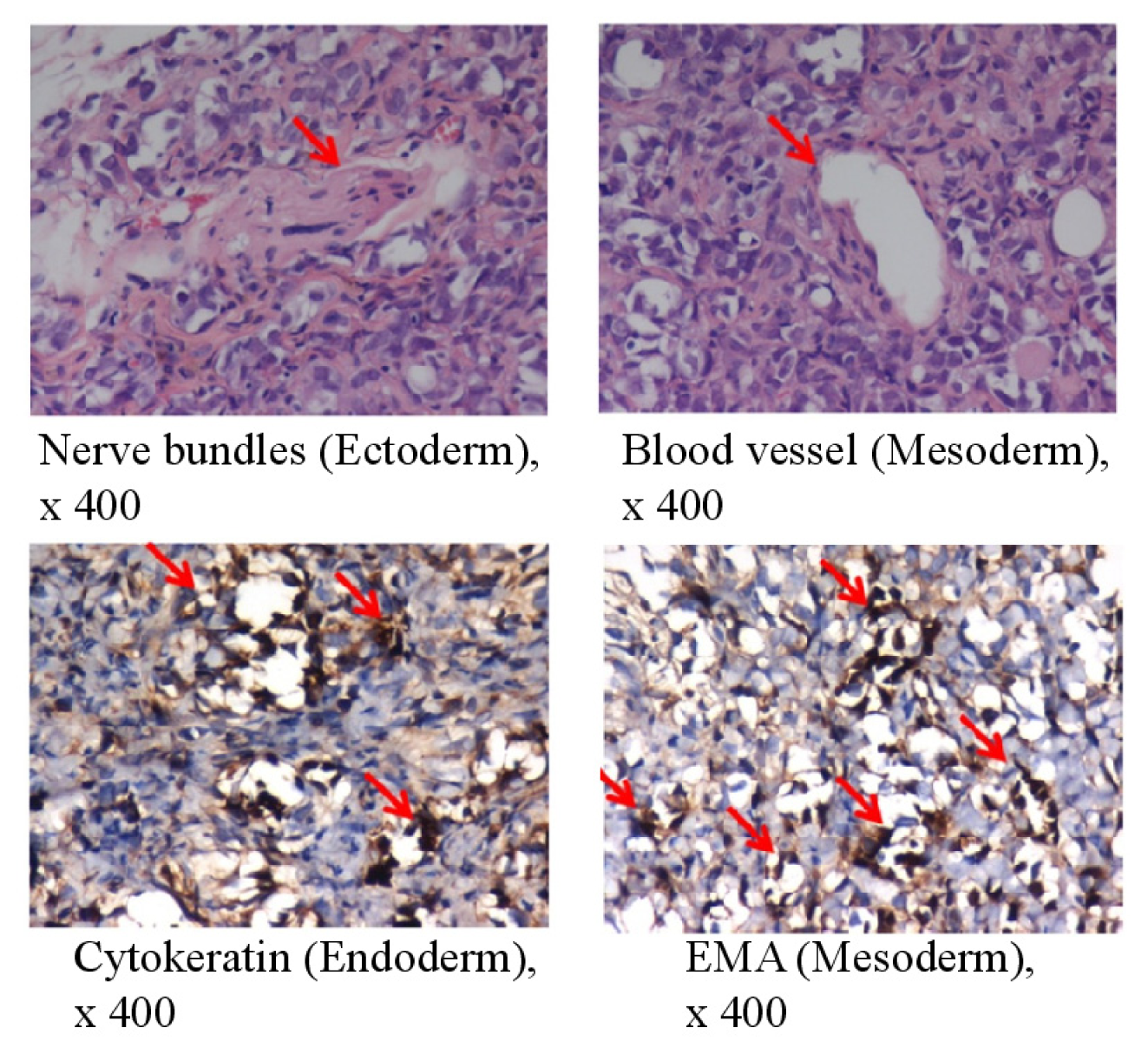


| Characterizationof bovine testicular cells and induced pluripotent stem cells | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Cell type | Teratoma formation | Cell-surface markers | Stem cell genes | |||||||
| SSEA-1 | SSEA-3 SSEA-4 | AP | 4-Oct | Nanog | Sox2 | Klf4 | C-Myc | Stat3 | ||
| Testicular cells | − | +/− a | +/− a | +/− a | − | − | − | + | + | + |
| Testicular iPSCs | + | + | + | + | + | + | + | + | + | + |
| Primers | Gene | 5′→3′ | Size of amplified DNA (bp) |
|---|---|---|---|
| 1 | oct3/4-F | ccctgaggagtcccaggacat | 356 |
| oct3/4-R | gcaggaacatgctctccaggtt | ||
| 2 | sox2-F | ctacagcatgatgcaggaccagct | 381 |
| sox2-R | tgctgggacatgtgaagtctgctg | ||
| 3 | gklf4-F | gttcgtgttgaaggcgtcgctg | 173 |
| gklf4-R | tgcacgaggagacagcctcct | ||
| 4 | c-myc-F | ccaagctcgtctcggagaagc | 334 |
| c-myc-R | tcagagtcgctactggtcgtgg | ||
| 5 | SALL4-F | catagacaaggccaccaccgacc | 276 |
| SALL4-R | atgtgcatgcggatgtgctgct | ||
| 6 | ID1-F | acgacatgaacggctgctactc | 142 |
| ID1-R | tgggattccgagttgagctccaa | ||
| 7 | EED-F | atagcaatacaagccatcccctgc | 223 |
| EED-R | aatattgccaccagagtgtccgtc | ||
| 8 | SUZ12-F | gcagttcactcttcgttggacagg | 449 |
| SUZ12-R | cctgaggatttcctgcataggagc | ||
| 9 | STAT3-F | gtctaacaatggcagcctctcagc | 405 |
| STAT3-R | aagagtttctccgccagcgtc | ||
| 10 | GADD45A-F | ctttggaggaattctcggctggag | 252 |
| GADD45A-R | cattctcacagcagaatgcctgg | ||
| 11 | SMAD4-F | ttcatgactttgagggacagcca | 438 |
| SMAD4-R | gctcattgtgaactggtggccag | ||
| 12 | DNMT1-F | cggtgttcacaaaggactgcaacg | 359 |
| DNMT1-R | gtactgaccagcctgcagcac | ||
| 13 | DNMT3A-F | tgcaagaactgcttcctggaatgc | 398 |
| DNMT3A-R | accagaagccctgtagcaattcc | ||
| 14 | TERT-F | cctacgtggtggagctgctcag | 155 |
| TERT-R | tgacagttctcgaagccgcac | ||
| 15 | MEF2A-F | atgcctccactgaatacccaaagg | 217 |
| MEF2A-R | acacctgtcccagagacagcat | ||
| 16 | MEF2C-F | ggtatggcaatccccgaaactcac | 408 |
| MEF2C-R | gccagccagttactgacccaagat |
| Primers | Gene | 5′→3′ |
|---|---|---|
| 1 | Androgen receptor-F | CAGTGGATGGGCTGAAAAAT |
| Androgen receptor-R | AGGAGCTTGGTGAGCTGGTA | |
| 2 | p21/Cip1-F | ATGGGTCTGGGAGATGTGAG |
| p21/Cip1-R | CATATGGGAGCCAGGAGAAA | |
| 3 | GSK3β-F | CGTGATCCTTCCGCCGCTTCC |
| GSK3β-R | TCCACTCCCTTTCCTTGGAGGGCA | |
| 4 | CTNNB1-F | CCATTCCATTGTTTGTGCAG |
| CTNNB1-R | TGCATATGTTGCCACACCTT | |
| 5 | Frizzled | 7-F TTGCCTCTGGACCTTTGCAC |
| Frizzled | 7-R CGTGGTCTGGCACTGAGATG | |
| 6 | LEF-F | CACCCTGAAGAGGAAGGTGAC |
| LEF-R | GAGGTTTGTGCTTGTCTGGC | |
| 7 | TCF3-F | CGTGGCAGCTGATACAGCCGA |
| TCF3-R | GGCCGCTTTAGGGTTCAGGTTACG | |
| 8 | GAPDH-F | GGGTCATCATCTCTGCACCT |
| GAPDH-R | GGTCATAAGTCCCTCCACGA |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lin, Y.-C.; Kuo, K.-K.; Wuputra, K.; Lin, S.-H.; Ku, C.-C.; Yang, Y.-H.; Wang, S.-W.; Wang, S.-W.; Wu, D.-C.; Wu, C.-C.; et al. Bovine Induced Pluripotent Stem Cells Are More Resistant to Apoptosis than Testicular Cells in Response to Mono-(2-ethylhexyl) Phthalate. Int. J. Mol. Sci. 2014, 15, 5011-5031. https://doi.org/10.3390/ijms15035011
Lin Y-C, Kuo K-K, Wuputra K, Lin S-H, Ku C-C, Yang Y-H, Wang S-W, Wang S-W, Wu D-C, Wu C-C, et al. Bovine Induced Pluripotent Stem Cells Are More Resistant to Apoptosis than Testicular Cells in Response to Mono-(2-ethylhexyl) Phthalate. International Journal of Molecular Sciences. 2014; 15(3):5011-5031. https://doi.org/10.3390/ijms15035011
Chicago/Turabian StyleLin, Ying-Chu, Kung-Kai Kuo, Kenly Wuputra, Shih-Han Lin, Chia-Chen Ku, Ya-Han Yang, Shin-Wei Wang, Sheng-Wen Wang, Deng-Chyang Wu, Chun-Chien Wu, and et al. 2014. "Bovine Induced Pluripotent Stem Cells Are More Resistant to Apoptosis than Testicular Cells in Response to Mono-(2-ethylhexyl) Phthalate" International Journal of Molecular Sciences 15, no. 3: 5011-5031. https://doi.org/10.3390/ijms15035011






